Abstract
Background:
Suprachoroidal surgery can lower intraocular pressure and medication use. There is currently no commercial suprachoroidal product on the market. Here, we report our 1 year results of a novel ciliary sulcus suprachoroidal microtube technique.
Purpose:
To determine the real-world efficacy of intrascleral ciliary sulcus suprachoroidal microtube technique in Black and Afro-Latinx patients with glaucoma refractory to topical ocular hypertensive medications.
Methods:
A retrospective non-comparative single center study of 36 Black and Afro-Latinx patients with glaucoma and pseudophakia who underwent intrascleral ciliary sulcus suprachoroidal microtube surgery for glaucoma as a stand-alone procedure at a single practice. Investigated parameters were number of medications, visual acuity (VA), intraocular pressure (IOP), mean deviation on visual field (VF) test. Success was defined as (a) IOP ⩽ 15 mmHg and or ⩾ 20% reduction in IOP and (b) a reduction in number of medications. We used paired t-test to compare baseline and follow-up parameters.
Results:
We reviewed a total of 36 patients who had undergone the procedure. Twenty had success with 12-month follow-up in the study. The mean number of medications decreased significantly from 4.2 ± 1.0 preoperatively to 2.4 ± 1.7 in 12 months (p = 0.021) with five patients being medication free. In addition, the IOP decreased significantly from 21 ± 8.2 to 13.5 ± 4.4 mmHg (p = 0.032). In the 20 patients who had 12-month follow-up, the VA remained stable from Log Mar 0.62 ± 0.6–0.46 ± 0.6 (p = 0.052). VF in patients with successful procedure (no further interventions) remained stable from baseline mean deviation of –16.53 ± 10.04 to –16.82 ± 9.80 dB at 6–12 months. Adverse effects were transient and included IOP spike, hypotony, hyphema, and cornea edema that were treated and resolved.
Conclusions:
This 12-month retrospective study demonstrated that intrascleral ciliary sulcus suprachoroidal microtube surgery can reduce IOP and medication burden in pseudophakic patients with glaucoma. However, despite several successes, surveillance should take place for IOP spikes, possible tube obstruction, and need for additional glaucoma surgery.
Keywords
Introduction
Glaucoma is a major cause of preventable blindness. This progressive optic neuropathy will affect about 110 million people globally by 2040.1–3 The treatment for glaucoma is lowering intraocular pressure (IOP) to reduce retinal nerve fiber layer and ganglion cell loss which leads to VF deterioration. Topical medical eye drop therapy is often the first line of treatment for glaucoma.4–7 Microinvasive glaucoma surgery (MIGS) and cataract surgery is rapidly gaining favor over the gold standard treatment for glaucoma – trabeculectomy and tube shunt surgery – due to its high safety and efficacy. Uncomplicated MIGS procedures are microinvasive, have minimal trauma, excellent performance, rapid recovery, and a high safety profile. Globally, affordability is an issue with the treatment for glaucoma with medication or surgery. In sub-Saharan African countries, the average per capita income is US$762 per year. According to the Census Bureau in the United States, there are great disparities in wealth. In 2016, the median net worth of African American households was US$17,113, while the comparable figure for white households was US$111,740.40. Medical devices are also very expensive due to regulations in the United States, research, clinical trials, patents, and the United States Food and Drug Adminisitration approval process. In addition, insurance companies require that devices be approved and then added to their formulary for use and payment through their reimbursement processes. 8
The suprachoroidal space (SCS) is involved in the uveoscleral aqueous outflow pathway.9–12 This space serves as the interface between the internal surface of the sclera and the external surface of the choroid. Physiologic studies have shown a negative pressure gradient between the anterior chamber (AC) of the eye and SCS.9–12 The drainage is hypothesized to drain through the SCS into the perivascular and perineural spaces of the sclera nerves and blood vessels. Ultimately, clearing via the episcleral venous plexus. Second, aqueous drains via choroidal absorption 13 into scleral veins. 9 Historically, cyclodialysis was used as a surgical approach to lower IOP. This was performed by creating a fistula between the AC and the SCS. 14 However, closure of the ciliary cleft can occur and result in an IOP spike. This is thought to be caused by a sudden reduction in outflow through the cleft with decreased trabecular meshwork functioning and decreased Schlemm’s canal and collector channel outflow. 14
Recent ab-interno supraciliary MIGS devices have been successful in IOP lowering, including CyPass (Alcon, Vernier-Geneva, Switzerland) 11 and iStent Supra (Glaukos Corporation, San Clement, CA). 15 The CyPass was withdrawn from the market due to increased corneal endothelial loss in a 5-year study and longer-term safety concerns. 16 The iStent Supra is currently not available on the market. Currently, there are no commercial readily available IOP reducing procedures that leverage the supraciliary space. There is another ab-interno MINIject system (iSTAR Medical SA, Wavre, Belgium) that is currently under clinical trials and is not yet available.17,18 We previously reported an initial case series with excellent lowering of IOP and medication burden with the intrascleral ciliary sulcus suprachoroidal microtube technique in patients with open angle glaucoma.19,20 Now, we report longer-term follow-up in a larger group of patients using an affordable supraciliary procedure.
Methods
This is a retrospective non-comparative study of 36 Black and Afro-Latinx glaucoma patients with pseudophakia who underwent intrascleral ciliary sulcus suprachoroidal microtube surgery at a single practice in New York, USA between December 2018 and December 2019. The ethnic demographic was reflective of the local community in Harlem New York City and Queens Village, New York.
All patients had a complete ophthalmological examination that included visual acuity (VA), Goldmann applanation tonometry, slit lamp examination, optic nerve assessment, and VF examination. The indications for intrascleral ciliary sulcus suprachoroidal microtube surgery were to reduce IOP and to reduce dependency on glaucoma medications. Disease progression or noncompliance with medications was an indication for surgery. This procedure was offered prior to trabeculectomy due to better safety profile and not having a lifelong bleb. As the superior conjunctiva was not altered, trabeculectomy could be performed if needed. Safety was evaluated by documenting adverse events from the postoperative period and on.
The patient’s glaucoma medications were halted in the operative eye immediately after the surgery. After the procedure, patients were prescribed prednisolone acetate 1% (Allergan; Irvine, CA) ophthalmic solution one drop qid, ofloxacin 0.3% (Rising) one drop every qid, and ketorolac 0.5% (Allergan) one drop tid. Pre-operative data were reviewed prior to the day of surgery, and the post-operative course was reviewed at 1 day, 1 month, 3 months, 6 months, and 12 months. Individual IOP goals were set for patient, and IOP-lowering medications were reintroduced as needed. Success was defined as (a) IOP ⩽ 15 mmHg and or ⩾ 20% reduction in IOP and (b) a reduction in number of medications ⩾ 1.
Statistics
We tested the hypothesis that intrascleral ciliary sulcus suprachoroidal microtube surgery would reduce glaucoma medicine use while maintaining acceptable IOP control. Data analysis was done with Microsoft excel using paired t-test of sample means.
Surgical device
A sterile medical grade silicone tube (Tube extender, New World Medical) measuring a 300 μm internal diameter and 600 μm external diameter. The tube can be cut to any size length, usually (7–10 mm). The tube can then be customized for smaller or large eyes. The silicone microtube is flexible and during insertion assumes the curvature of the globe. The tube is initially inserted into the ciliary sulcus. The tube in then sutured to the sclera to prevent it from migrating. Then, an incision if made beneath the sclera flap and the tube in then inserted into the SCS. The partial thickness scleral flap is then sutured closed to the sclera over the tube.
Surgical technique
Informed consent was obtained and witness for all patients. The surgical technique has been previously described.19,20 The patients received a peribulbar block inferotemporally with a mixture of 2% lidocaine and 0.75% Marcaine. This anesthetizes the posterior ciliary nerves travel in the SCS. The patient is dilated preoperatively to see the insertion of the microtube behind the iris, in front of the intraocular lens and not in the visual axis. An 8-0 double-armed vicryl suture was passed through the inferotemporal cornea to retract the globe into the superonasally. This is done from the temporal position and will increase visibility and access to the inferotemporal space. A conjunctival peritomy is made in the inferotemporal quadrant. Inferotemporal tube placement allows the superior conjunctiva to be preserved for any future surgery. Wet-field cautery was used to maintain hemostasis. A partial thickness scleral flap was created with .12 forceps and a crescent blade. The flap measured approximately 4 by 4 mm. A paracentesis was made into the clear cornea superiorly. Viscoelastic (Healon) was inserted into the AC to expand inferior ciliary sulcus space between the iris and intraocular lens. A 23-gauge needle was then used to make in incision 2 mm from the limbus inferotemporally beneath the scleral flap into the ciliary sulcus. The tip of the microtube is cut 45 degrees with the bevel down for insertion. The microtube was then implanted through the opening into the ciliary sulcus between the iris and ciliary body. The technique is similar to placing traditional glaucoma tubes into the ciliary sulcus. The tube is positioned behind the iris and in front of the intraocular lens, not in the visual axis, and bevel facing the intraocular lens. The microtube is then secured to the sclera centrally beneath the scleral flap with a 10-0 prolene suture through the tube. This ensures no tube migration. A cyclodialysis spatula was placed into the AC to confirm the position of the microtube in the sulcus in front of the intraocular lens and behind the dilated iris. Approximately, 5 mm from the sclera beneath the scleral flap a crescent blade was used to make a 2 mm incision through the sclera into the SCS (Figure 1). The posterior end of the microtube extending 3 mm over the posterior scleral incision was cut flush. The posterior 3 mm of the tube was gently inserted into the SCS. Two 8-0 vicryl sutures were used to close the scleral flap. Two 8-0 vicryl sutures were used to close the inferotemporal conjunctival peritomy. Approximately, 75% of the viscoelastic was left in the eye to keep the chamber formed during the anticipated transient hypotony. A balanced salt solution (BSS) hydration of the paracentesis site was used to close it. Diluted preservative-free Moxifloxacin (Alcon) (50/50 with balanced saline solution) was given intracamerally, and topical 0.1 cc of triamcinolone acetonide (Kenalog, Bristol Myers Squibb) was given. An eye shield was placed over the eye. Ultrasound biomicroscopy imaging technique can be used to confirm position of microtube.

Microtube sutured to sclera beneath scleral flap. Proximal end into the ciliary sulcus. Distal end about to be inserted into the suprachoroidal space.
Results
A total of 36 patients were included in the study. In Table 1, the baseline characteristics of patients are presented. The mean age (± standard deviation) of the study patients were 69 ± 13 years. Females outnumbered males 1.25:1 and left eye to right eye was even at 1:1. The mean baseline pre-operative IOP was 21 ± 8.2 mmHg with a mean use of 4.2 ± 1.0 ocular pressure lowering medications. All the patients were on two or more medications, while 83% were on four or more medications, 11.1% on three medications, and 5.6% on two medications. The average baseline VF mean deviation of the patients was –15.13 ± 12.55 dB and the baseline mean VA in logMAR was 0.62 ± 0.58. Primary open angle glaucoma (POAG) was the primary diagnosis in 66.7% of the sample population, chronic angle closure glaucoma in 22.2%, pigmentary glaucoma in 5.6%, uveitic glaucoma with 2.8%, and ocular hypertension in 2.8% of cases.
Baseline characteristics of study participants who underwent intrascleral ciliary sulcus suprachoroidal microtube surgery.

CACG, chronic angle closure glaucoma; IOP, intraocular pressure; MD, mean deviation; n, number of patients; POAG, primary open angle glaucoma; SD, standard deviation; VFT, visual field test.
There was a significant drop in IOP at each post-operative visit and in the number of medications compared with preoperative values in the patients who did not require an additional intervention. A significant decrease in mean IOP was seen at 1 month (IOP = 16.5 ± 7.2; p = 0.0015), 3 months (IOP = 14.4 ± 7.1; p = 0.0002), 6 months (IOP = 12.1 ± 5.1; p < 0.001), and 12 months (IOP = 13.2 ± 4.4; p = 0.032) (Table 2). The mean number of medications at 1 month decreased from baseline 4.2 ± 1.0–0.6 ± 1.2 (–76%; p < 0.001). At 3 months, the mean number of medications was 1.7 ± 1.8 (–60%; p < 0.001), 1.8 ± 1.7 (–57%; p < 0.001) at 6 months, and 2.4 ± 1.7 (–44%; p = 0.021) at 12 months (Table 2). In addition, five patients or 14% of the sample population were medication free at 12 months.
Main outcome measures, IOP, best-corrected visual acuity (BCVA), and number of ocular hypertensive medications at pre-operative and follow-up visits for all patients.
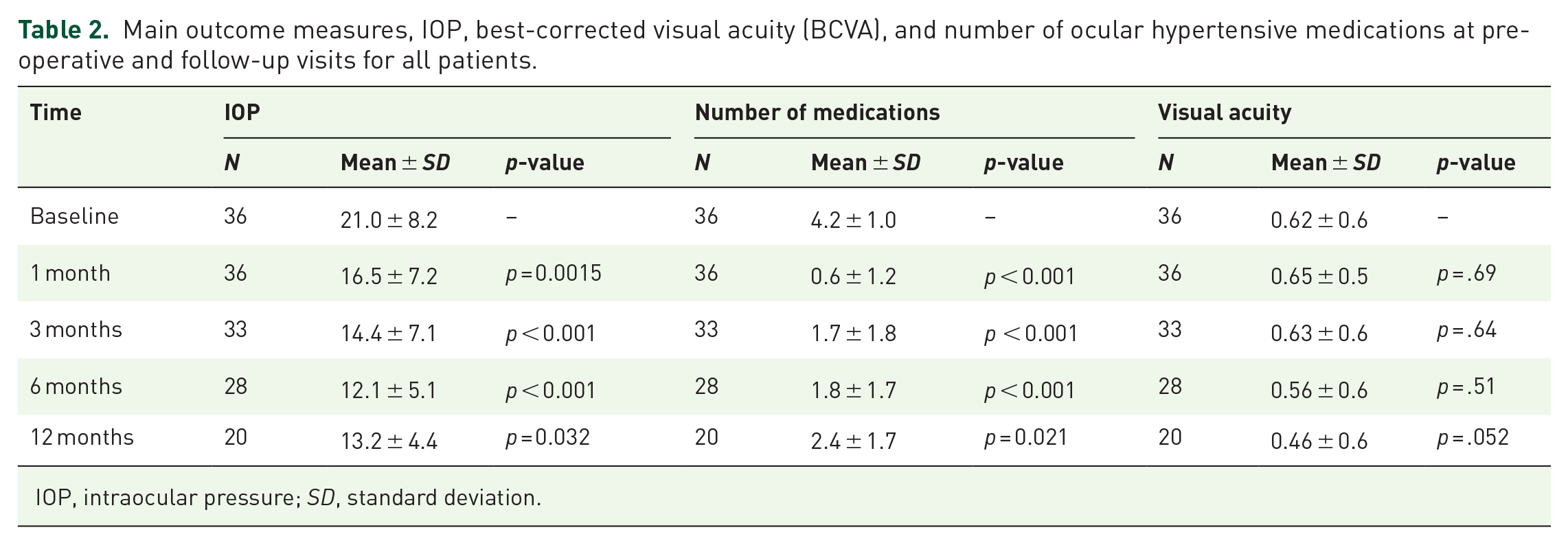
IOP, intraocular pressure; SD, standard deviation.
The average mean deviation on VF compared with baseline was stable in those patients who did not require an additional intervention (baseline: –16.53 ± 10.04 and –16.82 ± 9.8 dB at 6–12 months). Table 3 shows subgroup analysis of VF. There was more VF change in nine other patients who had failed and undergone subsequent surgeries (baseline VF: –13.1 ± 8.9 and –15.65 ± 11.1 dB at 6–12 months). Patient who underwent the intrascleral ciliary sulcus suprachoroidal microtube surgery did not experience any significant change in the VA. The mean logMAR at baseline was 0.62 ± 0.6. At 1-month follow-up, the mean logMAR was 0.65 ± 0.5 (p < 0.69), 0.63 ± 0.6 (p < 0.64) at 3 months, 0.56 ± 0.6 (p < 0.51) at 6 months and 0.46 ± 0.6 (p < 0.052) at 12 months. Although there was no significant change in VA during the 12-month follow-up, there was a trend toward better VA in patients who underwent the procedure as shown in Table 2 (p = 0.052 for logMAR vision at 12 months versus baseline).
Mean visual field MD (in decibels) pre-operatively and 6–12 months for all patients, patients with and without additional surgeries, and patients on no medications at 12 months who underwent intrascleral ciliary sulcus suprachoroidal microtube surgery.

MD, mean deviation.
Adverse events were self-limited and non-sight threatening. There were no sight threatening intraoperative complications nor ocular discomfort. Potential complications that could occur during the dissection of the scleral flap and incision through the scleral base to the SCS did not occur in this series. No implant migration occurred. The most common non-invasive adverse effects included IOP spike (IOP > 29 mmHg, 5.6%), hypotony (IOP ⩽ 5 mmHg, 2.8%), hyphema (2.8%), corneal edema (2.8%), and retained viscoelastic (2.8%). However, four patients had tube obstruction by the iris (11.1%) and three patients had elevated IOP due to presumed scarring in the SCS (8.3%) as shown in Table 4. Ultimately, nine patients required additional intervention by the 12-month mark. We had one patient deceased and five patients lost to follow-up.
Complications/adverse effects of intrascleral ciliary sulcus suprachoroidal microtube surgery.

IOP, intraocular pressure.
Discussion
This study presents a 12-month real-world retrospective study of patients who underwent the intrascleral ciliary sulcus suprachoroidal microtube technique. This procedure can successfully lower IOP and reduce glaucoma medication use in successful patients. There was no visible corneal edema although specular microscopy was neither performed nor available. This technique of diverting aqueous outflow through the SCS may be safer with less risk of corneal edema compared with other similar devices when placed in the AC. Future studies should include specular microscopy. The potentially reduced risk of corneal edema is due to the positioning of the proximal end of the tube in the ciliary sulcus and away from the cornea (Figure 2). The short-term efficacy is comparable with other microinvasive stents.16,21–23 The mean IOP of patients at 12 months was 13.2 mmHg (37% reduction) with additional reduction in intraocular medication use from 4.2 to 2.4 (44% reduction). The IOP and medication burden were able to be significantly reduced in 20 patients justifying the potential role of this procedure for patients who are concerned about having a lifelong bleb from trabeculectomy and associated risks of loss of two lines of central vision and hypotony. We note the VF stabilized in five patients who were medication free at 12 months. This study shows that suprachoroidal surgery can be successful in lowering IOP and medication burden in pseudophakic glaucoma patients (Figure 3).

Proximal end of tube seen in ciliary sulcus behind the iris.

Microtube illustration showing position intrasclerally from ciliary sulcus to suprachoroidal space.
Our initial approach to adult mild to moderate glaucoma is often cataract surgery/clear lensectomy and MIGS Schlemm’s canal angle surgery first. In patients with failed lens extraction and MIGS or advanced glaucoma, we then perform trabeculectomy. The suprachoroidal surgery is an attractive alternative in patients with scarred conjunctiva, failed trabeculectomy and failed tube shunts, since the procedure ‘bleb-less’. This suprachoroidal procedure can also be used in resource-poor areas where access to more expensive supraciliary devices may not be available. In patients with scarred conjunctival, we are currently combining this technique with goniotomy in pseudophakic patients with patients with uncontrolled mild to moderate glaucoma to lower IOP and medication burden before trabeculectomy. This technique has been published 24 and we believe this has the potential to reduce the need for trabeculectomy. In this procedure, the position of the tube beneath a scleral flap minimizes the risk of microtube exposure (Figure 4).

UBM of tube seen in ciliary sulcus, intrascleral, and into the suprachoroidal space.
Another advantage with this technique is, it can be performed in any quadrant of the eye. We usually perform this in the inferotemporal quadrant sitting temporally, we have also performed this in the superior quadrant as well. This can also be performed in eyes with scarred conjunctiva from previous failed surgery. The most common adverse effects in our patients were transient and included IOP spike, hypotony, hyphema, and cornea edema that all were self-limiting. However, nine patients required additional glaucoma surgery for uncontrolled IOP. There was no incidence of flattened AC, aqueous misdirection, choroidal effusions, choroidal hemorrhage, cystoid macular edema, or morphologic changes at the retina. The placement of the silicone tube in the ciliary sulcus may reduce the risk of corneal edema seen in SCS devices like CyPass. 11 To prevent tube migration, it is important to pass the 10-0 prolene suture through the tube to anchor and tie it to the sclera beneath the scleral flap.
It is known in the literature that the cell proliferation and expression patterns of the extracellular matrix proteins differ between the scleral and choroidal fibroblasts in the SCS compared with the conjunctival fibroblasts. 25 This suggests that there could be less fibrosis in the SCS compared with the subconjunctival space. However, it does not refute the fact it can occur. 25
During this study, there were IOP spikes over 29 mmHg in 5.6% of cases that responded well to topical medication. During these IOP spikes, there were no abnormal findings during the examination, the AC was quiet and deep, and the microtube had no apparent block at the proximal end upon dilation. These pressure spikes can go unnoticed and thus patients will need close monitoring. These pressure spikes usually do not cause blurred vision, pain, nor discomfort. Patients may experience increased pressures over long periods of time in between office visits creating a higher risk for progression. Thus, these patients still do require ongoing surveillance similar to patients who undergo traditional glaucoma incisional glaucoma surgeries. Most of the patients will need additional medial therapy to reach their target IOP post-operatively.
The VF remained stable for patients who had success with the procedure (baseline: –16.53 ± 10.04 dB versus –16.82 ± 9.80 dB at 12 months). In patients who successfully undergo the intrascleral ciliary sulcus suprachoroidal microtube technique, we have shown this can stabilize the VF and potentially reduce further progression. Overall, the mean visual defect of our patients at baseline was –15.85 ± 9.75 dB. However, at 12 months, the VF progressed to –16.58 ± 9.85 dB. On subgroup analysis, this deterioration could be attributed to the patients who failed the procedure and required secondary intervention (baseline: –13.1 ± 8.9 versus –15.65 ± 11.09 at 6–12 months).
This intrascleral ciliary sulcus suprachoroidal technique can be used in eyes that have milder glaucoma, not had glaucoma surgery or had previous failed glaucoma surgery with traditional trabeculectomy or glaucoma tube shunts or scarred conjunctiva. Outside of the United States in resource-poor areas, similar silicone microtubes can be obtained from the AADI (Aurolab, India) implant. It has a similar sized inner diameter silicone tube opening of 300 μm. As an example of cost-efficiency, Hurricane Medical makes a sterile silicone tube with similar dimensions that costs about US$10. 20 In resource-poor areas, further study with this technique and less-expensive microtube provides an opportunity for additional research.
In this retrospective study, the results are promising in the patients who were successful. This can avoid the bleb-related complications from traditional trabeculectomy. The failure rate was higher than trabeculectomy though (Figure 5, Kaplan–Meir curve; Figure 6, scatter plot; Figure 7, medication bar graph). However, this was a group of patients who already had cataract surgery with moderately advanced glaucoma. Intrascleral ciliary sulcus suprachoroidal microtube surgery could be considered after failed traditional MIGS in patients with mild to moderate glaucoma. Further research is required with longer follow-up data. We also do not recommend this technique in neovascular glaucoma. In mild to moderate glaucoma patients, we currently combine excisional goniotomy with a 23-gauge cystotome and the intrascleral ciliary sulcus suprachoroidal surgery to further enhance IOP lowering, reduce IOP spikes, and increase success. 24 Limitations of the study include non-randomized nature of study, retrospective study, limited follow-up period, and small sample. In addition, the loss to follow-up was high (14%) with no detailed data on corneal biomarker and definitive evidence of scarring in the SCS. In this challenging group of patients with moderately advanced glaucoma, the study provides real-world evidence of the efficacy and complications of intrascleral ciliary sulcus suprachoroidal microtube surgery.

Survival curve.

Scatter plot of intraocular pressure.

Bar graph of pre- and post-operative medications.
Conclusion
We have reported the outcomes of a novel technique using a silicone microtube for ab externo intrascleral ciliary sulcus suprachoroidal shunting of aqueous to lower IOP in patients with glaucoma. This surgical technique is potentially safer than other previous suprachoroidal surgeries due to the placement in the ciliary sulcus. This potentially reduces the risk of corneal edema that was not seen in this study. This technique can be used in pseudophakic glaucoma with uncontrolled IOP, angle closure patients who are pseudophakic, patients with other failed micro incisional glaucoma surgery, failed trabeculectomy, or failed tube shunt. In resource-poor areas without access to a microscope that tilts, it can be performed at the time of cataract surgery as well. We do not recommend this procedure be performed in phakic patients due to the presence of the natural lens in the posterior chamber, nor placement of the microtube in the AC due to the increased risk of corneal endothelial compromise. We do recommend that initial cataract extraction and trabecular bypass surgery be performed before this procedure to expand Schlemm’s canal and restore the physiologic outflow of the eye.26,27 In advanced glaucoma, we then perform trabeculectomy or traditional tube shunt surgery. Should these fail, then this suprachoroidal procedure can be considered to allow for aqueous bypass of the trabecular meshwork, to have greater success at lowering IOP. This can also be used in patients with conjunctival scarring as well. This supraciliary glaucoma technique appears manageable, and the learning curve for glaucoma surgeons seems to be acceptable.
In conclusion, this technique offers an additional procedure to access the suprachoroidal pathway. There continues to be the challenges of surveillance for IOP spikes and need for additional surgery.
Footnotes
Acknowledgements
The authors thank Louis R. Pasquale MD for his editorial assistance and Harriet Lloyd from the Einhorn Clinical Research Center at New York Eye and Ear Infirmary of Mount Sinai for administrative support. Finally, the authors thank the staff at Advance Eye Care of New York for their assistance and support.
