Abstract
Background:
Laser peripheral iridotomy (LPI) is the current standard of care for primary angle-closure glaucoma. The existing literature lacks evidence regarding the effects of LPI on contrast sensitivity (CS) after the procedure.
Objective:
This study evaluates central and peripheral CS in patients undergoing LPI using the computer-based, Spaeth/Richman Contrast Sensitivity (SPARCS) test.
Methods:
We performed a pilot, prospective, interventional cohort study including 30 patients of primary angle-closure suspect (PACS) or primary angle closure (PAC) in both eyes. LPI was performed after a detailed history and clinical examination using standard procedure in all eyes. Intraocular pressure (IOP) and CS testing using SPARCS was performed before, 2 weeks and 3 months after LPI.
Results:
Data analyses revealed female predominance (66.67%, 20/30); the mean age of enrolled patients was 49.93 ± 10.43 years, and presenting acuity was 0.02 ± 0.06 (Log of Minimum Angle of Resolution [LogMAR]). The mean vertical cup-to-disc ratio (VCDR), mean deviation (MD in dB) and pattern standard deviation (PSD in dB) were 0.34 ± 0.09, –2.36 ± 1.72 and 2.34 ± 0.81, respectively. There was a statistically significant decrease between the pre- (15.17 ± 3.83 mmHg) and 2 weeks post-LPI (11.70 ± 1.53 mmHg) IOP (p < 0.001). However, CS in the pre- (73.47 ± 9.88) and 3 months post-LPI (75.20 ± 11.98) SPARCS scores did not reveal any statistical difference. The group-wise analysis showed a similar trend between PAC and PACS patients.
Conclusion:
LPI does not affect central as well as peripheral CS assessment in patients with the primary angle-closure disease.
Introduction
Glaucoma is the second most common cause of blindness globally, and visual impairment in 1 in 45 and blindness in 1 in 15 patients are attributed to it. 1 Among the people diagnosed with glaucoma, primary angle closure disease (PACD) is the most common cause of glaucoma-associated blindness.2,3 Laser peripheral iridotomy (LPI) is the standard and primary management option for PACD.4–8 LPI very efficaciously relieves the pupillary block and also increases the angle width, which can be confirmed gonioscopically or using ultrasound biomicroscopy, and anterior segment optical coherence tomography in eyes with or without peripheral anterior synechiae (PAS). 1
Medicare data reports more than 80,000 LPI procedures (2003–2012) were performed every year in the United States. 2 This number reflects not only the role of the procedure in the management of the PACD but also its relative safety and efficacy.2,3 The current clinical evidence has reports implicating LPI in post-procedural complications like intraocular pressure (IOP) spikes, subjective visual dysphotopsias, anterior chamber bleeding, the progression of cataract and decrease in endothelial cell counts, but the effect on contrast sensitivity (CS) post-LPI has not been addressed. 1
CS is the ability to perceive slight changes in the luminance between the adjacent areas, which helps in determining the difference in colour and brightness of the objects in the real world. Thus, CS plays a critical role in visual acuity, visual field testing, dark adaptation, motion recognition and pattern recognition. Glaucoma affects the CS, and any additional alteration induced by either the laser procedure such as LPI or size and location of fenestra might play a role in visual compromise.9,10 Post-LPI accumulation of aqueous melanin granules and iris pigment in the anterior chamber especially on the corneal endothelium can clinically affect CS without influencing the visual acuity, thus affecting the visual function.
The changes in CS post-argon laser iridotomy was first reported by Anderson et al., 4 where the authors described a slight increase at low spatial frequencies and a small decrease at high spatial frequencies post-iridotomy. The focus of the report was to study the phototoxicity associated with the laser procedure and did not follow up on the patients specifically for CS. Short-term outcomes of LPI have indicated stability or improvement in visual parameters after the procedure. 5
The rationale of this pilot study was to fill in the knowledge gaps regarding the possible effect of LPI on both central and peripheral CS in patients with the primary angle closure (PAC) and primary angle-closure suspect (PACS). For measuring the CS, we used the Spaeth/Richman Contrast Sensitivity (SPARCS) test, which is a free, computer-based tool for CS. This tool is a reliable and repeatable test for CS assessment.6,7
Materials and methods
We recruited 33 consecutive Asian Indian patients over a period of 3 months from, at the Glaucoma Clinic of Government Medical College and Hospital, Chandigarh, India. The inclusion criteria were a recent diagnosis of PACS or with PAC in both eyes and having an age of ⩾40 years. We randomly selected one eye of each patient for analysis. This being a pilot study, prior sample size calculation was not performed.
Clinical assessment
All patients underwent a detailed ocular examination at the first visit, which included complete ocular history, best-corrected visual acuity (BCVA), IOP as measured with calibrated Goldman applanation tonometry (GAT), gonioscopy with four mirror lens (modified Schaffer’s grading), dilated fundus examination with stereoscopic biomicroscopy of the optic nerve head using slit lamp and +90 D lens after LPI, indirect ophthalmoscopy (when indicated) and white-on-white visual field testing using 24-2 SITA FAST on Humphrey Field Analyser II. A visual field test was deemed reliable only if the fixation losses and false-positive rates were less than 20%.
CS assessment using SPARCS test, IOP and BCVA assessment was done at index visit, 2 weeks and 3 months after LPI. Patient classification into PACS or PAC was done using Foster’s Classification cited in the American Academy of Ophthalmology Preferred Practice Guidelines. Briefly, under this classification, PACS is defined with ⩾180 degrees iridotrabecular contact (ITC), normal IOP and no optic nerve damage. PAC is defined with ⩾180 degrees ITC with PAS or elevated IOP, but no optic neuropathy. Primary angle-closure glaucoma (PACG) is defined as eyes with narrow angles (eyes in which the posterior trabecular meshwork was not seen for at least 180° on indentation gonioscopy in the primary position) with PAS, and/or raised IOP (IOP > 21 mmHg) with glaucomatous optic neuropathy [defined as a vertical cup-to-disc (C:D) ratio (VCDR) > 0.7 and/or C:D asymmetry > 0.2 and/or focal notching/thinning] and correlating visual field loss on static automated perimetry. PACD is a blanket term that includes PACS, PAC and PACG.
All the patients who had a history of prior cataract, incisional glaucoma or any other laser surgery were excluded. In addition, patients who presented with signs and/or symptoms of acute angle-closure attack or visually significant cataract [nuclear sclerosis more than grade 2 using Lens Opacity Classification System (LOCS) III grading], diabetes mellitus and neurological diseases were excluded from the study, as they could influence CS. Patients with BCVA of less than 20/40 or patients who had undergone refractive surgeries were also excluded.
CS testing
SPARCS test was performed on a standard computer system with Internet access. The test was initiated through the website https://www.sparcscontrastcenter.com, which provided each patient with a unique identification number. Before initiation of the test, patients were briefed about the procedure and explained in a step by step manner about the test. Since all patients were new to SPARCS, a trial test was given to the patients. The monitor was set to 1024 × 768 resolution, 256 grey levels, and a size of 22 cm width and 26.5 cm height. Light-emitting diode (LED) light: 22 W, colour temperature: 6500 K and Lumens: 1900 lm were used in the room without daylight to minimize glare and to standardize the test conditions (Figure 1(a)). The patients were seated at a distance of 50 cm from the screen, and the test covered the visual field area of 30 degrees horizontally and 23.5 degrees vertically (Figure 1(b)). The patients were asked to fixate on the centre of the test screen while various vertical square gratings were flashed randomly in one of the five test areas at the spatial frequency of 0.4 cycles per degree for 0.3 s (Figure 1(c)).
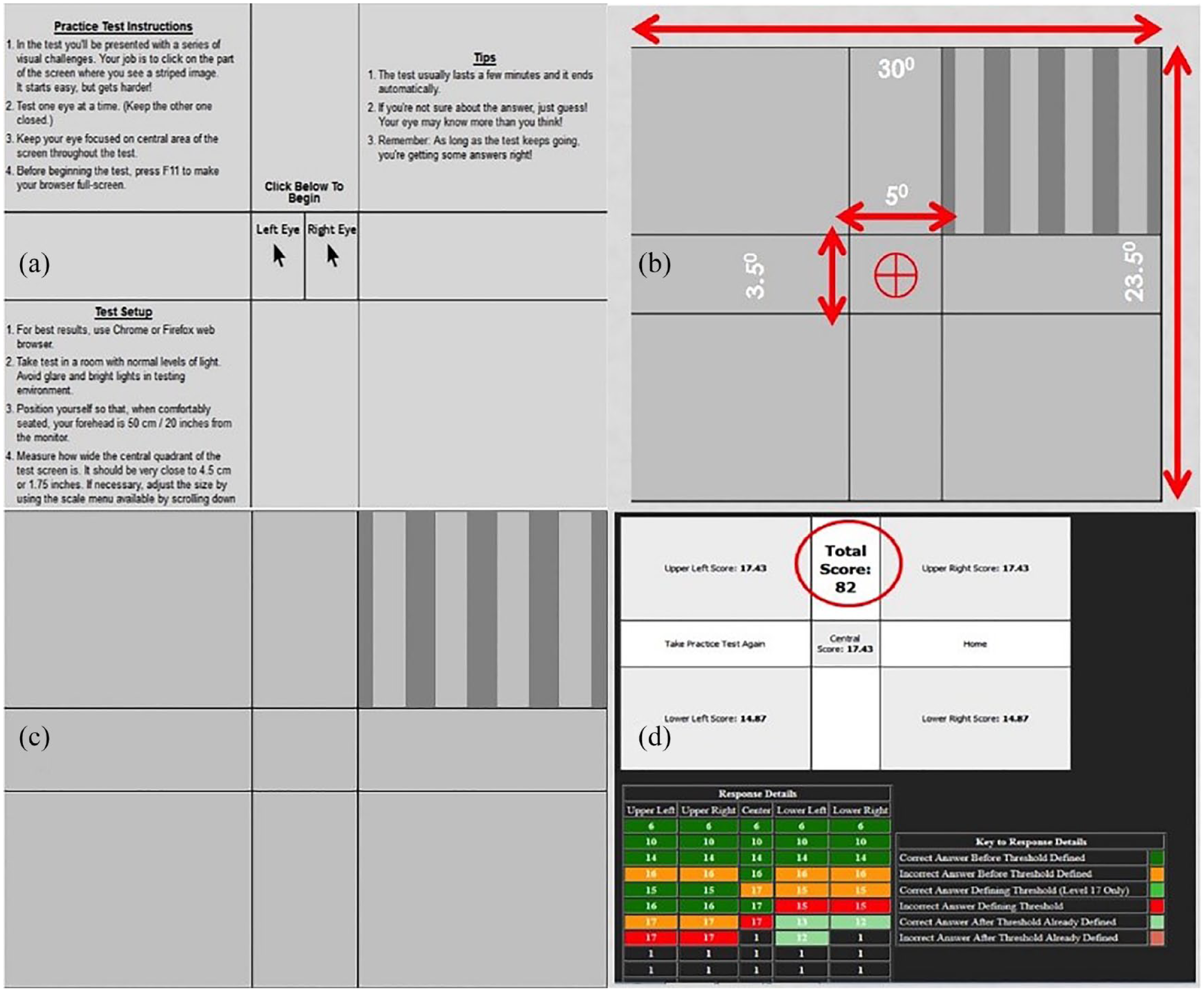
SPARCS test screen: (a) initial patient information screen with eye selection checkbox, (b) testing dimensions for the SPARCS test, (c) grating display in the upper right quadrant and (d) result screen.
While focussing on the centre, the patients were instructed to identify the area which would appear different. Every correct response would step up the contrast levels by four steps until the patient gives an inaccurate response, after which the contrast level would step down by two levels. Subsequently, the algorithm would advance or regress by one level at a time till the patient has given two incorrect responses for one particular level, which would determine the threshold for that patient in that specific area.
The contrast testing was performed in the range from 100% to 0.45% (log CS: 0.00–2.35) and decreased in the step of approximately 0.15 log units between levels. The average time taken to complete the test ranged from 5 to 10 min per eye. The scoring was done for central area (representing central contrast) and four peripheral areas separately (representing peripheral contrast) (Figure 1(d)); the scores from these five areas were summated together (a score of 20 per quadrant) to give the final scores with 100 being the perfect score. The first test was done on the initial visit after ensuring that the patient fulfilled the inclusion criteria. SPARCS test was then repeated in all patients 2 weeks (SPARCS2) and 3 months (SPARCS3) after the LPI procedure (Figure 1). We chose the timepoint of 2 weeks to re-evaluate the SPARCS test as by this point, the procedure-induced inflammation subsides significantly, and resultant fenestra also becomes pigment debris free. The final evaluation for CS was done at 3 months as by this time LPI-induced inflammation is negligible. Visual field test was also repeated at 3 months after LPI.
LPI procedure
All the patients received 2% pilocarpine twice and 0.2% brimonidine tartrate once before the procedure. LPI was done using neodymium-doped yttrium aluminium garnet (Nd:YAG) laser (Visulas Yag II; Carl Zeiss Meditec, Jena, Germany) with the help of an Abraham lens (Ocular Abraham Iridectomy YAG laser lens; Ocular Instruments, Bellevue, WA, USA) and using hydroxypropyl methylcellulose as a coupling agent. All iridotomies were performed by a single physician (P.I.). The LPI was made between the 12 and 2-o’clock positions or the 12- and 10-o’clock positions making sure that the LPI was covered entirely by the lid margin. While performing the procedure, the iris crypts were identified for the shot, giving special attention to avoid iris vessels.
LPI was performed for both the eyes at the same clinical visit, and the patency was confirmed after the procedure. LPI patency was confirmed post-procedure using standard visualization of aqueous gush, deepening on the anterior chamber and/or retro illumination through the LPI. Intraprocedural data collected included initial per-shot laser energy in millijoules, number of laser shots and total laser energy in millijoules. After 45 min of LPI, IOP was measured using the GAT. All the patients were prescribed prednisolone acetate 1% eye drops four times a day after the LPI for 10 days. In case of elevated IOP, before, after or at follow-up visit, the patient was treated with an appropriate IOP-lowering drug at the physician’s (P.I.) discretion. Patients were checked for LPI patency after 2 weeks, and patients requiring a repeat LPI procedure were excluded from the study.
Statistical analysis
All statistical analyses were done using IBM Statistical Package for the Social Sciences (SPSS, Version 23, for Windows). Descriptive data for quantitative variables were calculated as mean with standard deviation. The Shapiro–Wilk test was used to assess the normal distribution of data. The difference in the SPARCS scores and IOP before and after LPI was evaluated using paired t-test. Independent-samples t-test was used to compare these parameters between PAC and PACS eyes. The strength of linear association between the continuous variables was assessed by the Pearson correlation coefficient. A p value <0.05 was considered statistically significant.
Results
Out of 33 eyes (one eye each from each patient), 3 eyes with iris bleed following LPI were excluded from the study; therefore, data of only 30 eyes were analysed. The mean age was 49.93 ± 10.43 years, and presenting acuity was 0.02 ± 0.06 (LogMAR). Table 1 depicts the demographic profile of the study population. The patients included in this study cohort were followed up until 3 months post-LPI to ensure maintenance of target IOP, and the follow-up compliance was ensured with telephonic reminders. None of the patients included in our study required a repeat LPI procedure.
Demographic details of the study population.

BCVA, best-corrected visual acuity; IOP, intraocular pressure; LPI, laser peripheral iridotomy; MD, mean deviation; PAC, primary angle closure; PACS, primary angle-closure suspect; PSD, pattern standard deviation; SD, standard deviation; VCDR, vertical cup-to-disc ratio.
The p value is for pairwise comparison between PAC/PACS; all values in mean ± SD.
In the PAC group, three eyes had IOP elevation (but none of these three had an IOP >30 mmHg, as that could have resulted in corneal haze and altered CS, irrespective of LPI) and rest were diagnosed on the basis of ITC ⩾180 degrees with PAS. There was a statistically significant difference between the pre- and post-LPI IOP (p < 0.001) at both 2 weeks and 3 months, as assessed by paired t-test. The mean VCDR, mean deviation (MD in dB) and pattern standard deviation (PSD in dB) were 0.34 ± 0.09, –2.36 ± 1.72 and 2.34 ± 0.81, respectively.
There was only a moderate difference (statistically not significant) between the average SPARCS scores in the pre-LPI (73.47 ± 9.88), 2 weeks post-LPI (74.23 ± 12.99, p = 0.657) and 3 months post-LPI (75.20 ± 11.98; p = 0.135), if all 30 eyes (both PACS and PAC) were considered for analysis. Interestingly, SPARCS2IN and SPARCS3IN (post-LPI, contrast score from inferonasal quadrant) differed significantly from SPARCSIN (pre-LPI) (p < 0.001). Table 2 shows the mean values for the SPARCS scores in the study population. There was no statistically significant difference across the PAC and PACS groups except for SPARCSSN pre-LPI (p = 0.020) (SPARCS2SN and SPARCS3SN did not have a statistically significant difference).
SPARCS scores at different time points.

CC, centre; IN, inferonasal quadrant; IT, inferotemporal quadrant; LPI, laser peripheral iridotomy; PAC, primary angle closure; PACS, primary angle-closure suspect; SD, standard deviation; SN, supero-nasal quadrant; SPARCS, Spaeth/Richman Contrast Sensitivity; ST, supero-temporal quadrant.
The p value is for pairwise comparison between PAC/PACS; all values in mean ± SD.
There was a statistically significant decrease in the IOP after LPI (p < 0.001). SPARCS scores showed a marginal increase that was statistically not significant for both time points (2 weeks and 3 months post-LPI) on the paired t-test (p = 0.706 and p = 0.656).
Discussion
LPI has become the standard treatment option for IOP control in PACD. 1 It would be plausible to think that the small peripheral hole created in the iris after an LPI might result in stray light reaching the retina and lead to notorious visual disturbances in the form of blurring, ghost images, spots, haloes and shadows. In addition, the probable effect of Nd:YAG laser and the resultant fenestrae can result in compromised visual function. CS being a vital subset of visual function might also be affected by laser procedures. Possible mechanisms for change in CS may be direct damage to the crystalline lens, altered aqueous fluid dynamics and mild iris inflammatory changes, which may induce metabolic changes that accelerate cataract formation. Changes in CS were noted by Anderson et al. 4 as a small increase at low spatial frequencies and a slight decrease at high spatial frequencies. Hence, we undertook this pilot study to explore the effect of LPI on both central and peripheral CS in patients of the PACD.
We did not observe any previously reported immediate and short-term LPI-associated complications in our study like hyphaema, significant IOP spike, need for retreatment or any additional IOP-lowering drug.1,3,9 Single user LPI, pre-treatment (pilocarpine/brimonidine) and post-treatment (steroids) drugs may account for our observations. Pre-laser treatment with IOP-lowering agents is standard practice in our clinic, and it has been shown to significantly decrease post-laser IOP spikes.9–11 No patient in our study population required postoperative medication for IOP control as reported previously. 9 The reduction in IOP post-LPI could account for the slight improvement in average/total SPARCS score.
The location and size of LPI contributing to the visual symptoms have been a controversial topic. In a randomized control trial of 559 patients, Srinivasan et al. compared superior iridotomy and nasal/temporal iridotomy and reported no group differences regarding the proportion of patients showing visual disturbances based on iridotomy location, area or total laser energy. Their findings are in stark contrast to previously published literature that suggests more haloes with temporal laser iridotomy that are uncovered by the eyelid. 12
In another study that evaluated the light scatter and subjective visual symptoms in patients treated with LPI, Congdon et al. 13 also reported that neither iridotomy size nor location parameters were associated with stray light or subjective symptoms. These findings indicate that other factors like ocular surface, cataract and changes in CS may account for the visual symptoms reported classically after LPI, and iridotomy location or size may not be the cause of these.12,14,15 We performed supero-temporal LPI in all our patients, and, as previously demonstrated, the location or size of patent LPI does not seem to affect the occurrence of visual disturbances. 3 None of our patients reported the occurrence of any visual disturbances or dysphotopsias after the procedure. Also, no significant difference was seen in the CS values based on the power of the laser, number or shots, initial power setting or baseline visual field indices, both when analysed as subgroups (PACS, PAC) and when analysed together. The significant improvement in the inferonasal quadrant (quadrant diametrically opposite to the LPI site) on SPARCS, however, seems to be an incidental finding, which cannot be explained. A larger dataset is needed to see whether the improvement in this quadrant is noted consistently.
In a recent work, Trevino et al. evaluated CS with the Pelli–Robson chart and the CSV-1000 test (with and without glare) in PAC and PACS patients undergoing LPI. They found that CS values with both the tests were unchanged following either superior or temporal LPI. 16 Both Pelli–Robson and CSV-1000 look at only central CS, unlike SPARCS.
Erdurmus et al. have emphasized that the CS test may be a helpful method for evaluating the visual recovery in pigmentary glaucoma after LPI. 17 The authors observed increase in CS especially in moderate and high frequencies with Vistech Contrast Sensitivity Test System. We also believe that LPI has a similar impact on CS in PACD.
Our study, however, has certain limitations that need to be considered. Principally, the small sample size, stringent exclusion criteria and lack of CS values immediately after the procedure could have affected the outcome of the study. As regards the stringent exclusion criteria, we wanted to make sure that there are no confounders that could influence CS. PACS and PAC both do not have glaucomatous optic neuropathy; therefore, ‘degree of glaucoma’ was not a confounder. In cases with PACG, the CS would be less anyways because of glaucomatous optic neuropathy, so whether the CS is getting affected because of glaucoma or because of LPI will be difficult to ascertain on intergroup analysis (PAC/PACS versus PACG). However, future studies to study change in CS following LPI in PACG can be done. But there too, need for additional medications will act as a confounder.
Chief strength of the study was the use of an objective tool for assessing both central and peripheral CS, SPARCS, which has already been validated as a robust measure of CS. Other strengths were the uniformity in LPI technique and pre- or post-LPI management.
In conclusion, the importance of changes in CS as a factor that causes patients to have visual dysfunction post-LPI should not be understated until this avenue is further explored. 18 Our preliminary findings, however, demonstrate that both central and peripheral CS remain unaffected or may even show a marginal improvement after LPI. Future research looking at the effect of different locations/quadrants of LPI on the peripheral CS (in relation to quadrant affected on SPARCS) may help to effectively understand the change in this important visual parameter that is often neglected in routine glaucoma care.
Footnotes
Correction (February,2023):
“The study protocol was approved by the Government Medical College and Hospital Ethics Committee vide No. 2018/041 and was conducted in strict compliance with the tenets of the Declaration of Helsinki” has been added to the “Ethics statement”.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study protocol was approved by the Government Medical College and Hospital Ethics Committee vide No. 2018/041 and was conducted in strict compliance with the tenets of the Declaration of Helsinki.
Informed consent
We obtained written informed consent from the patients before enrolment in the study.
