Abstract
Patients with a keratoprosthesis often develop complications including glaucoma, requiring glaucoma drainage devices. In most of these patients, glaucoma drainage devices have been shown to be safe and effective. However, occasionally, a glaucoma drainage device in the setting of a keratoprosthesis can lead to conjunctival erosion with mechanical trauma. While repeat surgical intervention may appear necessary, we report a case of a patient who had improved conjunctival erosion and glaucoma drainage device exposure after refitting of a therapeutic contact lens. Therapeutic contact lenses can be used to maintain hydration and decrease exposure while improving cosmesis and refractive error. Complications following keratoprosthesis surgery are an understudied area, particularly regarding glaucoma drainage devices, and we seek to show that careful fitting of therapeutic contact lenses may avoid the risks of repeat surgical intervention.
Keywords
Introduction
Glaucoma progression or de novo glaucoma is the most common cause of permanent vision loss in patients with keratoprosthesis (KPro).1,2 Glaucoma drainage devices (GDDs) have been shown to be safe and effective in the treatment of this condition and most patients require GDD implantation either concurrent with their KPro surgery or during their postoperative period. 1 It has been shown that a posteriorly placed pars plana GDD with corneal patch graft in conjunction with KPro has a low risk of erosion and postoperative complications. 3 Nevertheless, complications occasionally occur and need to be addressed.
Therapeutic soft contact lenses are routinely used in the postoperative management of patients with recently implanted KPro. 4 By diffusing evaporation and maintaining local hydration, soft lenses can greatly enhance patient comfort and decrease exposure complications and possibly sterile keratolysis. Moreover, they can serve the additional purposes of cosmesis and refractive error correction. 5 However, continuous wear of the contact lens is also associated with potential complications. Mechanical trauma to the area of the GDD can accelerate breakdown of the conjunctiva overlying the tube shunt. 6 Although soft lenses have occasionally been associated with negative outcomes for GDDs, improved design and customization options translate into decreased rates of complications and improved outcomes for patients. In this case, we present a patient with an exposed pars plana Baerveldt shunt with a Boston KPro-type 1 that healed with lens refitting without surgical intervention.
Case presentation
A 62-year-old Caucasian man with a history of radial keratotomy in both eyes, fungal keratitis complicated by a perforated corneal ulcer in the right eye status post multiple penetrating keratoplasties, and a KPro-type 1 presented for routine 3-month follow-up to the cornea, glaucoma, and contact lens services. He had no visual complaints but presented with mild ocular pain and irritation in the right eye for 2 weeks.
He had secondary glaucoma in the right eye status after a failed inferonasal pars plana Ahmed tube implantation (New World Medical, Rancho Cucamonga, California, USA) 11 years prior and transscleral cyclophotocoagulation with diode laser and superotemporal pars plana Baerveldt shunt placement with a corneal patch graft 7 years prior (Pfizer, New York City, New York, USA). He also had a history of an epiretinal membrane status post a pars plana vitrectomy and membrane peel. He was using dorzolamide-timolol BID, brimonidine BID, travaprost BID, prednisolone forte daily, ofloxacin BID, and ketorolac QID, all in the right eye.
His visual acuity (VA) was counting fingers at 1 foot in the right eye without improvement with manifest refraction. His intraocular pressure (IOP) was 18 mmHg. He was using a large diameter therapeutic soft lens (Kontur Kontact Lens, Hercules, California, USA) on an extended wear schedule with weekly removal for cleaning. The lens was plano prescription, 8.0 mm base curve, 20 mm diameter with 11.5 mm brown iris tint, and 4 mm clear pupil (Figure 1(a) and (b)). It was well-centered with a defective jagged lens edge defect. The contact lens was removed, and on slit lamp examination, there was no epithelial defect or melt in the tissue surrounding the KPro (Figure 2). However, there was a conjunctival epithelial erosion with exposure of the glaucoma tube superotemporally (Figure 3(a)) and inferonasally (Figure 3(b)). His anterior chamber was quiet and he was aphakic.

(a) Slit lamp photograph of the tinted Kontur lens (temporal view), (b) slit lamp photograph of the tinted Kontur lens (nasal view).
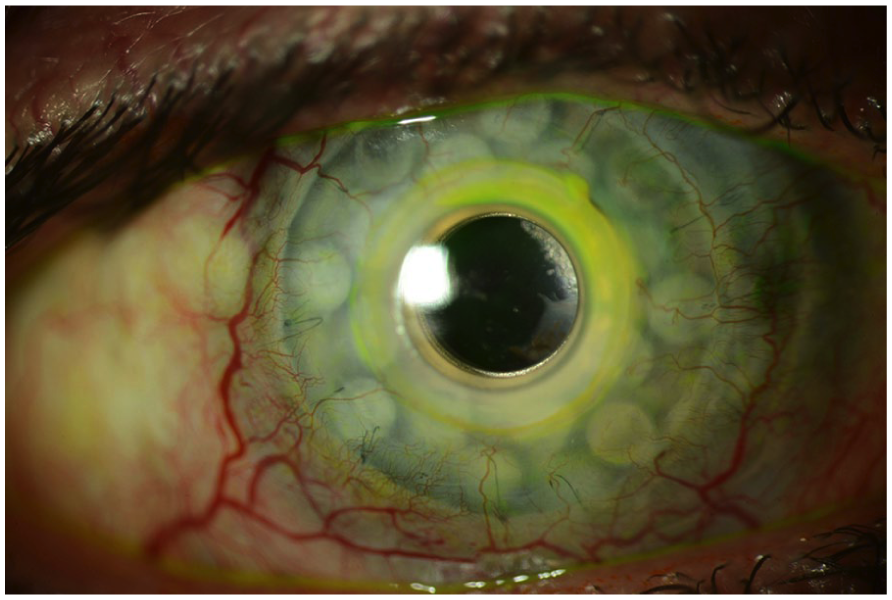
External photograph of the Boston keratoprosthesis type 1.

(a) Slit lamp photograph of the pars plana Baerveldt tube with overlying conjunctival erosion (superotemporal view), (b) slit lamp photograph of the pars plana Baerveldt tube with overlying conjunctival erosion (inferonasal view).
The patient was started on prophylactic vancomycin drops and switched to moxifloxacin drops and erythromycin ointment. The therapeutic contact lens was discarded exchanged for a smaller diameter therapeutic soft lens (Air Optix Night and Day, Alcon, Fort Worth, Texas, USA) with +0.50 prescription, 8.4 mm base curve and 13.8 mm to diameter to avoid mechanical interaction with the conjunctival defects. Two days later, the conjunctival epithelial defects were improved both superotemporally (Figure 4(a)) and inferotemporally (Figure 4(b)). The patient remained stable without complications or further surgical intervention.

(a) Improvement in the conjunctival erosion with a smaller Air Optix contact lens (superotemporal view), (b) improvement in the conjunctival erosion with a smaller Air Optix contact lens (inferonasal view).
Discussion
The Boston KPro type 1 includes both a front plate made of polymethylene methacrylate (PMMA) and a larger back plate made of PMMA or titanium. These devices can exacerbate preexisting glaucoma or lead to the development secondary glaucoma.4,7 In preoperatively non-glaucomatous patients, while only up to 30% of recipients of penetrating keratoplasty had elevated postoperative IOPs, 75% of recipients of KPro devices developed elevated IOP after their implantation. 7 Patients with KPro devices and comorbid glaucoma requiring a GDD are notoriously difficult to manage. In general, the literature advocates early glaucoma surgical intervention in patients with KPro devices, as it has been associated with enhanced vision retention when compared to later intervention.4,8 As in our patient, a pars plana approach is preferred in patients with a KPro, contact lens (CL), or both in order to decrease potential complications.4,9 Therapeutic soft CLs protect against evaporation, epithelial defects, stromal thinning and melt, and dellen formation while enhancing comfort in patients with implanted KPro devices and GDDs.5,10 The initial lens selection is often a Kontur lens, with plano prescription, 9.8 mm base curve and 16.0 mm diameter. The Kontur lens is made of 55% methafilcon A hydrogel. It can be refitted if needed as the base curve and diameter can range from 6.8 to 9.8 mm and 12.0 to 24.0 mm, respectively. Given that the donor cornea is a carrier, transparency and oxygen transmission are not prioritized. As such, there is relatively low oxygen permeability (dK). Tinting of the CL may reduce glare from a hazy corneal graft and can improve cosmesis, as was in this case. However, tube erosion is associated with these lenses,10–13 and patients may need to be refit with a smaller diameter, including CLs smaller 14.0 mm. Tube erosion can also be minimized with placement in the vitreous cavity versus the anterior chamber, and the tube can be reinforced with a patch graft made of fascia lata, pericardium, sclera, cornea, or dura mater.4,14,15
The removal and replacement of the contact lens will vary. In general, the contact lens should be worn continuously and removed for cleaning and disinfection in office at each follow-up visit. Follow-up should occur every 1–3 months, and more frequently if there is frequent lens loss or heavy deposits. Of note, this patient was unique in that he removed and disinfected his lens in a hydrogen peroxide system weekly. Soft contact lenses are often comprised of hydrogels, which are susceptible to cross-linking. Hydrogen peroxide is a known cross-linking agent. As such, it is plausible to believe that hydrogen peroxide–based contact lens cleaning solution may cross-link, and thus harden, contact lenses stored in solution for long periods of time, thus potentially giving rise to conjunctival erosions. Notably, surveillance cultures of the contact lens were negative. One study reported 57% of KPro eyes with chemical burns experienced lens loss. 5 Daily disposable lenses, when compared to extended wear lenses, reduce the risk of development of lens lipid deposits, contact lens–induced papillary conjunctivitis, and infectious keratitis. 5
CL evaluation especially in the setting of existing KPro device is critical. The evaluation should involve inspection of the lens fitting, including the contact lens (centration, edge fluting, bubbles, buckling, deposits), corneal epithelial defects and stromal melt, conjunctival erosions over tube shunts, conjunctival and scleral injection, anterior chamber reaction. Regardless of the choice of CL, lens centration and complete corneal coverage are critical. If the lens is too flat, there can be fluting of the edges. Conversely, if the contact lens is too tight, there can be presence of air bubbles and vascular compression. Fortunately, our patient who already had multiple ocular comorbidities and a complex ocular history improved with prompt CL refitting without development of endophthalmitis or need for additional surgical intervention. However, a conservative approach may increase the likelihood of endophthalmitis, and thus precautions must be taken to ensure clean devices and patient understanding and cooperation.
Footnotes
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This case report adhered to the ethical principles outlined in the Declaration of Helsinki. Collection and evaluation of the protected patient health information was Health Insurance Portability and Accountability Act compliant.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an unrestricted grant from Research to Prevent Blindness and an EY01792 from the National Eye Institute. The funders had no role in the study design, collection, analysis, and interpretation of data; writing of the article; nor decision to submit the article for publication.
Informed consent
Written consent was obtained from the patient for publication of this report and all relevant images and figures.
Study Approval
This is a single-case study and as such is exempt from necessary approval from the University of Illinois at Chicago Institutional Review Board.
