Abstract
Macular edema is a condition of retinal tissue treated with anti-inflammatory agents including placement of an intravitreal sustained-release dexamethasone device, designed to deliver a controlled amount of the medication for a prolonged time, representing an excellent therapy. Nonetheless, the implantation cannot be carried out without an anatomical barrier, such as the presence of posterior capsular support, lens, or intraocular lens. The absence of these barriers could lead to several complications, due to migration of the device from the vitreous cavity to the anterior chamber, causing corneal endothelial damage, corneal edema, glaucoma, and uveitis, among others. In consequence, a large number of patients cannot be treated with this useful surgical tool, resulting in chronicity of macular edema and severe visual acuity impairment. Therefore, we modified the conventional technique, through scleral fixation of the device providing a continuous delivering of dexamethasone, avoiding its migration to the anterior chamber in a patient without capsular support.
Introduction
Macular edema is the leading cause of poor visual acuity in many retinal disorders, including diabetic retinopathy, retinal vein occlusion, uveitis, and Irvine–Gass syndrome. 1 Macular edema can be treated with placement of a dexamethasone implant (Ozurdex; Allergan Inc., Irvine, CA, USA) into the vitreous cavity. This is a biodegradable implant containing 700 mcg of dexamethasone and measuring 6 mm in length and 0.46 mm in diameter.1–4 However, retina specialists have started expanding its use to other indications and in vitrectomized eyes with good results.5–7
A potential complication of the Ozurdex® implant is its migration to the anterior chamber in pseudophakic, aphakic, and vitrectomized eyes. This complication could cause permanent corneal decompensation 8 and, therefore, requires the immediate reposition in the vitreous cavity or complete removal of the implant. The case presented here involved a pseudophakic patient with subluxated bag–intraocular lens complex, previous posterior vitrectomy, and postoperative macular edema who was managed with an Ozurdex implant that was fixed to the sclera to prevent potential migration into the anterior chamber while achieving effective delivery of the drug and the desired pharmacologic effect. We did not consider the removal of this subluxated complex with the purpose of avoiding any kind of additional inflammatory stimulus.
Case report
The patient was a 75-year-old male with a history of left eye cataract surgery by phacoemulsification and toric intraocular lens (IOL) implantation that was complicated by zonular dialysis and vitreous prolapse requiring anterior vitrectomy. At 1 month after the surgery, the patient reported poor visual acuity, with a best-corrected visual acuity (BCVA) of 20/100 (Snellen equivalent). Slit-lamp examination showed a subluxated capsular bag–IOL complex, normal intraocular pressure (IOP), and macular edema. Macular optical coherence tomography (OCT) showed a central macular thickness of 890 m, cystoid macular edema, and subretinal liquid (Figure 1).
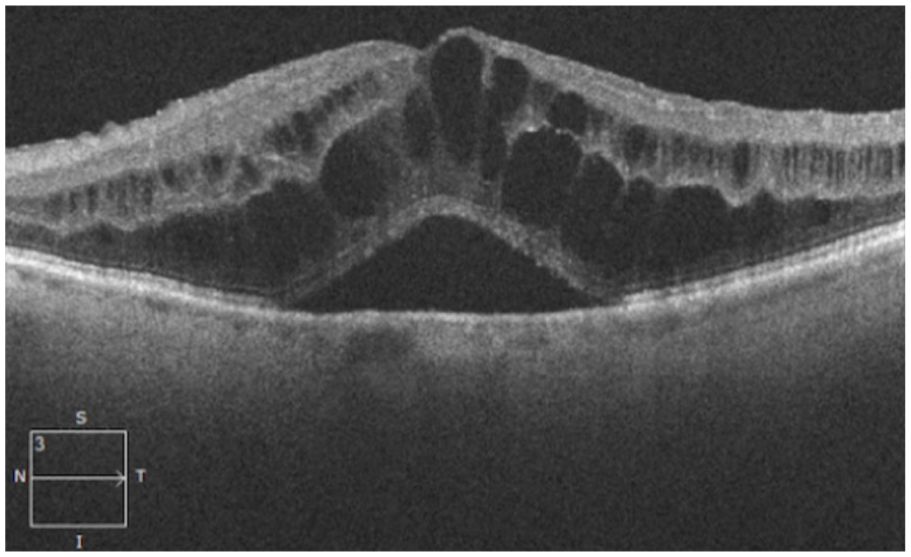
Preoperative macular edema.
To treat the macular edema, it was decided to proceed with placement of a dexamethasone intravitreal implant (Ozurdex). Due to the subluxated capsular bag–IOL complex, previous posterior vitrectomy, and the higher risk of migration to the anterior chamber with potential corneal decompensation, we decided to proceed by suturing the implant to the sclera to anchor it. To do so and prior to the procedure, we placed the implant into the sterile container to prevent its loss or damage. Then, one of the ends of the implant was sutured with 10-0 polypropylene (Prolene; Ethicon, UK) (Figure 2(a)). Under regional anesthesia, a 23-G sclerotomy was performed at a 3.5 mm distance from the limbus. The implant was then introduced through the sclerotomy into the vitreous cavity using forceps (Figure 2(b)). The sclerotomy was closed using the same polypropylene suture fixed to the implant. The conjunctiva was closed in standard fashion with 9-0 nylon suture.

Surgical technique: (a) we sutured one of the ends of the implant with nonabsorbable polypropylene (Prolene; Ethicon, UK) 10-0; (b) after performing a 23-G sclerotomy at the 6 o’clock position (3.5 mm from the corneal limbus), using forceps we introduced the implant through the sclerotomy into the vitreous cavity.
Complete ophthalmologic examination 1 and 7 days after the surgery did not show complications. Ozurdex implant was properly placed and fixed behind the iris at the 6 o’clock position (Figure 3(a)). At 1 month after the surgery, the BCVA improved to 20/60 (Snellen equivalent), while IOP remained at 14 mmHg. The cornea was clear and the measured central macular thickness decreased from 890 to 252 m, without cystoid macular edema or subretinal fluid (Figure 3(b)). There was no displacement with eye movements or changes in the patient’s position. In addition, the patient did not find the implant bothering. At 2 months after the placement of Ozurdex implant with scleral fixation, the BCVA remained 20/60, IOP was 13 mmHg, and the cornea was still clear. The implant was no longer detectable 4 months after the surgery. At 6 months after the surgery, visual acuity remained stable and the cornea maintained its clarity. We did not observe macular edema on the OCT scans and there were neither anterior nor posterior complications during the follow-up.

Postoperative period: (a) at 1 week after OZURDEX scleral fixation, the implant could be easily observed behind the iris at the 6 o’clock position on slit-lamp examination. (b) Swept source OCT shows no cystoid macular edema or subretinal fluid.
Discussion
The Ozurdex implant is an effective treatment for retinal pathologies complicated by macular edema.2–6 Nonetheless, migration is a described complication, especially in cases with aphakia, anterior chamber IOL, scleral-fixated posterior chamber intraocular lens (PCIOL), iris-fixated PCIOL, pars plana vitrectomy, and absence of lens capsule.8,9 Among those eyes undergoing surgical removal of the implant, earlier intervention reduced the likelihood of permanent corneal edema.
Therefore, the absence of the lens capsule and prior vitrectomy are common high-risk factors for migration of the implant into the anterior chamber and early removal of the implant is necessary to reduce the risk of corneal damage and potential need for corneal transplantation.
In view of the serious effects of the migration of the implant in the mentioned cases, it has been chosen not to use it in them, depriving those patients of an excellent therapeutic option for macular edema. Unfortunately, most of the cases with macular edema have the absence of lens capsular support, evidently placing the ophthalmologist in a difficult situation about the management.
Given the above, there is a need to design new surgical techniques that allow the implantation of the dexamethasone release device without the risk of migration. Recently, an article was published by Mateo and colleagues 10 reporting a case of an 80-year-old male who underwent intravitreal scleral fixation of Ozurdex to treat postoperative macular edema secondary to a complicated cataract surgery. The patient had a previous angle-supported lens implantation. The implant was introduced into the vitreous cavity and sutured to the sclera using a 10-0 polypropylene suture 4 mm away from the limbus. The implant exhibited a stable position during the follow-up, until it was completely reabsorbed with improved macular thickness and visual acuity. In our patient, we sutured the implant because he was a vitrectomized patient with subluxated bag–IOL complex, with a high risk of implant migration into the anterior chamber. We sutured the Ozurdex implant to the sclera using a nonabsorbable suture as well.
The scleral fixation of the Ozurdex implant is useful in cases of aphakia, pseudoaphakia without posterior capsular integrity, and previous posterior vitrectomy. We recommend this procedure in order to prevent the anterior migration of the implant while achieving its pharmacologic effect. The previous contraindication of the Ozurdex implant in these cases can be a relative one instead of an absolute one, with every case being considered individually for the candidacy and feasibility of the application of the technique proposed here.
Footnotes
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our study did not require an ethical board approval because it did not contain human trial. This is because our study was just observational and retrospective, describing the results of the previously performed procedure.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
We obtained a written informed consent signed by the patient for publishing the information and images. The written consent is held by the authors in a patient’s hospital record.
