Abstract
Background:
In this paper, our aim was to describe an imaging-based empirical approach for categorizing and initiating treatment of immunocompetent patients with posterior focal retinitis, prior to the availability of results of expensive laboratory investigations.
Materials and methods:
The hospital records of 13 patients with posterior focal retinitis were reviewed.
Results:
Of the 13 patients, 9 were women and 4 were men. The mean age was 24 ± 8 years. Based on similarities in clinical presentation and imaging, we categorized our cases into three groups with different first-line therapeutic strategies. In the first group, patients had presumed toxoplasmosis (treated with oral cotrimoxazole); in the second group, patients had presumed viral (herpetic) etiology (treated with oral valacyclovir); and in the third group, patients had presumed nonherpetic, nontoxoplasma retinitis (treated with oral doxycycline). Positive serology results included Rickettsia (two patients), Borrelia (one patient), Toxoplasma (two patients), and herpes simplex virus (one patient).
Conclusion:
An empirical approach for early initiation of therapy in retinitis cases based on imaging features is described.
Keywords
Introduction
Focal retinitis at the posterior pole is a vision-threatening condition and early initiation of appropriate therapy is important. Focal retinitis can be autoimmune in nature 1 or of infective origin. 2 Organisms implicated include Toxoplasma gondii, Rickettsia (Indian tick typhus- Rickettsia conorii; epidemic typhus- Rickettsia prowazekii), 3 Orientia tsutsugamushi (scrub typhus), 4 Borrelia burgdorferi (Lyme disease), Bartonella henselae, West Nile virus, dengue virus, chikungunya virus, Rift valley fever virus and herpetic viruses. Retinitis patches at the posterior pole may also be noted in measles virus (subacute sclerosing panencephalitis (SSPE)), 5 presumed mumps (cerebriform retinitis), 6 and metastatic endophthalmitis.
Possible strategies to confirm the infective agent include serological tests and molecular assays of the vitreous or aqueous humor.7–9 However, these techniques have certain limitations. Serological tests are costly, 10 may not be available in all countries, it may take few days to get the final results, and their interpretation varies with local endemicity of the disease. Molecular assays are invasive and require a vitreous/aqueous tap to obtain an adequate sample. These tests also have false positives and negatives and the results vary with quality and standardization protocols followed by the laboratory. 11
It is important for the clinician to start appropriate therapy before the results of such tests are available, to preserve the visual acuity and prevent progression of the disease. Clinical judgment, based on fundus findings and information obtained from investigations such as optical coherence tomography (OCT), may play a crucial role in the management of such patients. We present an empirical approach for the initiation of management of posterior focal retinitis in immunocompetent patients with a presumed infective etiology.
Materials and methods
We carried out a retrospective case review of 13 cases diagnosed as posterior focal retinitis at the uveitis services of Dr Rajendra Prasad Centre for Ophthalmic Sciences, New Delhi, India. Posterior focal retinitis was defined as solitary or multiple (but localized) patches of retinitis confined predominantly to the posterior pole. All patients were asked about a history of any prior febrile illness with or without rash. Best-corrected visual acuity (BCVA), detailed slit lamp and fundus examination findings were noted. Fundus imaging and swept-source OCT (SS-OCT) were performed using Optos (Optos, Inc., Marlborough, MA, USA) and Triton DRI OCT (Topcon Corporation, Tokyo, Japan). Serological testing was performed for patients who could afford it. Diabetes and hypertension were excluded in all patients. HIV status (history and serology) was also assessed in all patients to rule out the possibility of opportunistic infections in the setting of immunosuppression. All patients were negative for HIV and none had abscess or localized infection in any site of the body.
Serological tests were performed to confirm the etiology based on clinical suspicion and included serum immunoglobulin G (IgG) and IgM levels for Toxoplasma, IgM and IgG for herpetic viruses (herpes simplex, Varicella zoster and cytomegalovirus), IgG and IgM levels for Borrelia, Weil–Felix test (WFT) (OX 2, OX K, and OX 19) for Rickettsia, IgM and IgG for chikungunya and IgM and IgG for dengue. Bartonella serology is not currently commercially available in India and hence could not be performed. Patients with suspected toxoplasmosis were started on oral trimethoprim–sulfamethoxazole (160 mg + 800 mg) twice a day; patients with suspected viral retinitis were initiated with oral valacyclovir 1 g thrice daily; and other patients were started empirically on oral doxycycline 100 mg twice daily. Steroids were curtailed in patients who were already on exclusive steroid therapy prior to presenting to us. Repeat fundus imaging and OCT were performed at follow-up visits and change in the visual acuity was noted. Low-dose prednisolone was given in few cases after a clinical response on antibiotics was documented to aid the early resolution of the disc and macular edema.
BCVA was converted to logarithm of minimum angle of resolution (LogMAR) for statistical analysis. 12 The eyes (two) with perception of light (PL) or no PL were excluded from the analysis.12,13 A LogMAR value of 2 was allotted for finger counting close to face (FCCF) and hand movement close to face (HMCF) received a value of 3. 12
Written informed consent was obtained from all patients. An ethical approval was not sought, as this study is a retrospective, noninterventional analysis of imaging findings with the final likely diagnosis. As this study correlated imaging characteristics in patients of focal retinitis with their final clinical diagnosis, the findings of the study did not change the management protocol of the patients. No intervention was performed on the patients, and their treatment was not modified on the basis of the findings described in this article. Rather, the insights obtained from this paper are an attempt to help clinicians in establishing an empirical diagnosis from the imaging findings themselves.
Results
Demographic profile
The presentation was unilateral in 8 patients and bilateral in 5 cases (18 eyes of 13 patients). Of the 13 patients in our series, 9 were women and 4 were men. None of the women was pregnant or lactating. Mean age was 24 ± 8 years (range: 13–36 years). History of febrile illness (2–3 weeks prior to presentation) was present in six patients. Four patients also gave a history of rash associated with the fever. In one patient (case 12), the rash could be documented on the legs at the time of presentation. A physician review of the cases with fever and rashes was inconclusive of any specific disease. Five patients had a prior history of oral steroid use (cases 6, 9, 10, and 13). Presenting BCVA was 1.43 ± 0.66 LogMAR (HMCF to 6/24). All affected eyes had mild nongranulomatous anterior chamber reaction (0.5 to 1+ cells). Fundus examination revealed focal patches of retinitis, which were solitary in 6 eyes and multiple (but confined predominantly to the posterior pole) in the remaining 12 eyes. Table 1 summarizes demographic data, BCVA and investigation profile of patients with posterior focal retinitis.
Clinical details of the patients.
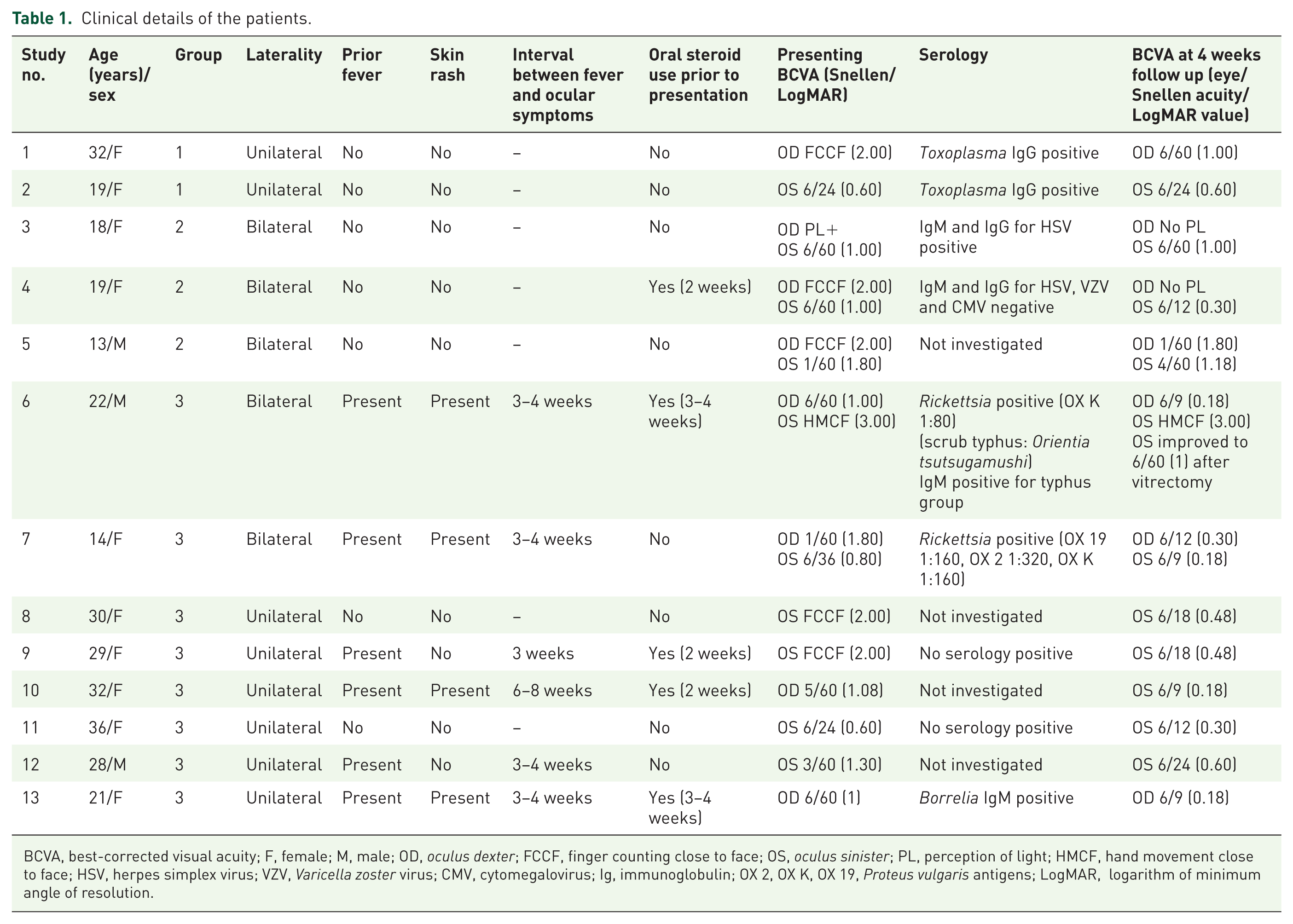
BCVA, best-corrected visual acuity; F, female; M, male; OD, oculus dexter; FCCF, finger counting close to face; OS, oculus sinister; PL, perception of light; HMCF, hand movement close to face; HSV, herpes simplex virus; VZV, Varicella zoster virus; CMV, cytomegalovirus; Ig, immunoglobulin; OX 2, OX K, OX 19, Proteus vulgaris antigens; LogMAR, logarithm of minimum angle of resolution.
Based on ocular examination, fundus findings and OCT features, these patients were divided into three groups.
Group 1 (two eyes of two patients; cases 1 and 2): cases of solitary focal retinitis with vitritis (presumed toxoplasmosis)
The retinitis patch in these cases was whitish and fluffy in appearance, with minimal or absent hemorrhage. There was a presence of overlying vitritis and an adjacent small whitish retinitis patch (case 2). These cases lacked an adjacent scar, which is typically considered a hallmark of Toxoplasma retinitis. Serous detachment, involving inferior retina was noted in one eye (case 1). There was a lack of disc edema or macular star in both of these cases. Vascular cuffing involving both arteries and veins was seen in one case (case 1, Figure 1).

Images of Case 1 of Group 1 (presumed toxoplasmosis).
OCT of these cases revealed increased hyperreflectivity of inner retinal layers restricted from the inner surface of retina to the outer plexiform layer in most sections. In some sections, it extended more posteriorly up to the retinal pigment epithelium with localized disruption of the external limiting membrane and ellipsoid zone (inner segment–outer segment junction; IS–OS junction). Hyper-reflective dots in the retinal layers were not much appreciable (Figure 1). Both serum IgM and IgG for Toxoplasma were positive. Serum IgG was strongly positive in both of these cases. Oral cotrimoxazole (trimethoprim–sulfamethoxazole; 160 mg + 800 mg) twice a day was given and oral steroids (prednisolone 1 mg/kg) were added after 3–5 days of cotrimoxazole therapy and slowly tapered over 3–4 weeks. On resolution, OCT showed a reduction in the vitreous cells with a marked decrease in retinal hyper-reflectivity (Figure 1). The overall retinal thickness also reduced with some scarring. The overlying internal limiting membrane (ILM) remained intact.
Group 2 (six eyes of three patients; cases 3, 4, and 5): cases of focal retinitis with placoid lesion (presumed viral/herpetic retinitis)
The appearance of retinitis lesions mimicked cases of cilioretinal arterial occlusion. The lesions appeared like a dry plaque with evidence of deep retinal necrosis. Associated intralesional hemorrhages were observed. There was no overlying vitritis in these cases [Figure 2(a)]. Description of two of these cases (cases 3 and 4) has been published earlier. 14 Two of these patients were already on steroids prior to presenting to us. The lesions in these eyes were seen to rapidly progress centrifugally, probably as a result of steroid therapy.

The right eye of case 3 (Group 2- presumed viral/herpetic retinitis).
OCT showed significant thickening of the inner retinal layers with underlying neurosensory detachment, with full-thickness involvement noted in some of the scans (Figures 2 and 3). There was a partial ILM separation 15 at the fovea with hyporeflective spaces seen (necrosis with tissue loss) in the retinal layers [Figures 2(a), 2(b), and 3(a)]. Vitreous cells were also seen (Figures 2 and 3). Retina showed a few hyporeflective spaces on OCT at resolution [Figures 2(d) and 3(b)]. Of the three cases of suspected herpetic viral etiology, two patients could afford serological testing. Of these, IgM and IgG for herpes simplex was positive in one patient (case 3) and the rest of the serological tests were negative.

The left eye of case 5 (group 2- presumed viral/herpetic retinitis).
In two cases (cases 3 and 4), steroids were stopped, and they were started on oral valacyclovir. In both the patients, retinal detachment was noted in the eye with worse retinitis with complete loss of PL. 14 However, the fellow eye of these patients had visual improvement compared with at baseline. The third of these patients (case 5, Figure 2) directly presented to us. He had not received oral steroid. He was started on oral valacyclovir. On this therapy, there was complete resolution of lesions in 3 weeks (Figure 2). OCT during resolution showed marked thinning of the retina at the fovea with the loss of distinction of individual retinal layers [Figures 2(d) and 3(b)]. Vitreous cells also reduced.
Group 3 (10 eyes of 8 patients; cases 6–13): cases of focal retinitis with macular star (presumed nonherpetic, non-Toxoplasma retinitis)
The retinitis lesions were solitary in two eyes and multiple but confined predominantly to the posterior pole in eight eyes. Superficial retinal hemorrhages on or adjacent to the lesions were seen in all eyes. Nine eyes had some amount of hard exudates deposited in a radial orientation at the fovea (macular star) 16 at presentation [Figures 4(a) and 5(a)]. No patient gave a history of insect bite or exposure to a cat. One patient who was on oral steroids and presented late had vitreous hemorrhage in one eye and multifocal retinitis with hemorrhages and macular star in the other eye (case 6). 4 Some amount of disc edema was seen in all patients (Figures 4 and 5). Clinically, there was no vitritis in these patients. One patient had a visible focal vitreous adhesion over the patch of retinitis (case 10). Small segments of Kyrieleis’ arteriolitis 17 or long segments of retinal arterial wall whitening were seen adjacent to the patches of resolving retinitis in 6 of these 10 eyes (Figure 6).
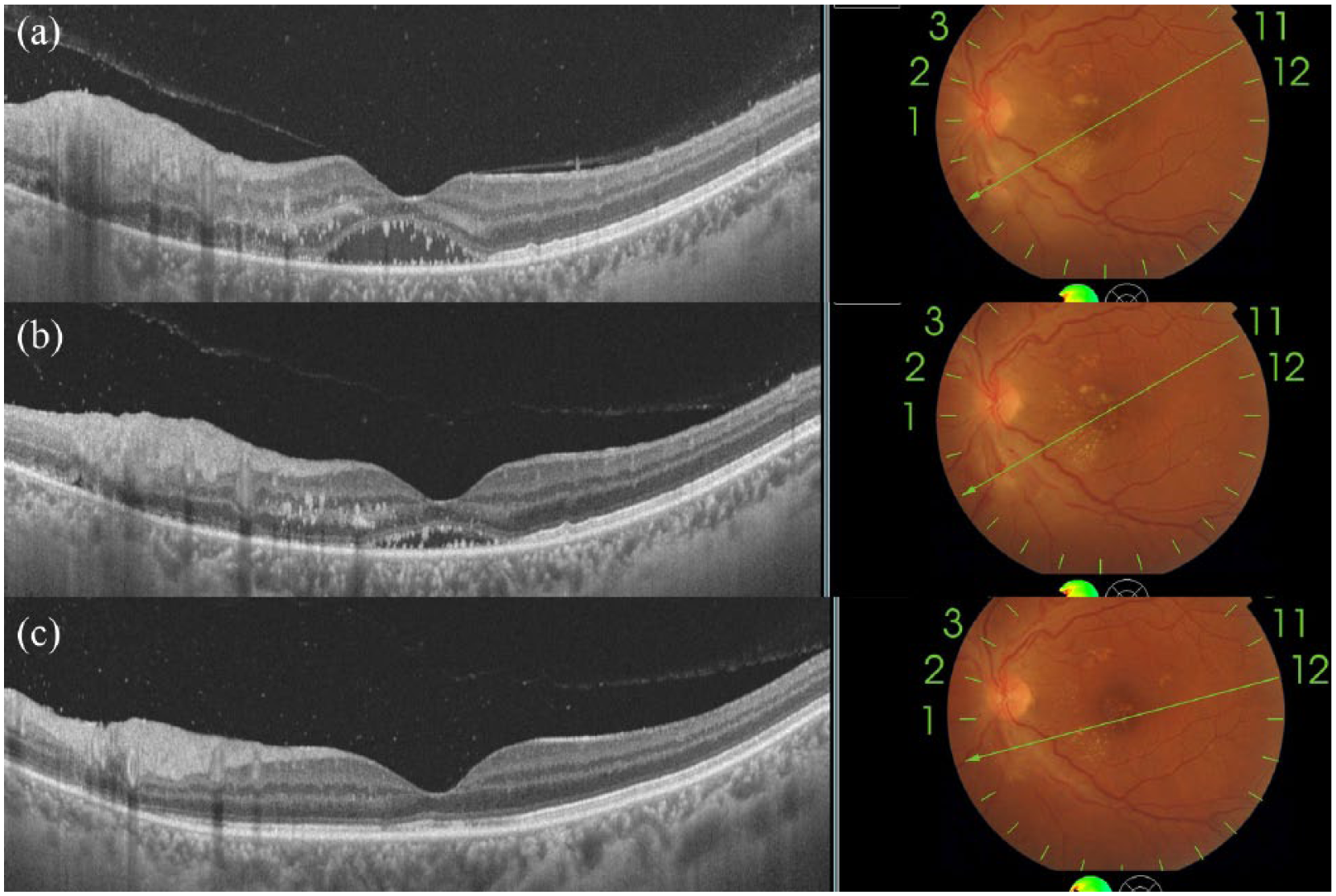
Images of case 11 (group 3- presumed nonherpetic, non-Toxoplasma retinitis).

Images of case 12 (group 3- presumed nonherpetic, non-Toxoplasma retinitis).

Periarteriolar Kyrieleis’ plaques (*) in case 6 (a), case 9 (b), case 11 (c), and case 12 (d).
On OCT, there was increased reflectivity of the inner retinal layers up to the outer plexiform layer (Figures 4 and 5). There was associated neurosensory retinal detachment (nine eyes, absent in case 10) and cystoid intraretinal changes (eight eyes, absent in cases 11 and 12). Vitreous cells were noted in all cases with partial hyaloid separation [Figured 4(c) and 5(c)]. On the resolution of retinitis, the focal vitreous adhesion and traction over the patch of retinitis got released in case 10. Additional hyper-reflective dots (other than those due to hard exudates) were seen in the retinal layers. Of these eight cases of suspected nonherpetic, non-Toxoplasma retinitis, serological tests could be obtained in five patients, of which two patients had positive serology for Rickettsia (cases 6 and 7), one had positive serology for Borrelia (case 13) and two did not have any positive serological tests (cases 9 and 11). Bartonella serology could not be performed, as it is not, at present, commercially available in India.
These patients were treated with oral doxycycline 100 mg twice a day. Four of these patients were already on steroids and the dose of their steroids was tapered. The remaining six patients were given doxycycline alone initially and after a week, when the clinical response was evident, 0.5 mg/kg body weight of prednisolone was added to aid the early resolution of disc and macular edema. In case 6, where a vitreous hemorrhage had developed in the left eye, vitrectomy was performed and the BCVA improved to 6/60. 4 Pigmentary changes were evident at the posterior pole of this eye, suggestive of resolved retinitis.
Improvement in visual acuity (p = 0.001) was noted in patients of all three groups compared with baseline (1.43 ± 0.66 LogMAR versus 0.73 ± 0.75 LogMAR).
Discussion
It is often difficult to determine the correct line of initial therapy in cases of focal posterior retinitis. Differential diagnosis of retinitis at the posterior pole includes cotton-wool spots. An appearance of multiple whitish patches around the optic disc or posterior pole may also be noted in hypertensive retinopathy, lupus retinopathy, Purtscher retinopathy, and HIV retinopathy. All our cases were nondiabetic, nonhypertensive and negative for serology of HIV. There was no clinical evidence of systemic lupus erythematosus in any of our patients. To diagnose the cause of focal posterior retinitis, various serological and molecular assays are required. These are expensive and time consuming. However, empirical therapy needs to be started in the clinic early so that the precious time is not lost, as delay in initiation of therapy may cause irreversible damage to the retina. From a retrospective analysis of the clinical features, imaging characteristics, response to therapy, and serological results of 13 cases, we categorized the patients into three broad groups with a different first line of therapy (Table 1). This empirical therapy can be modified later based on the results of serological assays or response to therapy.
Group 1 patients in our study were cases of suspected toxoplasmosis. The cases included by us did not have typical features of toxoplasmosis such as active retinitis adjacent to a previous scar and the presence of very severe vitritis.18,19 Lack of an adjacent scar or extensive vitritis leads to a diagnostic dilemma in these cases. Common clinical features found in our series in this group are whitish and fluffy patches of retinitis, lack of significant hemorrhages in the patch of retinitis, early arteriolar involvement, the absence of disc edema and macular hard exudates. IgG for toxoplasma was strongly positive in both the patients. However, it is known that the overall seroprevalence rate for toxoplasmosis may be as high as 45% in India. 20 The clinical response to cotrimoxazole may indicate toward toxoplasmosis as the possible origin, though the possibility of spontaneously resolving retinitis cannot be excluded in these cases. OCT in these cases showed the involvement of the entire thickness of the retina in at least a few sections. On the resolution of retinitis, OCT revealed a thinner retina.
Group 2 patients were characterized clinically by plaque-like focal retinal whitening simulating some cases of cilioretinal arterial occlusion. 21 The lesions progressed to develop intralesional hemorrhages and started spreading in a centrifugal manner. OCT characteristically showed significant retinal thinning and necrosis. Overlying ILM still remained intact in some of these cases.
This characteristic of the ILM acting as a potential barrier to spread of retinal infection into the vitreous has been previously described by the authors. 15 The necrotizing and rapidly progressing characteristic of the retinitis in such cases may denote an origin from the herpetic viruses.22,23 In addition, one patient had serum IgM and IgG positivity for HSV. SSPE 5 may be another differential diagnosis for such cases. However, none of the cases showed any behavioral changes or seizures up to the last follow up.
Group 3 cases had single or multiple patches of retinitis predominantly confined to the posterior pole. They were additionally associated with retinal hemorrhages, disc edema and macular hard exudates forming a partial or complete macular star. Some of these cases were diagnosed as neuroretinitis and started on steroid therapy alone by ophthalmologists who saw them prior to presentation to us. However, these cases did not improve on steroids alone. All of them showed a good response to oral doxycycline. The bacteria causing such a retinitis (Rickettsia, Borrelia, and Bartonella) have a similar spectrum of antibiotic sensitivity (e.g. to doxycycline).24–26 Two of our patients (cases 6 and 7), had positive WFT results (titers of 1:80 and above). The WFT often lacks specificity for distinguishing between various subtypes of Rickettsia. As per the local guidelines, a probable rickettsial case has been defined as ‘…a suspected clinical case showing titres of 1:80 or above in OX2, OX19 and OXK antigens by Weil–Felix test and an optical density (OD) > 0.5 for IgM by enzyme-linked immunosorbent assay is considered positive for members of typhus and spotted fever groups of Rickettsiae’. 27 However, compared to the gold standard immunofluorescent antiblody test, the WFT has low sensitivity and specificity. Also, the WFT may be positive even in healthy individuals and in those with nonrickettsial fever. 10 IgM against Borrelia was positive in one patient (case 13)
A limitation of this study is that we could not evaluate for cat-scratch disease as the serological test for Bartonella is commercially not available in India. Early institution of doxycycline ensured good visual outcome in our cases indicating a probable infective etiology. Even cases worsening on empirical steroid therapy started elsewhere improved on antibiotic therapy and tapering of the high dose of steroids. Empirical initiation of oral steroid in a similar case of retinitis resulted in worsening. 28 This patient responded well once the oral steroid was stopped and oral doxycycline started. 28 Though we consider most of our cases as presumed bacterial (related to Rickettsia, Lyme disease or Bartonella), for the purpose of categorization, we have also included presumed dengue, chikungunya and West Nile fever retinitis in this group as a similar presentation may be seen following infection by these viruses. 29
Infections related to these viruses presently do not have any specific treatment and may resolve spontaneously. 29 Clinically, it may be difficult to differentiate retinitis due to such virus from presumed bacterial retinitis. Considering the safety of short-term oral doxycycline and the diagnostic difficulty, it may be prudent and not harmful to start oral doxycycline in such cases initially. Six cases in this group had a history of fever within 3–4 weeks of the diagnosis of retinitis. Serological positivity was evident in three cases and all six patients received systemic doxycycline for 3–4 weeks.
There are some reports of spontaneous resolution or resolution only on steroids of cases of focal retinitis, thus presuming the manifestations to be an immune response. 3 However, in most reported case series with similar clinical features, systemic steroid was used only under the cover of an antibiotic. In the series of rickettsial retinitis studied by Kawali and colleagues, 3 ‘only three patients were treated with “steroid-only” regimen for retinitis but they received antibiotics during their fever.’ Kahloun and colleagues 30 reported 14 patients with rickettsial retinitis, all of whom were treated successfully with a 2-week course of oral doxycycline (200 mg/day), including a patient (patient number 10) who showed initial spontaneous improvement while the serological tests were pending (Table 1 30 ). Four cases in this series also received oral prednisolone (1 mg/kg) which was tapered gradually. Khairallah and colleagues reported ‘white retinal lesions’ in 30% eyes of 30 patients with serologically proven rickettsial infection. 31 Six of the 13 eyes in a recently presented series of 10 patients with cat-scratch disease 32 showed retinal infiltrates, all of whom received systemic antibiotic with or without systemic steroid. Though periocular and systemic steroids have been used successfully for posterior segment involvements associated with dengue fever, chikungunya fever, and Rift valley fever, many of such lesions resolve spontaneously with time.33–35 Thus, there is an unmet need for large randomized control trials evaluating the role of systemic steroid and systemic antiviral in such cases.
All our patients responded promptly to oral antibiotics and antivirals, including those worsening on steroids alone. Thus, we presume that there is often an active infective component in this entity and it is not purely an immunological phenomenon. 4 Systemic or periocular steroid is a known cause for activation of ocular infections 36 including retinitis 29 and endophthalmitis. Based on our experience, we suggest that steroids should be reserved as a second line of management in focal retinitis for cases in which inflammation does not respond to antibiotics/antivirals or where features are suggestive of an autoimmune etiology. They can also be added to aid in early resolution of disc and macular edema after an initial response to antibiotics/antivirals is noted. However, an important limitation of our study is the small number of cases.
Posterior focal retinitis is often due to an infective etiology and appropriate anti-infective agents need to be initiated to prevent visual loss. Categorizing these patients into groups based on clinical examination and OCT findings as described helped to initiate early empirical therapy, enabling resolution of retinitis lesions and attainment of good visual outcomes. Steroids in these cases should never be used as first-line treatment as this can lead to worsening of the disease with progression of retinitis.
