Abstract
Background
Menstrual-related migraine (MRM) is a subtype of migraine that significantly impacts women. Despite its high prevalence, MRM remains under-researched compared to other migraine types. This study investigates the phenotypic characteristics and treatment outcomes of MRM compared to non-menstrual migraine (non-MRM).
Methods
This cross-sectional study included women aged 18 years or older with a diagnosis of migraine and an established menstruation pattern. Informed consent was properly obtained from all participants from the Stanford Headache Clinic and surrounding community between 2014 and 2024 prior to participation in the study. Data were collected from self-reported surveys, including demographic information, headache characteristics, physical and mental health status, and treatment outcomes from the Stanford Headache Clinic and surrounding community. Participants were then categorized into MRM and non-MRM groups. An exploratory analysis was performed to investigate phenotypic features, treatment outcomes, and brain volume through magnetic resonance imaging (MRI) study for each group. Statistical analyses included descriptive statistics, analysis of covariance (ANCOVA), and logistic regression.
Results
A total of 84 subjects were included, with 38 in the MRM group and 46 in the non-MRM group. The mean age of participants was 34 (SD = 9) years. The MRM group (mean age 38 ± 9) showed significantly higher prevalences of recent change in migraine frequency, migraine attacks occurring in the same site, prodromal signs, speech aura, nausea, and vomiting associated with migraine, and a higher prevalence of reported family history of headache as compared with the non-MRM group (mean age 31 ± 10) (p < 0.05). MRM group also showed significantly higher rate of migraine improvement following the use of triptans and the overall acute and preventive medications (p < 0.05). No significant differences were found in brain volumes between the groups (p > 0.05).
Conclusion
The MRM group was associated with significantly more bothersome symptoms associated with migraine, higher family history of headache, and more favorable response to overall acute medication especially triptan as compared with the non-MRM group.
This is a visual representation of the abstract.
Introduction
Migraine is a highly prevalent and disabling neurological condition with various subtypes. Among them, menstrually-related migraine (MRM), which is defined as migraine attacks that occur in a menstruating woman on day 1 ± 2 of menstruation in at least two out of three consecutive menstrual cycles. Attack patterns may be limited exclusively to the menstrual window, which is regarded as pure menstrual migraine (PMM) or associated with menses as well as other times of the month (MRM). 1
According to the literature, approximately 50% of female patients with migraine have a coexisting MRM. 2 While migraine has a similar prevalence of 4% in prepubertal ages among both male and female genders, prevalences after puberty increase to 18% among females compared with 6% among males.3–5 With the high prevalence of hormonally influenced migraine attacks specifically among females, studies have suggested a potential link between female hormonal changes and migraine attacks.1,2
Female sex hormones especially estrogen and progesterone are reported to significantly influence the migraine-related disability levels and attack frequency.6,7 Moreover, studies have shown a significant correlation between estrogen and progesterone levels during menstrual periods, pregnancy, and postmenopausal ages with the frequency and severity of migraine attacks.6–8
The pathophysiology of MRM has also been attributed to the increased sensitivity of pain pathways and enhanced release of CGRP and inflammatory cytokines following rapid estrogen depletion, and lack of nociceptive suppression following progesterone drop.9–11 Moreover, higher CGRP levels have been detected in plasma and tear fluid during the menstruation phase of female migraine patients, indicating a potential link between hormonal levels during menstruation and migraine pain pathways. 12
While studies have reported overall similar phenotypic and imaging features among MRM and non-MRM patients, clinical management and outcome of the two conditions are suggested to be different. Triptans alone or in combination with NSAIDs are commonly used therapeutically to treat MRM and have demonstrated favorable clinical improvement as acute and preventive treatment of MRM. However, uncertainties remain regarding the underlying causes, actual differences that put some people at a higher risk of MRM, and widely practical management of the condition.13–17
Our study aims to explore phenotypic characteristics, brain volumetric differences, and treatment outcome differences among females with MRM and non-MRM to investigate and compare different aspects of the two groups in more detail and increase the understanding regarding the nature of MRM.
Methods
We conducted a cross-sectional detailed phenotypical comparison of clinical features and treatment outcomes among female patients with and without MRM.
The inclusion criteria consisted of female gender, age ≥ 18 years, a stable menstrual pattern, and a diagnosis of migraine at the time of the study. Diagnoses were made based on self-reported surveys and according to the ICHD-3 criteria.
Participants were recruited from the Stanford Headache and Facial Pain Clinic and surrounding community between 2012 and 2024. All participants were consented prior to any study-related procedures. This study is reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies. 18
Data were collected from online self-reported surveys and included demographic information, phenotypic characteristics of migraine, mental health status, and treatment outcomes.
The validated instruments used in the study to assess and compare general mental health included the Patient Health Questionnaire-9 (PHQ-9) to screen for depression, 19 the Generalized Anxiety Disorder-7 (GAD-7) 20 to assess anxiety, and the Pittsburgh Sleep Quality Index (PSQI) 21 to evaluate sleep quality. Additional tests were used to assess pain-coping cognitions and lifestyle behaviors, including the pain catastrophizing scale (PCS) to measure pain catastrophizing, 22 the pain self-efficacy questionnaire (PSEQ), 23 and the regular lifestyle behavior (RLB) assessment which includes regular timing and constant habits in three areas of daily meals, sleep habits, and exercise minutes. 24 The tests were selected in order to assess levels of the most common comorbid psychiatric conditions of migraine including general anxiety disorder, depression, and sleep disorders, as well as pain perception, pain coping ability, and lifestyle behavior to compare across the groups, in order to further explore psychiatric underpinnings of group differences.
Acute migraine medication categories included triptans, ergotamine, non-steroidal anti-inflammatory agents (NSAIDs), and over-the-counter medications, consisting of Acetaminophen, Aspirin, and combination products containing acetaminophen, aspirin, and caffeine. Preventive medication categories included anti-hypertensive agents, anti-depressive agents, neuropathic pain modulators, and botulinum toxin injection.
Treatment outcomes were assessed by asking participants about all available acute and preventive migraine medications. If the subject had used any of these medications, they were asked whether the treatment had improved their migraine through a yes/no question. A positive response was regarded as a favorable outcome. The rate of favorable outcomes for each treatment category was calculated as the percentage of favorable responses within that medication family.
Statistical methods included descriptive statistics, as well as analysis of covariance (ANCOVA) and logistic regression to assess group differences with age as a covariate. Variables with inadequate sample were removed from the study. A p value of less than 0.050 was considered significant. Post-hoc power analysis was performed to confirm sampling adequacy.
Structural MRI data were acquired at Stanford University using previously described protocols 25 using a GE MRI scanner (GE Healthcare, Milwaukee, WI, USA) with an eight-channel phased-array head coil. Structural MRI (sMRI) was performed using high-resolution T1-weighted 3D axial FSPGR IR-prepared sequences with the following parameters: repetition time (TR) = 5.9 ms, echo time (TE) = minimum, flip angle = 15°, voxel size = 0.9 × 0.9 mm, matrix size = 256 × 256, and slice thickness = 1 mm (no gap). For resting-state functional MRI (rsfMRI), T2*-sensitive spiral-pulse sequences were used with parameters: TR = 2000 ms, TE = 30 ms, flip angle = 80°, voxel size = 3.4 × 3.4 mm, slice thickness = 4 mm, and interslice spacing = 0.5 mm. Menstrual cycle information was not recorded at the time of MRI acquisition. Imaging data were processed using FreeSurfer v7.4.1. Cortical 26 and subcortical 27 volumes were extracted and adjusted for each participant's intracranial volume. Exploratory group comparisons between MRM and non-MRM participants were conducted using ANCOVA with age as a covariate. Resulting p-values were corrected for multiple comparisons using the Benjamini-Hochberg false discovery rate (BH-FDR) method.
Results
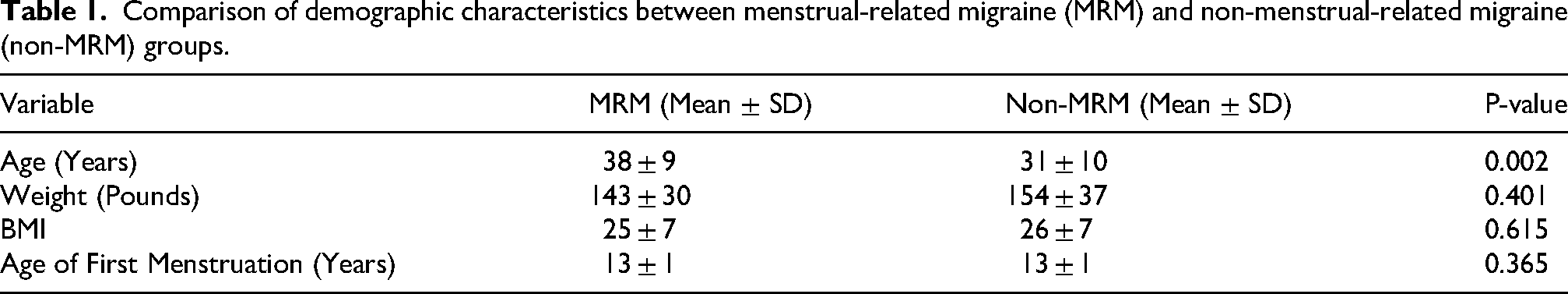
A total of 84 subjects were included in phenotype analysis, with 46 categorized in the non-MRM group and 38 in the MRM group. Demographic information analysis showed that mean age of the participants was 34 years, with a significant difference (p < 0.01) in mean age between the two groups: the MRM group had a mean age of 38 years (SD = 9), while the non-MRM group had a mean age of 31 years (SD = 10). No significant difference was observed in terms of age of menstruation onset, Body Mass Index (BMI), and weight in pounds (p > 0.05) between the two groups (Table 1).
Comparison of demographic characteristics between menstrual-related migraine (MRM) and non-menstrual-related migraine (non-MRM) groups.
Clinical migraine characteristics, as presented in Table 2, showed generally similar trends in years with migraine, time to maximum severity, monthly frequency, and intensity (p > 0.05). Moreover, migraine location details showed that the non-MRM group had significantly higher rate of migraine attacks occurring at the same site as compared with the MRM group (p < 0.05). However, rates of other migraine location trends and laterality were similarly reported by both groups (p > 0.05). Additionally, the two groups are compared in terms of migraine prodromes, allodynia, triggers, and aura. According to the results, MRM group generally reported higher rates of the recognized migraine triggers and prodromes, and aura and allodynia as compared with the non-MRM group (p > 0.05). The difference is significant in-terms of speech aura (p < 0.05) and prodromal signs including yawing, changes in stomach habits and appetite, and head discomfort (p < 0.05), with higher rates among the MRM as compared with the non-MRM group. Results of comparison of specific signs and symptoms associated with migraine showed no significant difference across the groups (p > 0.05).
Comparison of clinical characteristics and triggers between MRM and non-MRM groups.

Analysis of covariance (ANCOVA) was used for these variables with age as covariate; effect size is reported as partial eta-squared.
Logistic regression was used for these variables with age as covariate; effect size is reported as odds ratio (OR).
Note: CI [0.025, 0.975] indicates 95% confidence interval for the effect size.
Moreover, subjects have reported whether a recent change was experienced in clinical characteristic of migraine, as shown in Table 3. Accordingly, the MRM group has reported significantly higher rates of recent changes noticed in the migraine frequency (p < 0.05), and higher rates of recent changes in migraine severity and overall characteristics as compared with the non-MRM group (p > 0.05).
Comparison of recent changes in migraine characteristics between MRM and non-MRM groups.

Logistic regression was used for all variables with age included as covariate; effect size is reported as odds ratio.
CI [0.025, 0.975]: 95% confidence interval for the effect size.
“Recent Change” refers to any noticed recent increase or decrease in the above-mentioned qualities within the “last few months” from the time of survey completion.
Table 4 shows the prevalence rates of individual and familial medical history among the groups. The prevalence of the most common migraine comorbidities including psychiatric, cardiovascular, and autoimmune disorders were higher among the MRM group as compared with the non-MRM group (p > 0.05) (Table 4). Moreover, significantly more frequent reports of a family history of headache (p < 0.05), in addition to more reports of a family history of cardiovascular and/or neurological conditions (p > 0.05) was observed among the MRM group as compared with the non-MRM group.
Comparison of individual and familial medical history between MRM and non-MRM groups.
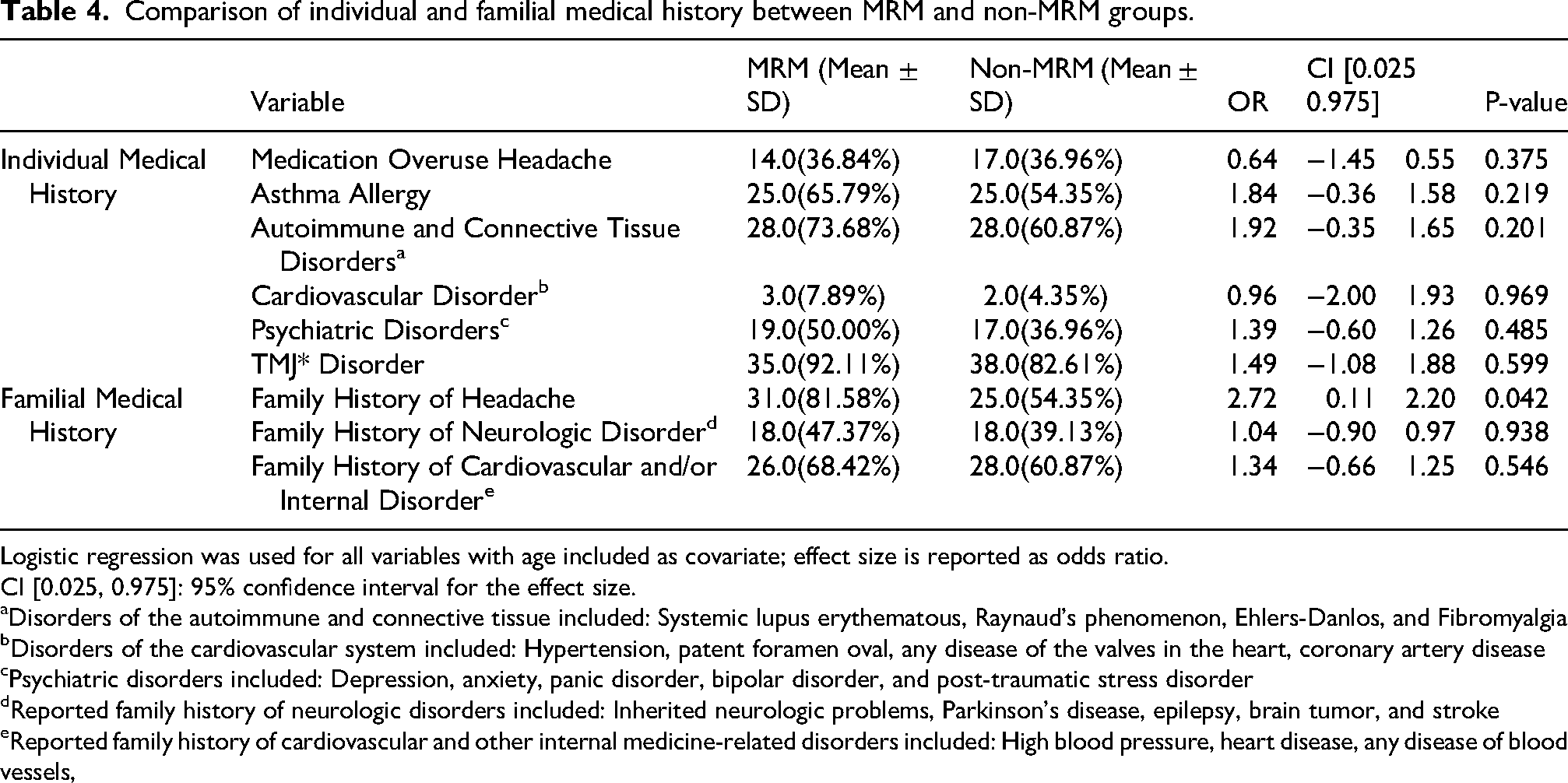
Logistic regression was used for all variables with age included as covariate; effect size is reported as odds ratio.
CI [0.025, 0.975]: 95% confidence interval for the effect size.
Disorders of the autoimmune and connective tissue included: Systemic lupus erythematous, Raynaud's phenomenon, Ehlers-Danlos, and Fibromyalgia
Disorders of the cardiovascular system included: Hypertension, patent foramen oval, any disease of the valves in the heart, coronary artery disease
Psychiatric disorders included: Depression, anxiety, panic disorder, bipolar disorder, and post-traumatic stress disorder
Reported family history of neurologic disorders included: Inherited neurologic problems, Parkinson's disease, epilepsy, brain tumor, and stroke
Reported family history of cardiovascular and other internal medicine-related disorders included: High blood pressure, heart disease, any disease of blood vessels,
Additionally, results of the comparison of PSEQ, PCS, PHQ-9, GAD, PSQI, and RLB scores in Table 5 showed no significant differences between the two groups (p > 0.05).
Comparison of pain-coping cognitions and comorbid mental health status between MRM and non-MRM groups.

ANCOVA was used with age as covariate to compare scores across the groups.
Treatment outcome results in Figure 1 showed significantly higher rate of favorable clinical improvement following the administration of triptans among MRM groups as compared with the non-MRM group. Moreover, MRM group showed significantly higher rate of clinical improvement following the overall use of acute migraine medication. No difference was observed among the groups in terms of clinical outcome following the use of preventive medication (Figure 2).

Proportion of users reporting migraine improvement by medication type.

Overall treatment outcome among acute vs preventive medication groups.
For exploratory imaging analyses, a subset of 67 participants with available structural MRI data was included, comprising 33 individuals in the MRM group and 34 in the non-MRM group. The non-MRM group exhibited significantly higher average volumes in the bilateral pericalcarine cortex and the left lingual gyrus compared to the MRM group. However, these differences did not remain significant after correction for multiple comparisons.
Discussion
This study shows that while MRM and non-MRM show several overlapping features, they tend to be distinct conditions requiring targeted treatment approaches for each of them.
Our demographic findings show that the prevalence of MRM in this cohort is 45% among female migraine patients, which is consistent with the previously reported prevalence rate between 22% and 70% through different studies.28–30 With the noticeable prevalence and burden, MRM is a subset of migraine that significantly affects women's quality of life and is important to be explored in more detail. The prevalence rate of MRM is higher than PMM with reports between 3.5 and 21.2, which indicates the burden and importance of studying MRM as a separate condition than PMM and non-MRM to address the specific patient needs.28,31,32
After exploring phenotypic characteristics and imaging comparisons between female migraine patients with and without MRM, this study shows generally different trends of clinical characteristics including significantly higher rates of attacks occurring on the same site, more prevalence rate of bothersome migraine symptoms, greater family history of headache, and more prevalence rate of favorable treatment outcome following the use of acute medication in the MRM group. A more detailed review of the results indicates a lower frequency, longer time to maximum severity, and greater intensity among the MRM as compared to the non-MRM group. This is consistent with several diary-based studies, which have shown that MRM attacks are usually more disabling and persistent than non-MRM attacks.33–37 Similar to our findings, studies have shown that MRM tends to be more severe and longer lasting,33,38,39 with generally high rates of favorable clinical outcome following the use acute medications such as NSAIDs and triptans.40–42 These findings show the importance of a comprehensive patient evaluation prior to diagnostic and therapeutic decision making, because while the conditions have several overlapping features they represent distinct clinical characteristics as well as different therapeutic outcome, which is highlighting the differences between underlying causes of the two conditions. Further studies have reported no significant improvement of MRM following the administration of preventive medications such as botulinum toxin A and anti-calcitonin gene-related peptide monoclonal antibodies,40,47 which in-line with our findings suggest the superiority of the effect of acute treatment especially triptans and NSAIDs for MRM and the importance of accurate and preferably diary-based diagnosis of MRM during the clinical visit. Moreover, as our results show a significantly higher prevalence rate of reported family history of headache among MRM as compared with the non-MRM patients, which is consistent with several other papers,52–54 the importance of obtaining a comprehensive medical history prior to any clinical decision making.
Further comparison of pain-coping cognitions including pain catastrophizing and pain self-efficacy scores across the groups was performed to investigate any personality-related pain experiences and perceptions that may contribute to the observed group differences, however, it did not reveal any cognitive and psychological base to explain the group differences in migraine experiences. Additionally, the two groups showed generally similar trends in lifestyle behavior scores, which includes maintaining consistent and healthy patterns in sleep, mealtimes, and exercise, and the psychiatric conditions that most commonly are associated with migraine, including anxiety, depression, and sleep disorders. Therefore, patients with and without MRM have distinct migraine experiences which are less likely to be affected by how they interpret and explain the pain and the lifestyle and comorbid factors which generally affect migraine. For instance, studies have reported high rates of maladaptive coping strategies, an externalized locus of control, and a fear of pain among migraine patients, all of which can exacerbate their condition and complicate treatment outcomes, particularly in those with medication overuse.43–46 To the best of our knowledge, this is the first study to investigate psychiatric and life-style related differences between MRM and non-MRM.
Our phenotype-based data suggests that MRM patients mostly experience migraine pain as peri-orbital, one-sided pain with varying sites of pain. Moreover, MRM patients have reported a greater number of recognized precipitants, prodromes, triggers, aura, allodynia, and associated signs and symptoms as compared with the non-MRM group in this study. While these findings did not reach to a statistically significant level, they are consistent with the existing literature, suggesting one-sided pain location and more recognized bothersome features such as nausea, vomiting, allodynia, and pain exacerbation by physical activity associated with MRM as compared to non-MRM patients.40,48 As an exploratory study, our findings are a basis for further assessment of the underlying causes of MRM, possibly by addressing the observed differences through pathophysiological and translational research as the exact underlying cause remains unclear.49–51
Prior imaging studies have reported structural and functional brain alterations associated with PMM,55,56 however, our exploratory brain volumetric analysis did not detect significant differences between MRM, non-MRM, and healthy controls. This can be due to potential overlapping features of the two conditions, which do not cause significant structural differences. Probably when it becomes only pure menstrual migraine then we can see differences.
Limitations
This study has several limitations that should be considered. One major limitation is the self-reported and cross-sectional design of the study, which may introduce recall bias and affect the accuracy of the data. Additionally, the analyses were based on the participants’ overall migraine profiles rather than on menstrual-related attacks specifically. Therefore, the conclusions are not specifically regarding menstrual-related attacks.
Another limitation is that the study was initiated before the public use of gepants and anti-CGRP monoclonal antibodies for the treatment of migraine, so it does not include data regarding treatment outcomes following the administration of these newer agents, which are now being widely used for migraine.
Moreover, in the exploratory imaging analysis, functional connectivity data were not included due to the absence of tracking the menstrual cycle stage at the time of MRI acquisition, which is believed to influence functional connectivity results. This omission may have affected the findings related to brain function and connectivity in the context of menstrual-related migraine.
Conclusion
This study reveals distinct clinical characteristics and treatment outcomes for MRM compared to non-MRM. These differences suggest potentially different pathophysiological pathways for these conditions and highlight the importance of careful and comprehensive patient evaluation in making accurate clinical decisions regarding diagnosis and treatment plans, especially when encountering female migraine patients.
Article highlights
Menstrual-related migraine (MRM) group experienced significantly higher rates of recent changes in migraine frequency, same-site occurrence of attacks, and family history of headaches compared to non-MRM.
MRM group reported greater incidence of prodromal signs, speech aura, nausea, and vomiting during migraine episodes.
MRM group demonstrated a significantly higher rate of improvement following the use of acute medications, particularly triptans.
Footnotes
Acknowledgements
We sincerely appreciate the participants for their time and cooperation through this research.
ORCID iDs
Ethical considerations
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. The study protocol was reviewed and approved by the Institutional Review Board (IRB) at Stanford University. All data collected were de-identified to ensure participant confidentiality and privacy. Participants were informed of their right to withdraw from the study at any time without any impact on their medical care.
Consent to participate
Written informed consent was obtained from all participants prior to their involvement in the research.
Consent for publishing
All authors agree to publish the article with Cephalalgia Reports
Author contributions
MB contributed in conceptualization, data curation, methodology, data analysis, visualization, writing, review and editing the draft; JS contributed in data analysis, and review and editing the draft; BMS contributed in data curation, methodology, review and editing the draft; AMP contributed in methodology, writing, review and editing the draft; RCK contributed in conceptualization, writing, review and editing the draft; RPC contributed in data curation, funding acquisition, supervision, resources, methodology, review and writing the draft.
Funding
This study was supported by the SUNSTAR foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
De-identified clinical and imaging data can be obtained by contacting the corresponding author.
Open practices
Not applicable.
