Abstract
Background
Whole-body resistance exercise is a popular training method that enhances strength and muscle hypertrophy. Little is known about the effect of this type of exercise on primary headaches. This scoping review and exploratory meta-analysis aim to elucidate the existing state of knowledge of whole-body resistance exercise as a non-pharmacological treatment of primary headaches compared to non-exercise.
Methods
Following the PRISMA Extension for Scoping Reviews guidelines and the Cochrane handbook, we conducted a literature search in Embase, Medline, Amed, PEDro, and Web of Science. Methodological quality was assessed using the Cochrane 2.0 tool to characterize the evidence base. An exploratory meta-analysis was conducted using a random-effect model.
Results
We identified notable gaps in the evidence base, with only two studies on migraine (18 participants) and vestibular migraine (286 participants), respectively, meeting the inclusion criteria. No studies were found for cluster headache or tension-type headache. The interventions consisted of 8 to 12 weeks of whole-body resistance exercise with two to three sessions per week, utilizing 45% to 80% of 1-repetition-maximum. The exploratory meta-analyses showed a standardized mean difference of −1.69 [95% CI −2.26, −1.13] for headache/vertigo intensity and −1,31 [95% CI −2.05, −0.56] for headache/vertigo frequency between exercise and no exercise. Quality assessments revealed major methodological limitations in both studies.
Conclusion
This scoping review identified substantial evidence gaps regarding whole-body resistance for primary headaches. While exploratory analyses suggest benefits for people with migraine, the limited and low-quality evidence base indicates a need for well-designed studies—particularly for tension-type headaches and cluster headaches.
Trial registration
The registration can be found under CRD42022343692 in PROSPERO. Registration details. Amendments from this protocol are explained in detail in the supplemental materials (Appendix 1).
This is a visual representation of the abstract.
Introduction
The use of pharmacological treatment in primary headaches is often limited by side effects, lack of availability, costs, or lack of efficacy. Patients often suffer from comorbid conditions, reducing the number of pharmacological options due to contraindications or interactions between treatments.1–4 Additionally, patients of childbearing potential using headache preventives are at risk for teratogenicity.5,6 Finally, some patients simply do not wish to use pharmacological therapies. Increased knowledge and availability of non-pharmacological therapies might expand treatment options for these patients. An example of a non-pharmacological therapy is exercise, which includes modalities such as endurance exercise and resistance exercise. A recent meta-analysis found that exercise could reduce migraine frequency, intensity, and duration. 7
Endurance exercise, involving prolonged isotonic contractions of large muscle groups, mainly influences cardiovascular and metabolic functions. 8 In contrast, whole-body resistance exercise targets large muscle groups using intermittent bursts of patterned movements against an external load. Such exercise regimens usually involve fewer than 15 repetitions per set to promote strength and muscle hypertrophy.9,10 Due to the differences between exercise types, it is possible that the effect, if any, on the different types of primary headaches vary. This makes it unclear how exercise should be tailored to patients.11,12 Considering that exercise modalities affect organ systems differently, there might also be modality-specific effects on primary headache disorders.
Unlike prolonged endurance activities that may trigger acute headache symptoms, 13 resistance exercise typically involves brief bursts of activity separated by rest periods. Different forms of resistance exercise may also engage distinct physiological pathways. For instance, whole-body resistance exercise likely induces broader systemic changes of hormonal, inflammatory, and neuroendocrine adaptions14–16 that could address common mechanisms in primary headache disorders such as central sensitization.17–20 In contrast, neck- and shoulder-specific resistance exercise primarily targets local musculoskeletal structures, which might not fully induce such systemic benefits. Based on these differing effects, we hypothesize that whole-body resistance exercise can reduce headache without triggering short-term exercise-induced headaches. Given the limited understanding of how whole-body resistance exercise affects different primary headache disorders, this scoping review and exploratory meta-analysis aims to map the existing evidence base regarding whole-body resistance exercise.
Methods
This scoping review adheres to the PRISMA Extension for Scoping Reviews 21 and follows the five-stage framework proposed by Arksey and O’Malley: (1) identifying the research question, (2) identifying relevant studies, (3) study selection, (4) charting the data, and (5) summarizing the results. 22 For the quality assessment component, we followed the Cochrane Handbook for Systematic Reviews of Interventions. 23 The registration of this review can be found under CRD42022343692 in PROSPERO. Amendments from this protocol are included in the supplemental materials (Appendix 1).
Inclusion and exclusion criteria
Inclusion and exclusion criteria were developed to map the available evidence on whole-body resistance exercise for primary headaches. Randomized controlled trials published in English or Scandinavian languages with patients diagnosed with cluster headache, migraine disorder, or tension-type headache by a neurologist or in accordance with the ICHD-3 criteria (including ICHD-3 appendix diagnoses such as vestibular migraine) without former experience with resistance exercise, were included. Our intervention of interest was whole-body resistance exercise compared to non-exercise for a minimum of eight weeks. Studies that delivered multiple interventions, for example, resistance exercise and endurance exercise, were excluded. Outcomes of interest were headache indices such as intensity, frequency, and duration, applying validated scales such as the visual analogue scale for intensity and headache diaries for frequency and duration in specified time periods.
Literature search
The literature search was conducted on 25 October 2023. Ovid was used to search Allied and Complementary Medicine Database (Amed), Excerpta Medica Database (Embase), and Medical Literature Analysis and Retrieval System Online (Medline). Additionally, Web of Science and Physiotherapy Evidence Database (PEDro) were also searched. The full literature search can be found in the supplemental materials (Appendix 2). Additionally, a briefer, updated PubMed search was performed on 3 February 2025, to identify any new studies published since the original search. Two reviewers (EM and ATD) worked independently and screened all titles and abstracts. If the abstract didn’t provide enough information to assess eligibility, the full-text was retrieved. If discrepancies occurred, the two reviewers discussed them. If an agreement could not be reached, a third reviewer (MHB) was recruited.
Data extraction
We extracted the following variables from studies that met the eligibility criteria using a data extraction form: study inclusion and exclusion criteria, participant demographics (age and sex), and characteristics of the intervention (exercise selection, number of sets and repetitions, time of pauses, and weight-increment protocol). Our predefined outcomes were headache attack intensity, headache attack frequency, and duration of headache attacks. We recorded the mean difference in outcome values at baseline and after the intervention was delivered.
Quality assessment of included studies
We performed qualitative assessments using the Cochrane Risk of Bias 2.0 tool and its guidance document 24 to characterize the methodological quality of the evidence base. This tool assesses risk of bias in five domains: randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result. 24 Because the methodologies used for different outcomes in the studies were similar, the risk of bias was evaluated for each study rather than for individual outcomes. This method streamlined the assessment, concentrating on general biases affecting the entire study rather than those related to specific outcomes. Additionally, the GRADEpro software and the GRADE handbook25,26 were employed to assess the certainty of the body of evidence. This involved evaluating factors such as study design, risk of bias, inconsistency, indirectness, imprecision, and a summary of findings. Each variable was given a rating to measure the quality of evidence for each outcome. 25
Exploratory meta-analysis
To explore the potential role of whole-body resistance exercise as a non-pharmacological therapy, we performed an exploratory meta-analysis on the frequency and intensity of migraine using standardized mean difference, also known as Cohen's D, as the effect measure to consider differences in measurement scales. The standardized mean difference can be interpreted as the mean difference expressed in standard deviation units. It describes the size of the difference between two groups. A standardized mean difference of under 0.5 can be understood as small, 0.5–0.8 is medium, 0.8–2.0 is large, and over 2.0 can be considered huge. 27 The meta-analysis, as well as an assessment of statistical heterogeneity, was conducted in R using the metafor package. 28 The code and data used are included in the supplemental materials (Appendices 3 and 4). We chose to use a random-effect model because of the variability in methods used for assessing outcome measures across the studies. 28 Furthermore, we used the tau2-metric to investigate statistical heterogeneity across studies as it might be less sensitive to the number of studies compared to I2. 29 However, tau2 does, such as all metrics of statistical heterogeneity, become increasingly uncertain when applied to a small number of studies. 23
Results
Study selection and characteristics
We identified a total of 74 studies in Amed, Embase, and Medline, with an additional 20 and five results from Web of Science and PEDro, respectively. We imported the 99 identified studies into an EndNote library and deduplicated to 85 results. Based on titles and abstracts, we identified five potentially eligible papers that were further assessed for inclusion. Two of these fulfilled the eligibility criteria.30,31 The remaining studies were excluded as the intervention only consisted of neck-specific resistance training or combined resistance training with endurance exercise32–34 (Figure 1 and Table 1).

This figure is a flow diagram demonstrating the process of inclusion and exclusion of identified studies.
Study characteristics.
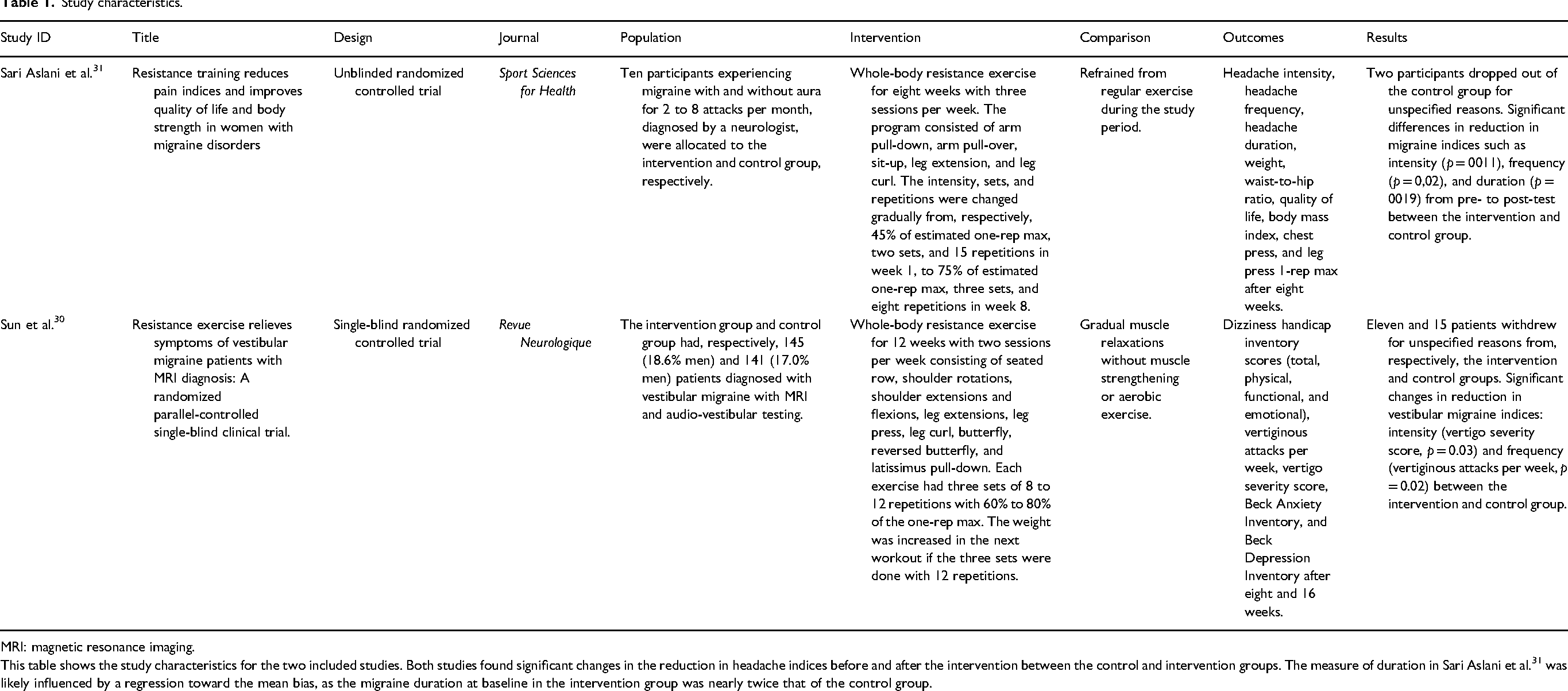
MRI: magnetic resonance imaging.
This table shows the study characteristics for the two included studies. Both studies found significant changes in the reduction in headache indices before and after the intervention between the control and intervention groups. The measure of duration in Sari Aslani et al. 31 was likely influenced by a regression toward the mean bias, as the migraine duration at baseline in the intervention group was nearly twice that of the control group.
The first study by Sari Aslani et al. 31 investigated resistance exercise in women with migraine. The second study by Sun et al. 30 investigated resistance exercise in a mixed-sex population with vestibular migraine. No eligible papers on tension-type headache or cluster headache were identified. The studies included investigated 8 to 12 weeks of whole-body resistance exercise with two to three sessions per week, utilizing 45% to 80% of 1-repetition-maximum (Table 2). However, the study characteristics (Table 2) and study results (Table 3) revealed substantial heterogeneity between the two studies, including intervention duration, participant characteristics, and outcome measurement methods. No additional studies meeting the eligibility criteria were identified in the briefer PubMed search on 3 February 2025.
Study results.

VAS: Visual Analogue Scale.
This table shows the results of the two studies that were included in our report. Headache intensity was measured using a VAS 1–10. In the paper by Sari Aslani et al., 31 headache duration was described as days with migraine. However, we have interpreted this as a typographical error, as hours with migraine is the standard unit of measurement of migraine duration. Headache frequency was measured as the number of attacks in a four-week period before and after the intervention using headache diaries. The frequency of vestibular migraine was measured as vertiginous attacks per week. Vestibular migraine intensity was measured using the vertigo severity score.
Risk of bias assessment.
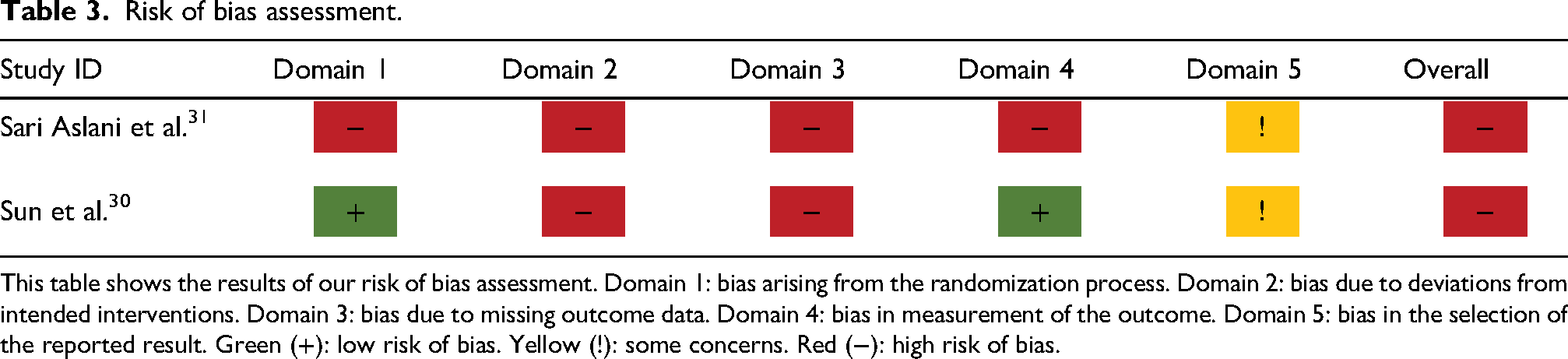
This table shows the results of our risk of bias assessment. Domain 1: bias arising from the randomization process. Domain 2: bias due to deviations from intended interventions. Domain 3: bias due to missing outcome data. Domain 4: bias in measurement of the outcome. Domain 5: bias in the selection of the reported result. Green (+): low risk of bias. Yellow (!): some concerns. Red (−): high risk of bias.
Quality assessment
Both studies identified had major limitations, which resulted in an overall high risk of bias (Table 3). The most significant domains were domains 2 and 3, studying, respectively, deviations from intended interventions and missing outcome data. A detailed assessment of our analysis can be found in the supplemental materials (Appendix 5).
We downgraded the certainty of evidence by two levels for the outcome of frequency due to a high risk of bias in both studies. The certainty of evidence comparing whole-body resistance exercise to non-exercise for headache/vertigo frequency was accordingly rated as low. While we found a moderate tau2-value for the frequency outcome, this alone cannot be used to conclude with a serious inconsistency across studies due to the very low number of included studies.23,26 However, given the consistent direction of effect in both studies, and the partially overlapping confidence intervals between the two studies (Figure 2), we did not find it appropriate to downgrade the certainty of evidence an additional step for inconsistency. We do, however, acknowledge that this may overlook underlying differences in intervention effects across migraine subtypes. For the outcome of intensity, we downgraded the certainty of evidence two levels because of a high risk of bias. The certainty of evidence comparing whole-body resistance exercise to non-exercise for headache/vertigo intensity was accordingly rated as low. In conclusion, we found a low certainty of evidence for both change in frequency and intensity (Table 4).

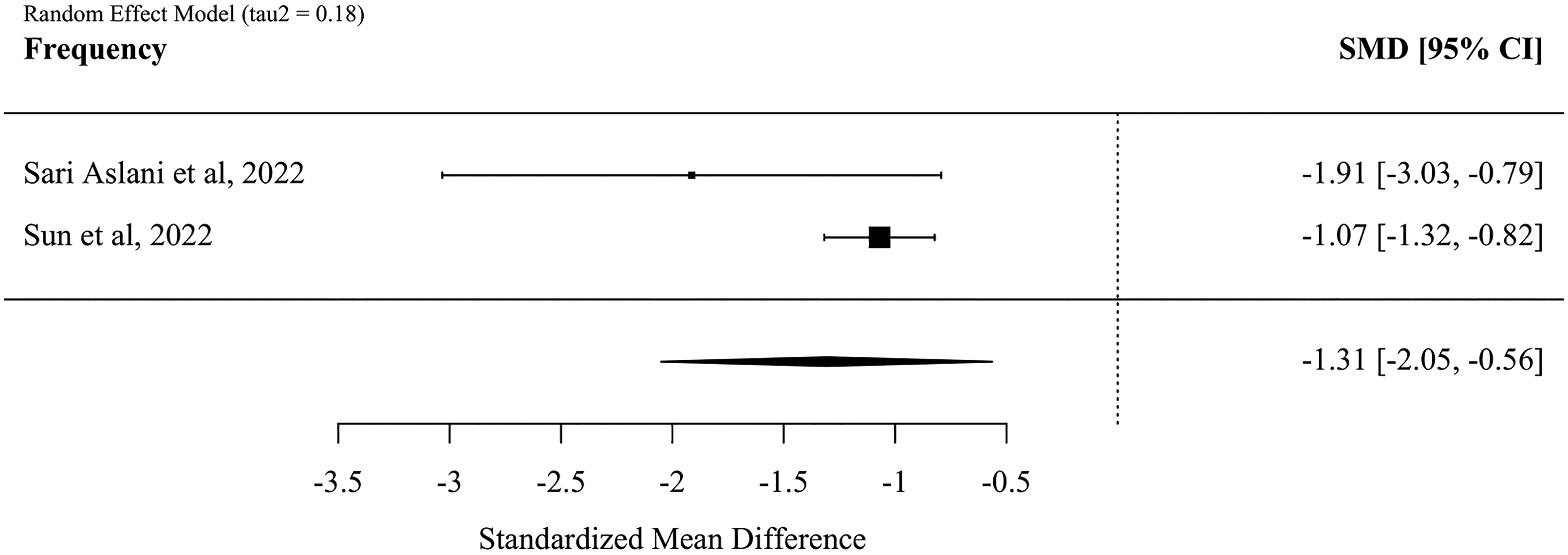
This figure is a forest plot and demonstrates the standardized mean difference in the reduction of headache/vertigo intensity. We found a large standardized mean difference of −1.31 [95% CI −2.05, −0.56]. The tau2-value of 0.18 suggests a moderate heterogeneity between studies.
Certainty of evidence (GRADE) assessment.

SMD: standardized mean difference.
This table shows our assessment of the certainty of evidence in the included studies across two outcomes: frequency and intensity. We found a low certainty of evidence for both outcomes because of a high risk of bias in both studies.
Exploratory meta-analysis
Standard deviations for headache frequency and intensity in the study by Sari Aslani et al. 31 were not reported for the change scores, but separately for the pre-test and post-test. To estimate the standard deviation of the change, which was necessary for the exploratory meta-analysis, we calculated a pooled standard deviation and assumed a moderate correlation between scores. Varying this coefficient revealed substantial differences, which indicates that our findings lack robustness (Supplemental Appendix 6). 35
The exploratory meta-analysis showed a standardized mean difference in headache/vertigo intensity of −1.69 [95% CI −2.26, −1.13] (Figure 3). Headache/vertigo frequency had a similar reduction in standardized mean difference of −1.31 [95% CI −2.05, −0.56] (Figure 2). The individual study results shown in the forest plots represent a natural subgroup analysis by migraine type, with classic migraine demonstrating larger frequency reductions but smaller intensity reductions compared to vestibular migraine, though with considerably wider confidence intervals (Figures 2 and 3). We found statistical heterogeneity (tau2) values of 0.09 for frequency and 0.18 for intensity, but these estimates should be interpreted with care because of the low number of studies.
This figure is a forest plot and demonstrates the standardized mean difference in the reduction of headache/vertigo intensity. We found a large standardized mean difference of −1.69 [95% CI −2.26, −1.13]. The tau2-value of 0.09 suggests a low heterogeneity between studies.
Quality of life outcomes
Both studies reported increased quality of life in participants, but used different outcome measures. Sari Aslani et al. 31 used the Headache Impact Test-6 (HIT-6) and concluded with an increased quality of life in the intervention group, but reported contradictory interpretations of HIT-6 scores (Supplemental Appendix 1). 31 Sun et al. 30 used the Dizziness Handicap Inventory and reported a statistically significantly larger reduction in the intervention group, which translates to improved quality of life. 30
Discussion
Summary of findings
This scoping review identified substantial evidence gaps regarding whole-body resistance exercise for primary headaches. Only two studies met our inclusion criteria, both focusing on migraine populations. No evidence was identified for tension-type headache or cluster headache. While an exploratory meta-analysis suggested potential benefits for people with migraine, methodological concerns and heterogeneity in the populations examined preclude strong conclusions.
Comparison of findings
Our exploratory analysis aligns with broader evidence on exercise interventions for primary headache disorders, which consistently demonstrate a reduction in headache burden.7,36–38 However, previous reviews have not differentiated between whole-body resistance exercise and neck-specific resistance exercise. A recent umbrella review found that both neck-specific resistance exercise and endurance exercise are effective in reducing the intensity, frequency, and duration of migraine and tension-type headaches. 36 A systematic review and meta-analysis examined the compared efficacy of endurance exercise and resistance exercise in people with migraine. 37 Resistance exercise was identified as the most effective exercise modality for reducing migraine outcomes, but the high risk of bias in resistance exercise studies was not addressed. Another systematic review investigated the effect of different exercise intensities on migraine and tension-type headaches. 38 Consistent with the previously discussed reviews, they found that exercise reduced migraine and tension-type headache symptoms. However, they could not present conclusive data on the impact of different exercise intensities on these conditions.
Resistance exercise may lead to a lower production and greater clearance of lactate compared to endurance exercise. This would make it an effective exercise modality as it allows the benefits of physical activity while reducing the risk of triggering migraine through lactate accumulation. A retrospective observational study identified exercise as a trigger for attacks in some patients with migraine, but did not differentiate between different forms of exercise. 39 Increased lactate induced by exercise has been suspected as a trigger of migraine attacks. 40 Another potential trigger is calcitonin gene-related peptide (CGRP), a neuropeptide that is released during migraine attacks. CGRP has been shown to rise during high-intensity exercise. 41 Because of its nature of short bursts of high-intensity activity with inter-set pauses, resistance exercise relies primarily on the anaerobic adenosine triphosphate-creatine phosphate energy system. 42 The rest time between sets allows for clearance of lactate produced during the preceding sets. 43 Conversely, endurance exercise primarily engages the aerobic energy system. 44 However, when the intensity of the exercise reaches or surpasses the lactate threshold, the body relies increasingly on anaerobic metabolism, leading to the production of lactate. 45
Contrary to isolated resistance exercise regimens such as neck-specific resistance exercise, whole-body resistance exercise likely promotes systemic effects to a greater extent. 46 We believe that it is these systemic effects that most likely explain the reduction in headache burden from resistance exercise, but the mechanisms are not understood in detail. One possible mechanism is that resistance exercise reduces pain sensitivity, possibly by the release of endogenous opioids such as beta-endorphin.47,48 Interestingly, beta-endorphin has been shown in some studies to be lowered in people with migraine. 49
Strengths and limitations
The studies that were identified had a well-defined and transparent study design of whole-body resistance exercise and are easily replicable. The scoping review methodology allowed mapping of the limited evidence base, identifying multiple research gaps. However, there are several limitations to the evidence included in the review. In summary, the identified studies demonstrated considerable heterogeneity that limits the interpretation of the findings in our exploratory meta-analysis. Another caveat was the high risk of bias and low certainty of evidence identified in our assessment of methodological quality. Furthermore, the studies differed markedly in populations (exclusively women vs. mixed sex), sample sizes (18 vs. 286), migraine subtypes (migraine vs. vestibular migraine), outcome measurements, intervention duration (8 vs. 12 weeks), and exercise selection. These differences, and in particular the heterogeneity between the interventions, limit the generalizability and certainty of the results in our exploratory analysis. There is also a possibility of publication bias, where studies with negative results may remain unpublished, potentially overestimating the true effect of whole-body resistance exercise.
The study by Sari Aslani et al. 31 had a small sample size with a substantial attrition rate and did not report standard deviations for change scores. This necessitated statistical approximation using an established formula that incorporates pre- and post-test standard deviations with an assumed correlation. While this provides a pragmatic solution, it may not fully capture the true variability.
The outcomes in the included studies were measured using different methods. The study by Sari Aslani et al. 31 measured the frequency of migraine as the total number of attacks during a four-week period before and after the training period using a headache diary. Sun et al. 30 measured frequency as the number of vertiginous attacks per week. It is unclear whether this included headache days. Nevertheless, these metrics are likely less precise than measuring migraine days. Conversely, Sun et al. 30 measured intensity using a vertigo severity score, while Sari Aslani et al. 31 measured intensity using a visual analogue scale.
Pooling headache frequency with vertiginous attack frequency, and headache intensity with vertigo severity, introduces clinical heterogeneity and constitutes a limitation in our exploratory analysis. This approach may oversimplify complex differences between migraine subtypes and exercise outcomes. However, we do believe that this pragmatic approach is useful for several reasons. First, the pathogenesis of vestibular migraine is still uncertain, and the diagnosis of vestibular migraine is not universally recognized as a separate disease by the headache community. 50 The disease is thought to have shared pathophysiological mechanisms with typical migraine attacks involving the trigemino-vascular system.51,52 Second, the use of the standardized mean difference accounts for the differences in measurement scales and allows for the comparison of effect sizes across different outcome measures. Third, the random-effects model employed in our analysis accounts for between-study variability through the assumption that the included studies estimate different, but related, intervention effects. 23 However, the substantial heterogeneity underscores the early stage of research in this area and the need for more standardized approaches.
Future research directions
A notable challenge in studies examining exercise and primary headaches is the difficulty of blinding. Lack of blinding increases the risk of placebo and other contextual effects associated with the intervention. This issue could be addressed by utilizing control groups engaged in non-specific activities. For example, a possible study design might include an intervention group engaging in a traditional resistance exercise regimen, compared to a control group engaging in a non-traditional resistance exercise regimen with non-specific exercises and low intensities, which is presumed to be less effective.
Future studies should also examine how variables such as CGRP and lactate profiles vary with different resistance exercise regimens (variability in pauses, repetitions, sets, and exercise selection) and explore their potential relationships to headache outcomes, ideally for an extended period (>8 weeks) with prospective daily headache diary entries. Standardized, validated quality-of-life measures should be incorporated to provide comprehensive patient-centered outcome assessment and enable meaningful comparison across trials.
It is especially prudent to conduct studies on resistance exercise in tension-type headache and cluster headache, as our systematic review did not identify any such studies. Finally, to address individual exercise preferences and different impacts of exercise modalities on the body, future studies should aim to compare the efficacy of endurance and resistance exercise in managing different types of primary headaches. Given the belief that there are sex-specific differences in exercise responses,53,54 future studies should also address potential sex-specific differences in headache alleviation through exercise.
Conclusions
This scoping review identified large evidence gaps regarding whole-body resistance exercise for primary headaches. Exploratory analysis of two migraine studies suggests benefits, but these studies had notable clinical heterogeneity and methodological concerns, precluding clear conclusions. Future research should focus on well-designed randomized controlled trials examining whole-body resistance exercise for primary headache disorders, particularly tension-type headache and cluster headache. This includes standardized outcome measurement approaches and strategies to address blinding challenges inherent in exercise interventions.
Key findings
We identified only two randomized controlled trials with whole-body resistance training in people with migraine.
The certainty of evidence in the included studies was rated as low due to high risk of bias and methodological limitations.
Studies investigated 8 to 12 weeks of whole-body resistance exercise (2 to 3 sessions per week, 45% to 80% of 1-repetition-maximum), but there was large heterogeneity between studies.
The mechanisms for how resistance exercise may benefit people with migraine include reduced lactate accumulation, increased endorphin release, and systemic anti-inflammatory effects.
Supplemental Material
sj-docx-1-rep-10.1177_25158163251383149 - Supplemental material for The effect of whole-body resistance exercise versus non-exercise on primary headaches: A scoping review and exploratory meta-analysis
Supplemental material, sj-docx-1-rep-10.1177_25158163251383149 for The effect of whole-body resistance exercise versus non-exercise on primary headaches: A scoping review and exploratory meta-analysis by Einar Naveen Møen, Anne-Mari Torgersen Dalgeir, Mattias Linde, Tore Wergeland, Erling Tronvik, Jannicke Igland and Marte-Helene Bjørk in Cephalalgia Reports
Footnotes
Acknowledgments
None.
ORCID iDs
Author contributions
All authors have made a substantial contribution to the work reported. ENM, MHB, ET, and TW conceived the study. ENM, ATD, and MHB defined the eligibility criteria. ENM and ATD conducted the literature search. ENM conducted the RoB analysis, which was revised by ML. ENM conducted the GRADE analysis, which was revised by MHB. ENM conducted the statistical analysis, which was revised by JI. ENM, JI, and MHB interpreted the results. ENM drafted the first version of the manuscript. ATD, ML, TW, ET, and JI revised the writing. All authors have critically revised and approved the final version of the manuscript.
Consent to participate
The information in this study is derived from previously published data, thus not requiring ethics approval according to our regional regulations.
Consent for publishing
Yes, all authors consent to the publication of this work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: NorHEAD is financed by the Research Council of Norway. Grant number 328615.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Erling Tronvik has received personal fees for lectures and advisory boards from the following: Novartis, Eli Lilly, AbbVie, TEVA, Roche, Lundbeck, Pfizer, Biogen, and Organon. Consultant for and owner of stocks and IP in Man & Science and Nordic Brain Tech, and holds shares in Keimon Medical. Non-personal research grants from several sources, including the European Commission, Norwegian Research Council, DAM Foundation, KlinBeForsk, and Nordic Innovation. Commissioned research (non-personal): Lundbeck, Eli Lilly. Jannicke Igland is the head of the Core Facility for Biostatistics and Data Analysis at the University of Bergen, which is doing commissioned research for Novartis on the use of prophylactic migraine medications, not related to the current project. Tore Wergeland reports research grants from The Liaison Committee for Education, Research and Innovation in Central Norway (Samarbeidsorganet) and The DAM Foundation. He has received lecture honoraria from TEVA, Roche, Lundbeck, and Lilly, and is a shareholder in two medical device start-up companies (Vilje Bionics AS and Keimon Medical AS). Marte-Helene Bjørk has received honoraria for giving lectures from Organon, AbbVie, UCB, Pfizer, Eisai, BestPractice, Angelini Pharma, and Novartis; honoraria for serving on advisory board from Jazz Pharmaceuticals, Angelini Pharma, Lundbeck, Pfizer, and Eisai; consultancy honoraria and research support, not related to the present study, from Novartis; and institutional fees for contract research from Lundbeck and marked authorization holders of valproate.
Data availability statement
The supplemental material includes amendments from the protocol registered in PROSPERO (![]() Appendix 1), literature search (Supplemental Appendix 2), code in R (Supplemental Appendix 3), data extracted from the included studies (Supplemental Appendix 4), risk of bias assessment (Supplemental Appendix 5), and the calculations of pooled standard deviations (Supplemental Appendix 6).
Appendix 1), literature search (Supplemental Appendix 2), code in R (Supplemental Appendix 3), data extracted from the included studies (Supplemental Appendix 4), risk of bias assessment (Supplemental Appendix 5), and the calculations of pooled standard deviations (Supplemental Appendix 6).
Open practices
None.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
