Abstract
Background
Migraine is a prevalent neurological disease that has socioeconomic and functional impact. It is also frequently associated with sleep disorders. The intricate relationship between migraine and sleep disturbances involves complex neurobiological circuits within the brain, including those that govern the sleep-wake transition. Observational evidence suggests that sleep disruption may trigger migraine episodes and contribute to their progression from episodic to chronic state. Our study aimed to explore the impact of fremanezumab, a calcitonin gene-related peptide (CGRP) monoclonal antibody (mAb), on sleep in patients with migraine.
Methods
Employing a prospective, observational, open-label treatment approach, this preliminary case series assessed sleep quality using daily e-diaries and the Insomnia Severity Index over four months: one month pre-treatment and three months during treatment with fremanezumab.
Results
Analyzing data from six cases, we found associations between improvements in migraine and sleep quality in five cases and no association in one case in which improvement in sleep was recorded, despite a lack of improvement in migraine frequency and intensity.
Conclusion
This study suggests that the primary mechanism by which fremanezumab improves sleep involves a reduction in headache frequency and severity. The possibility that fremanezumab acts directly on sleep-promoting neurons in the hypothalamus is also discussed. These findings emphasize the need for further research to elucidate the precise mechanisms by which CGRP mAbs influence sleep pathology.
Trial registration
NCT04693533.
This is a visual representation of the abstract.
Background
Migraine, which affects ∼15% of the population, is a disabling condition with substantial social, economic, and functional impact. 1 It often coexists with psychiatric and medical comorbidities, including frequently reported sleep disorders.2–6 The exact mechanisms linking migraines and sleep disturbances are not fully understood. Previous studies have shown that brainstem cholinergic and monoaminergic neuronal circuits regulate wakefulness, including the pedunculopontine and laterodorsal tegmental nuclei and serotoninergic, noradrenergic, dopaminergic, and histaminergic neurons, which collectively form the ascending arousal system.7–12 These circuits interact with sleep-promoting neurons in the hypothalamus and preoptic area, suggesting a complex network that controls the transition between sleep and wake states, potentially mediated by a flip-flop switch mechanism.10,13
Observations from migraine patients have attributed their attacks to various sleep-related factors, such as insufficient sleep, interrupted sleep, and poor sleep quality, leading to feeling unrested in the morning, which sparked the belief that sleep disturbances are common migraine triggers and may contribute to the progression from episodic to chronic migraine. A cross-sectional association has been reported between migraines and several sleep disorders, including sleep apnea, insomnia, sleep disturbance, snoring, shortness of breath, and excessive sleepiness. 14 Sleep disturbances are known to be risk factors for the chronification or progression of migraine. 15
Calcitonin gene-related peptide (CGRP) may play a direct role in sleep modulation. CGRP may influence sleep by promoting wakefulness through pathways similar to diuretic hormone 31 (DH31) in animal models, suggesting a potential role in modulating sleep patterns. 16 The emergence of more migraine-specific medications, including CGRP-targeted treatments, reflects a growing understanding of migraine pathophysiology. 17 CGRP monoclonal antibodies (mAb) have been suggested to be effective not only in alleviating migraine symptoms but also in improving sleep quality in patients with both sleep disorders and migraine.18–20 Furthermore, treatment with galcanezumab affects sleep by reducing the incidence of headache, which typically follows various triggers, including sleeping too little. 6 The mechanisms by which CGRP mAbs may affect sleep are not fully understood. Migraine treatment with CGRP mAb may improve sleep disturbances by targeting hypothalamic neurons involved in the sleep-wake cycle, given the hypothalamus's more permeable blood-brain barrier. In contrast, there are also reports of no improvement in sleep quality after treatment with CGRP mAb.21,22 Our working hypothesis suggests that if fremanezumab interacts directly with hypothalamic sleep neurons, improvements in sleep would be unrelated to improvements in migraine. However, if fremanezumab does not interact directly with sleep neurons, improvements in sleep would be associated with reductions in the headache intensity. The aim of the present study was to examine whether fremanezumab, a CGRP mAb, improves sleep disturbances and, using clinical correlates, to determine whether fremanezumab can directly interact with hypothalamic neurons that regulate sleep.
Methods
This was a prospective, observational, open-label treatment, preliminary descriptive case series study (Clinical Trial Identifier NCT04693533).
Patients
Patients were recruited from the Comprehensive Headache Center, Beth Israel Deaconess Medical Center (Brookline, MA, USA). The study's inclusion criteria were individuals aged ≥18 years with a prior diagnosis of migraine (with or without aura), experiencing 10 or more days with headache per month over at least the past 3 months, with at least 8 of those days being migraine days lasting more than 4 hours if untreated. The inclusion criteria were migraine onset at age 50 years or younger and a score of 10 or higher on the insomnia severity index (ISI) (Table 1). All patients agreed not to initiate or modify the type, dosage, or frequency of any preventive medications for conditions other than migraine that could impact the study outcomes (e.g. anticonvulsants, antidepressants, and beta-blockers).
Demographics and vital signs of patients.
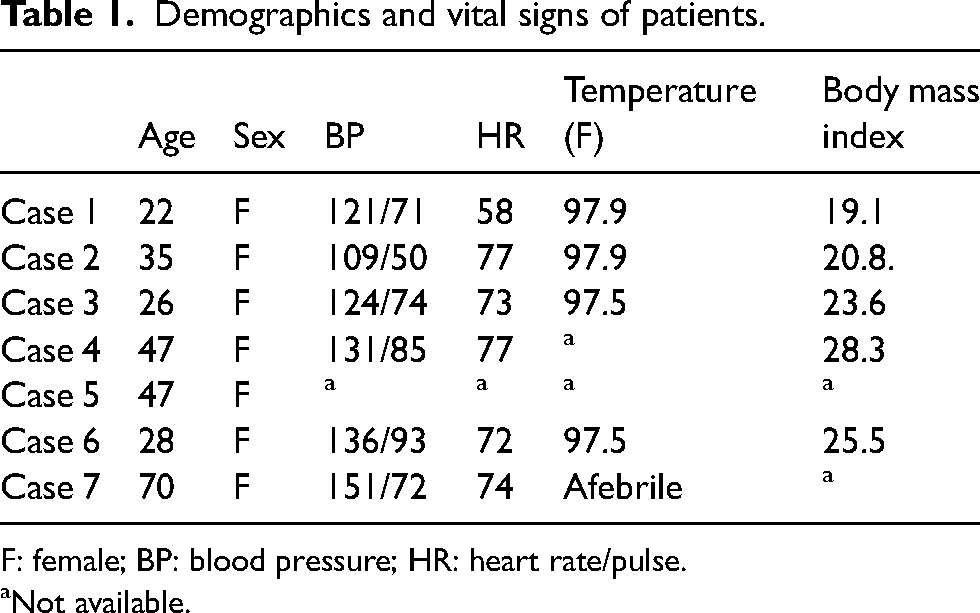
F: female; BP: blood pressure; HR: heart rate/pulse.
Not available.
Exclusion criteria for the study included patients currently on one or more migraine preventive therapies; those who were pregnant or breastfeeding; and individuals with significant psychiatric, cognitive, or behavioral disorders that could interfere with study participation. Other exclusion criteria included significant pain conditions (e.g. fibromyalgia, cancer pain, or other head or facial pain disorders), known or suspected severe cardiac disease (e.g. symptomatic coronary artery disease, history of myocardial infarction, or heart failure), and cerebrovascular disease (e.g. history of transient ischemic attack, stroke, or symptomatic carotid artery disease). Patients with a report of an abnormal electrocardiogram (ECG) in the past year (e.g. second- or third-degree heart block, prolonged QT interval, atrial fibrillation, or clinically significant ventricular arrhythmias) or those with uncontrolled hypertension (systolic 160 mm Hg, diastolic >100 mm Hg) were also excluded. The study did not include patients with known or suspected secondary headaches, a history of substance abuse or addiction within the past 5 years, or current marijuana or cannabidiol oil use (including medical marijuana) within the last year. Additional exclusion criteria included the use of simple analgesics or NSAIDs for more than 15 days per month; triptans, ergots, or combination analgesics for more than 10 days per month; and current opioid use for headache or other pain. Patients who had undergone pericranial nerve blocks within the last 3 months or who had received botulinum toxin or CGRP mAb treatment within the past 6 months were also ineligible. Individuals using daily sleep aids (e.g. benzodiazepines, barbiturates, eszopiclone, zaleplon, zolpidem, or over-the-counter sleep medications) or those with diagnoses of restless legs syndrome or obstructive sleep apnea were excluded.
Study design
The patients visited the headache center five times (Figure 1). During the first visit, patients received a thorough explanation of the study, signed informed consent forms, and were assessed for eligibility. The patients provided their medical and headache histories and were trained on how to use a daily electronic diary (e-diary) to record information on both sleep and headaches through REDCap. At the second visit, patients returned to review their e-diaries and received initial treatment with subcutaneous injection of fremanezumab 225 mg (1.5 mL volume). They were also instructed on how to self-administer the future doses at home. Patients continued to complete their daily e-diaries for an additional 3 months. During this period, patients were either self-administered or returned to the clinic for their second and third fremanezumab injections (225 mg). In addition, they participated in virtual appointments for their third and fourth check-ins, in which their headaches and sleep e-diaries were reviewed. In total, six patients completed the four-month e-diary and were scheduled for the last visit, during which they were able to ask questions, share their thoughts on the treatment, and review a summary of the data they had entered in the e-diary.

Flowchart of research design.
Headache assessment and diagnosis
All participants completed two daily e-diaries for a minimum of 4 months: 1 month before and 3 months after beginning treatment (Figure 1). The e-diaries were administered through a REDCap survey, accessed via an email link on the participants’ personal computers or electronic devices. The e-diary includes a questionnaire designed to capture the daily and monthly frequency of headaches and migraines, headache laterality (unilateral or bilateral), headache intensity, associated symptoms (e.g. nausea, vomiting, sensitivity to light, sound, and odors), and the use of headache-abortive medications. 6
Patients were diagnosed with migraine by a neurologist and headache specialist (SA) after reviewing their completed four-week e-diary. The diagnosis was made using the criteria from the third edition of the International Classification of Headache Disorders (ICHD-3). 23 Patients who experienced 14 or fewer monthly migraine days (MMD) were diagnosed with episodic migraine. Those with 8–14 MMD were diagnosed with high-frequency episodic migraine (HFEM), whereas those with 15 or more MMD were diagnosed with chronic migraine (CM).
Headache treatment response
Fremanezumab responders were defined as patients who experienced a reduction of ≥ 50% in the number of migraine days during the three-month treatment period compared to the pre-treatment month. In contrast, non-responders showed less than a 50% reduction in migraine days over the same period. The 50% reduction threshold is the standard for identifying responders in phase 3 placebo-controlled trials for preventive migraine treatment. 24
Sleep assessment
Daily assessments of sleep were collected over a period of 4 months: 1 month before treatment and 3 months during treatment. To assess sleep, the participants were asked to fill out an e-diary every morning about their experience with sleep the night before using the first three questions of the ISI. 25 The patients were asked to evaluate the following three aspects of sleep: (1) difficulty falling asleep, (2) difficulty staying asleep, and (3) problems waking up too early. For each question, patients responded using the following scale: 0 = none, 1 = mild, 2 = moderate, 3 = severe, and 4 = very severe. Movement from a higher severity category to a lower category was defined as improvement.
Results
Case 1: No improvement in migraine or sleep
Clinical background: A 22-year-old right-handed female with a history of headache since the age of 8 years and difficulty falling or staying asleep. Her headache fulfilled the ICHD-3 criteria for migraine without aura, and she was diagnosed with CM (21 MMD). She reported a family history of migraine. Headaches were described as unilateral but alternating sides, bilateral, frontal, temporal, periorbital, throbbing, pulsating, pressing, sharp, or stabbing. The attacks lasted 24–48 hours at an intensity of 7–10 (0–10 visual analog scale [VAS]). The Migraine Disability Assessment (MIDAS) score was 76. The associated symptoms included nausea, vomiting, photophobia, phonophobia, osmophobia, tearing of the eyes, nasal congestion, lightheadedness, spinning sensation, ringing in the ears, neck pain, neck stiffness, jaw pain, abdominal pain, decreased appetite, nervousness, anxiousness, irritability, difficulty with concentration, and memory. Prodromal/premonitory symptoms include sensitivity to sound/light/odor, skin sensitivity, and poor appetite. Past medical history included anxiety, and back and neck pain. The past pharmacological treatments included ibuprofen, naproxen, diphenhydramine, codeine, escitalopram, melatonin, sumatriptan, eletriptan, amitriptyline, acetaminophen, sertraline, and topiramate. At the time of the study, she used aspirin-acetaminophen-caffeine (Excedrin Migraine®) or ibuprofen 800 mg every 4–6 hours as needed. Treatment outcomes are shown in Figure 2(A).

(A) Treatment outcome: (A) Migraine—at baseline, the number of monthly migraine days (MMD) was 20 (57.1%), and the mean (IQR) pain intensity was 5.7 (4–7). After 1, 2, and 3 months of treatment, MMD was 36.3%, 46.1%, and 37.5% (40.6% for all 3 months), and the pain intensity was 5.8 (5.2–6.7), 4.8 (3–6), and 5 (3.5–6.5), respectively. (B) Sleep—throughout the study, this patient scored his difficulty with sleep at 4, 3, 2, 1, but not 0. Scores of 4 were reported at 34.2% of the nights at baseline versus 36.3%, 50%, and 50% of the nights during the first, second, and third months of treatment. Scores of 3 were reported at 34.2% of the nights at baseline versus 50%, 42.3%, and 31.2% of the nights during the first, second, and third months of treatment. Scores of 2 were reported at 31.4% of the nights at baseline versus 13.6%, 7.6%, and 12.5% of the nights during the first, second, and third months of treatment. Scores of 1 were reported at 0% of the nights at baseline versus 0%, 0%, and 6.2% of the nights during the first, second, and third months of treatment. (B) Treatment outcome: (A) Migraine—at baseline, the number of MMD was 25 (73.5%), and the mean (IQR) pain intensity was 3.4 (2–5). After 1, 2, and 3 months of treatment, MMD was 33.3%, 23.5%, and 27.5% (27.7% for all 3 months), and the pain intensity was 1.7 (1–2), 1.6 (1–2), and 2.1 (1–2.7), respectively. (B) Sleep—throughout the study, this patient scored his difficulty with sleep at 2, 1, or 0, but not 3 or 4. Scores of 2 were reported at 26.4% of the nights at baseline versus 3.7%, 8.8%, and 3.4% of the nights during the first, second, and third months of treatment. Scores of 1 were reported at 44.1% of the nights at baseline versus 25.9%, 26.4%, and 24.1% of the nights during the first, second, and third months of treatment. Scores of 0 were reported at 29.4% of the nights at baseline versus 70.3%, 64.7%, and 72.4% of the nights during the first, second, and third months of treatment. (C) Treatment outcome: (A) Migraine—at baseline, the number of MMD was 29 (93.5%), and the mean (IQR) pain intensity was 5.3 (4–7). After 1, 2, and 3 months of treatment, MMD was 91.2%, 36.6%, and 53.5% (57.7% for all 3 months), and the pain intensity was 2.7 (1–4), 2.6 (2–3), and 2.3 (1–3), respectively. (B) Sleep—throughout the study, this patient scored his difficulty with sleep at 3, 2, 1, or 0, but not 4. Scores of 3 were reported at 0% of the nights at baseline versus 3.1%, 0%, and 0% of the nights during the first, second, and third months of treatment. Scores of 2 were reported at 25.8% of the nights at baseline versus 0%, 0%, and 0% of the nights during the first, second, and third months of treatment. Scores of 1 were reported at 12.9% of the nights at baseline versus 9.3%, 10%, and 21.4% of the nights during the first, second, and third months of treatment. Scores of 0 were reported at 61.2% of the nights at baseline versus 87.5%, 90%, and 78.5% of the nights during the first, second, and third months of treatment. (D) Treatment outcome: (A) Migraine—at baseline, the number of MMD was 21 (63.6%), and the mean (IQR) pain intensity was 6.5 (6–7). After 1, 2, and 3 months of treatment, MMD was 27%, 9.6%, and 24% (20.4% for all 3 months), and the pain intensity was 5.6 (3.2–8), 7 (7–7), and 8.1 (7.5–9), respectively. (B) Sleep—throughout the study, this patient scored his difficulty with sleep at 4, 3, 2, 1, or 0. Scores of 4 were reported at 3% of the nights at baseline versus 2.7%, 0%, and 0% of the nights during the first, second, and third months of treatment. Scores of 3 were reported at 9% of the nights at baseline versus 5.4%, 0%, and 0% of the nights during the first, second, and third months of treatment. Scores of 2 were reported at 33.3% of the nights at baseline versus 8.1%, 3.2%, and 12% of the nights during the first, second, and third months of treatment. Scores of 1 were reported at 24.2% of the nights at baseline versus 29.7%, 6.4%, and 4% of the nights during the first, second, and third months of treatment. Scores of 0 were reported at 30.3% of the nights at baseline versus 54%, 90.3%, and 84% of the nights during the first, second, and third months of treatment. (E) Treatment outcome: (A) Migraine—at baseline, the number of MMD was 20 (52.6%), and the mean (IQR) pain intensity was 3.5 (2.2–5). After 1, 2, and 3 months of treatment, MMD was 32.2%, 30.4%, and 19% (26% for all 3 months), and the pain intensity was 2.7 (2–3.2), 3.2 (2–5), and 4.3 (2.2–6.5), respectively. (B) Sleep—throughout the study, this patient scored his difficulty with sleep at 3, 2, 1, or 0, but not 4. Scores of 3 were reported at 2.6% of the nights at baseline versus 3.2%, 4.6%, and 0% of the nights during the first, second, and third months of treatment. Scores of 2 were reported at 13.1% of the nights at baseline versus 35.4%, 8.6%, and 2.3% of the nights during the first, second, and third months of treatment. Scores of 1 were reported at 42.1% of the nights at baseline versus 19.3%, 4.3%, and 11.9% of the nights during the first, second, and third months of treatment. Scores of 0 were reported at 42.1% of the nights at baseline versus 41.9%, 82.6%, and 85.7% of the nights during the first, second, and third months of treatment. (F) Treatment outcome: (A) Migraine—at baseline, the number of MMD was 10 (31.2%), and the mean (IQR) pain intensity was 2.2 (1–3). After 1, 2, and 3 months of treatment, MMD was 33.3%, 34.4%, and 5.2% (22.3% for all 3 months), and the pain intensity was 2.5 (1–2), 2.8 (2–4), and 1.6 (1–2), respectively. (B) Sleep—throughout the study, this patient scored his difficulty with sleep at 3, 2, 1, or 0, but not 4. Scores of 3 were reported at 0% of the nights at baseline versus 18.5%, 13.7%, and 0% of the nights during the first, second, and third months of treatment. Scores of 2 were reported at 25% of the nights at baseline versus 14.8%, 20.6%, and 34.2% of the nights during the first, second, and third months of treatment. Scores of 1 were reported at 62.5% of the nights at baseline versus 51.8%, 44.8%, and 34.2% of the nights during the first, second, and third months of treatment. Scores of 0 were reported at 12.5% of the nights at baseline versus 14.8%, 20.6%, and 60.5% of the nights during the first, second, and third months of treatment. (A) Case 1, (B) Case 2, (C) Case 3, (D) Case 4, (E) Case 5, and (F) Case 6.
Case 2: Immediate improvement in migraine and sleep
Clinical background: A 35-year-old right-handed female (body mass index [BMI] 20.8) with a history of headaches since the age of 14 years and difficulty with sleep. Her headache fulfilled the ICHD-3 criteria for migraine with and without aura, and she was diagnosed with CM (25 MMD). A family history of migraine has been reported in first-degree relatives. Past medical history included back and neck pain. No previous trauma was reported. The headaches were described as bilateral, frontal, temporal, occipital, throbbing, achy, dull, or shooting. If untreated, the attacks lasted for 4–24 hours at an intensity of 4/10 (0–10 VAS). The MIDAS score was 18. The associated symptoms included nausea, vomiting, photophobia, phonophobia, osmophobia, lightheadedness, spinning sensation, neck pain, neck stiffness, jaw pain, decreased appetite, nervousness, anxiousness, difficulty concentrating or difficulty with memory, and confusion. Triggers included menses. The prodromal/premonitory symptoms include tiredness, neck stiffness, dizziness, sensitivity to light, nausea, poor appetite, and irritability. The past pharmacological treatments included ibuprofen, aspirin, butalbital/acetaminophen/caffeine, sumatriptan, promethazine, prednisone, eletriptan, acetaminophen, and topiramate. Acute treatment included ibuprofen and acetaminophen as needed or aspirin-acetaminophen-caffeine (Excedrin Migraine®) 250 mg–250 mg–65 mg 10–15 days per month. The treatment outcomes are shown in Figure 2(B).
Case 3: Improvement in sleep but not migraine
Clinical background: A 26-year-old right-handed female with a history of headache since the age of 9 years and difficulty sleeping. Her headache fulfilled the ICHD-3 criteria for migraine with and without aura, and she was diagnosed with CM (29 MMD). A family history of migraine has been reported in first-degree relatives. Headaches were described as bilateral, frontal, temporal, sometimes occipital, pressing, tight, achy, or dull. They lasted for up to 24 hours at an intensity of 3/10 (0–10 VAS). The MIDAS score was 15. The associated symptoms include photophobia, irritability, and difficulty with concentration. Triggers included menses, physical activity, working with computers, weather changes, smoking, and lack of sleep. Prodromal symptoms included tiredness, irritability, and difficulty concentrating. Past medical history included anxiety, constipation, and hay fever. Past pharmacological treatments included ibuprofen, amitriptyline, sumatriptan, and naratriptan. The acute treatment included naproxen (220 mg, 1–2 capsules). Acute treatment included naproxen, magnesium, coenzyme Q, and riboflavin. Treatment outcomes are shown in Figure 2(C).
Case 4: Immediate improvement in migraine and sleep
Clinical background: A 47-year-old right-handed female (BMI 28.3) with a history of headaches since the age of 9 years and difficulty sleeping. Her headache fulfilled the ICHD-3 criteria for migraine with and without aura, and she was diagnosed with CM (21 MMD). A family history of migraine has been reported in first-degree relatives. The headaches were described as bilateral or unilateral, but with alternating sides, frontal, temporal, occipital, throbbing, pulsating, pressing, sharp, and shooting. If untreated, the attacks lasted for > 4 h at an intensity of 4/10 (0–10 VAS). The MIDAS score was 30. The associated symptoms include photophobia, phonophobia, osmophobia, nasal congestion, spinning sensation, ringing in the ears, neck pain, neck stiffness, jaw pain, decreased appetite, nervousness, anxiousness, difficulty with concentration, and increased sensitivity to touch on the head during headaches. Triggers included menses, physical activity, car riding, airplane riding, weather changes, and sunlight. Prodromal symptoms included neck stiffness, sensitivity to sound, sensitivity to light, sensitivity to odor, nausea, feeling sad, irritability, difficulty concentrating, and restlessness. Past medical history included neck pain, vulvar cancer, and hay fever. The past pharmacological treatments included ibuprofen, codeine, sumatriptan, acetaminophen/aspirin/caffeine, acetaminophen/pamabrom/pyrilamine maleate, and ketorolac. Acute treatment included sumatriptan 25 mg and naproxen 500 mg, as needed. The preventive treatment included amitriptyline (20 mg). Treatment outcomes are shown in Figure 2(D).
Case 5: Delayed improvement in sleep and migraine
Clinical background: A 47-year-old woman with a history of headaches since the age of 13 years was diagnosed with migraine and difficulty falling asleep. Her headache fulfilled the ICHD-3 criteria for migraine without aura, and she was diagnosed with CM migraine (20 MMD). A family history of migraine has been reported in first-degree relatives. Headaches were described as bilateral, unilateral, or alternating sides, frontal, temporal, throbbing, pulsating, or shooting. If untreated, the attacks lasted for up to 24 hours at an intensity of 5/10 (0–10 VAS). The MIDAS score was 4. The associated symptoms included nausea, photophobia, phonophobia, abdominal pain, decreased appetite, irritability, and difficulty concentrating. Triggers included menses, sexual activity, hunger, odors, and alcohol. No premonitory symptoms have been reported. Past medical history included anxiety, depression, Raynaud's phenomenon, asthma, and premenstrual syndrome. Past pharmacological treatments include ibuprofen, naproxen, Excedrin®, sumatriptan, erenumab, and acetaminophen. The acute treatment included ibuprofen. Treatment outcomes are shown in Figure 2(E).
Case 6: Delayed improvement in migraine and sleep
Clinical background: A 70-year-old left-handed female with a history of headaches since the age of 14 years and difficulty sleeping. Her headache fulfilled the ICHD-3 criteria for migraine with and without aura, and she was diagnosed with HFEM (10 MMD). A family history of migraine has been reported in first-degree relatives. Headaches were described as holocephalic, frontal, temporal, occipital, throbbing, pulsating, pressing, and achy. If untreated, the attacks lasted for 24 hours at an intensity of 8–9/10 (0–10 VAS). The MIDAS score was 18. The associated symptoms included nausea, vomiting, photophobia, nasal congestion, lightheadedness, spinning sensation, diarrhea, decreased appetite, concentration difficulties, memory disturbances, and confusion. Prodromal symptoms include tiredness, dizziness, pale face, nausea, weakness, and difficulty concentrating. Triggers included car riding and weather changes. Past medical history included anxiety, gastroesophageal reflux, gastric ulcer, sensorineural hearing loss, anemia, hypothyroidism, basal cell carcinoma, and head trauma. Past pharmacological treatments include aspirin, butalbital, and caffeine. Acute treatment included acetaminophen 1000 mg as needed. Treatment outcomes are shown in Figure 2(F).
Discussion
Using daily headache and sleep e-diaries to document the effects of fremanezumab therapy on sleep latency and the frequency and severity of migraine, we described in this case series four patients whose sleep and migraine improved during the same treatment cycle, one patient whose sleep and migraine did not improve, and one patient whose sleep improved despite no change in migraine. For the five cases in which sleep and migraine improved or did not improve together, it is reasonable to propose that neural circuits involved in migraine and the regulation of sleep may interact such that a reduction in migraine load can improve sleep, and improved sleep can alter the excitability of neurons involved in the chronification of the migraine headache phase. For one participant in whom fremanezumab improved sleep without reducing migraine frequency or intensity, it is reasonable to consider the possibility that, in this participant, fremanezumab may have crossed the blood-brain barrier and affected hypothalamic neurons that regulate sleep onset. A limited number of previous studies have investigated the relationship between CGRP mAbs and sleep.
In agreement with our findings, two studies reported that sleep quality did not change during the treatment period with erenumab and galcanezumab, and one study reported that treatment with galcanezumab, erenumab, or fremanezumab improved sleep and migraine severity and MIDAS scores.20–22 These findings suggest that the overall impact of CGRP mAbs on sleep quality remains inconclusive.
Although the mechanism by which CGRP mAbs, such as fremanezumab, reduce migraine frequency has been extensively investigated by many researchers, the mechanisms by which this class of drugs affects sleep have not been fully studied. Following the principles of neural science and preclinical studies, it is reasonable to assume that the ability of fremanezumab to reduce migraine frequency is mediated by its inhibitory effects on trigeminovascular neurons that convey nociceptive information from the dura to the cortex.26–28 Following similar principles, it is reasonable to assume that improvements in sleep depend on the medication's ability to reduce activity in neurons that promote wakefulness and/or enhance activity in neurons that promote sleep. The network of neurons promoting wakefulness, commonly termed the ascending arousal system, consists of cholinergic and monoaminergic neurons in the brainstem and hypothalamic nuclei such as the pedunculopontine and laterodorsal tegmental nuclei, dorsal and median raphe, locus coeruleus, ventral periaqueductal gray, lateral parabrachial, and tuberomammillary nuclei.8–10,12,29 These cholinergic and monoaminergic neurons are regulated by wake-promoting orexinergic neurons in the lateral hypothalamus and cholinergic neurons in the horizontal limb of the diagonal band nucleus, as well as by sleep-promoting melanin-concentrating hormone neurons in the hypothalamus and neurons containing GABA and galanin in the ventrolateral preoptic area. Particularly relevant to fremanezumab, a drug that selectively targets CGRP, is the current understanding of the role played by external lateral parabrachial neurons containing CGRP (PBel-CGRP) and glutamate, which project to the substantia innominata and nucleus basalis in the basal forebrain, central nucleus of the amygdala, and lateral hypothalamus in arousal. 29 Given that this is the only component of the complex network that regulates sleep and involves CGRP, it seems reasonable to propose that if fremanezumab is capable of crossing the blood-brain barrier and blocking CGRP in the basal forebrain, amygdala, and lateral hypothalamus, it could promote sleep by reducing arousal. However, these two datasets challenged this possibility. First, all PBel neurons containing CGRP also contain glutamate, and their arousal-mediating effects in the basal forebrain, amygdala, and hypothalamus are blocked solely by the elimination of glutamate receptors. 30 The second challenge is the absence of evidence for fremanezumab's ability to cross the blood-brain barrier and reach neurons in any of these areas.31,32 Another intriguing pathway by which fremanezumab may improve sleep is the inhibition of histaminergic neurons in the ventral tuberomammillary nucleus. This possibility is based on several factors: (a) the location of these neurons along the ventral surface of the hypothalamus, which is closer to the cerebrospinal fluid (CSF) where the blood-brain barrier is most compromised; (b) the presence of fremanezumab in the CSF; and (c) the well-known role of histamine receptor blockers, such as diphenhydramine, in promoting sleep.33–36 To further explore this theoretical option, it is necessary to demonstrate CGRP's ability to activate histaminergic neurons and the potential of CGRP mAbs to reduce their activity.
Our study has limitations, particularly the small sample size of only six patients and the absence of a placebo-controlled group, which raises concerns about the potential influence of placebo effects on the observed improvements in sleep and migraine frequency. It is also possible that fremanezumab may improve sleep indirectly by reducing sleep-disrupting factors, including a decrease in headache frequency and intensity.
In summary, the findings of the current case series suggest that improvement in sleep in migraine patients treated with fremanezumab is tightly correlated with headache improvement. However, in rare cases, it may be independent in patients with significantly compromised hypothalamic blood-brain barrier, but this needs to be confirmed in preclinical and clinical studies. If future studies confirm that fremanezumab can promote sleep and thereby reduce the excitability of trigeminovascular neurons and the overall activity along this pathway, our understanding of the mechanisms by which CGRP mAbs prevent migraines will need to be redefined. The study also suggests that prophylactic treatments, particularly anti-CGRP mAbs, not only reduce migraine frequency but also potentially improve sleep quality. These findings highlight the importance of considering sleep comorbidity when selecting preventive therapies, guiding clinicians toward more personalized and effective treatment approaches.
Clinical implications
Fremanezumab primarily enhances sleep by reducing the frequency and severity of headaches. Fremanezumab potential acts directly on sleep-promoting neurons in the hypothalamus. In individuals with migraine and a hypothalamic blood-brain barrier, sleep improvement with fremanezumab may be independent of headache relief.
Footnotes
Acknowledgements
Mario Ortega and Verena Ramirez Campos provided a medical accuracy review at the request of the authors.
Authors’ contributions
1RB, SA, and AMC contributed substantially to the conception and design of the study. RB, AMC, SA, and JG contributed to data acquisition and analysis. All authors (AMC, JG, AM, RB, and SA) contributed to the interpretation of data and the drafting and revision of the manuscript. SA and RB were contributing equally. All authors have read and approved the final manuscript.
Availability of data and materials
The datasets generated and/or analyzed during this study are not publicly available but may be obtained from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Rami Burstein: Research support from the NIH, AbbVie, Modulight, Eli Lilly, and Teva. The reviewer for NINDS holds the stock options in AllayLamp and Percep. Serves as consultants, advisory board members, or has received honoraria from Allergan, Amgen, Biohaven, Brise Mutual, CGRP diagnostic, Dr Reddy's Laboratory, ElectroCore, Eli Lilly, Escient, Johnson & Johnson, Merck, NeuroRays, Pernix, Theranica, Teva, and Ventus. CME fees from Healthlogix, Medlogix, WebMD/Medscape, and Patents 9061025, 11732265.1, 10806890, US2021-0015908, WO21007165, US2021-0128724, and WO21005497. Sait Ashina received honoraria for consulting and teaching from Allergan/AbbVie, Eli Lilly, Impel NeuroPharma, Linpharma, Lundbeck, Pfizer, Satsuma, Teva, and Theranica. All other authors declare no competing interests.
Ethical approval and consent to participate
The study was approved by the Institutional Review Board of the Beth Israel Deaconess Medical Center and was conducted in accordance with the Good Clinical Practice guidelines and the principles of the Declaration of Helsinki. All participants provided written informed consent before enrollment in the study.
Consent for publication
Not applicable.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Teva Pharmaceutical Industries Ltd, Petach Tikva, Israel.
