Abstract
Cyclic vomiting syndrome (CVS) is characterized by episodic vomiting and shares similarities with migraine headache. Guidelines recommend using triptans as abortive therapy in CVS, but there are limited data on its efficacy.
Introduction
Cyclic vomiting syndrome (CVS) is a chronic disorder of gut brain interaction, characterized by episodes of severe nausea, vomiting, and often abdominal pain, and is defined by Rome criteria. 1,2 The prevalence of CVS in adults is 2%, based on a recent population-based study. 3 The pathogenesis of CVS remains unclear, but the close relationship between migraine and CVS suggests that they share a common pathophysiology. 4,5 Clinically, CVS and migraine have similarities: both are episodic in nature, are triggered by stress, and have a prodromal phase when patients can use medications to try and abort an episode. 4 Symptoms such as nausea, vomiting, abdominal pain, headache, and photophobia can be seen in both disorders. 5
Due to the similarities between these two disorders, the management of CVS overlaps with migraine. Prophylactic therapies including antidepressants and anticonvulsants have been shown to be effective in preventing migraines and CVS attacks. 6 –8 Options for abortive therapy for CVS attacks include medications used for acute migraines. Sumatriptan, a medication commonly used to abort migraine attacks, has been found to be effective for acute CVS episodes. 4,9,10 It is a serotonin agonist and binds to the 5-HT1B and 5-HT1D receptor subclasses in the meninges and modulates neuropathic pain sensation. 11 However, data using sumatriptan in CVS are limited to small studies in children and some case reports in children and adults. 9,10,12 Given this paucity of data as well as recent recommendations for the use of sumatriptan for aborting CVS episodes in the management of CVS attacks, we sought to characterize sumatriptan use and investigated differences between responders and nonresponders.
Methods
A cross-sectional study of patients with CVS using Rome criteria diagnosed at a single tertiary referral center was performed between November of 2018 and April of 2019, after approval by the Institutional Review Board at the Medical College of Wisconsin. Only patients who had an episode of CVS within the last 6 months were included to minimize recall bias. A detailed questionnaire was administered to patients who consented to participate and only those who had an attack of CVS within the past 6 months prior to the study were included. This questionnaire included specific questions about symptoms that patients experienced during the most recent episode such as nausea, vomiting, abdominal pain, and other symptoms (including diarrhea, headache, light or sound sensitivity, excessive sweating, hot/cold flashes). Each participant was asked about the effects of sumatriptan on individual symptoms within 2 h after administration (defined as the response at baseline) as well at 24 h. Finally, participants were asked about health-care utilization (emergency department (ED) visits and hospital admissions) and the use of other abortive therapies. Other therapies used in aborting episodes of CVS included ondansetron, promethazine, diphenhydramine, benzodiazepines, aprepitant, and opioids were also recorded. Current use of cannabis, defined as any use within the past 6 months was ascertained. Questionnaires were distributed to patients via e-mail through a patient registry stored in
Statistical analysis
The overall sample and those indicating use of sumatriptan were characterized using descriptive statistics in the sample of 101 individuals indicating a CVS attack over the past 6 months. Within the 39 individuals that indicated use of sumatriptan, responses were compared for responders vs. nonresponders. Response to therapy was defined as ≥50% improvement in symptoms including response to nausea, vomiting, abdominal pain, and other symptoms (diarrhea, headache, light or sound sensitivity, excessive sweating, hot/cold flashes). For vomiting, this was defined as >50% reduction in episodes of vomiting. Responses for nausea, abdominal pain, and other symptoms were a subjective global response as per patient report. Response was considered within 2 h and at 24 h after the first sumatriptan dose. Categorical variables were compared using χ 2 test and continuous compared using the Student’s t test. Statistical analysis was performed using SAS 9.4 software (SAS Institution). A p value <0.05 was considered significant.
Results
Demographics and clinical characteristics
Of 115 participants who consented, 101 (88%) reported having at least one CVS attack over the past 6 months and were included in the analysis. Mean age was 41 ± 15 years, 75 (74%) were female, and 84 (83%) were Caucasian. Mean duration of the most recent CVS attack was 2.2 ± 3.6 days. Many participants had comorbid conditions: 62 (61%) had anxiety, 51 (50%) had depression, and 45 (45%) had a personal history of migraines. Of 52 patients who were prescribed sumatriptan, 13 (25%) patients did not take it as directed. Of the 11 patients who responded to the question about why they did not use sumatriptan,4 (7.6%) patients did not have the medicine with them, 1 patient did not like the side effects, and 6 (11.5%) felt it did not work for them in the past. Thirty-nine (37%) participants reported using sumatriptan during a CVS attack in the past 6 months. The demographics and clinical characteristics of patients with CVS are presented in Table 1.
Demographics, clinical course, and medication use of patients with CVS based on triptan use.

CVS: cyclic vomiting syndrome; ED: emergency department.
a Other abortive medications include tizanidine, compazine, and caffeine.
b Other prophylactic medications include mirtazapine, gabapentin, and valproic acid.
Use of sumatriptan
The average time to taking the first dose of sumatriptan was 3.68 ± 8.45 h. The intranasal route was most commonly used (33.84%), followed by the oral (3.8%), and subcutaneous routes (3.8%). A dose of 20 mg was used in 92% of patients who used intranasal sumatriptan. Eleven (28%) of the sumatriptan users reported using a second dose. Of the 39 sumatriptan users, 20 (51%) were completely disabled from their symptoms during an episode, 16 (41%) were moderately impaired and could perform some activities, while 3 (7.69%) were mildly impaired and could perform most of their usual activities.
Response to sumatriptan use
The response rates to sumatriptan within 2 h and at 24 h are shown in Figure 1. The majority of patients noted a response to sumatriptan with reduction in nausea (19/32, 59%), vomiting (16/29, 55%), and other symptoms (19/36, 53%) within 2 h. The response to abdominal pain was less robust with 13/30 (43%) noting a response. This response rate continued to improve over the course of 24 h with improvement in nausea (22/33, 67%), vomiting (22/30, 73%), abdominal pain (19/30, 63%), and other symptoms (26/36, 72%). The time to feeling the first effects of sumatriptan and its full beneficial effects are shown in Figure 2. Despite using sumatriptan and other rescue medications, 11(28%) were seen in the ED and treated for symptoms, while 4 (10%) were admitted to the hospital for management of their CVS flare. Of those who responded to questions about ED visits and hospitalizations, 18 of 28 (64%) reported that using sumatriptan helped them avoid ED visits and 21 of 35 (60%) reported that it helped avoid being hospitalized for CVS symptoms. There were 17 patients who responded to questions about both ED visits and hospitalizations.

Response to sumatriptan at baseline* and at 24 h after use *baseline was defined as within 2 h after sumatriptan dose. **Response to sumatriptan was defined as ≥50% improvement in symptoms. ***Other symptoms include diarrhea, excessive sweating, headache, light or sound sensitivity, hot or cold flashes, fatigue, and muscle aches.
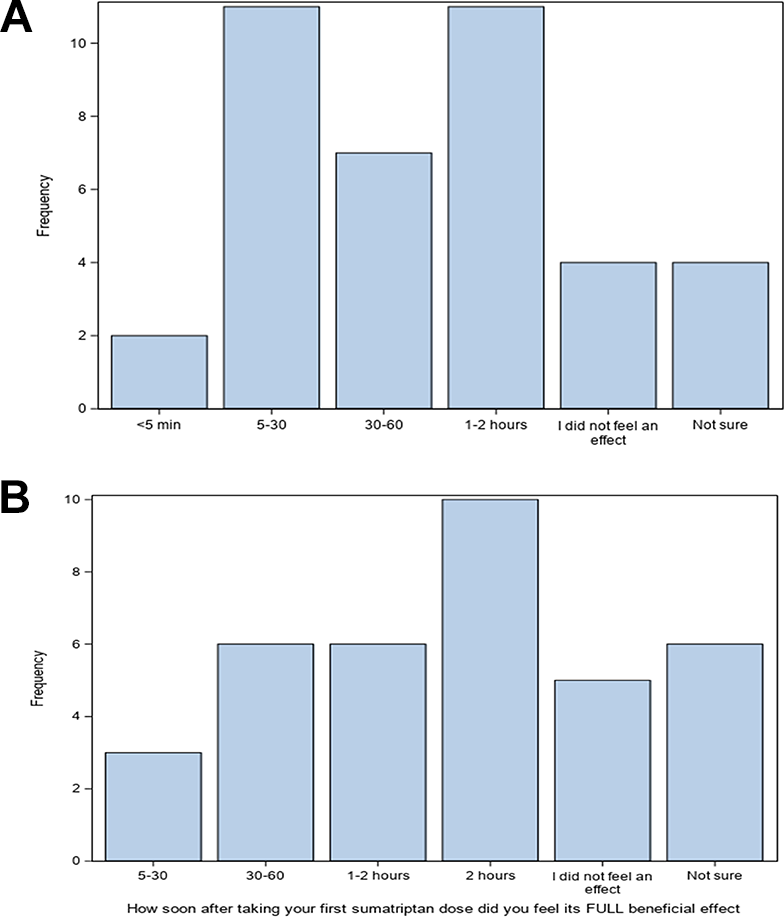
Time to relief of symptoms noted by patients when asked, (a) “How soon after the first sumatriptan does did you start to feel an improvement? (b) “How soon after the first sumatriptan does did you feel its FULL beneficial effect?
Differences between responders and nonresponders to sumatriptan
The clinical characteristics of responders and nonresponders at baseline (2 h after first sumatriptan dose) and at 24 h after first dose are shown in Tables 2 and 3, respectively. Triptan users who did not experience an improvement in vomiting within 2 h were more likely to suffer from depression (82% vs. 33%, p = 0.019) and were more likely to report using marijuana to alleviate symptoms (50% vs. 13%, p = 0.025). At 24 h, marijuana use was higher in those who did not feel an improvement in nausea (60% vs. 18%, p = 0.024) and those who did not experience significant relief from other symptoms (63% vs. 18%, p = 0.019). Benzodiazepine use was higher among triptan users who did not experience an improvement in vomiting (63% vs. 23%, p = 0.041), and opioid use was higher among those who continued to have significant abdominal pain (36% vs. 5%, p = 0.028) at 24 h.
Differences between responders and nonresponders to triptans at baseline.

aSignificant with p < 0.05.
Differences between responders and nonresponders to triptans at 24 hours.
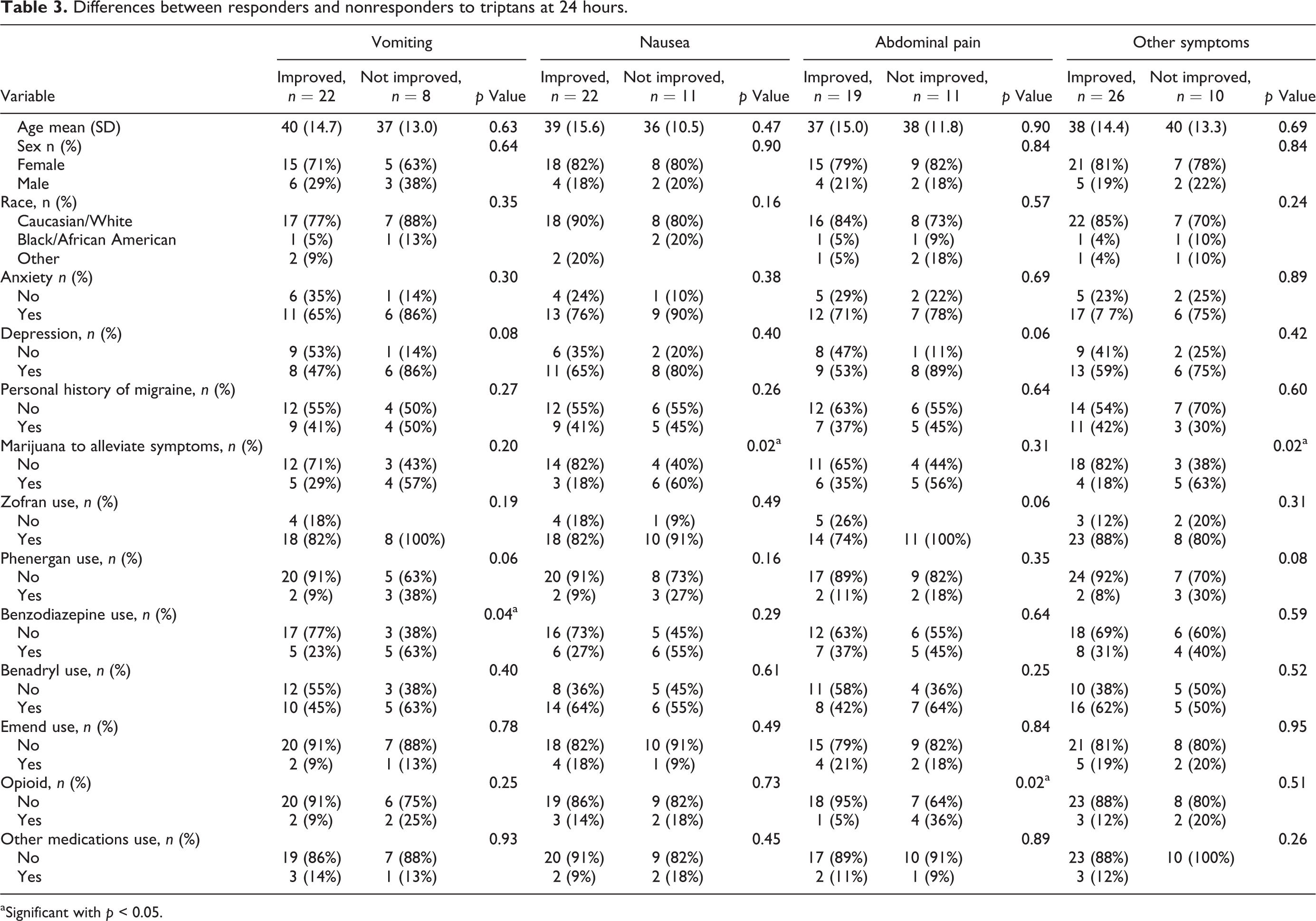
aSignificant with p < 0.05.
Adverse effects due to the use of triptans
Twenty-four (62%) of sumatriptan users reported at least one adverse effect after sumatriptan use. The most common adverse effect reported was fatigue, in 16 (41%) of sumatriptan users. Other commonly reported side effects included flushing/warmth in nine (23%), weakness in nine (23%), and dizziness in six (15%). Of all triptan users, 32 (92%) stated that they would use sumatriptan again for their next CVS attack.
Discussion
This cross-sectional study provides us with valuable information about the response to sumatriptan in adults with CVS. Sumatriptan was effective in reducing nausea, vomiting, and other associated symptoms of CVS in about half of the patients (43–59%) within 2 h of administration. Sumatriptan was less effective in alleviating abdominal pain, with only 43% noting a response. Patients continued to improve over the course of the day, with further reduction in individual symptoms (67–73%). Of note, approximately two-thirds of the patients reported that it helped them avoid both ED visits and hospitalizations. However, 17 patients responded to questions about both ED visits and hospitalizations; it would be impossible to clarify this as this was a subjective response based on patient perception about their CVS disease experience. Nevertheless, many patients report long wait times and substandard care in the ED and hospital, likely due to a lack of knowledge about CVS among the medical community. 13 The ability to avert ED visits and hospitalizations itself can empower patients and provides them with a degree of control over their disease course. This can also reduce the frustration and health-care costs that these patients undergo and improve overall well-being.
Sumatriptan was well tolerated overall and 92% of users reported that they planned to use sumatriptan again for their next CVS attack. Other notable findings in our study were that nonresponse to treatment with sumatriptan within 2 h was associated with psychological comorbidity such as depression and use of cannabis. Factors associated with a nonresponse at 24 h included use of cannabis, opioids, and benzodiazepines for relief of symptoms. Contrary to previous data, there were no differences in the presence of migraine headache between responders and nonresponders.
This is the first study to our knowledge to determine the efficacy of triptans in adults with CVS and offers several insights into its use. The response rates in our study were modest but were comparable to the smaller study by Hikita et al. which showed a 54% response rate. 9 However, the outcomes and the methodology in the Hikita study were different from our study. The primary outcome in the Hikita study was the reduction of vomiting by >50%; our study by contrast assessed response to individual symptoms including nausea, vomiting, abdominal pain, and other associated symptoms. While vomiting is a key feature of CVS, other symptoms such as nausea and particularly abdominal pain can be quite debilitating in adults, and it would be important to understand how sumatriptan affects these symptoms. Also, the investigators in the study by Hikita et al. analyzed a total of 35 attacks in 11 patients while we analyzed the response to sumatriptan during a CVS attack experienced within the prior 6-month period. The response rate to sumatriptan continued to improve at 24 h but this may have been due to the additional effects of other rescue medications which may have augmented the overall response. Also, it appears that contrary to advice about the early use of triptans, the mean time to using sumatriptan was 3.68 h which could have mitigated its effects. These findings suggest that further efforts to educate patients on the proper use and timing of sumatriptan are important. While we did not explore reasons for this delay in using sumatriptan as prescribed in this study, reiterating recommendations and further patient education may help maximize its efficacy.
Depression and cannabis use were associated with a nonresponse to triptans within 2 h of administration and highlights the potential role that psychological comorbidity plays in the propagation of symptoms in CVS. 14,15 It is well known that cannabis use is associated with mood disorders, and patients with CVS often use cannabis to improve mood symptoms. 16,17 These findings underscore the importance of addressing psychological comorbidity which can help alleviate much of the anxiety and panic which is associated with an acute episode of CVS which in turn can improve outcomes.
Our findings also reinforce recommendations that a combination of rescue medications is more likely to be effective in aborting symptoms rather than a single drug as these can treat multiple aspects of a CVS attack. It is common practice to use triptans in combination with antiemetics and sedatives which not only treat nausea and vomiting but also alleviate the stress and anxiety that often accompanies a CVS episode. 6 It is important to note that benzodiazepines are not recommended for daily use and must be used sparingly and cautiously. The findings from this study would support such an approach. Further, it is important to counsel patients about behavioral techniques and working with a psychologist to address stress which can both precipitate and propagate CVS attacks.
Other findings that the use of cannabis, opioids, and benzodiazepines were associated with a nonresponse at 24 h need to be interpreted with caution. Rather than being a cause for the lack of response, it is possible that these patients were more likely to resort to other therapies for symptom relief when standard medications like ondansetron and triptans failed. The use of cannabis in CVS is quite common and given its antiemetic effects and purported health benefits, it is not surprising that several patients used this for aborting symptoms.
Other findings in our study were that sumatriptan nasal spray was well tolerated by most patients and most stated that they would use it for future CVS attacks. Due to uncertain oral absorption during an attack of CVS, either the nasal or subcutaneous route is usually recommended: The route of administration was intranasal in 85% of patients in this study. While patients may be averse to a nasal spray, ensuring that they follow the proper technique and flexing the neck while self-administering will prevent dripping down the back of the nasopharynx and avoid a bitter taste. 18
There are limitations to this study inherent to all retrospective studies including recall bias. However, we tried to minimize this by studying the most recent CVS attack within 6 months prior to the study, which would have mitigated some of these issues. Another weakness of this study was that our questionnaire did not ask patients how they self-administered nasal sumatriptan. It is well established that using a “face forward” technique will allow the drug to be delivered to the anterior nares where the receptors for the drug are located. 18 Recent guidelines on the management of CVS also reiterate using this technique. 6 The PI routinely instructs patients to use this technique, but it is unclear if subjects complied.
Other limitations of our study include the use of other rescue medications such as ondansetron and benzodiazepines in aborting symptoms. It is difficult to ascertain the independent effects of sumatriptan and this would require future prospective studies. However, conducting such studies can be challenging as withholding rescue medications would likely be unethical and patients may be reluctant to avoid using other rescue medications in the throes of an acute episode. For the same reason, a placebo-controlled trial would be both unethical and difficult to conduct. However, studies comparing newer treatments with standard treatment may address this concern. Other limitations include the small sample size and tertiary referral bias with sicker patients. Again, future prospective trials with a larger cohort and inclusion of patients from the community are required to confirm these findings.
Conclusions
Sumatriptan is effective in controlling symptoms of nausea, vomiting, abdominal pain, and other associated symptoms within 2 h of administration during a CVS attack. The response rate continues to improve over the course of 24 h which may be due to the use of other rescue medications. Psychological factors such as depression and use of cannabis are associated with a nonresponse and behavioral strategies aimed at reducing anxiety and stress before and during an attack of CVS can be helpful in improving outcomes. Prospective studies are needed to better define its efficacy and understand whether its use can reduce health-care utilization.
Clinical implications
Guidelines recommend using triptans as abortive therapy in CVS, but there are limited data on its efficacy in adults.
Sumatriptan was effective in reducing symptoms of nausea, vomiting, abdominal pain, and other symptoms in CVS in most patients.
Nonresponse to sumatriptan was associated with depression, cannabis use, and the use of opioids and benzodiazepines during an episode.
Footnotes
Acknowledgement
The authors would like to gratefully acknowledge Mark Oium, who assisted with the development of questionnaires in REDCap.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TV is a consultant for Takeda Pharmaceuticals and has received funding for a study from Alnylam Pharmaceuticals. These do not represent a conflict of interest. The other authors do not have any relevant disclosures.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
