Abstract
The aim of this article is to provide neuroimaging data on cases with familial hemiplegic migraine (FHM). A 14-year-old male presented normal diffusion-weighted magnetic resonance imaging (DWI) and fluid-attenuated inversion recovery (FLAIR) findings during his first hemiplegic migraine attack. However, magnetic resonance angiography (MRA) showed diffuse narrowing of the right middle cerebral artery. Cerebral blood flow–single-photon emission computed tomography showed right hypoperfusion. A follow-up study showed no abnormal findings. His mother had normal DWI, FLAIR, and MRA findings during her migraine attack. Both patients were diagnosed with FHM2 (p.R763H mutation in ATP1A2). This study highlights the importance of neurovascular examinations during the FHM2 headache phase. Further cases are required to clarify the pathophysiology of migraine.
Introduction
Hemiplegic migraine (HM) aura is defined as unilateral, fully reversible, visual, sensory, motor, or other central nervous symptoms. Brainstem symptoms, disturbance of consciousness, confusion, fever, and cerebrospinal fluid pleocytosis can also occur. 1 Mutations in three genes have been linked to familial HM (FHM): 1 CACNA1A, ATP1A2, and SCN1A, causing FHM1, FHM2, and FHM3, respectively. Since these genes control ion channel functions, the mutations cause neuronal hyperexcitability, leading to cortical spreading depression (CSD). Approximately 70 ATP1A2 mutations have been identified, 2 but the clinical features and physiological mechanism underlying aura symptoms are understood poorly. Further, there is limited neuroimaging evidence of genetically identified FHM attacks. Here, we report neuroimaging findings during an attack in an FHM2 case with a known ATP1A2 mutation (p.R763H).
Methods
Case reports
Patient 1, a 14-year-old male, developed sudden left-sided hemiplegia and sensory disturbance several hours before admission. He then developed nausea, vomiting, and visual disturbance of his left side. Hemiplegia and sensory disturbance improved after ∼10 min, but he gradually developed a right-sided occipital headache. Neurological evaluation on admission revealed mild left-sided hemiplegia, sensory disturbance, and increased deep tendon reflex in the left arm and leg but no pathologic reflexes or cerebellar ataxia. He was unable to walk due to headache and nausea. All clinical symptoms resolved within 1 day. This was his first and only HM attack, but he had experienced occasional migraines without apparent aura. Therefore, patient 1 did not fully meet the International Classification of Headache Disorders (ICHD)-3 criteria for HM. His mother (patient 2) had been admitted with similar symptoms.
Patient 2 had no major past history but had occasionally experienced migraine with aura from a young age, accompanied by sudden syncope, transient dysarthria, and vomiting. She experienced her first HM attack at 39 years. She presented with sudden headache, dysarthria, aphasia, and left-sided hemiplegia but was misdiagnosed with hysteria. All clinical symptoms resolved within 1 day. Patient 2 fulfilled the ICHD-3 criteria for HM.
Neuroimaging
For patient 1, brain magnetic resonance imaging (MRI) was performed at 3 h after symptom onset, when hemiplegia and headache were present, using a 1.5-T scanner. Standard acute imaging for suspected stroke was performed using the following sequences: fluid-attenuated inversion recovery (FLAIR), diffusion-weighted MRI (DWI), apparent diffusion coefficient, and three-dimensional time-of-flight magnetic resonance angiography (MRA). Follow-up MRI was obtained at 24 h after onset, when hemiplegia had resolved, using a 3-T scanner.
Cerebral blood flow (CBF)–single-photon emission computed tomography (SPECT) was performed with 99mTc-
For patient 2, MRI was performed at about 2 h after onset when hemiplegia and headache were present, using a 1.5-T scanner with the same sequences as for patient 1. Follow-up MRI after hemiplegia resolution was not obtained.
Genetic analysis
Mutational analysis of genomic DNA for CACNA1A, ATP1A2, and SCN1A was performed using direct nucleotide sequencing with specific primers. 2 Written informed consent was obtained from both patients.
Results
Genetic analysis
Both patients had a heterozygous p.R763H (c.2288G>A) mutation in ATP1A2. 3
Neuroimaging
For patient 1, MRI revealed normal DWI and FLAIR findings, but MRA showed diffuse narrowing of the right middle cerebral artery (Figure 1). CBF-SPECT revealed a large area of hypoperfusion in the right cerebral hemisphere, which did not respect anatomical vascular boundaries. CBF-SPECT also revealed crossed cerebellar diaschisis (CCD) in the left cerebellar region. Follow-up MRI and CBF-SPECT showed complete resolution.
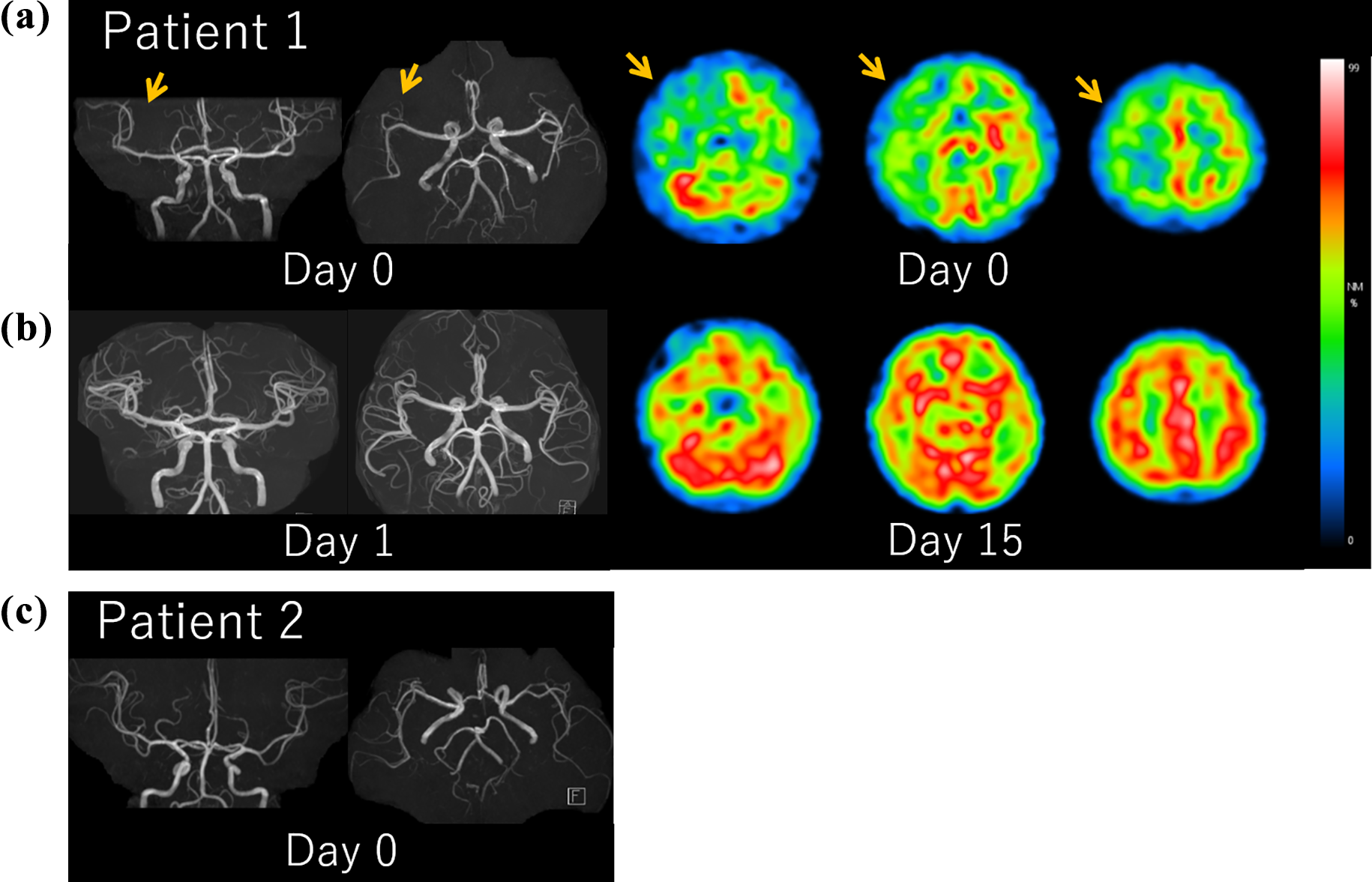
Time-of-flight MRA and 99mTc-
For patient 2, MRI revealed normal DWI, FLAIR, and MRA findings. CBF-SPECT was not conducted.
Discussion
Here, we reported neurovascular changes in an FHM2 family. CBF-SPECT played an important diagnostic role, as the CBF changes in patient 1 were suggestive of HM, while he did not fully meet the ICHD-3 criteria for HM. Conversely, patient 2 was misdiagnosed as hysteria without CBF-SPECT, although she presented with typical HM symptoms and fulfilled the ICHD-3 criteria for HM. Another diagnostic clue in patient 1 was that the aura symptoms occurred gradually and in succession, suggesting a relationship with the propagation of CSD. Patient 1 demonstrated CCD, which is another unique aspect of our case; CCD has been reported frequently in acute stroke but rarely in migraine. 4
Two FHM2 patients in the same family with the p.R763H gene mutation showed similar clinical characteristics. 3 They presented with unilateral headache and contralateral hemiplegia, which persisted for 1 day and a few hours, respectively. Although their characteristics were similar to those of our cases, MRI or CBF-SPECT findings were not presented.
CBF changes during migraine have been well studied. The first observable event in an attack of migraine with aura is a posterior decrease in regional CBF in one hemisphere, followed by headache onset. 5 Regional CBF then gradually changes from abnormally low to abnormally high without an apparent change in headache, which are referred to as biphasic CBF changes.
However, the neurovascular changes in genetically determined FHM remained unclear until recently, due to its low prevalence. Associations between FHM and hyperperfusion, 6 normoperfusion, 7 and hypoperfusion 8 have been reported, but the timings of the scans from migraine onset varied. Iizuka et al. 9 assessed neurovascular changes over time in the affected cerebral hemisphere corresponding to aura symptoms during the acute phase of FHM2. They repeatedly measured CBF during each migraine attack in FHM2 patients, confirming the same biphasic CBF pattern as Olesen et al. 5 (Figure 2). The pathological basis of biphasic CBF changes is postulated to start with the diffusion of neuronal and glial cell depolarization in the cerebral cortex during the aura phase, with subsequent inhibition of neural activity. This phenomenon is called CSD. Hypoperfusion may represent the suppression of neural activity and the concomitant decrease of CBF during CSD. Thereafter, CBF increases gradually during the headache phase. Although the mechanism underlying hyperperfusion remains unknown, it is postulated that the activation of the trigeminovascular system causes neurogenic inflammation in the affected hemisphere. 10

This figure shows the biphasic cerebral blood flow change according to each phase of migraine attack proposed by Olesen et al. 5 During the aura phase, hypoperfusion initially occurs representing suppression of neural activity, that is, CSD. During the headache phase, subsequent hyperperfusion is caused by trigeminal nerve activation, which causes neurogenic inflammation and increases vasoactive neuropeptides, such as astrocyte-derived vasodilator, CGRP, and substance P. Vasogenic leakage of blood plasma proteins and blood vessels extension leads to focal hyperperfusion of affected hemisphere. CSD: cortical spreading depression; CGRP: calcitonin gene-related protein; CBF: cerebral blood flow.
In conclusion, our cases demonstrated the value of neuroimaging to identify neurovascular changes during the early headache phase of FHM2. For patient 1, CBF-SPECT was performed during the acute headache phase when left hemiplegia and right hemisphere hypoperfusion were present. Right hemisphere hypoperfusion is thought to correspond to CSD; this finding is consistent with previously reported “biphasic CBF changes.” However, our findings should be interpreted carefully due to the cross-sectional nature of this study. Observing dynamic CBF changes using functional brain imaging during a migraine attack is crucial to clarify the pathophysiology of migraine.
Clinical implications
Our cases demonstrated the value of neuroimaging to identify neurovascular changes during the early headache phase of FHM2. The neuroimaging finding in our cases is consistent with previously reported “biphasic CBF changes” in HM attack. Patient 1 demonstrated CCD, which is another unique aspect of our case; CCD has been reported frequently in acute stroke but rarely in migraine.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
