Abstract
Background:
Migraine therapy with sumatriptan may cause adverse side effects like pain at the injection site, muscle pain, and transient aggravation of headaches. In animal experiments, sumatriptan excited or sensitized slowly conducting meningeal afferents. We hypothesized that sumatriptan may activate transduction channels of the “irritant receptor,” the transient receptor potential ankyrin type (TRPA1) expressed in nociceptive neurons.
Methods:
Calcium microfluorometry was performed in HEK293t cells transfected with human TRPA1 (hTRPA1) or a mutated channel (TRPA1-3C) and in dissociated trigeminal ganglion neurons. Membrane currents were recorded in the whole-cell patch clamp configuration.
Results:
Sumatriptan (10 and 400 µM) evoked calcium transients in hTRPA1-expressing HEK293t cells also activated by the TRPA1 agonist carvacrol (100 µM). In TRPA1-3C-expressing HEK293t cells, sumatriptan had hardly any effect. In rat trigeminal ganglion neurons, sumatriptan, carvacrol, and the transient receptor potential vanillod type 1 agonist capsaicin (1 µM) generated robust calcium signals. All sumatriptan-sensitive neurons (8% of the sample) were also activated by carvacrol (14%) and capsaicin (48%). In HEK293-hTRPA1 cells, sumatriptan (100 µM) evoked outwardly rectifying currents, which were almost completely inhibited by the TRPA1 antagonist HC-030031 (10 µM).
Conclusion:
Sumatriptan activates TRPA1 channels inducing calcium inflow and membrane currents. TRPA1-dependent activation of primary afferents may explain the painful side effects of sumatriptan.
Keywords
Introduction
Sumatriptan is the first clinically used triptan, a selective agonist at 5-HT1B/D receptors, which has beneficial therapeutic effects in migraine and cluster headache. 1 In the beginning, it was only available for subcutaneous injection, but now, it is mostly used as an orally available formula. 2 Nevertheless, the subcutaneous administration route is more effective and still used if a rapid onset is required, as it is usually the case in cluster headache attacks. 3
Adverse side effects of sumatriptan and other triptans include transient pain phenomena like chest pressure and pain, 4,5 muscle pain, jaw and neck pain, 6 and even headache starting some minutes after triptan administration and lasting up to 30 min. 7 Sumatriptan injection can also cause local pain. 8 Particularly when sumatriptan was given in patients suffering from an injury like a fracture or an inflammatory disease like sunburn, aggravation, or activation of pain at the site of the previously or actually injured site was noted. 9 This is reminiscent of a hyperalgesic state, in which nonpainful or weakly painful stimuli cause allodynia or hyperalgesia.
To examine this phenomenon on a preclinical basis, Burstein et al. have recorded from slowly conducting meningeal afferents in the rat and administered 3–300 mg/kg sumatriptan intravenously, which caused a transient increase in spontaneous discharge activity in 10 of 18 units and increased firing to mechanical stimulation of the cranial dura mater in 13 of 18 units with a minimal effective dose of 3 mg/kg. 7 Previously, Strassman and Levy have used this preparation to observe that sumatriptan at local concentrations of 0.24–24 µM induced a dose-dependent increase in activity, which was dependent on extracellular calcium. 10 In trigeminal ganglion cultures, Durham and Russo found that sumatriptan administration caused a slow and sustained increase in intracellular calcium. 11
Sumatriptan effects on a cellular and molecular level have later been examined with patch clamping in dissociated trigeminal neurons retrogradely labeled from rat cranial dura mater. 12 Sumatriptan suppressed voltage-gated Ca2+ currents, but a 2-min sumatriptan application increased dural afferent excitability, while 30 min preincubation with sumatriptan partially prevented the sensitization of afferents by inflammatory mediators. These changes were blocked by preincubation with a 5-HT1D antagonist.
The molecular mechanisms of these complex actions of sumatriptan remained unknown but reminded us to a similar phenomenon depending on the activation of transient receptor potential transduction channels of the ankyrin type (TRPA1), which can be activated by a variety of irritating substances 13 and have also been associated with noxious cold transduction. 14 The molecular structure of sumatriptan indicates that it could be an electrophilic compound, which may be able to activate TRPA1 through covalent modification of cysteine residues located in the N-terminus of the channel. 15
Therefore, we set out to examine whether sumatriptan is able to activate HEK293 cells transfected with human TRPA1 receptor using calcium imaging and patch clamp recordings and compared the response with known TRPA1 receptor agonists. In addition, we used dissociated rat trigeminal ganglion neurons for calcium imaging to test sumatriptan responses. The results strongly suggest that sumatriptan is a TRPA1 agonist, which may explain its acute pronociceptive effects.
Materials and methods
Culturing of trigeminal ganglion neurons and transfection of HEK293 cells
Experiments were done in accordance with the ethical guidelines of the International Association for the Study of Pain and in compliance with the guidelines for the welfare of experimental animals of the Federal Republic of Germany and the European Commission (Directive 2010/63/EU). Wistar rats (both sexes, 3 days postnatal) bred in the animal facility of our institute were killed by cervical dislocation. The skull was divided into halves along the midline, and the trigeminal ganglia (TG) were excised, exposed to collagenase and protease (Sigma-Aldrich, Taufkirchen, Germany) and mechanically dissociated. Cells were plated on poly-
HEK293t cells were subcultured in Dulbecco’s modified Eagle’s medium, supplemented with penicillin and streptomycin, and regularly controlled for the absence of mycoplasma contamination. HEK293t were transiently transfected with hTRPA1 plasmids in a pcDNA3 vector using jetPEI transfection reagent from Polyplus Transfection (Illkirch, France). Cells were replated after 6 h onto poly-
Calcium microfluorimetry
HEK293t expressing the human isoform of TRPA1 (h-TRPA1) or human TRPA1-C621S-C641S-C665 S (TRPA1-3C), a plasmid in which the activation site for covalent agonists is mutated, 16 and TG neurons, all plated on glass coverslips, were loaded with Fura-2 AM (3 µM) supplemented with 0.02% Pluronic (Life Technologies GmbH, Darmstadt, Germany) for 30 min at 37°C, and left to recover for about 10 min in extracellular solution (ES) prior to recording. Coverslips were then mounted on an Olympus IX71 inverted microscope and imaged using a 10× objective. Cells were permanently superfused with ES using a custom-built software-controlled seven-channel gravity-driven common-outlet system. Fura-2 was excited at 340 and 380 nm using a Polychrome V monochromator (Till Photonics, Gräfelfing, Germany), with a full-width half-maximum wavelength range of about 10 nm. Fluorescence emission was long-passed at 495 nm and pairs of images were acquired at a rate of 1 Hz with an exposure time of 4 ms with a 12-bit CCD camera (Imago Sensicam QE, Till Photonics). Data were processed off-line using the TILLvisION 4 (Till Photonics) using self-written macros. The background intensity was subtracted before calculating the ratio between the fluorescence emitted when the dye was excited at 340 and at 380 nm (F340/F380 nm). The time course of this ratio was analyzed for regions of interest adapted to individual cells.
Whole-cell patch clamp recordings
Voltage clamp experiments were performed on HEK293t cells transiently transfected with a hTRPA1::YFP fusion plasmid for selecting successfully transfected cells. 16 Currents were acquired using an Axopatch 200B patch clamp amplifier (Molecular Devices, Sunnyvale, CA, USA), low-passed at 1 kHz (2-kHz sampling rate); pCLAMP 10 was used for voltage control, data acquisition, and off-line analysis. Patch pipettes were pulled from borosilicate glass tubes (TW150F-3, World Precision Instruments, Berlin, Germany) and heat-polished to a final resistance of 1.7–4.0 MΩ. Cells were held at –60 mV and probed every 4 s by voltage ramps from –100 mV to +100 mV of 400 ms duration.
Substances
The composition of the ES (in mM) was NaCl 140, KCl 4, CaCl2 1.25, MgCl2 1, and Glucose 5, Hepes 10. Stock solutions of sumatriptan (sumatriptan succinate, 40 mM in saline), carvacrol (100 mM in dimethyl sulfoxide (DMSO), HC-030031 (50 mM in DMSO), capsaicin (10 mM in ethanol), and ionomycin (2 mM in DMSO) were diluted to the final concentration in ES and therefore contained at maximum 0.1% of the solvent. All substances were obtained from Sigma-Aldrich.
Results
Calcium microfluorimetry in HEK293 cells
HEK293 cells transiently transfected with human TRPA1 (HEK293-hTRPA1) were challenged with sumatriptan (400 µM, 3 min). Changes in the intracellular calcium concentration ([Ca2+] i ) were monitored using Fura 2 calcium microfluorimetry. The cells were also stimulated with the TRPA1 agonist carvacrol (100 µM, 30 s) and with the calcium ionophore ionomycin (2 µM, 20 s) as a positive control. Sumatriptan evoked robust calcium transients in HEK293-hTRPA1 cells, and these cells were also activated by carvacrol, indicating functional expression of hTRPA1 (Figure 1). Activation by sumatriptan caused a slow onset and the calcium signal reached a plateau within 1 min. Untransfected HEK cells were not activated by sumatriptan (not shown). The same stimulation protocol was applied to HEK293 cells transiently transfected with the hTRPA1-3C mutant, in which three cysteines important for the action of covalent agonists were replaced by serines. As shown in Figure 1, the sumatriptan-evoked calcium transients mediated by hTRPA1-3C were substantially delayed and diminished compared to those mediated by wild-type hTRPA1 channels.
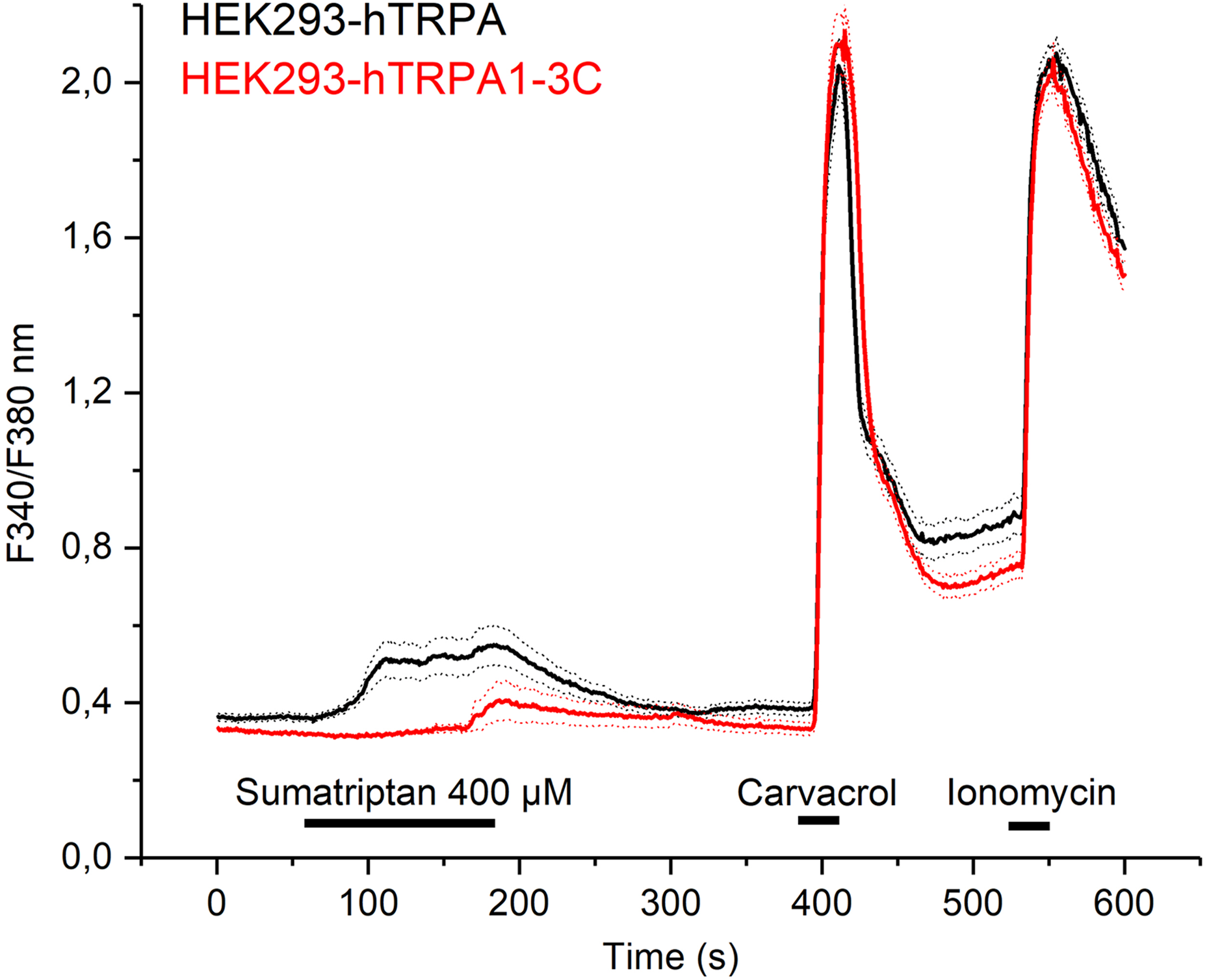
Sumatriptan activates hTRPA1 and, to a lesser extent, HEK293-hTRPA1-3C. HEK293t cells transiently transfected with hTRPA1 were stimulated with sumatriptan (black trace, n = 54). Responses to carvacrol (100 µM) confirmed the functional expression of TRPA1. Ionomycin (2 µM) was applied at the end of the experiment as a positive control. The same stimulation protocol was applied to HEK293t cells expressing the hTRPA1-C621S-C641S-C665 S mutant (red trace, n = 52). Data are represented as mean ± SEM, horizontal bars indicate duration of application. SEM: standard error of the mean.
In order to reflect the sumatriptan concentration range measured in human plasma, HEK293-hTRPA1 cells were also stimulated with a lower concentration of sumatriptan for an extended duration. As shown in Figure 2, sumatriptan (10 µM) evoked transient responses in HEK293-hTRPA1 cells, which peaked after about 10 min.
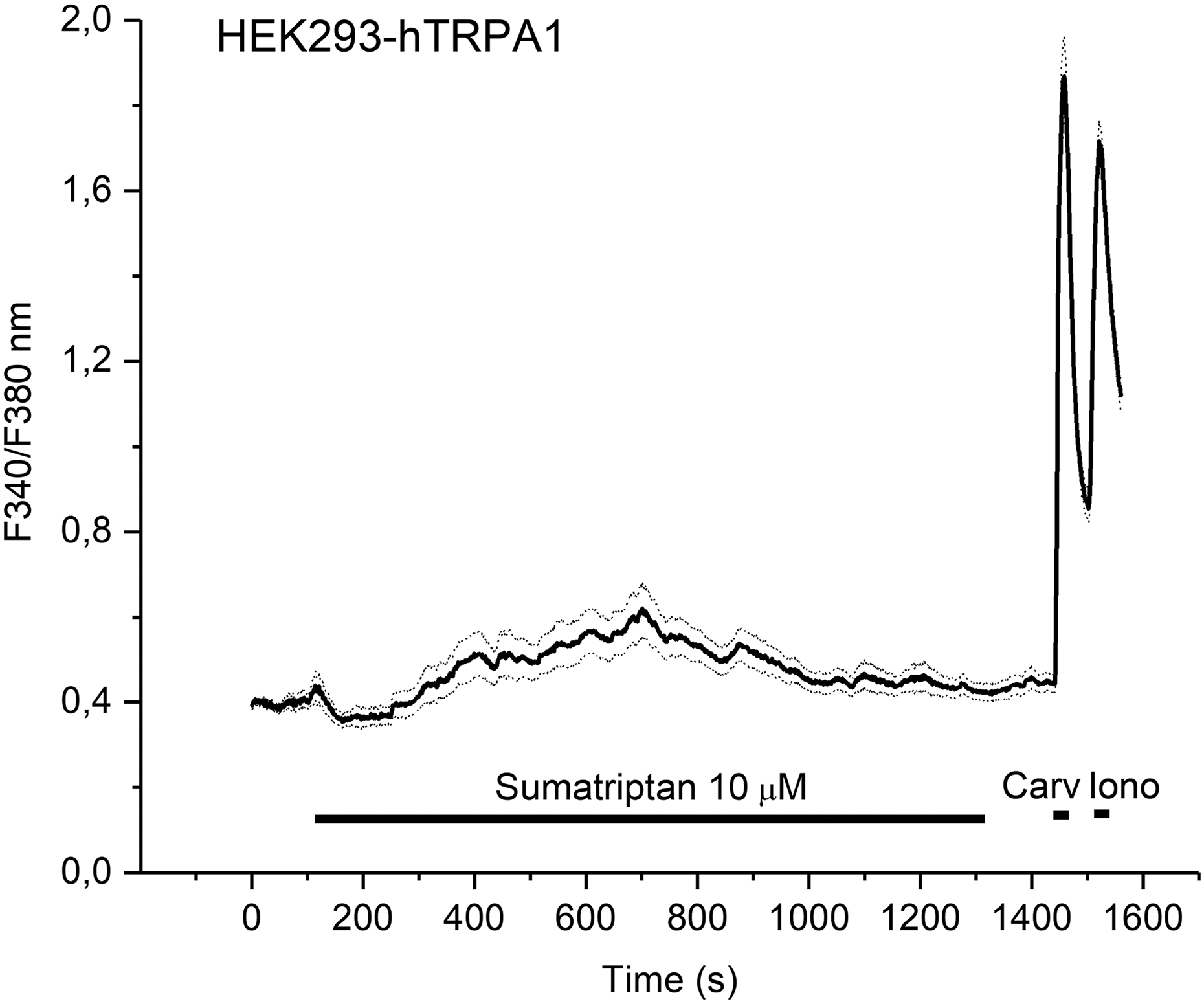
Sumatriptan activates recombinant hTRPA1 at low concentrations. HEK293t cells transiently transfected with hTRPA1 (n = 55) were challenged with sumatriptan for 20 min, followed by carvacrol (Carv, 100 µM, 30 s) and ionomycin (Iono, 2 µM, 30 s). Note the robust activation of HEK293t-hTRPA1 cells by sumatriptan, followed by slow desensitization after about 10 min. Horizontal bars indicate duration of application.
Calcium microfluorimetry in trigeminal ganglion neurons
Sumatriptan (400 µM; 4 min) was also applied to cultured rat TG neurons. Following sumatriptan, the neurons were stimulated with carvacrol (100 µM; 30 s) and capsaicin (1 µM; 30 s). KCl (60 mM; 30 s) was applied at the end of the experiment to identify viable neurons. Sumatriptan generated robust increases in [Ca2+] i in a subpopulation of TG neurons (Figure 3). Within 337 tested neurons, 161 (48%) were activated by capsaicin and 47 (14%) responded to carvacrol. All 27 sumatriptan-sensitive neurons (8%) were also activated by capsaicin and carvacrol (Figure 4).

Sumatriptan activates a subpopulation of carvacrol-sensitive rat TG neurons. Rat TG neurons in primary culture were stimulated with sumatriptan, carvacrol (Carv, 100 µM), capsaicin (Caps, 1 µM) and KCl (60 mM). The averaged responses of all 47 carvacrol-sensitive neurons are illustrated. Note the robust calcium transient evoked by sumatriptan in these neurons. Data are represented as mean ± SEM, horizontal bars indicate the duration of application. SEM: standard error of the mean.

Venn diagram of sensitivity to capsaicin, carvacrol, and sumatriptan in rat TG neurons. All 27 sumatriptan-sensitive neurons were a subset of the 47 carvacrol-sensitive neurons. All carvacrol-sensitive neurons were a subset of the 161 capsaicin-sensitive neurons.
Whole-cell patch clamp recordings
We have also used the patch clamp technique in the voltage clamp mode to record membrane currents induced by sumatriptan in HEK293-hTRPA1 cells. Sumatriptan (100 µM, 2 min) evoked large outwardly rectifying currents with a reversal potential close to 0 mV, which were almost completely inhibited by the selective TRPA1 antagonist HC-030031 (10 µM; n = 3, Figure 5).
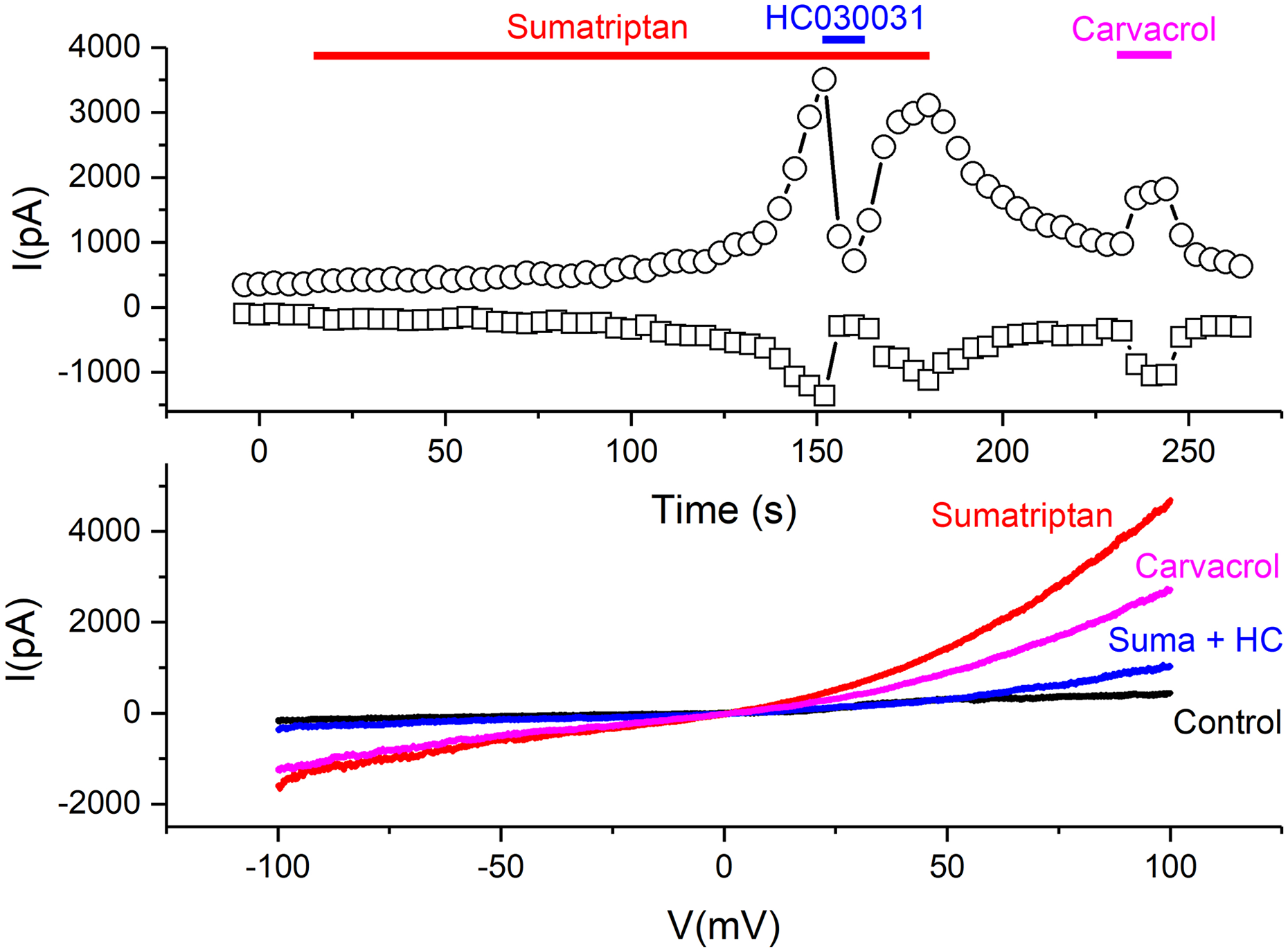
Membrane currents evoked by sumatriptan in HEK293-hTRPA1 cells. Illustrative whole-cell patch clamp recording from a HEK293t cell transiently transfected with hTRPA1 voltage-clamped at −60 mV, and subjected to 400-ms voltage ramps from −100 mV to +100 mV every 4 s. Under the exposure to sumatriptan (100 µM), a slowly developing but in the end, large current developed. The current at +80 mV (circles) and −80 mV (squares) are visualized in the upper panel. The current–voltage relationship before and at the end of the respective application bars showed a TRPA1-typical outward rectification in response to sumatriptan and carvacrol (lower panel). This current was almost completely inhibited by the TRPA1 antagonist HC030031 (10 µM), see blue trace Suma + HC. All sumatriptan-sensitive cells (n = 3) were also activated by the TRPA1 agonist carvacrol (100 µM). Horizontal bars indicate the duration of application.
Discussion
Our experiments employing calcium microfluorimetry and the patch clamp technique rendered clear evidence that sumatriptan at micromolar concentrations causes transient activation of human TRPA1 receptor channels expressed in HEK293 cells. The covalent mechanism of activation is demonstrated by the large reduction of this effect in the hTRPA1-3C mutant. Sumatriptan also evoked calcium transients in cultured TG neurons suggesting the same effect on rat TRPA1 in trigeminal primary afferents innervating extra- and intracranial trigeminal tissues. As an additional control, all sumatriptan-sensitive cells responded to the TRPA1 agonist carvacrol.
Activation of TRPA1 channels and involvement in pain generation
Using immunohistochemistry and in situ hybridization, TRPA1 receptor protein was localized in a major fraction of rodent TG neurons, peptidergic as well as IB4-positive ones. 17,18 In mice, TRPA1 immunoreactive TG neurons were clustered around retrogradely labeled dural afferent neurons, 19 which may indicate a close association of TRPA1 with meningeal nociception and headache pathophysiology. From calcium imaging experiments in rodent dorsal root ganglia, it was concluded that TRPA1 is functionally expressed primarily in nonpeptidergic IB4-positive small neurons. 20
TRPA1 receptors are activated by substances mediating oxidative, nitrative, and electrophilic stress, 21 including endogenous and exogenous irritants like umbellulone, an extract of the Californian “headache tree.” 22 Since these stimuli can trigger migraine attacks, TRPA1 receptor channels have been assumed to contribute to migraine pain generation. 23 Application of TRPA1 agonists to the rat cranial dura mater caused tactile facial and hind paw allodynia and pronociceptive behavior that was attenuated by pretreatment with the TRPA1 antagonist HC-030031 and sumatriptan. 24 Our group has found that rat meningeal afferents in vitro are activated by TRPA1 agonists such as acrolein and mustard oil only in the presence of intact transient receptor potential transduction channels of the vanillod type (TRPV1). 25 TRPV1 is expressed in most of the slowly conducting nociceptive afferents. 26,27 The mechanisms underlying this cooperative effect are probably the calcium dependence of TRPA1, and the respective calcium is provided by concomitant TRPV1 activation. 28
TRPA1 receptors can be sensitized by inflammatory mediators such as prostaglandins 29 through the activation of protein kinase A. 30 In addition, nociceptor sensitization may go along with an upregulation of TRPA1 receptors, as it was shown in visceral and oral pathologic states. 31,32 On the other hand, in an orofacial formalin model, the activation of TRPA1 receptors was found counteracting hyperalgesia. 33 TRPA1 activation in nociceptors can be followed by neuronal desensitization, as it was induced by isopetasin, an extract of butterbur. 34 Therefore, the activation of TRPA1 receptors can have nociceptive and antinociceptive effects, 35 in a time-dependent manner involving different intracellular pathways.
Sumatriptan effects through 5-HT1B/D receptor activation
Sumatriptan was the first specific 5-HT1B/D agonist used in the therapy of migraine. 36 Although it was the standard triptan for several years and is still used as a very effective antimigraine drug in different pharmaceutical forms, 1 the molecular mechanisms underlying its effects are not completely known. Like later developed triptans acting at 5-HT1B/D receptors, sumatriptan causes constriction of cranial arteries and inhibits the release of neuropeptides from trigeminal primary afferents in peripheral tissues and at the first synaptic level in the trigeminocervical complex, which is thought to counteract the nociceptive mechanisms involved in migraine pain generation. 37 These inhibitory effects have originally been explained by opening of K+ channels via pertussis toxin-sensitive G proteins. 38 However, 5-HT1 receptors are not only linked to inhibitory pathways regulating ion channels via Gi/o inhibiting adenylyl cyclase but can also stimulate phospholipase C and mitogen-activated protein kinase. 39 Accordingly, sumatriptan has been found inhibiting forskolin-stimulated increases in cyclic adenosine monophosphate (cAMP) but also elevating inositol phosphates and intracellular calcium concentrations, 40 which offers several possibilities that may explain the nociceptive side effects of sumatriptan, although the mechanisms seem to be contradictory. While the ability of sumatriptan to induce calcium-dependent discharges in meningeal sensory neurons was documented in rat, 10 optical imaging on afferent fiber terminations in mouse dura mater showed that sumatriptan, via 5-HT1D receptors, caused dose-dependent inhibition of calcium transients mediated by N-type calcium channels. 41 In a recent examination, intradermal injection of sumatriptan at low doses (1 pg–10 ng) in rats produced hyperalgesia, which was dependent on protein kinase A. 42
Nociceptive effects of sumatriptan
We add now a competing explanation for the local pronociceptive actions of sumatriptan via the direct activation of TRPA1 receptor channels, as indicated by calcium transients and membrane currents in HEK293 transfected with human TRPA1 receptors. The agonistic effect of sumatriptan on TRPA1 receptors may provide an explanation for the induction of local pain by this drug, 8 particularly in pathological states associated with sensitization of nociceptors. 9 The clinically used subcutaneous dose of 6 mg/0.5 ml sumatriptan has a concentration of about 40 mM, which is far above the applied dose in our experiments (400 µM). Thus, it is very likely that local injections of sumatriptan cause massive transient activation of TRPA1 receptors of the surrounding nociceptors. The systemic drug concentration during pain events like chest, muscle, and neck pain and aggravation of headache 7 reported after sumatriptan therapy is certainly much lower. However, the minimal sumatriptan dose activating meningeal afferents in the rat 7 was 3 µg/kg, which approximates the subcutaneous therapeutic dose of 6 mg in humans assuming that 3% of this dose enters the circulation during the first 10 min after injection. The low dose of sumatriptan (10 µM) used in our study caused a small but still substantial transient activation of several minutes in HEK293t–hTRPA1 cells. Thus, we conclude that the activation of TRPA1 receptors can well contribute to the adverse pain events following sumatriptan application, particularly if TRPA1 receptors are upregulated and sensitized.
Footnotes
Acknowledgements
We thank Birgit Vogler and Annette Kuhn for their excellent technical assistance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Friedrich-Alexander-University Erlangen-Nürnberg (Emerging Fields Project: Medicinal Redox Inorganic Chemistry), the Alexander von Humboldt Foundation (travel grant to AB), and the CNCS/UEFISCDI grant PN-III-P4-ID-PCE-2016-0475 from the Romanian Ministry of Research and Innovation.
