Abstract
Objective:
To assess the feasibility of a randomized controlled trial protocol that aims to determine the efficacy and safety of oral dexamethasone compared to placebo for the prevention of migraine recurrence in children and adolescents visiting the pediatric emergency department (ED) with migraine.
Methods:
This study was a two-arm, parallel-group, randomized, placebo-controlled, double-blind pilot trial of patients presenting to the pediatric ED with migraine. Eligible participants were randomized at 1:1 ratio to receive either oral dexamethasone 0.6 mg/kg (maximum 15 mg) or matched placebo as a single dose. Efficacy and safety outcomes were assessed at discharge, 48 h and 7 days after discharge. The primary outcome of the trial was feasibility and was assessed through participant recruitment rate, follow-up completion rates, participant satisfaction ratings and comparison of enrolled versus non-enrolled participants. Efficacy and safety outcomes were not analyzed given that this was a pilot study.
Results:
Twelve participants were enrolled over the 6-month recruitment period. This represents 60% of the planned sample size and a 10.5% recruitment rate. No other feasibility issues were identified and patients expressed high satisfaction rates with their treatment: 90.9% were satisfied with their treatment at discharge and at 48-h follow-up and 81.8% were satisfied with their treatment at 7-day follow-up (81.8%). There were no significant differences observed when comparing enrolled participants to those not enrolled.
Conclusion:
This pilot randomized controlled trial is the first to assess dexamethasone in the pediatric ED for the prevention of migraine recurrence. The protocol is feasible but recruitment in a single center was lower than expected. Future pediatric ED migraine studies may use innovative or pragmatic trial designs to maximize feasibility from a recruitment standpoint.
Keywords
Background
Headache is very common in the pediatric population, with an estimated worldwide prevalence of 58.4% among children and adolescents. 1 Not only is headache highly prevalent in the community, but it also accounts for a significant proportion of pediatric emergency department (ED) visits, anywhere from 0.8% to 1.3% of all pediatric ED visits are due to headache. 2 –5 Although the majority of pediatric ED visits for headache are due to secondary causes, migraine accounts for between 5.4% and 28.7% of these visits. 2 –7 Despite how common it is, the evidence for how to manage migraine in the pediatric ED is very limited, with only three randomized controlled trials having been carried out in this area. 8 –10
The recurrence of headache and migraine relapse following treatment in the ED is also very common; 26% to 68% of pediatric patients have recurrence of their migraine within one week of ED discharge. 8,9,11 In addition, approximately 5.5% of pediatric patients presenting to the ED with migraine have a return visit to the ED within 3 days of discharge. 12 Unfortunately, there are no clinical trials to support evidence-based treatments to prevent recurrence in this population. A few studies have assessed migraine recurrence as a secondary outcome 8,9,13 but have failed to yield evidence for an effective therapy. Furthermore, a large retrospective study in a pediatric ED failed to show an association between the specific migraine treatment administered and rates of migraine recurrence within 1 month of ED discharge. 14 In one retrospective pediatric study, steroid administration did not yield any difference in migraine recurrence rates at 48 and 72 h, but minimal conclusions can be drawn from this data given that it was non-randomized and the patients who received steroids were more likely to have ongoing pain at discharge as compared to those who did not receive steroids. 15 Therefore, despite the prevalence of the problem, there is no evidence to guide clinicians seeking to prevent migraine recurrence after ED discharge in pediatric patients.
There is well-established literature supporting the efficacy of steroids for the prevention of migraine recurrence in adults presenting both to outpatient settings and to the ED with migraine. 16 Four systematic reviews 17 –20 have shown that dexamethasone reduces migraine recurrence in this setting and that it is well tolerated in this population. Furthermore, dexamethasone has a plausible mechanism of action in preventing migraine recurrence in that it attenuates the inflammatory response, and in theoretical frameworks, migraine is believed to be associated with neurogenic inflammation. 21
Based on the adult evidence, dexamethasone is the most promising therapy for preventing migraine recurrence in children and adolescents presenting to the ED with migraine. This study was designed as a randomized controlled trial protocol to explore whether oral dexamethasone is superior to placebo in preventing migraine recurrence 48 h after ED discharge in a sample of pediatric patients visiting the ED for migraine. The aim of this pilot project was to explore the feasibility of implementing a randomized controlled trial in a single pediatric ED. We hypothesized that the protocol would be feasible and acceptable to participants. Specifically, we aimed to (1) estimate recruitment and follow-up rates, (2) evaluate differences between enrolled and non-enrolled participants, and (3) evaluate participant satisfaction with treatment.
Methods
Study population
Participants were recruited between December 2016 and June 2017 from the ED at the Children’s Hospital of Eastern Ontario, a tertiary care academic pediatric hospital located in Ottawa, Canada. Patients were eligible to participate if they were aged between 8.0 and 18.0 years, met criteria B–E of the International Classification of Headache Disorders-3 (ICHD-3) diagnostic criteria for migraine, 22 spoke English or French, and had a negative urine pregnancy test (for females who have had menses). The ICHD-3 criteria were modified given that prior studies have shown that removal of criterion A (at least five attacks fulfilling criteria B–D) increases the sensitivity of these criteria in the ED setting. 23,24 Exclusion criteria included dose of a steroid medication in the past 7 days, known allergy to dexamethasone, immunosuppression, Cushing’s syndrome, known diabetes mellitus, known peptic or duodenal ulcer or other major gastrointestinal illness (e.g. ulcerative colitis), known myasthenia gravis, glaucoma, febrile at triage, history of head trauma in the past 7 days, presence of any known active infection, current secondary headache, and pregnant or breastfeeding.
Study design
This study was a two-arm, parallel-group, randomized, placebo-controlled, double-blind pilot trial. Because this was a pilot trial, a 6-month recruitment period was predetermined and the study was closed to enrollment once this period was complete. The trial protocol was registered prior to participant enrollment (ClinicalTrials.gov identifier NCT02794441) and the Children’s Hospital of Eastern Ontario Research Ethics Board approved the study protocol (protocol 16/27E). This project was funded by The Physicians’ Services Inc. Foundation.
Patients presenting to the ED with a triage diagnosis of headache or migraine were approached by research volunteers to introduce the study. A research assistant met with interested patients to further discuss the study and carry out eligibility screening and consent. At this time, basic demographic data were collected on all screened participants, including age, sex, Canadian Triage and Acuity Scale (CTAS) score, date of triage and time of triage. Parents or guardians and patients deemed to be capable of consenting gave verbal and written consent and patients who were not deemed to be capable of consenting gave verbal and written assent.
For consenting participants, baseline interviews were carried out by research assistants and included a structured headache history, medical history, and assessment of baseline pain using a four-point rating scale where 0 = no headache, 1 = mild headache, 2 = moderate headache, and 3 = severe headache, as per the recommendations in the International Headache Society guidelines. 25 During baseline interviews, participants answered structured questions about their migraine history, including duration of time with migraines, baseline headache frequency, migraine characteristics, and Pediatric Migraine Disability Assessment Scale (PedMIDAS) scores. 26 Participants were given an ICHD-3 diagnosis based on their migraine history completed by the research assistants. Participants were also asked questions about their current migraine, including intensity, severity (using the four-point rating scale described above), and at-home treatment strategies. In addition, the treating ED physician completed a baseline history and physical examination.
After the baseline evaluations, patients were randomized to their study intervention. In addition, participants received acute migraine treatment that was selected by their treating physician. Following randomization, research assistants assessed participant outcomes at discharge. Follow-up was completed 48 h and 7 days after ED discharge using emailed questionnaires or telephone follow-up, depending on participant preference. At all time points, participants were asked about presence of headache, headache intensity, patient-reported treatment satisfaction, and adverse events. Participants who completed all follow-up questionnaires were given a CAD $10 gift card. Chart reviews were completed 7 days after admission to record any return visits to the ED and return visits were coded as either headache-related or unrelated.
Interventions
Participants were randomized at 1:1 ratio to receive either an oral compound of dexamethasone (1 mg/mL suspension) given as a one-time dose of 0.6 mg/kg (maximum of 15 mg) or placebo matched for taste, volume, and appearance. The treating nurse administered the study intervention to the participants in the ED. The dexamethasone dosing was chosen based on both the range of dexamethasone doses administered for this indication in the adult literature, which ranged from 8 to 24 mg intravenous (IV), 17 –19 and based on the doses used in pediatric patients for other indications, which ranged from 0.03 to 2 mg/kg/dose. 27 Acute interventions for migraine were selected at the discretion of the treating physician.
Randomization
A statistician with no role in participant recruitment or clinical care prepared the allocation sequence prior to the study initiation using a computer-generated randomization schedule. The randomization sequence was composed of permuted blocks of four, and randomization was stratified according to the length of current migraine duration (
Outcomes
Efficacy and safety data were collected for the purposes of trial protocol implementation but are not reported here given that this is a pilot trial that is underpowered to assess safety and efficacy outcomes. The primary outcome of interest was trial feasibility, assessed with participant recruitment rate, follow-up completion rates, and participant satisfaction ratings. The recruitment rate was calculated in two ways: (1) the proportion of patients screened during research assistant hours who met eligibility criteria and were enrolled and (2) the proportion of eligible patients screened during research assistant hours who were enrolled. Follow-up completion rates were calculated as the proportion of enrolled participants who completed the follow-up questionnaires. Participant satisfaction was measured using a five-point Likert-type scale (5 = very satisfied, 4 = satisfied, 3 = neutral, 2 = unsatisfied, 1 = very unsatisfied), and scores were dichotomized into satisfied (Likert = 4 or 5) or dissatisfied (Likert = 1–3).
Statistical analyses
We aimed to enroll 20 participants over the 6-month recruitment period. Demographic characteristics of the participants and outcome data were summarized using descriptive statistics. Comparisons between enrolled and non-enrolled participants with regards to basic demographic characteristics (i.e. age, sex, CTAS score, time, and month of presentation to triage) were made using either Fisher’s exact tests or Wilcoxon rank sum tests, depending on the distribution of the variable. Percentages were used to characterize recruitment rates, follow-up completion rates, and participant satisfaction ratings.
Data availability policy
Anonymized data will be shared by request from the qualified investigators.
Results
Figure 1 describes the flow of participants through the study. The overall recruitment rate was 10.5%, with 60% of eligible participants enrolled and 60% of the planned sample size achieved. Seven participants were randomized to dexamethasone and five participants were randomized to placebo. One participant in the dexamethasone group withdrew from the study prior to receiving the study intervention.

Consolidated Standards of Reporting Trials (CONSORT) diagram.
Table 1 describes participant baseline characteristics. Of note, the majority (83.3%) of recruited participants presented in the morning or in the afternoon. The mean age of participants was 13 years (SD = 2.6), and 75% of the participants were female. The sample was composed primarily of patients with migraine with aura (75%). Participants had an average on 10.3 (SD = 12.6) headaches per month in the past three months. The majority of patients had attempted at home treatment prior to visiting the ED (83.3%), and the baseline duration of the headache was 29 h (SD = 23.5).
Descriptive statistics for enrolled participants (N = 12).
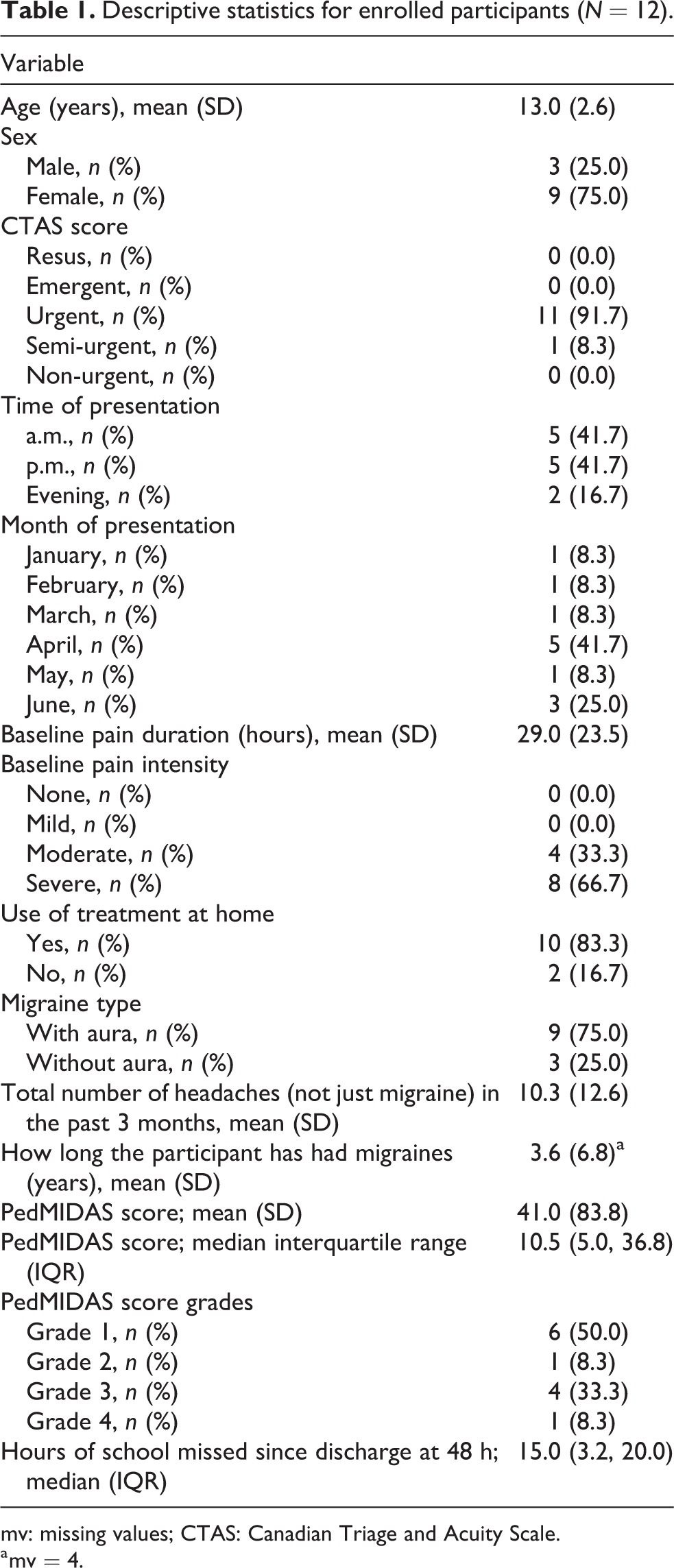
mv: missing values; CTAS: Canadian Triage and Acuity Scale.
a mv = 4.
Among eligible participants, enrolled participants and those not enrolled did not differ significantly in any of the measured variables (Table 2).
Enrolled participants versus eligible but not enrolled (N = 20).

CTAS: Canadian Triage and Acuity Scale.
All of the 11 participants who received a study intervention completed all of the follow-up questionnaires (100%). The preferred method of follow-up was emailed questionnaires, with 11 of the 12 participants who were randomized (91.7%) selecting this method of follow-up. The vast majority of participants reported being satisfied with their treatment at discharge (90.9%), at 48-h follow-up (90.9%), and at 7-day follow-up (81.8%).
Discussion
We successfully implemented the planned protocol for a clinical trial on the efficacy and safety of oral dexamethasone for the prevention of migraine recurrence in the pediatric ED at a single center. During this pilot trial, high follow-up rates were observed and participant satisfaction rates were high, though significant challenges were experienced in participant recruitment. Although efficacy and safety data are presented here, conclusions about dexamethasone’s role in this population cannot be drawn from this pilot study due to lack of statistical power in the context of a very small sample size.
Recruitment in pediatric clinical trials can often be challenging and even more so in acute care environments like the ED. While there are few pediatric migraine clinical trials in the ED, the enrolment challenges seem to be unique to the pediatric population. To our knowledge, only three other published randomized controlled trials have been carried out among patients with migraine in the pediatric ED: one of the trials reported a recruitment rate of 13.4% overall, with enrollment of 45.6% of eligible participants. 9 The other two trials did not report their recruitment rates, though one of the studies required 3 years to recruit 62 participants from two pediatric EDs, 8 while the other study required 26 months to recruit 74 participants from two pediatric EDs. 10 Our recruitment rate and the published rates appear to be considerably lower than the rates observed in the seminal adult migraine trials assessing the efficacy of dexamethasone for the prevention of migraine recurrence in the ED. Though most of those trials unfortunately did not publish their recruitment rates, the two that did reported higher rates as compared to the present study: recruitment rates in these studies were 31.3% overall, with 76.2% of eligible participants enrolled, 28 and 42.4%, with 50.2% of potentially eligible participants enrolled. 29
The low recruitment rates seen in pediatric ED migraine trials should not discourage investigators from pursuing intervention studies in this population, as there is an urgent need for evidence on how to manage migraine in the pediatric ED. 30 Given these low recruitment rates, future studies assessing migraine interventions in the pediatric ED may apply lessons learned from the present study and could utilize pragmatic or innovative trial designs (Figure 2) to answer the many clinical questions that remain in this area.

Examples of strategies to enhance feasibility in pediatric ED migraine trials. ED: emergency department.
In the present study, we made several observations about factors that may have limited enrollment in this trial. Of note, 83.3% of the recruited participants were patients who presented to the ED in the morning or in the afternoon. Research assistant hours encompassed the period between 10 a.m. and 10 p.m., and this may have influenced recruitment due to timing issues related to limited staff coverage. Another factor that appeared to influence recruitment was month of presentation. Seventy-five percent of the enrolled participants were recruited between April and June, with the winter months (January–March) having yielded a lower recruitment rate. Previous studies have also found seasonal patterns to pediatric ED headache presentations with a higher frequency of visits during certain months of the academic year as compared to other calendar months. 31 –33 Additionally, we used slightly modified ICHD-3 criteria (criterion A removed) to define migraine in this study. However, these criteria may still have posed a challenge to recruitment given that the ICHD criteria have been shown to lack sensitivity in the ED setting. 23,24 In addition, it has been shown that only 26% of ED physicians use the ICHD criteria, 34 thereby rendering trials that define migraine in this way problematic in terms of external validity. Therefore, future trials of ED interventions for pediatric migraine may optimize recruitment by extending research assistant hours, targeting recruitment to calendar months where ED presentations for headache tend to peak, and using less stringent criteria for migraine that are more sensitive and pragmatic in the ED setting.
One of the most promising approaches to enhancing feasibility in pediatric ED migraine trials would be to shift toward pragmatic trial designs that aim to be more reflective of “real world” circumstances as compared to explanatory trials, which are carried out in more ideal circumstances. 35 While the International Headache Society’s guideline for controlled trials of drugs in migraine 25 provides an essential framework for the design of migraine trials, these guidelines are weighted toward explanatory designs, and some research questions may lend themselves to more pragmatic designs. The ED setting poses particular challenge to the use of traditional explanatory trial methods and there is opportunity for future studies to adapt their protocols to enhance applicability in ED settings. For example, the use of ICHD criteria in the ED setting poses a challenge, as discussed above. Another important design factor to consider here is setting: while all existing pediatric ED migraine trials have been carried out in one or two tertiary-care pediatric EDs, a more pragmatic approach would involve recruiting from multiple EDs 35 and ideally from both pediatric and general EDs, given that many children are seen outside of specialized pediatric centers. Several other design aspects could also be reconsidered in a pragmatic pediatric ED migraine trial such as reducing the intensity of follow-up, using native ED resources for recruitment, and selecting patient-centered outcomes (Figure 2). A reexamination of trial design, paying particular attention to shifting the balance toward a more pragmatic approach, may not only serve to enhance external validity of the trial but may also help with feasibility by reducing the burdens associated with protocol implementation and by increasing the number of eligible participants.
One innovative approach that could be considered in the design of future pediatric ED migraine trials would be to apply Bayesian statistics to optimize feasibility by potentially reducing the required sample size. Bayesian statistics use historical knowledge about the treatment effect to create an assumed distribution of the model’s parameters. This assumed distribution is then merged with the distribution derived from the results of the trial using a weighted average. The merged distribution is called the posterior distribution and is used for data analysis. 36,37 In order to apply this approach, one has to assume that the historical data used to create the assumed distribution is applicable to the study population. One way of applying Bayesian statistics to pediatric trials is using data from uncontrolled pediatric studies or from adult studies as historical data. For example, a group of investigators recently simulated the use of a Bayesian model for pediatric diabetes trials using data from adult diabetes trials. In this simulation study, Huff et al. demonstrated that pediatric diabetes trial sample sizes could be reduced anywhere from 30% to 78% depending on the assumptions of the model and the desired type I error. 37 Given that certain ED migraine interventions have been well studied in the adult population and given that the assumption of shared pathophysiology between adult and pediatric migraine may be reasonable, Bayesian statistics could be considered for future pediatric ED migraine trials. In the case of dexamethasone, the large body of adult data could be used in a Bayesian model to enhance the feasibility of a future randomized controlled trial in the pediatric migraine population. Of course, this approach is relatively novel, untested, and should only be considered after careful discussion among both clinical and statistical experts, given its complexity.
Another innovative approach to trial design that could be applied to pediatric ED migraine studies involves the use of adaptive trial methods. Adaptive trials make use of accumulated study data to modify aspects of the ongoing trial. Many of the methods used in adaptive designs can enhance trial feasibility by potentially reducing the required sample size. For example, treatment-adaptive randomization can reduce the sample size of the trial by preferentially assigning participants to the more successful intervention over time. Adaptive designs can also be applied to group sequential trials, for example, by reestimating the required sample size at interim analyses, which can result in a reduction in the sample size target, though also carries the potential of increasing sample size requirements. 38 Thus, several adaptive methods could be applied to future pediatric ED migraine trials to potentially reduce sample size requirements and to optimize trial success in the face of limited resources and recruitment challenges.
Conclusions
This pilot trial comparing oral dexamethasone to placebo for the prevention of migraine recurrence in a sample of pediatric ED patients with migraine demonstrated feasibility at the level of protocol implementation and acceptability to participants but challenges at the level of participant recruitment. In designing future pediatric ED migraine intervention studies, trialists may want to consider some of the lessons learned in the present study or use pragmatic or innovative trial designs to optimize feasibility and recruitment. Despite the inherent challenges associated with pediatric ED migraine studies, the paucity of data on how to manage migraine in this setting should compel ongoing efforts at finding ways to generate high quality evidence for this patient population.
Footnotes
Authors’ note
Dr Orr receives royalties from Cambridge University Press; Dr Richer is supported by the Women and Children’s Health Research Institute and Northern Alberta Clinical Trials and Research Center, University of Alberta; Dr Barrowman does not have any financial disclosures to report; Dr Zemek is supported by a Clinical Research Chair by the University of Ottawa.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by The Physicians’ Services Inc. Foundation.
