Abstract
Background
Tacca chantrieri (Andre), or the black bat flower, is commonly found in Southeast Asia; however, its effects on immunity have not been studied. This study aimed to investigate the effects of T. chantrieri crude extract on CD4+ T cell phenotypes and on the phagocytosis and oxidative burst of polymorphonuclear neutrophils (PMNs) in the elderly (ex-vivo). Its ability to inhibit opportunistic and pathogenic bacteria was also evaluated.
Methods
T chantrieri crude extract was obtained by ethanolic extraction. A total of eight elderly (age ≥65 years) were enrolled in the study. Investigations used flow cytometric analysis. Antimicrobial effects were explored by agar well diffusion and minimum inhibition concentration (MIC) testing.
Results
CD4+ T cell proportion significantly decreased at the 100 μg/ml concentration (p = 0.015). CD4+NKG2D+ T cells decreased at all concentrations (50, 100, 200, 300 μg/ml). Regarding PMNs, phagocytosis and oxidative burst in the treated group were significantly increased at 15 min compared with the untreated group (p = 0.039 and 0.015, respectively). Regarding antimicrobial effect, 100 mg/ml crude extract inhibited Staphylococcus epidermidis, Staphylococcus aureus, Neisseria sicca, and Acinetobacter lwoffii. MIC values were 1.23, 3.70, 33.33, and 100 mg/ml, respectively.
Conclusions
This work reports preliminary findings of immunity promotion in the elderly following treatment with T. chantrieri crude extract. In future, the extract may be developed into a natural herb product or a form of alternative medicine for immune status enhancement, as well as a potential antimicrobial agent, particularly in the elderly.
Introduction
Aging is characterized by a reduction of physical fitness, cell and tissue dysfunction, and immune cell deterioration (known as immunosenescence). Several changes in both innate and adaptive immunity lead to chronic low-grade inflammation and are likely associated with disease pathogenesis. 1 Studies on the elderly populations have demonstrated decreases in phagocytic cell function and superoxide production and alterations of T cell frequencies and phenotypes.2–7
Various studies have reported immune system deterioration, particularly in cell functions, in the elderly with weakened immunity. Compared with younger individuals, the elderly exhibited lower phagocytic activity8,9 and decrease in T cell proliferation. T cell response to mitogen in the elderly has also been found to be lower than in younger individuals. 10 Regarding T cell phenotypic changes, studies have reported a higher accumulation of CD28null T cells (an immunological biomarker of immunosenescence). 6 Additionally, natural killer group 2 member D (NKG2D) receptors were predominantly expressed on the CD4+ T cells of the elderly, particularly on the CD28null T cell subpopulation, and likely acted as a surrogate co-stimulatory receptor. 3
Natural plant products are commonly used as complementary therapies in conjunction with modern medical treatments. 11 Tacca chantrieri (Andre), known as the black bat flower, is a flowering plant in the Taccaceae family commonly found in Southeast Asia. 12 Previously, a decoction made from the rhizome of this plant was used to treat muscle aches, gastrointestinal pain, and as a food poisoning antidote.13,14 This rhizome extract has been analyzed for its anti-inflammatory, anti-tumor, antioxidant, anti-cancer, and antimicrobial properties14,15; however, the effectiveness of the plant in immunity promotion has not been investigated and its potential antimicrobial properties are poorly explored. 16
This work therefore aimed to evaluate the effects of a T. chantrieri crude extract on immunity in the elderly (age ≥65 years) by investigating CD4+ T cell phenotype and subsets as well as the performance of PMNs (phagocytosis and oxidative burst). Antimicrobial effects were also investigated.
Materials and Methods
T. chantrieri Extraction
T. chantrieri extract was obtained from the rhizomes using the ethanolic extraction method. The rhizomes were acquired from Lamphun Province, finely chopped, and dried in a hot air oven at 60 °C. A total of 500 g of the powdered plant was macerated in 1500 ml absolute ethanol for 7 days prior to filtering with Whatman #No.2 filter paper under cooling conditions. The solution was filtered again with Büchner funnels and the residue washed twice with ethanol. The combination ethanolic extraction was evaporated at 40 °C and stored in an amber glass bottle at 2 °C. Finally, the combined ethanolic extract was dissolved in sterile water for injection as a working stock and stored at −20 °C until use.
Participants and Sample Collection
In total, eight of the elderly (age ≥65 years) were enrolled in this study. 17 This project was approved by the ethics committee of Khon Kaen University, Khon Kaen, Thailand (code HE-661164). All subjects provided written informed consent prior to enrollment. Peripheral whole blood samples with EDTA anticoagulant were collected for the complete blood count. Samples were also collected using a clotted blood tube for blood chemistry testing. Lithium heparinized blood was collected for immune cell phenotyping and functional assay. Participants with hypertension and/or type 2 diabetes mellitus and/or dyslipidemia were also enrolled, since the elderly without these cardiovascular disease risk factors are rare in the population. However, participants taking anti-inflammatory medications and those with autoimmune diseases, hematological diseases, coronary artery disease, heart failure, stroke, kidney diseases, liver diseases, type 1 diabetes mellitus, infectious diseases, cancer, or severe accidents, etc, were excluded.
PBMC Isolation
Peripheral blood mononuclear cells (PBMCs) were isolated from heparinized blood by standard gradient centrifugation using Lymphocyte Separation Medium (Lymphosep, Biowest, France). A total of 2 ml whole blood was diluted with 2 ml of 1X phosphate-buffered saline (1X PBS) prior to overlaying on 3 ml of Lymphosep at a ratio of 4:3 and centrifugation at 400 g without breaks for 40 min. The PBMC layer was harvested, transferred to a new 15 ml sterile conical tube, and washed twice with 7 ml of 1X PBS for 10 min at 280 and 150 g, respectively. Finally, the supernatant was discarded and the sample was resuspended with 1 ml of R10 medium [10% fetal bovine serum with 90% RPMI-1640 (Gibco, USA)].
PMN Isolation
Heparinized blood samples were sedimented to remove red blood cells using the HeptaSep™ reagent kit (Stem Cell Technologies Inc., Canada) at a ratio of 1:5 for 30 min prior to transferring the leukocyte-rich plasma to the top of 3 ml Lymphosep (isolated by standard gradient centrifugation) at a ratio of 1:1. This was then centrifuged at 400 g for 30 min and the pellet was harvested from the lowest layer. Following this, 1 ml of R10 medium and 9 ml of ammonium chloride lysis buffer (13.4 mM KHCO3, 155 mM NH4CL, 96.7 μM EDTA) were added to the isolated PMNs for 3 min, followed by centrifugation at 400 g for 3 min. Finally, the supernatant was discarded and the sample was resuspended with 2 ml of R10 medium.
Cell Viability by MTT Assay
Isolated PBMCs and PMNs were co-cultured with 100 and 300 μg/ml of T. chantrieri crude extract (Treated) or without T. chantrieri crude extract (Untreated) for 30 min. Three independent experiments were conducted (triplicate). MTT solution (ThermoFisher Scientific Inc., USA) was added and the solution was incubated at 37 °C for 3 h. The crystals were dissolved with dimethyl sulfoxide (AppliChem GmbH, Germany) prior to measuring the absorbance at 540 nm using a Varioskan™ LUX Multimode Microplate Reader (ThermoFisher).
Surface Staining for CD4+ T Cell Phenotyping by Flow Cytometry
PBMCs were co-cultured with (Treated) and without (Untreated) T. chantrieri crude extract for 30 min prior to staining by monoclonal antibodies conjugated with different fluorochromes as follows: anti-human CD3-FITC (BD Pharmingen, USA), anti-human CD4-APC-Cy7 (BD Pharmingen), anti-human CD28-PE-Cy7 (BioLegend, USA), CD314 (NKG2D)-APC (BioLegend), and CD56-PerCP-Cy5.5 (BioLegend). The samples were then incubated at room temperature for 10 min in the dark and washed with 1X PBS. Finally, the samples were analyzed using a BD FACSLyric™ flow cytometer (Becton Dickinson, USA). The immunophenotyping was analyzed using BD FACSuite™ RUO v1.5. The representative data were demonstrated as Figure 1. Lymphocytes and singlet cells were gated. CD3+CD4+ cells were identified as T helper (Th) cells. Subsequently, Th cells were categorized into two subsets: CD28+ Th cells and 28null Th cells. Each group was assessed for CD314 (NKG2D) expression. Isotype controls were performed based on the fluorescence minus one (FMO) isotype.
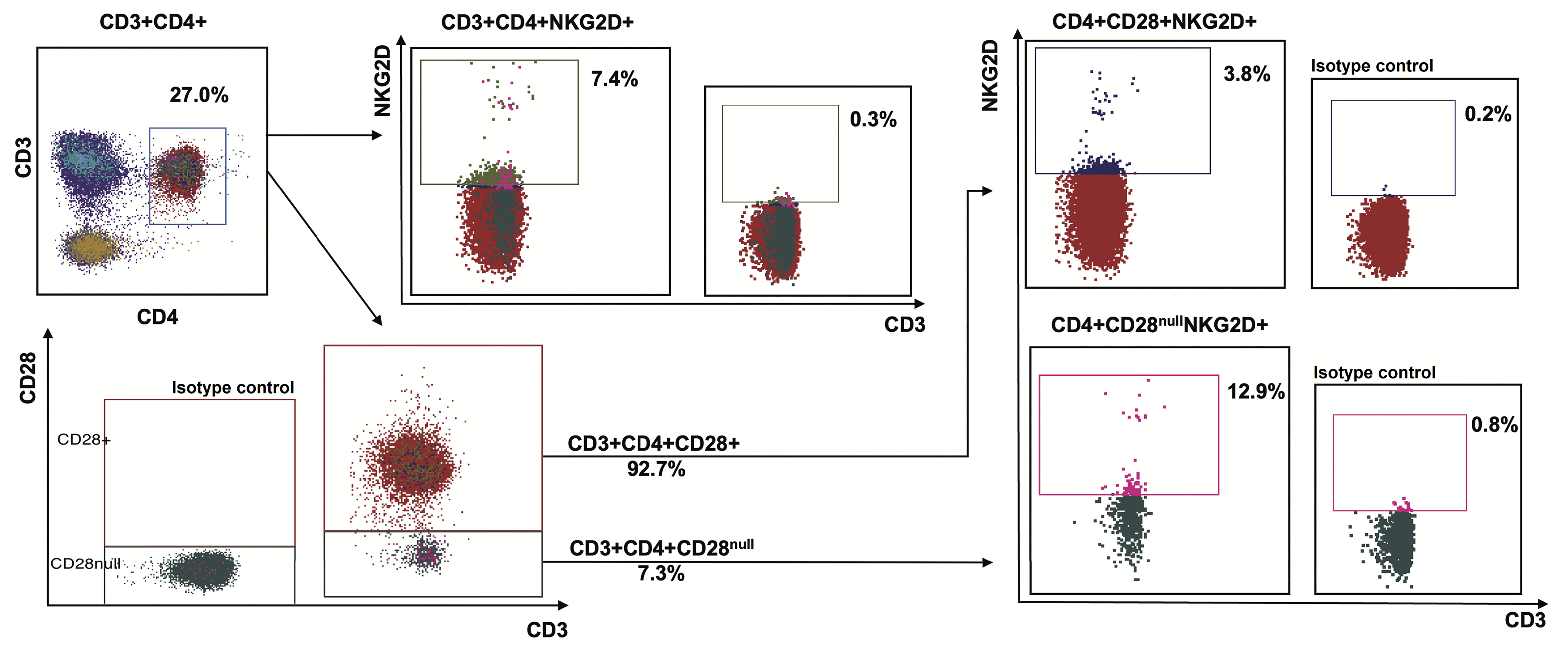
Representative data of CD4+ T cells and their subsets by flow cytometric analysis. After gating on lymphocytes and singlet cells, CD3+CD4+ cells were identified as helper T cells prior to separation into subsets, including the classical CD4+CD28+ T cells and aberrant CD4+CD28null T cells. Each population was analyzed for NKG2D (CD314) expression. CD28+ and NKG2D+ population gating was based on the minus one isotype control.
Phagocytosis and Oxidative Burst
Phagocytosis and oxidative burst were investigated by flow cytometric analysis. Isolated phagocytosed cells were incubated with (Treated) or without (Untreated) 100 μg/ml 18 of T. chantrieri crude extract for 30 min prior to co-culturing with FITC-labeled bacteria for 5, 15, and 30 min. 19 For the bacterial preparation, Staphylococcus aureus (strain ATCC 29223) was cultured overnight in Tryptic Soy Broth (TSB) at 37 °C, subsequently inoculated into fresh TSB, and grown to OD600 of 0.6–1. The bacterial cells were then preserved in 25% glycerol at −20 °C for later use. During the experimental phase, S. aureus glycerol stocks were washed twice with 1X PBS, centrifuged, and the cell concentration adjusted to 1.25 × 106 colony-forming units. Bacterial cells were then stained in the dark with 1 mg/ml of FITC for 60 min at 25 °C [Multiplicity of infection (MOI) = 5]. The fluorescently labeled bacterial cells were washed twice with 1X PBS before use in the experiment. PMNs were then stained with hydroethidine (HE) to detect oxidative burst (PE channel) and later measured with a flow cytometer.
The interpretative data of phagocytosis and oxidative burst was shown in Figure 2. Un-stained PMNs without bacteria were used for cut-off determination (Figure 2A). Figure 2B, quadrant (Q) 4 shows phagocytosis only; Q1 represents both phagocytosis and oxidative burst; Q2 shows oxidative burst only; Q3 represents non-phagocytosis. Q1+Q4 represents total phagocytosis and Q1+Q2 represents total oxidative burst. For the gating strategy, samples were gated on the granulocyte population using the forward scatter (FSC) and side scatter (SSC) flow cytometry properties (Figure 3A). Unstained PMNs without bacterial co-culturing were used for medium control and cut-off determination (Figure 3B). PMNs stimulated with phorbol 12-myristate 13-acetate (PMA; Sigma-Aldrich, USA) were used as positive controls for oxidative burst (Figure 3C). PMNs incubated with FITC-labeled bacteria and without HE staining were used as positive controls for phagocytosis (Figure 3D). The spontaneous oxidative burst at each time (5, 15, and 30 min) was independently measured (Figures 3E–3G, respectively). PMNs were co-cultured with T. chantrieri crude extract for 30 min and later co-cultured with FITC-labeled bacteria for 5, 15, and 30 min (Figures 3H–3J, respectively).

Representative data of the flow cytometry gating strategy for PMN function in the elderly. (A) Firstly, the PMN population was gated. (B) On FITC and HE (PE channel), uninfected PMNs were used as the cut-off. (C) PMA-stimulated PMNs were used as the positive control (HE+) for oxidative burst. (D) PMNs engulfing FITC-labeled bacteria without HE staining were used as the positive control (FITC+) for phagocytosis. (E-G) The spontaneous oxidative burst at each time point (5, 15, and 30 min) was investigated for subtraction. (H-J) PMNs treated with Tacca chantrieri crude extract were co-cultured with FITC-labeled bacteria and later stained with HE at (H) 5, (I) 15, and (J) 30 min.

Representative Data of the Flow Cytometry Gating Strategy for PMN Function in the Elderly. (A) Firstly, the PMN Population was Gated. (B) on FITC and HE (PE Channel), Uninfected PMNs Were Used as the cut-off. (C) PMA-Stimulated PMNs Were Used as the Positive Control (HE+) for Oxidative Burst. (D) PMNs Engulfing FITC-Labeled bacteria Without HE Staining Were Used as the Positive Control (FITC+) for Phagocytosis. (E–G) the Spontaneous Oxidative Burst at Each Time Point (5, 15, and 30 min) was Investigated for Subtraction. (H–J) PMNs Treated with Tacca chantrieri Crude Extract Were co-Cultured with FITC-Labeled bacteria and Later Stained with HE at (H) 5, (I) 15, and (J) 30 min.
Agar Well Diffusion and MIC Testing
Various pathogenic and opportunistic bacteria (gram positive, N = 6; gram negative, N = 8) were cultured overnight and adjusted to McFarland 0.5 (OD ≈ 0.1). Gram-positive bacteria included Bacillus subtilis, Staphylococcus aureus (ATCC 29223), Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus pneumoniae, and Enterococcus faecalis. Gram-negative bacteria included Escherichia coli, Neisseria sicca, Klebsiella pneumoniae, Proteus mirabilis, Proteus vulgalis, Pseudomonas aeruginosa, Acinetobacter lwoffii, and Vibrio cholerae. Mueller–Hinton agar plates were swabbed with the bacteria and 6-mm diameter wells made on the agar plate using cork borer prior to the addition of 100 μL of 100 mg/ml T. chantrieri crude extract into each hole. After overnight incubation at 37 °C, the inhibition zone of each sample was measured. The experiments were performed in duplicate. MIC was investigated using three-fold dilutions, with T. chantrieri crude extract concentrations as follows: 100.00, 33.33, 11.11, 3.70, 1.23, 0.41, and 0.14 mg/ml. Distilled water was used as the non-inhibition control.
Statistical Analysis
MTT assay data were represented as mean
Results
Participants
Due to the concealment of anti-inflammatory drug use, 2 of 10 participants were excluded; therefore, blood samples from only eight the elderly (age ≥ 65 years) were tested. Participant clinical characteristics were summarized in Table 1. Average participant age was 71 years (range 65-75 years); seven participants were female, and one was male. Five of eight participants had hypertension (systolic blood pressure ≥140 mm Hg); two participant had type 2 diabetes mellitus (HbA1C ≥ 7.0%); and six had dyslipidemia (TC ≥ 200 mg/dL, and/or HDL-C < 40 mg/dL, and/or LDL-C ≥ 120 mg/dL, and/or TG ≥ 150 mg/dL). Dyslipidemia was determined according to the Japan Atherosclerosis Society (JAS) guidelines.20–22 Herein, two of eight participants took oral medications for their underlying illness treatments.
Participant (the Elderly) Clinical Characteristics.

Note: HbA1C, the glycated hemoglobin A1c; ALT, alkaline phosphatase; TC, total cholesterol; TG, triglyceride; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; WBC count, white blood cell count.
Cytotoxicity Testing
The effects of T. chantrieri crude extract treatment (100 and 300 μg/ml) on PBMCs and PMNs from three participants were evaluated by MTT assay after 30 min of incubation. The average viability of PBMCs at 100 (T100) and 300 (T300) μg/ml was greater than 90% (Figure 4A). The average viability of PMNs at 100 and 300 μg/ml was also greater than 90% (Figure 4B).

Cytotoxic effect of 100 and 300 μg/mL Tacca chantrieri crude extract on PBMC and PMN viability as assessed by MTT assay. Samples were collected from three the elderly. Untreated (black bar) and treated (gray bar) group data are shown. (A) PBMCs treated with 100 (T100) and 300 μg/mL (T300) of T. chantrieri crude extract showed greater than 90% cell viability. (B) Treated PMNs also displayed more than 90% cell viability.
T. chantrieri Crude Extract Changed the Phenotypes of CD4+ T Cells and Their Subsets
PBMCs obtained from the eight participants were incubated without (Untreated) or with 50 (T50), 100 (T100), 200 (T200), and 300 (T300) μg/ml of T. chantrieri crude extract for 30 min. The median percentage of CD4+ T cells at 100 μg/ml was significantly decreased (p = 0.015) compared with the untreated group (Figure 5A). No significant effects were observed with 50, 200, and 300 μg/ml (Figure 5A). There was no statistically significant difference in CD28null T cell frequency (Figure 5B). Regarding NKG2D expression, the median percentage of CD4+ T cells expressing NKG2D was significantly decreased compared with the untreated group at 50, 200, and 300 μg/ml (p = 0.015, p = 0.007, and p = 0.007, respectively; Figure 5C). There was also reduced at 100 μg/ml. Moreover, aberrant CD4+CD28+NKG2D+ T cells were significantly decreased at 50, 200, and 300 μg/ml compared with the untreated group (p = 0.023, p = 0.007, and p = 0.007, respectively; Figure 5D). Statistical significance was also attained for the CD4+CD28null NKG2D+ T cell subset. At 50 and 200 μg/ml, the median percentage of CD4+CD28null NKG2D+ T cells was significantly decreased compared with the untreated group (p = 0.007 and p = 0.031, respectively; Figure 5E).

Effect of Tacca chantrieri crude extract on phenotypes of CD4+ T cells and their subsets in the elderly. Peripheral blood mononuclear cells from the elderly (N = 8) treated with 50 (T50), 100 (T100), 200 (T200), and 300 μg/mL (T300) of crude extract (gray bars) compared with the untreated group (U; black bar). (A) The percentage of CD3+CD4+ T cells at 100 μg/mL significantly decreased compared with the untreated group. (B) Statistical significance not attained for CD28null T cell changes. (C) CD3+CD4+NKG2D+ T cells significantly decreased at 50, 200, and 300 μg/mL compared with the untreated group. (D) The percentage of non-classical CD4+CD28+NKG2D+ T cells significantly decreased at 50, 200, and 300 μg/mL compared with the untreated group. (E) At 50 and 200 μg/mL, the median percentage of CD4+CD28null NKG2D+ T cells significantly decreased compared with the untreated group.
T. chantrieri Crude Extract Improved PMNs (Phagocytosis and Oxidative Burst)
Isolated PMNs were obtained from the eight participants. The data were analyzed in a pairwise manner, comparing the untreated group (black box) and the treated group (gray circle) with 100 μg/ml of T. chantrieri crude extract. The percentages of phagocytosis, phago-burst, and oxidative burst at 5, 15, and 30 min were analyzed. The percentages of phagocytosis and phago-burst at 15 min in treated samples were significantly increased compared with untreated samples (p = 0.039; Figure 6A and p = 0.015; Figure 6B, respectively). However, the percentages of oxidative burst did not attain statistical significance at any time point (Figure 6C).

Effect of Tacca chantrieri crude extract on phagocytosis and oxidative burst. Isolated PMNs were collected from eight the elderly and co-incubated with 100 μg/mL of crude extract for 30 min. Phagocytosis and oxidative burst experiments were performed at 5, 15, and 30 min. Treated samples are indicated (gray circles). (A) Samples show a significant increase in phagocytosis compared with untreated samples (black box) at 15 min only. (B) Phago-burst significantly increased at 15 min in treated samples. (C) Statistical significance not attained for oxidative burst at any time point.
Inhibitory Effect of T. chantrieri Crude Extract Against Pathogenic and Opportunistic bacteria
The agar well diffusion method was used to evaluate antimicrobial effect. Antimicrobial effects and MIC values were shown in Figures 7A and 7B, respectively. S. aureus, S. epidermidis, N. sicca, and A. lwoffii were suppressed by 100 mg/ml of T. chantrieri crude extract. Average inhibition zones were 14.9, 14.2, 16.0, and 11.1 mm, respectively (Table 2). The MIC investigation found that T. chantrieri crude extract inhibited S. epidermidis growth at 1.23 mg/ml, S. aureus at 3.70 mg/ml, N. sicca at 33.33 mg/ml, and A. lwoffii at 100.00 mg/ml (Table 3).

Inhibitory effect and the minimum inhibition concentration (MIC) investigation of Tacca chantrieri crude extract against pathogenic and opportunistic bacteria. (A) At 100 mg/mL, T. chantrieri crude extract inhibited Staphylococcus aureus (positive inhibition); Enterococcus faecalis was not inhibited (negative inhibition). Distilled water (DW) was used as the non–inhibition control. (B) The MIC was investigated from positive inhibition samples. Lanes 1 to 7 represent 100.00, 33.33, 11.11, 4.70, 1.23, 0.41, and 0.14 mg/mL (three–fold dilutions), respectively. The lane marked with a hyphen (–) represents the non–inhibition control using DW. The MIC for S. aureus was 3.70 mg/mL.
Antimicrobial Activity of 100 mg/ml Tacca chantrieri Crude Extract Against Tested Bacteria.

Minimum Inhibition Concentration (MIC) Evaluation of Tacca chantrieri Crude Extract Against Tested Bacteria.

Note: −, no growth; +, growth.
Discussion
T. chantrieri extract, rich in phenolic and flavonoid compounds, is frequently utilized in alternative/complementary medicine due to its known analgesic, antipyretic, anti-inflammatory, antioxidant, 23 anti-cancer, 15 and antimicrobial effects. 16 Flavonoids have been reported to inhibit arachidonic acid metabolism through enzyme pathways, conferring their anti-inflammatory and anti-thrombogenic properties.24,25 Key phytochemicals in T. chantrieri extract include diarylheptanoids, 26 steroidal saponins, 27 and sterol saponins. 28 Saponins are amphipathic molecules which can enter the cells via endocytosis and inhibit the activation of AP-1, NF-κB, and protein kinase C (PKC) involved in inflammatory signaling pathways. 29 Saponins also decrease pro-inflammatory mediators, enzymes, and cytokines.30,31
As is well known, organ dysfunction and impaired immune function (immunosenescence) are well documented in the elderly, along with aberrant T cell phenotypes and phagocytosis. Therefore, this study investigated the effects of T. chantrieri crude extract on CD4+ T cell phenotypes and PMN function in this population. This work reported the preliminary outcomes of T. chantrieri crude extract on innate and adaptive immune system function.
Eight elderly participants were enrolled in the study (age ≥65 years, range 65-75 years). Of the eight participants, 62.5% had hypertension (5/8), 25.0% had type 2 diabetes mellitus (2/8), and 75.0% had dyslipidemia (6/8). Normal creatinine and ALT levels were observed in all participants, suggesting normal kidney and liver function. However, three participants had mild anemia, which is common in this population due to the aging process. In the elderly, conditions such as dyslipidemia and/or hypertension and/or diabetes can be associated with higher chronic low-grade inflammation and drive dysregulation of immune cell functions.32–35
Of the eight participants, two (2/8) took oral medications to manage underlying conditions: one took only metformin for hyperglycemia, and another person took metformin, losartan combined with amlodipine for blood pressure control and simvastatin for the regulation of lipid profiles. Some reports describe the effects of these pharmacologic agents on immune responses evaluated in this study. Metformin [which reduces blood sugar via activating the adenosine monophosphate-activated protein kinase (AMPK) pathway] could exert an anti-inflammatory effect on CD4+ T cells by restraining the proliferation of isolated human CD4+ T cells (in vitro) under hyperglycemic conditions 36 ; whereas its effect on NKG2D-expressing CD4+ T cells and their subsets has not been reported. Interestingly, Park DW et al (2013) 37 showed that pretreating PMNs with metformin could improve phagocytosis and bacteria killing. For simvastatin, the distribution of human CD4+ T cells after 6 weeks of after treatment did not change 38 ; whereas its effects on NKG2D expression have not been investigated. Regarding PMN function, it was reported that simvastatin did not negatively impact phagocytic and bacteria killing ability. Simvastatin potentially improved PMN function via the neutrophil extracellular traps formation (NETosis) and chemotaxis. 39 Studies on the effects of losartan and amlodipine remain inconclusive. However, these drugs are unlikely to have impacted the outcomes because the experiment analyzed only changing after treatment compared to the untreated state observed within the same individual under identical conditions.
The cytotoxic effect of T. chantrieri crude extract on PBMCs and PMNs was evaluated by MTT assay. This method is more reliable than trypan blue staining as it measures cell activity in linear relation to absorbance and has a higher sensitivity than trypan blue exclusion assay. 40 The cell viability of most PBMCs and PMNs after incubation with 100 μg/ml crude extract for 30 min was greater than 90%, indicating low cytotoxicity (acceptable: greater than 90%. 41 The 300 μg/ml concentration also showed a cell viability greater than 90%. However, some results showed more than 100% of viable PBMCs and PMNs in the treated group compared with the untreated group. This is probably because some of the plant extract's components interfere with MTT, reducing MTT to formazan and leading to an overestimation of MTT results. 42
We next investigated the effect of T. chantrieri crude extract on changing CD4+ T cells and their subset phenotypes by comparing the treated group (using concentrations of 50, 100, 200, and 300 μg/ml) to the untreated group. Flow cytometric analysis of cell surface staining was performed. At low-cytotoxicity dose, the results displayed a statistically significant decrease of CD4+ T cells after treatment with 100 μg/ml T. chantrieri crude extract. It is possible that at this dose of crude extract might promote downmodulation of CD4 molecules on their surface via transient internalization that might be a consequence of cell activation, while the cells remain alive. Another interesting aspect, downmodulation of CD4 molecules might be a noteworthy point in terms of anti-inflammatory effect and maintaining of homeostasis. The evidence represented that a periphery T cell could downmodulate CD4 or CD8 molecules and sequentially convert into CD3+CD4-CD8- double negative (DN) T cells, affecting as acquire regulatory functions to promote peripheral tolerance, limiting autoimmune response, modulation of inflammation, anti-tumor immunity, and anti-infection effect.43–46 However, these mechanisms and associations have not been conclusively established and therefore warrant further study in the future. Previously, there were many natural herbal extracts that affect the decreasing of CD4+ T cell number, CD4 molecules or downmodulation of CD4+ T cell activation including root extract from several Thymus species, 47 and leave extract from Arrabidaea chica species 48 and Azadirachta indica. 49
NKG2D (CD314) is normally expressed on NK, CD8+ T, and NKT-like cells, but not on CD4+ T cells. Its expression on CD4+ T cells is considered as aberrant phenotype and is associated with higher chronic low-grade inflammation and age-related diseases which induce higher pro-inflammatory cytokines.2,50,51 Our results exhibited mostly a reduction of CD4+NKG2D+T cells, CD4+CD28+NKG2D+T, and CD4+CD28null NKG2D+T subsets following treatment with 50, 100, 200, and 300 μg/ml of T. chantrieri crude extract. We indicated that this crude extract potentially promoted anti-inflammatory properties by modulating surface NKG2D expression; however, the mechanisms/pathways of this phenomenon require further investigation. Functional cytokine release assays could confirm this anti-inflammatory effect. Increased pro-inflammatory cytokine secretion (such as IL-17 and IFN-γ) has been reported in T cells expressing NKG2D, 3 particularly in older adult populations and individuals with chronic inflammatory conditions. 23 Interestingly, this work represented a pilot study of this crude extract that potentially enhanced immunity through its biological activity by decreasing pro-inflammatory cell surface markers. However, the effect on the CD28null T cell population was not attain statistical significance.
In the innate immune system, PMNs play an important role in eliminating extracellular bacteria and fungi. Their main functions include phagocytosis and oxygen radical production. Phagocytosis is a cellular process which includes pathogen ingestion, activation of internal signaling, phagosome formation, phagolysosome maturation, and digestion. Phagocytic cells produce oxygen species to diminish the engulfed pathogen, known as the oxidative burst process. 52 This study investigated the effect of 100 μg/ml T. chantrieri crude extract on PMNs via phagocytosis and oxidative burst using flow cytometric analysis. This optimal concentration was selected based on a previous report indicating the highest stimulatory activity on phagocytosis of leukocytes. 18 Three time points (5, 15, and 30 min) were selected to observe the phagocytic response, with the aim of detecting the initial ingestion process at 5 min, the climax of ingestion at 15 min, and the end of the digestion process at 30 min. Prior studies reported that S. aureus phagocytosed by PMNs had a lag phase of 2.5–5 min, was killed at a constant rate for 10–30 min, and that bacterial killing declined after 30 min. 53
The current study showed that T. chantrieri crude extract significantly increased phagocytosis and oxidative burst (ex-vivo) at 15 min, suggesting that it could enhance innate immunity in the elderly. Possible mechanisms include promoting the recognition step by upregulating opsonic phagocytic receptors (such as CR and FC receptors) and non-opsonic receptors to ingest bacteria directly 54 and promoting actin polymerization, which is key for phagosome formation via increased Rac1-GTP levels. 55 Moreover, any active compounds in T. chantrieri crude extract possibly enhance PMNs through promoting protein signaling via PKC, leading to the upregulation of reactive oxygen species such as the superoxide anion.56,57 Similarly, previous studies have reported the enhancement of phagocytosis by PMNs following treatment with Guarea kunthiana bark extract; PMNs treated with G. kunthiana exhibited increased phagocytosis of E. coli. 58
Finally, this study investigated the antimicrobial effect of T. chantrieri crude extract on pathogenic and opportunistic bacteria (both gram positive and negative). Using the agar well diffusion technique, 100 mg/ml of T. chantrieri crude extract was found to inhibit gram-positive (S. aureus and S. epidermidis) and gram-negative bacteria (N. sicca and A. lwoffii). MIC values for each bacterium were also determined. The findings suggested that T. chantrieri crude extract could inhibit pathogenic and opportunistic bacteria. A possible mechanism may be the inhibition of enzymes involved in cell wall synthesis, resulting in cell death. 59 The MIC values indicated that T. chantrieri crude extract inhibited gram-positive bacteria more effectively than gram-negative bacteria. This reasonable cause may be attributable to the difference in bacterial structure: unlike gram-positive bacteria, gram-negative bacteria contain an outer membrane which acts as a permeability barrier. 60 Both N. sicca and A. lwoffii cause opportunistic infections in several organs, particularly in immunocompromised patients or patients with debilitations. T. chantrieri extract may therefore be a promising growth-inhibiting ingredient in natural antiseptics/disinfectants. In previous studies, partially purified T. chantrieri extract inhibited gram-positive bacteria such as S. aureus, S. pyogenes, E. faecalis, and B. cereus, and gram-negative bacteria such as E. cloacae, S. marcescens, and A. hydrophila. Evidently, S. aureus and S. pyogenes exhibited MIC values of 1.56 and 25.0 mg/ml, respectively. 16 Compared with previous studies, our findings showed MIC value for S. aureus was 3.7 mg/ml; S. pyogenes was not inhibited, which may be due to differences in extraction methods.
Overall, our findings indicated that T. chantrieri crude extract possibly produced a coordinated immunomodulatory effect with low cytotoxicity: it seemingly altered CD4+ T cell numbers in a manner consistent with transient CD4 internalization and downmodulated stress- or aberrant NKG2D-expressing CD4+ T subsets, probably suggesting an anti-inflammatory outcome. Simultaneously, this extract possibly enhanced the innate effector functions, markedly increasing phagocytosis and oxidative burst (particularly at 15 min), which could possibly improve early microbial clearance in the elderly. Additionally, T. chantrieri crude extract also exhibited direct antimicrobial activity against pathogenic and opportunistic gram-positive and gram-negative bacteria. Collectively, these outcomes suggest that the extract potentially helps restore immunological balance and homeostasis by dampening maladaptive adaptive inflammation while augmenting frontline innate defenses and providing complementary bacteriostatic activity. Nevertheless, further mechanistic and dose optimization studies should be conducted and analyzed.
Despite these findings, this study was only a pilot ex-vivo exploration. Investigations of the mechanisms of intracellular signaling pathways and other PBMC and PMN functional assays are required, as well as in-vivo studies and larger sample sizes. Specific active compounds affecting immunity responses in the elderly should also be evaluated.
Conclusions
T. chantrieri crude extract could possibly promote immunity in the elderly, recovering CD4+ T cell phenotypes and increasing phagocytosis and the oxidative burst process by PMNs. Protection against immunosenescence and immune dysfunction may reduce the burden of age-related health problems. Moreover, T. chantrieri also demonstrated antimicrobial activity against pathogenic/opportunistic bacteria. These findings indicated its potential utility in alternative and complementary medicine and as an antibacterial agent for infection prevention/treatment. However, as this is an ex-vivo study, functional assays in-vivo and a larger sample size are required to validate the findings.
Footnotes
Acknowledgments
We would like to express thankfulness for all supporting of all staffs of the Microbiology Laboratory Unit and Flow Cytometry Unit of the Department of Microbiology, Faculty of Medicine, Khon Kaen University.
ORCID iDs
Ethical Approval
This project was approved by the Ethics Committee of Khon Kaen University, Khon Kaen, Thailand (HE-661164) on April 11, 2023. All subjects provided written informed consent prior to enrollment in the study.
Author's Contributions
Chitsanucha Chatsakulphanit; designs and conducts the study, collects the sample, analyses the data, prepared the first draft of the manuscript. Supranee Phanthanawiboon, Malinee Thanee, Kanin Salao, Patcharaporn Tippayawat, Chanvit Leelayuwat, Nisa Chuangchot, Auttawit Sirichoat, Arnone Nithichanon design the study, edited the manuscript. Chatcharin Kamsom, Saitharn Papalee, Chalida Chuenchom; collected the sample and analyses the data. Wisitsak Phoksawat; designs and conducts the study, edited and finalized the manuscript, and co-responding. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Invitation Research Funding (grant number IN66067), Faculty of Medicine, Khon Kaen University, The Centre for Research and Development of Medical Diagnostic Laboratories (CMDL), Faculty of Associated Medical Sciences, Khon Kaen University, Research and Diagnostic Center for Emerging Infectious Diseases (RCEID), Khon Kaen University, Khon Kaen, Thailand.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
The data generated in the present study are included in the figures and/or tables of this article.
