Abstract
Background
Warfarin, a commonly used anticoagulant, interacts with medicinal plants, potentially complicating patient management. Clinical Decision Resources (CDRs) provide guidance on these interactions, but inconsistencies across resources may impact clinical decisions. This study aims to evaluate the consistency of interaction reports between warfarin and medicinal plants across four online CDRs.
Methods
A cross-sectional study was conducted. Medicinal plants assessed were selected from the World Health Organization's Phytotherapy Manual. Interaction reports were manually reviewed across four CDRs: UpToDate, ClinicalKey, DynaMed, and DrugBank. Interactions were categorized by severity as mild, moderate or severe.
Results
Of 141 medicinal plants reviewed, 28 were documented as interacting with warfarin in at least one of the four assessed CDRs. DynaMed reported interactions for 18 plants, ClinicalKey for 15, UpToDate for 13, and DrugBank for 3. Only one plant (Ginseng - Panax ginseng) was consistently identified across all CDRs, although discrepancies in the direction of the interaction (whether it increased or decreased anticoagulant effect) were observed. Regarding interaction severity classifications, there was substantial variability, with ClinicalKey identifying the highest proportion of severe interactions (32.1%).
Conclusion
We identified significant variability among CDRs in reporting interactions between warfarin and medicinal plants, which may lead to inconsistencies in clinical decision-making. To ensure more consistent and reliable patient care, standardized and comprehensive methodologies for assessing these interactions are needed.
Introduction
Warfarin is a widely used, low-cost anticoagulant, 1 that inhibits the enzyme epoxide reductase, thereby reducing active vitamin K levels, which are essential for the synthesis of coagulation factors II, VII, IX, and X. This disruption interferes with the coagulation cascade, prolonging clotting time.2,3
However, its use carries considerable risks, such as severe bleeding due to its narrow therapeutic margin and the need for constant monitoring through the International Normalized Ratio (INR). 4 In addition, the interaction of warfarin with various drugs, foods, and medicinal plants can complicate patient management, emphasizing the importance of proper patient education to minimize risks and maximize therapeutic benefits. 5
Medicinal plants are widely used in both developing and developed countries. 6 As of 2019, about 50,000 species of plants with potential medicinal properties have been identified. Also, according to the World Health Organization (WHO), 80% of the population in developing countries may have used traditional medicine. 7 In this context, interactions between conventional drugs and medicinal plants are frequent. 8
Clinical Decision Resources (CDRs) are important tools for supporting healthcare decision-making. Many of these resources include data on drug-plant interactions, enabling healthcare professionals to effectively manage and advise patients who use medicinal plants alongside conventional treatments, thereby enhancing both treatment safety and effectiveness. 9 However, the information on interactions provided by different CDRs may vary, leading to clinical variability that can jeopardize patient management. 10
Therefore, the aim of this study is to assess the consistency of recommendations across four online CDRs regarding the interactions between warfarin and medicinal plants.
Methods
Study Design
A cross-sectional study was carried out.
Selection of Medicinal Plants and CDRs
We selected medicinal plants that may have pharmacological interactions with Warfarin. For this, we reviewed the four volumes of the “WHO Monographs on Selected Medicinal Plants”, which are official publications of the WHO that compiled the most used medicinal plants worldwide, describing their biological properties, therapeutic effects, and forms of use. 11
We compiled a list of all the plants included in these manuals. Then, between June and August 2024, four researchers (LJUC, PVM, EBE, and ASH) identified potential interactions between each plant and warfarin, by searching in four CDRs: UpToDate-Lexicomp Online (https://www.uptodate.com/drug-interactions), 12 ClinicalKey (https://www.clinicalkey.es), 13 DynaMed (https://www.dynamed.com), 14 and DrugBank (https://go.drugbank.com/drug-interaction-checker). 15
We selected UpToDate, ClinicalKey, and DynaMed as they are considered among the most comprehensive CDRs available online.16–19 Additionally, we included DrugBank due to its free access, which provides an invaluable resource for healthcare professionals with limited access to paid tools who need to investigate plant interactions. 20 All four CDRs feature a section on medical interactions, including those involving plants.
Procedures and Variables
Each researcher was assigned one CDR to extract data regarding interactions between the plants and warfarin. Later, each researcher was randomly reassigned to a different CDR than their original assignment, to review the data extraction. This approach ensured that every interaction was assessed by two independent researchers.
We accessed each CDR's website and utilized their specific tools for drug interaction assessments. For UpToDate, DynaMed, and DrugBank, we employed their respective “Drug Interaction Checker” tools, allowing us to select both warfarin and the medicinal plant of interest to obtain detailed interaction results. For ClinicalKey, we navigated to the “Drug Information” section for warfarin and reviewed the “Interactions” subsection, which provided a comprehensive list of potential interactions with warfarin, including drugs, plants, and other substances. We thoroughly analyzed this information to identify the relevant plants. In each CDR, we used both the commercial and scientific names of the plants, as listed in our WHO manuals. Additionally, we compiled a complete list of all names found across the CDRs (Supplementary Table 1).
For each identified interaction, we recorded its severity. Since categories of interaction severity varied between CDRs, we applied the Common Terminology Criteria for Adverse Events (CTCAE) 21 to uniformly classify them as mild, moderate, or severe (Table 1).
Severity Levels of Adverse Events According to the Common Terminology Criteria for Adverse Events (CTCAE) and Standardization of Terms Across CDRs.

We also extracted the direction of the interaction (whether it increased or decreased anticoagulant effect, according to the CDRs information), as well as the type of evidence cited by each CDR to support the interaction information (systematic reviews, clinical trials, observational studies, case reports, or preclinical studies).
Statistical Analysis
The results were extracted into an Excel database for further analysis. Results were reported descriptively using absolute and relative frequencies.
Results
Of the 141 medicinal plants included in the “WHO Monographs on Selected Medicinal Plants”, 28 plants have been documented to interact with warfarin in at least one of the four assessed CDRs.
Our analysis of these CDRs revealed a great variability in interaction reporting. DynaMed identified interactions for 18 plants, including 3 (10.7%) severe interactions. ClinicalKey reported interactions for 15 plants, with 9 severe interactions (32.1%). UpToDate documented interactions for 13 plants but did not report any severe interactions. DrugBank reported interactions for only 3 plants, with no severe interactions (Figure 1).
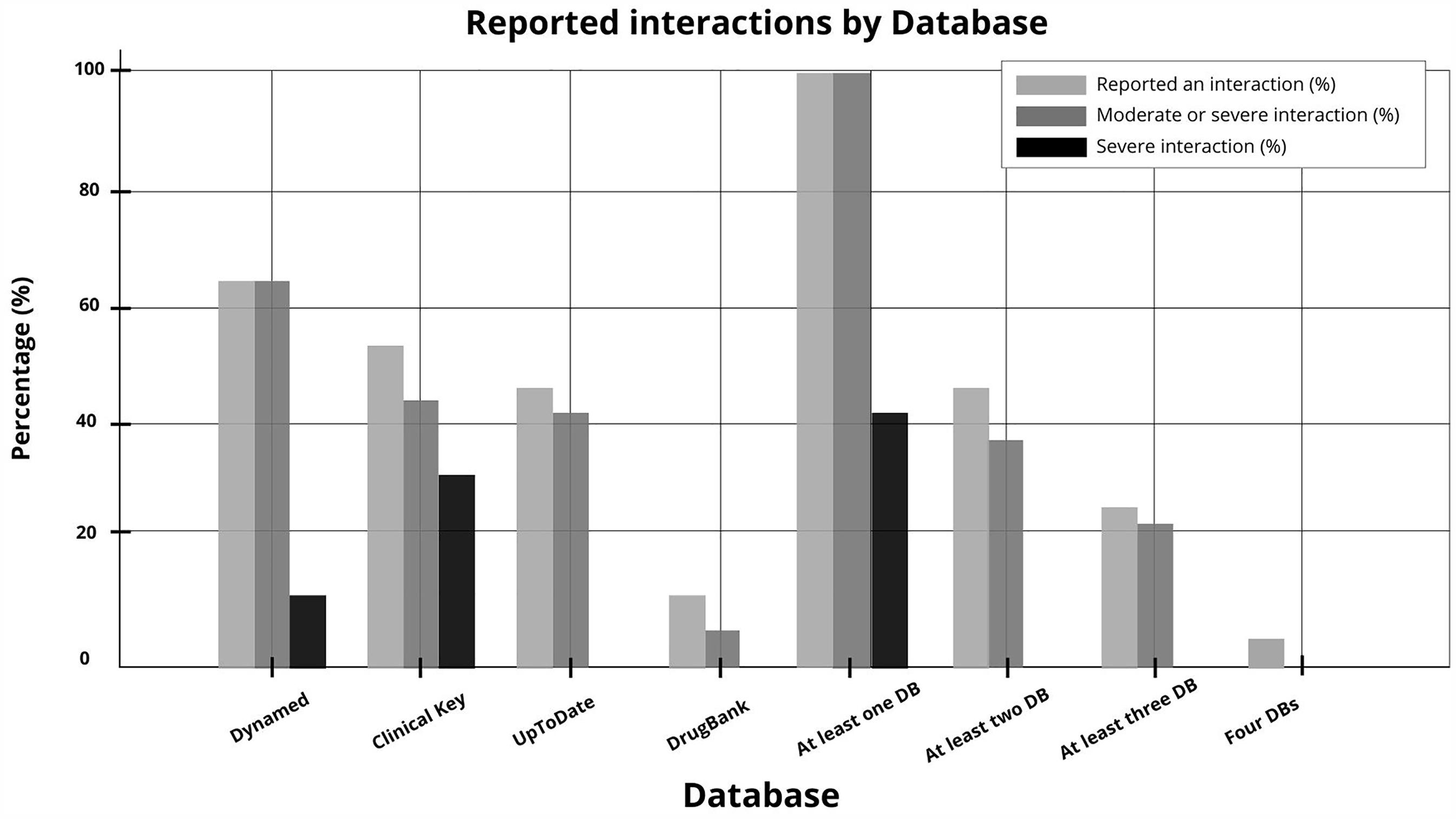
Percentage of plants identified with interactions with warfarin, based on the studied CDRs (n = 28).
In terms of overlap, 28 plants were reported to have interactions with warfarin in at least one CDR, 13 in at least two, 7 in at least three, and only one plant—Panax ginseng—was noted to interact with warfarin across all four CDRs. Regarding the direction of interaction, of the 28 plants, in 10 (35.7%) the CDRs indicated a reduction in warfarin's anticoagulant effect, in 14 (50.0%) the CDRs indicated an increase in the anticoagulant effect, and in 4 (14.3%) the CDRs provided conflicting results (with some indicating an increase and others a decrease in warfarin's anticoagulant effect) (Table 2).
Interaction Between Warfarin and Medicinal Plants According to Assessed CDRs.
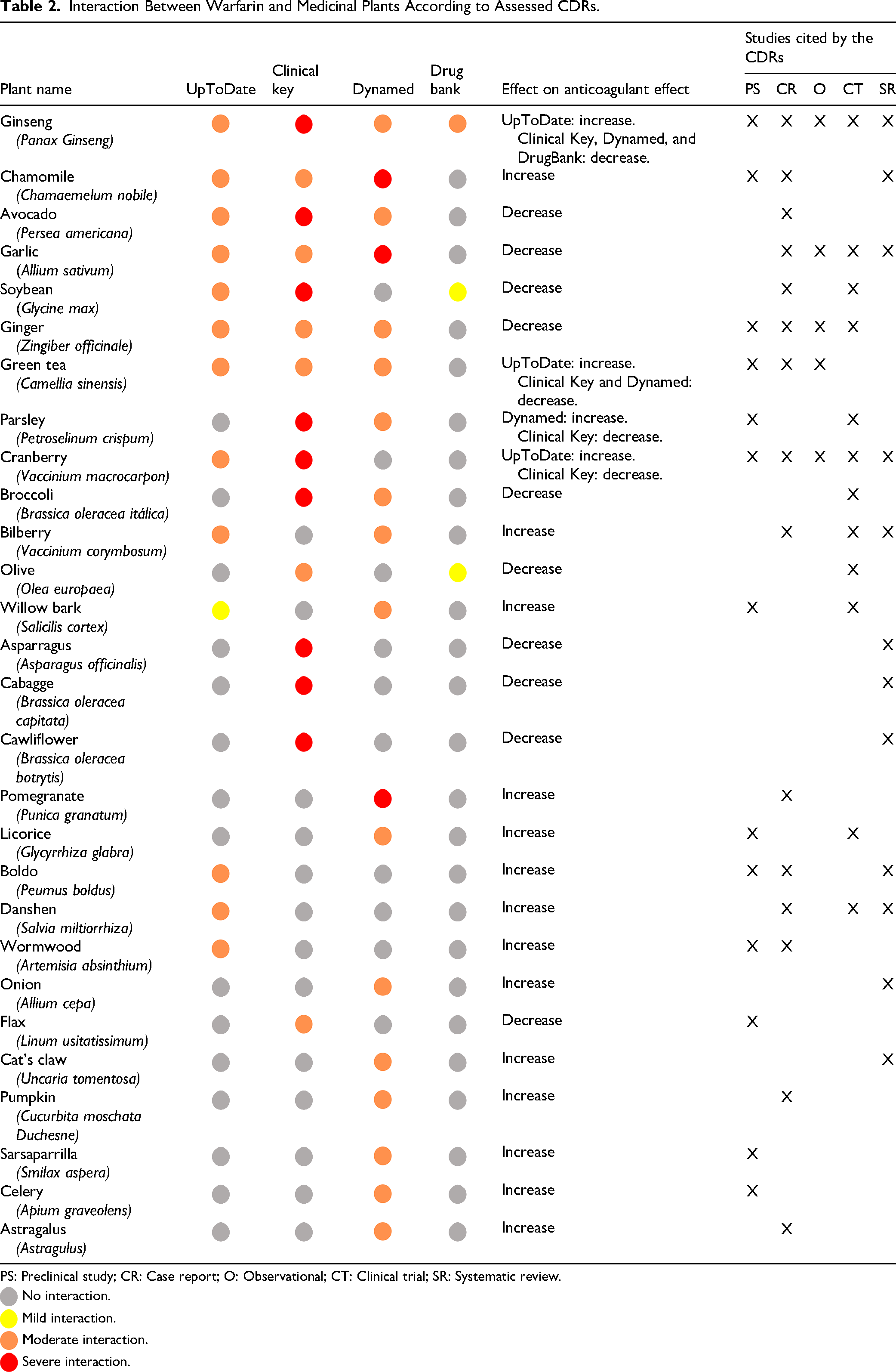
PS: Preclinical study; CR: Case report; O: Observational; CT: Clinical trial; SR: Systematic review.
 No interaction.
No interaction.
 Mild interaction.
Mild interaction.
 Moderate interaction.
Moderate interaction.
 Severe interaction.
Severe interaction.
Regarding the types of studies cited by the CDRs to support interaction statements, case reports were referenced for 15 plants (53.6%), preclinical studies for 13 plants (46.4%), clinical trials for 12 plants (42.9%), systematic reviews for 12 plants (42.9%), and observational studies for 5 plants (17.9%) (Table 2).
Discussion
Main Results
This study identified significant inconsistencies in the reported interactions between medicinal plants and warfarin across the four assessed CDRs. Each CDR differed substantially in both the number of plants identified as having interactions and the severity of these interactions. Notably, only one plant was consistently reported across all CDRs. Additionally, for four plants, the CDRs provided conflicting statements regarding the direction of the interaction. Case reports were the most frequently cited type of publication to support these interaction statements.
Discrepancies Across CDRs
We identified notable inconsistencies across CDRs regarding interactions between warfarin and medicinal plants. While previous studies have highlighted discrepancies among CDRs22–26 we found no studies that specifically assess these inconsistencies in drug-plant interactions as ours did. This evidence underscores the challenge of reaching consensus on recommendations across CDRs, particularly in drug-plant interactions, where CDRs tend to have less information compared to drug-drug interactions. 27
Notably, although DynaMed documented the highest number of interactions between warfarin and medicinal plants, it only identified interactions for 18 of the 28 plants reported by any of the CDRs (64.3%). Additionally, it recognized severe interactions for only 12 of the 28 plants that were classified as having severe interactions by any CDR. This suggests that even the most comprehensive CDR may miss important interactions, underscoring the variability and limitations across resources. Such discrepancies raise concerns about the completeness and reliability of CDRs when making clinical decisions, especially in the context of drug-plant interactions.
In addition to inconsistencies in identifying interactions, we also found disagreement regarding the direction of these interactions. Of the 13 interactions reported in at least two CDRs, four showed discrepancies in whether the plant increased or decreased warfarin's anticoagulant effect. A key issue lies in understanding how conclusions are drawn about drug-plant interactions. Currently, there is no standardized consensus for developing drug interaction information in CDRs. This lack of uniformity leads to variability in methodologies, information sources, and data interpretation across different CDRs, contributing to the observed inconsistencies.28–30
Clinical Implications
Variability in reports of warfarin-plant interactions can directly impact clinical decision-making, increasing the risk of adverse events.31,32 Given the critical nature of anticoagulant therapy, discrepancies in CDR information may compromise both treatment safety and effectiveness, potentially leading to inadequate risk assessments by healthcare professionals.
In our study, ginseng (Panax ginseng) was the only plant reported to interact across all four CDRs, underscoring its clinical relevance. However, it is concerning to observe contradictions in the recommendations regarding ginseng's potential mechanisms of action. For instance, DynaMed, ClinicalKey, and DrugBank suggest a decrease in anticoagulant effect, primarily based on preclinical studies, case reports, and randomized clinical trials.33–37 One such study is a clinical trial by Yuan, 37 which found that ginseng use in patients taking warfarin decreased INR levels after two weeks, thereby diminishing its anticoagulant effect.
In contrast, UpToDate reports that ginseng increase in anticoagulant effect, citing observational studies, randomized clinical trials, and systematic reviews.38–40 One such study is Ulbricht's systematic review, 40 which examines the pharmacokinetic properties and interactions of ginseng with various drugs, suggesting that ginseng inhibits platelet activity, potentially increasing the risk of bleeding.
These discrepancies, likely stemming from the divergent sources of evidence each CDR relies on, can cause confusion when adjusting concurrent therapies or determining contraindications based on specific pathologies.
Limitations and Strengths
While we focused on prominent CDRs, there may be other valuable resources not included in our analysis. Additionally, our search was limited to plants listed in the WHO Monographs on Selected Medicinal Plants. Although this is a comprehensive list of medicinal plants, it does not encompass other types of herbs and foods. Furthermore, we only assessed interactions with warfarin, so our results cannot be extrapolated to other drug-plant or drug-food interactions.
Nevertheless, this study enhances our understanding of the discrepancies among widely used CDRs concerning the interactions studied. We conducted a thorough review process, with two authors independently reviewing each search and all extracted data.
Conclusion
In conclusion, this study revealed significant inconsistencies in the reported interactions between medicinal plants and warfarin among the four assessed CDRs. There was substantial variation in both the number of plants identified as having interactions and the severity of these interactions across the different resources. Furthermore, for four plants, the CDRs provided conflicting information regarding the direction of the interactions. Case reports emerged as the most commonly cited publications supporting these interaction statements..
Supplemental Material
sj-docx-1-chp-10.1177_2515690X251334445 - Supplemental material for Inconsistent Reporting of Interactions Between Warfarin and Medicinal Plants Across Clinical Decision Support Resources
Supplemental material, sj-docx-1-chp-10.1177_2515690X251334445 for Inconsistent Reporting of Interactions Between Warfarin and Medicinal Plants Across Clinical Decision Support Resources by Leonardo J. Uribe-Cavero, MS, Patricia J. Vera-Maccha, MS, Anthony Siguas-Huasasquiche, MS, Elias E. Bohorquez-Espino, MS, and Alvaro Taype-Rondan, MD in Journal of Evidence-Based Integrative Medicine
Footnotes
Acknowledgments
None.
Author Contributions
L.U. conceived the work. L.U., P.V., A.S. and E.B. conducted the data extraction L.U., P.V., A.S., E.B. and A.T. contributed to data interpretation. L.U., P.V., A.S., E.B. and A.T. performed the analysis and drafted the original manuscript. A.T. provided critical feedback. All authors contributed to revising the paper and approved the final manuscript.
Data Avaibility Statement
Data from the CDRs were derived from the following web sources: UpToDate-Lexicomp Online (https://www.uptodate.com/drug-interactions, available by subscription); ClinicalKey (https://www.clinicalkey.es, available by subscription), DynaMed (https://www.dynamed.com, available by subscription) and DrugBank (![]() , available in the public domain).
, available in the public domain).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent statements
This study did not involve human subjects, patients, or the administration of any substances.
Funding
This study was self-funded by the authors.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
