Abstract
Hypertrophic scarring is an aberrant wound-healing response to reestablish dermal integrity after an injury and can cause significant abnormalities in physical, aesthetic, functional, and psychological symptoms, impacting the patient's quality of life. There is currently no gold standard for preventing and treating hypertrophic scars. Therefore, many researchers have attempted to search for antihypertrophic scar agents with greater efficacy and fewer side effects. Natural therapeutics are becoming attractive as potential alternative anti-scarring agents because of their high efficacy, safety, biocompatibility, low cost, and easy accessibility. This review demonstrates various kinds of natural product-based therapeutics, including onion, vitamin E, Gotu kola, green tea, resveratrol, emodin, curcumin, and others, in terms of their mechanisms of action, evidence of efficacy and safety, advantages, and disadvantages when used as anti-scarring agents. We reviewed the literature based on data from in vitro, in vivo, and clinical trials. A total of 23 clinical trials were identified in this review; most clinical trials were ranked as having uncertain results (level of evidence 2b; n = 16). Although these natural products showed beneficial effects in both in vitro and in vivo studies of potential anti-scarring agents, there was limited clinical evidence to support their efficacy due to the limited quality of the studies, with individual flaws including small sample sizes, poor randomization, and blinding, and short follow-up durations. More robust and well-designed clinical trials with large-scale and prolonged follow-up durations are required to clarify the benefits and risks of these agents.
Introduction
Hypertrophic scarring is an aberrant wound-healing response to reestablish dermal integrity after injury. Hypertrophic scars present as erythematous, indurated, and raised dermal scars that remain within the original injury margins.1,2 They are characterized by the expressive amounts of fibroblast-derived extracellular matrix (ECM) at the site of a skin injury, resulting from an imbalance between ECM protein deposition and breakdown during wound repair.2-4 When a hypertrophic scar develops, it can cause significant abnormalities in physical, aesthetic, functional, and psychological symptoms, impacting the patient's quality of life.1,2,4
Currently, there are numerous therapeutic strategies to reduce and prevent the development of hypertrophic scars, such as silicone gel, pressure therapy, intralesional corticosteroid injections, and surgical revision. However, most of these therapeutic approaches remain clinically unsatisfactory for both patients and their providers. They are associated with high recurrent rates, high costs, and adverse effects such as pain, skin atrophy, contact dermatitis, and further scar formation. So far, there is no gold standard in preventing scarring. 5 For this reason, the research and development of antihypertrophic scar agents with greater efficacy and fewer side effects remain an area of significant research interest at present.
In the last decade, the traditional use of natural products for healthcare, including wound and scar care, has renewed global interest due to the high cost and difficult accessibility of orthodox medicines. 6 Almost 80% of people in developing countries have used herbal medicine to treat wounds and prevent scars. 7 Currently, there is a large amount of research demonstrating the therapeutic potential of natural products for wound healing and scar management.8-11 Several reviews summarize the wound-healing effect of natural products.6,7 It is widely accepted that wound healing time is essential in predicting scar development. 3 However, few reviews have focused on the scar management properties of natural products in the stage after wound healing. 12 Furthermore, levels of evidence have not yet been assigned to these reviews on the summarized data. The present study aims to gather the available clinical evidence supporting natural products as potential therapeutic agents for scar development, particularly hypertrophic scars, focusing on the management after wound healing. The potential mechanisms, side effects, and current administration of natural therapeutics are also described in this review. As knowledge of the wound healing process is crucial to understand better the effects of natural product-based therapeutics on hypertrophic scars, the pathomechanism of wound healing and hypertrophic scarring are also reviewed. This review will benefit future research on natural products for potential use as antihypertrophic scar agents.
Methods
To conduct the present narrative review, the literature from PubMed, Science Direct, and Google Scholar was searched, focusing on the effects of natural product-based therapeutics on hypertrophic scars in vitro, in vivo, and clinical trials (only randomized controlled trials; RCTs). The search keywords included “scar”, “hypertrophic scar”, “natural products”, and “medicinal plants”. The individual therapies’ names identified during the search were included, and the identified articles’ reference lists were further searched for relevant literature. All available English articles were reviewed. Studies in cell culture and animal models were considered in association with mechanisms of action. The level of evidence was assigned to each clinical trial using the Oxford Centre for Evidence-based Medicine.13,14
Based on the findings from existing data, we have demonstrated and discussed the following issues: 1) pathomechanism of wound healing and scarring, particularly hypertrophic scarring, and 2) the effects of natural product-based therapeutics on hypertrophic scars, including phytochemicals, mechanisms of action, therapeutic efficacy, safety profile (if any), and examples of commercially available natural-based products.
Results
Pathomechanism of Wound Healing and Scarring, Particularly Hypertrophic Scarring
The wound healing process is comprised of three overlapping phases: inflammation (days 1-3), proliferation (days 4-21), and remodeling (days 21 to year 1). These three phases involve complicated interactions between cells (platelets, inflammatory cells, fibroblasts, endothelial cells, myofibroblasts, and keratinocytes), growth factors (transforming growth factor-beta (TGF-β), fibroblast growth factor (FGF), platelet-derived growth factor (PDGF), and connective tissue growth factor (CTGF)), cytokines, ECM components (collagen, hyaluronan, and fibronectin), and proteolytic enzymes (matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs)). Hypertrophic scars can occur due to an aberration in these processes. 4
The crucial wound-healing process for scar formation begins during the proliferation phase, where the granulation matrix provides a structural framework for wound bridging and vascular ingrowth by angiogenesis. Approximately one week after injury, some fibroblasts differentiate into myofibroblasts, activated by TGF-βI, which synthesize and deposit ECM components. Myofibroblasts, which contain alpha-smooth muscle actin (α-SMA), play a significant role in wound contraction, reducing the wound size. Simultaneously, re-epithelialization occurs by keratinocyte migration and proliferation across the injury site to close the wound surface. When re-epithelialization is complete, the immature scar can progress into the last remodeling phase, where the granulation tissue formation stops via apoptosis of the responsible cells. During this phase, collagen is remodeled and rearranged. The immature collagen type III found in the initial wound is broken down and replaced with mature collagen type I. The equilibrium between collagen deposition and breakdown is vital, regulated by matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs). When this process is disrupted, it results in hypertrophic scarring (Figure 1).2,4,15

The mechanisms of wound healing and scarring.
Hypertrophic scar formation results from an imbalance between ECM protein deposition and breakdown during wound repair. The underlying mechanism is likely due to the aberrant and over-release of growth factors and cytokines and a lack of molecules responsible for cell apoptosis or ECM remodeling. The excessive inflammatory cytokines, including tumor necrosis factor-alpha (TNF-α), interleukin-1beta (IL-1β), IL-6, and IL-8, promote fibroblast proliferation and ECM production, suppress collagenase activity, and enhance collagenase inhibitors activity, leading to abnormal collagen deposition and eventually to scar formation. 15 TGF-β is the most representative cytokine in regulating wound scarring via the TGF-β/Smad signaling pathway. All three TGF-β isoforms (TGF-β1, -β2, and -β3) are released by numerous cells involved in wound healing, mainly macrophages, neutrophils, and platelets. Exaggerated TGF-β1 and -β2 levels stimulate collagen synthesis and prevent its breakdown by reducing MMP activity while promoting TIMP expression, resulting in scar formation. Conversely, TGF-β3 has an antagonist effect and thereby reduces scarring.2,4
There are numerous hypertrophic scar-inducing signaling transduction pathways that play a role in the induction of cell proliferation, migration, and differentiation and the inhibition of cell apoptosis (Figure 2). Among them, the TGF-β/Smad signaling pathway is essential in hypertrophic scar development as it promotes ECM synthesis on fibroblast stimulation and induces fibroblast differentiation into myofibroblasts.2,4,15,16 Other vital signaling pathways are the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt) and mitogen-activated protein kinase (MAPK) pathways. MAPKs, consisting of the extracellular signal-regulated kinase (ERK), p38, and c-Jun N-terminal kinase (JNK) pathways, participate in hypertrophic scars formation and promotion through their involvement in TGF-β signaling. 16 Nonetheless, the complex mechanism of hypertrophic scarring is still not well understood.

TGF-β/Smad signaling pathway and other related signaling pathways (Akt, protein kinase B; ERK, extracellular signal-regulated kinase; JNK, c-Jun N-terminal kinase; MAPK, mitogen-activated protein kinase; PI3K, phosphatidylinositol 3-kinase; TGF-β, transforming growth factor-beta).
Effects of Natural Product-Based Therapeutics on Hypertrophic Scars
Some natural products have shown beneficial effects in the management of scars, particularly hypertrophic scars. In this study, natural product-based therapeutics, including onion (Allium cepa L.), vitamin E, Gotu kola (Centella asiatica (L.) Urb.), green tea (Camellia sinensis (L.) Kuntze), resveratrol, emodin, curcumin, and others, are reviewed. The preclinical evidence of the proposed mechanisms of action and the clinical evidence of these natural product-based therapeutics are summarized in detail (Tables 1, 2, and 3, respectively). The pros and cons of these natural product-based therapeutics are also shown in Table 4. Examples of commercially available natural-based products for scars are also provided in this review (Table 5).
Proposed Anti-Scarring Mechanisms of Natural Product-Based Therapeutics in This Review.

Abbreviations: 70S6K, ribosomal protein S6 kinase; Akt, protein kinase B; CBP, CREB-binding protein; COX-2, cyclooxygenase-2; CREB, cAMP response element binding protein; CTGF, connective tissue growth factor; d, day; ECG, epicatechin-3-gallate; ECM, extracellular matrix; EGCG, epigallocatechin-3-gallate; ERK, extracellular signal-regulated kinase; GPx, glutathione peroxidase; HSFs, hypertrophic scar-derived fibroblasts; IGF-I, insulin-like growth factor-I; IL, interleukin; iNOS, inducible nitric oxide synthase; JNK, c-Jun N-terminal kinase; KF, keloid scar-derived fibroblast; MAPK, mitogen-activated protein kinase; MMP-1, matrix metalloproteinase-1; mRNA, messenger ribonucleic acid; mTOR, mammalian target of rapamycin; NFs, normal skin fibroblasts; PAI-1, plasminogen activator inhibitor-1; P-cofilin, phosphorylated cofilin; PDGF-BB, platelet-derived growth factor-BB; PI3K, phosphatidylinositol 3-kinase; PPAR-γ, peroxisome proliferator-activated receptors-gamma; PSF, pathological scar-derived fibroblast; PVA, polyvinyl alcohol; Rheb, Ras homolog enriched in brain; ROS, reactive oxygen species; SIRT1, sirtuin 1; STAT3, signal transducer and activator of transcription-3; TGF-β, transforming growth factor-beta; TGF-βR, transforming growth factor-beta receptor; α-SMA, alpha-smooth muscle actin.
Summary of Proposed Anti-Scarring Mechanisms of Natural Product-Based Therapeutics in This Review.

Abbreviations: 70S6K, ribosomal protein S6 kinase; Akt, protein kinase B; CBP, CREB-binding protein; CREB, cAMP response element binding protein; CTGF, connective tissue growth factor; EGCG, epigallocatechin-3-gallate; ERK, extracellular signal-regulated kinase; GPx, glutathione peroxidase; IGF-I, insulin-like growth factor-I; IL, interleukin; JNK, c-Jun N-terminal kinase; MAPK, mitogen-activated protein kinase; MMP, matrix metalloproteinase; mTOR, mammalian target of rapamycin; PDGF, platelet-derived growth factor; PI3K, phosphatidylinositol 3-kinase; Rheb, Ras homolog enriched in brain; ROS, reactive oxygen species; STAT3, signal transducer and activator of transcription-3; TGF-β, transforming growth factor-beta; TGF-βR, transforming growth factor-beta receptor; TNF-α, tumor necrosis factor-alpha; α-SMA, alpha-smooth muscle actin.
Clinical Trials of Natural Product-Based Therapeutics Identified in This Review.
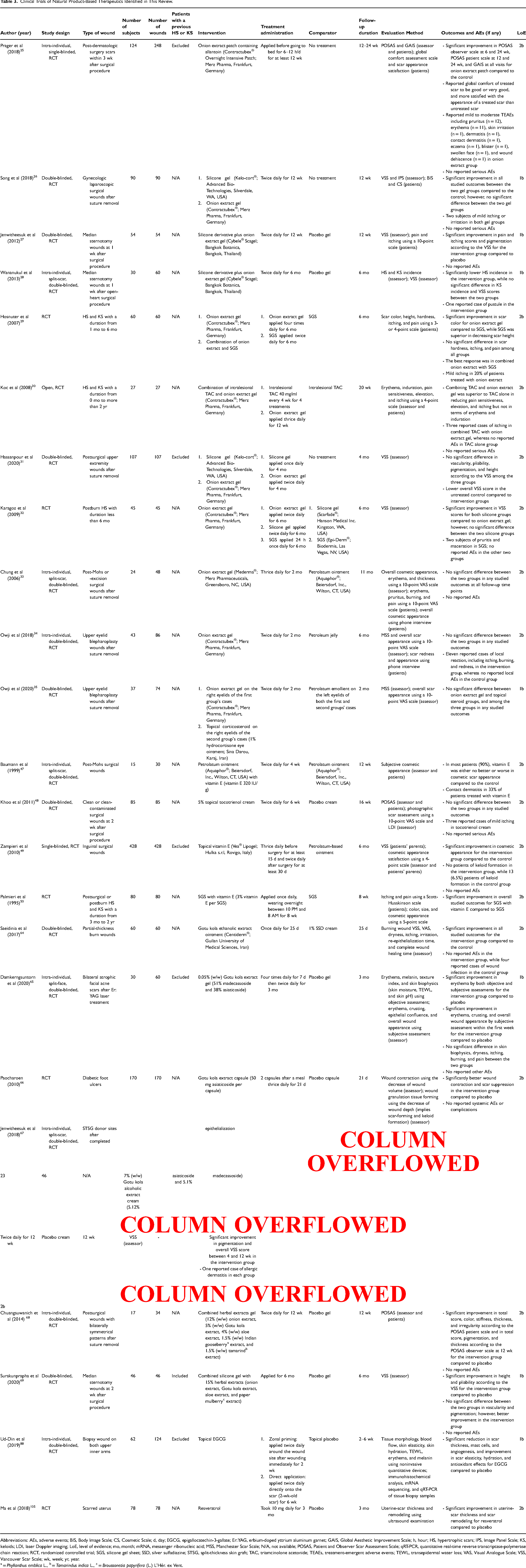
Abbreviations: AEs, adverse events; BIS, Body Image Scale; CS, Cosmetic Scale; d, day; EGCG, epigallocatechin-3-gallate; Er:YAG, erbium-doped yttrium aluminum garnet; GAIS, Global Aesthetic Improvement Scale; h, hour; HS, hypertrophic scars; IPS, Image Panel Scale; KS, keloids; LDI, laser Doppler imaging; LoE, level of evidence; mo, month; mRNA, messenger ribonucleic acid; MSS, Manchester Scar Scale; N/A, not available; POSAS, Patient and Observer Scar Assessment Scale; qRT-PCR, quantitative real-time reverse transcriptase-polymerase chain reaction; RCT, randomized controlled trial; SGS, silicone gel sheet; SSD, silver sulfadiazine; STSG, split-thickness skin graft; TAC, triamcinolone acetonide; TEAEs, treatment-emergent adverse events; TEWL, transepidermal water loss; VAS, Visual Analogue Scale; VSS, Vancouver Scar Scale; wk, week; yr, year.
= Phyllanthus emblica L., b = Tamarindus indica L., c = Broussonetia papyrifera (L.) L'Hér. ex Vent.
Pros and Cons of Natural Product-Based Therapeutics in Scar Management.

Abbreviations: EGCG, epigallocatechin-3-gallate.
Examples of Commercially Available Natural-Based Products for Scars.

Abbreviations: N/A, not available.
= Tamarindus indica L., b = Phyllanthus emblica L., c = Broussonetia papyrifera (L.) L'Hér. ex Vent., d = Theobroma cacao L., e = Carthamus tinctorius L., f = Bulbine frutescens (L.) Willd., g = Magnolia officinalis Rehder & E.H.Wilson, h = Salvia miltiorrhiza Bunge, i = Panax notoginseng (Burkill) F.H.Chen.
Onion
Onion extract is one of the most frequently used constituents in nonprescription scar treatment products. The active ingredients in onion extract are the flavonoids quercetin and kaempferol, which demonstrate anti-inflammatory, antioxidant, antibacterial, antiproliferative, and collagen downregulatory properties.5,17-21 Onion extract and quercetin have been shown to induce the upregulation of MMP-1 expression in vitro and in vivo studies, which is vital for ECM remodeling during wound healing. 22 Thus, this agent may play an antifibrotic role. In addition, quercetin has been demonstrated in vitro to suppress the TGF-β/Smad and insulin-like growth factor-I (IGF-I) signaling pathways.23,24 TGF-β/Smad and IGF-I signalings are known to have a role in pathologic fibroproliferation and abnormal scar formation. Thus, onion extract is a promising anti-scarring agent.
In this review, 11 clinical trials were identified in the current literature regarding the efficacy of onion extract on scars.25-35 The evidence was found to be inconsistent (Table 3). Four of these studies were ranked as level of evidence 1b,26-28,35 and the rest were ranked as level 2b. Some clinical trials of onion extract reported positive experiences with scar appearance. The randomized, single-blinded split scar study of an onion extract patch containing allantoin versus no treatment on post-dermatologic surgery scars (n = 124) found that the overall appearance and cosmesis were significantly better in the treatment group, as assessed by the Global Aesthetic Improvement Scale (GAIS) and Patient and Observer Scar Assessment Scale (POSAS). 25 The double-blinded RCT by Song et al (n = 90) comparing onion extract gel with silicone gel to treat postsurgical scars concluded that both had more efficacy than the untreated control, improving the overall appearance and cosmetic satisfaction. 26 However, the onion extract gel and silicone gel had similar efficacy. Two randomized studies by Jenwitheesuk et al in 2012 (n = 54) 27 and Wananukul et al in 2013 (n = 30), 28 following median sternotomy incision, concluded that onion extract in silicone derivative gel was more effective than placebo gel, with respect to hypertrophic scars incidence, scar pigmentation, and scar symptoms.
In some studies, it was concluded that combining onion extract with other therapies had a more remarkable improvement in scar conditions than individual treatment. The randomized comparative study of hypertrophic and keloid scars by Hosnuter et al (n = 60) concluded that a combination of onion extract gel with silicone gel sheet was the most effective therapeutic agent in improving scar appearance and symptoms, especially for scar height. 29 Another randomized, controlled, comparative study evaluating 27 patients with hypertrophic scars or keloids showed that a combination of intralesional triamcinolone acetonide (TAC) and onion extract gel was superior to TAC alone concerning pain or sensitiveness, elevation, and itching; however, not in erythema and induration. 30
Some clinical trials of onion extract on scar management gave neutral or negative findings compared to other commonly used topical treatments. In a double-blinded RCT of onion extract gel compared with silicone gel in patients with postsurgical upper extremity wounds (n = 107), Hassanpour et al concluded no significant difference in vascularity, pliability, pigmentation, and height, as subjectively assessed using the Vancouver Scar Scale (VSS). 31 However, in an RCT of 45 patients with postburn hypertrophic scars, Karagoz et al reported that both silicone gel and silicone gel sheet were significantly more effective than onion extract in improving cosmesis by using the VSS. 32 Other studies utilizing onion extract gel following Mohs surgery (n = 24) 33 and upper blepharoplasty (n = 43) 34 all showed no significant difference in scar cosmesis between topical onion extract and petroleum emollient. However, some of the possible limitations were that all patients were elderly Caucasians, 33 and the studied surgical sites were eyelids, 34 which are considered low-risk for scar formation. Another double-blinded comparative study comparing onion extract, topical steroid, and petrolatum emollient also showed no significant difference regarding scar appearance after upper blepharoplasty. 35 However, the follow-up time of this study was only 3 months, which may need to be longer to observe clinically evident effects.
In summary, according to the review of published studies, onion extract was superior to no treatment, but was not superior to other scar management treatments. Onion extract may be most effective when used in combination with other therapies. In terms of the safety of onion extract, there have been reported mild to moderate adverse reactions in human studies, including itching, burning, skin irritation, and contact dermatitis; however, they have resolved spontaneously.25,26,29,34
Vitamin E
Vitamin E, an essential micronutrient with lipid-soluble antioxidants, consists of tocopherols and tocotrienols.36,37 Vitamin E is a plant-derived compound. Green leafy vegetables, vegetable oils, nuts, and seeds are good vitamin E sources. 38 The possible mechanism of action of vitamin E on scar management involves the anti-inflammation effect and the inhibitory effects on fibroblast proliferation and collagen synthesis.37,39-41 Additionally, the antioxidant effect may prevent scar formation by decreasing cell damage from free oxygen radicals available during the inflammatory phase and preventing lipid peroxidation in membranes. Therefore, vitamin E acts as a cellular membrane-stabilizing agent.41-44
Vitamin E has been primarily used in cosmetics and in treating acute and chronic dermal wounds.37,45 Though many patients and medical professionals believe that vitamin E may promote wound healing and improve scar cosmesis, there is little scientific evidence supporting these claims.46,47 Four clinical trials were considered in this study, all of which were classified as level of evidence 2b.47-50 A double-blinded RCT of 15 patients undergoing Mohs surgery demonstrated that a petrolatum-based ointment containing vitamin E did not improve scar cosmetic appearance compared to the petrolatum-based ointment alone. 47 Furthermore, almost a third of patients developed contact dermatitis to the treatment with ointment containing vitamin E. In a double-blinded, randomized, placebo-controlled trial on 85 patients with surgical scars less than 2-week-old, 5% topical tocotrienol showed no significant effect on any of the scar parameters using laser Doppler imaging (LDI), POSAS, and a photographic scar assessment by a visual analogue scale (VAS) at 4-month follow-up. 48
In contrast, a large-scale, single-blinded RCT by Zampieri et al (n = 428) evaluated the before-and-after use of vitamin E on pediatric patients undergoing inguinal surgery. 49 Treatment with topical vitamin E provided a significant enhancement in cosmetic scar appearance as assessed by VSS and a significantly lower incidence of keloid formation. However, it is worth mentioning that the study population consisted of Caucasian children aged between 2–9 years old, who are less prone to develop keloids. This could also potentially limit the generalizability of this study's findings to the adult population. Similarly, an RCT conducted by Palmieri et al (n = 80) found a significant improvement in scar symptoms (pain and itching), color, size, and cosmetic appearance for keloid and hypertrophic scars treated with a combining vitamin E and silicone gel sheeting compared to the silicone gel sheeting alone. 50 This study concluded that combining vitamin E and silicone gel sheeting is efficacious for scar treatment, probably due to a sequential synergistic effect.
In summary, the current scientific evidence still lacks strong support for improving scar appearance with topical vitamin E alone. In addition, several side effects, such as contact dermatitis,47,51-53 urticarial eruptions, 52 and erythema multiforme-like reaction,52,54 possibly worsening scars, have been reported following its use. The early use of vitamin E in scar treatment may also reduce wound tensile strength due to its ability to inhibit collagen production, which could result in wound dehiscence and widened scars. 55 The topical application of vitamin E should thereby be discouraged. Further investigations of vitamin E used alone and/or in combination therapy are needed to confirm its clinical application in scar management.
Gotu Kola
C. asiatica, also commonly known as Gotu kola, is a medicinal plant used in both Asian traditional medicine and Western medicine. It has traditionally been used for wound healing promotion, scar reduction, and anti-inflammation. Pentacyclic triterpenes are active compounds, primarily asiaticoside, madecassoside, asiatic acid, and madecassic acid.56-58 Madecassoside has been reported to diminish scar formation by inhibiting keloid scar-derived fibroblast (KF) proliferation, inducing KF apoptosis by a mitochondrial-dependent pathway, 59 and inhibiting KF migration. 60 Of the other active components, both asiaticoside and asiatic acid have been demonstrated in vitro and in vivo studies to suppress KF proliferation and collagen synthesis by inducing Smad7 expression and inhibiting TGF-βI and TGF-βII expression, thus blocking the TGF-β/Smad signaling pathway.61-63 Gotu kola could therefore be of great use in preventing and treating scar formation.
Following the literature search, six articles were considered.64-69 Of these, just two were ranked as level of evidence 1b,65,69 and the remainder were ranked as level 2b. In clinical trials, the local application of Gotu kola extract has been demonstrated to have beneficial effects in reducing re-epithelialization and the complete wound healing time, as well as improving the wound appearance in patients with partial-thickness burn wounds (n = 60) 64 and facial acne scars underwent laser treatment (n = 30). 65 The oral administration of Gotu kola extract has been demonstrated to improve wound healing in diabetic foot ulcer patients due to fast wound contraction compared to placebo (n = 170). 66 It has also been shown to suppress scar formation due to a significant change in the granulation tissue formed between the study and placebo groups. A limited number of studies have shown that Gotu kola could be efficacious in scar management. The randomized, double-blinded, split-scar study utilizing Gotu kola alcoholic extract cream following a split-thickness skin graft (STSG) operation (n = 23) indicated a significant improvement in pigmentation and overall VSS scores between 4 and 12 weeks. 67 Two studies concluded that combining Gotu kola with herbal extracts gel could be beneficial in scar prevention and amelioration. The first, a randomized split-scar trial in patients with postsurgical wounds (n = 17), demonstrated that the combination of polyherbs with Gotu kola effectively improved scar appearance and hyperpigmentation, as evaluated using the POSAS. 68 The second, an RCT combining Gotu kola with herbal extracts gel plus silicone gel compared with placebo gel for the scar prevention of median sternotomy wounds (n = 46), indicated that the scarring in the study group was better in all items of the VSS, especially in scar height and pliability. 69 However, it is hard to determine the efficacy of this treatment regimen to Gotu kola extract alone, because silicone gel and the other combined herbal extracts, such as onion extract and aloe (Aloe vera (L.) Burm.f.) extract, have been independently demonstrated to improve scar cosmetic appearance.
In conclusion, although many preclinical studies have shown the therapeutic potential of Gotu kola with respect to scar prevention and amelioration, clinical evidence is sparse. A few clinical trials have demonstrated a trend effect in relation to the application of Gotu kola in scar management, but the small sample size and insufficient information on the herbal preparation used are inadequate to substantiate a well-established use. Therefore, more research is needed to clarify its therapeutic efficacy for scar prevention and treatment. In terms of the safety of Gotu kola, possible side effects occur less frequently, but it may lead to gastrointestinal system disorders, including abdominal pain, flatulence, constipation, dry mouth, nausea, and vomiting, when used orally. The topical application of Gotu kola may result in allergic reactions and a burning sensation. 70 Allergic contact dermatitis has also been indicated in a few case-reports following topical application;71-73 however, further evaluation disclosed that this reaction might have been caused by other ingredients in the products. 74 Due to little or no information being available regarding its safety during pregnancy and lactation, the use of Gotu kola is not recommended during these periods.57,70
Green Tea
Green tea, produced from the C. sinensis leaves, is one of the most widely consumed beverages. Its polyphenolic catechins, including (-)-epicatechin, (-)-epigallocatechin, (-)-epicatechin-3-gallate (ECG), and (-)-epigallocatechin-3-gallate (EGCG), have natural healing, photoprotective, free radical scavenging, antioxidant, anti-inflammatory, and antibacterial activities.75,76 EGCG, the most abundant green tea catechins, has shown a potential therapeutic effect in wounds and fibrosis/scarring. In KF cultures, EGCG has been demonstrated to suppress mast cell-stimulated collagen type I production by blocking the PI3K/Akt signaling pathway.77,78 EGCG has also been demonstrated to suppress the proliferation, migration, and collagen synthesis in KFs by disrupting the signal transducer and activator of transcription-3 (STAT3) signaling pathway.78,79 In the fibroblast-populated collagen lattice (FPCL) model, EGCG has been shown to inhibit PDGF- and TGF-βI-stimulated collagen gel contraction.80-82 It directly interacts with PDGF-BB, blocking its specific receptor binding and thereby inhibiting cell proliferation and collagen contraction.80,81 Furthermore, EGCG has been demonstrated to interrupt TGF-β signaling in some cell types by inhibiting the gene expression of TGF-β type I and II receptors, resulting in reduced CTGF and ECM gene expression.83,84 All of these profibrotic pathways, including TGF-βI, TGF-βII, PDGF, CTGF, PI3K/Akt, and STAT3, play an essential role in fibrosis/scarring.
Several studies on animal models have demonstrated the beneficial effects of polyphenolic catechins, especially ECG and EGCG, in wound healing and scarring84-87; nonetheless, little clinical trial has been performed on their potential for scar management. An RCT (n = 62) ranked as level of evidence 1b compared the topical administration of EGCG with placebo on the area surrounding a wound (zonal priming) and the fully formed scar. 88 Both systems of the topical EGCG were more effective than the placebo at improving scar thickness, elasticity, redness, and skin hydration.
Based on the present findings, there is inadequate evidence supporting the beneficial effect of the clinical application of EGCG on scarring. More clinical research needs to substantiate the anti-scarring effect of EGCG. In terms of safety, EGCG is classified as a “generally recognized as safe (GRAS)” substance by the United States Food and Drug Administration (USFDA). 89 It appears well tolerated with oral90-93 and topical administration94,95 in human studies. The most frequently reported adverse events are mild gastrointestinal issues, including abdominal pain/discomfort and nausea.90,92,93 Nevertheless, the clinical application of EGCG has some limitations because of its low bioavailability. 75 Therefore, topical or intralesional administration might be a more effective route to accomplish the functionality of EGCG. It is also unstable under high temperature and pH conditions, which could be a problem for developing dermatological and cosmetic products. 96
Resveratrol
Resveratrol, a natural plant polyphenolic compound, is abundantly present in grapes (Vitis vinifera L.) skin and seeds, red wine, peanuts, and soy. It is known to have numerous biological effects and pharmacological actions, such as antioxidant, anti-inflammatory, photoprotective, anti-aging, anticancer, and cardiovascular properties.97-99 There is also growing evidence that resveratrol has a beneficial effect against pathological scars. At the cellular level, Zeng et al demonstrated that resveratrol treatment suppressed cell proliferation in hypertrophic scar-derived fibroblasts (HSFs) by arresting the G1-phase cell cycle and inducing apoptosis. 98 Resveratrol further decreased the mRNA expression of procollagen type I and III in HSFs, reducing collagen deposition. 98 Zhai et al showed that resveratrol inhibited proliferation and triggered apoptosis in pathological scar-derived fibroblasts (PSFs) by altering the TGF-βI/Smad signaling pathway. 100 Ikeda et al reported similar findings regarding the proliferation inhibition and the apoptosis induction of KFs by resveratrol.99,101 Pang et al discovered that resveratrol induced HSF autophagy by upregulating microRNA-4654 expression, leading to the downregulation of Ras homolog enriched in brain (Rheb). 102 MicroRNA-4654 is known to regulate cell proliferation, differentiation, apoptosis, and autophagy, by directly binding to the GTP-binding protein Rheb, a target gene of microRNA-4654. 102 In addition to TGF-βI/Smad signaling and the microRNA-4654/Rheb axis, the mammalian target of the rapamycin (mTOR)/ribosomal protein S6 kinase (70S6K) signaling pathway also seems to be pivotal in PSF proliferation. 103 The mRNA and protein expression of mTOR and 70S6K was downregulated by resveratrol, leading to the inhibition of PSF proliferation. 103 Resveratrol has also been shown to upregulate sirtuin1 (SIRT1) in HSFs, resulting in the suppression of TGF-βI-induced dermal fibroblast transition, and the reduction in the expression of fibrotic markers such as collagen type I, collagen type III, and α-SMA. 104 Resveratrol further improved collagen density and arrangement in a mouse model of excisional wound healing. 104 Similar to the study by Ma et al, an RCT on women with a scarred uterus (n = 78) (ranked as level of evidence 2b) showed that oral resveratrol treatment promoted scar remodeling and reduced uterine-scar thickness compared to the placebo at 3 months post-treatment. 105
From these findings, it could therefore be concluded that resveratrol might be a potential therapeutic agent to counteract excessive scarring. Although the anti-scarring effects of resveratrol have been reported in numerous preclinical studies within various targets, further animal and clinical research is necessary to confirm these resveratrol-associated effects. Currently there is, to the best of our knowledge, only one published clinical trial regarding the anti-scarring properties of resveratrol. An obstacle that has limited the clinical administration of resveratrol is its fast metabolization and excretion. 97 Hence, improving its bioavailability during systemic intake or developing appropriate formulations for local and topical use is a subject of substantial research and may help in the targeted delivery of resveratrol.
Emodin
Emodin, a natural anthraquinone derivative, is found in numerous Chinese herbs, such as rhubarb (Rheum palmatum L.), Japanese knotweed (Polygonum cuspidatum Siebold & Zucc.), and Chinese climbing knotweed (Polygonum multiflorum Thunb.). It has been reported to exert various biological effects, including antiviral, antibacterial, anticancer, immunosuppressive, anti-inflammatory, anti-allergic, and wound-healing effects.106-108 Previous preclinical studies have shown that emodin inhibits fibrotic activity in many organs, suggesting that it might be beneficial in fibrosis treatment.109-112 Nevertheless, there are few studies examining the efficacy of emodin in treating hypertrophic scars.
Liu showed that emodin inhibited inflammation in mechanical stress-induced hypertrophic scar by suppressing the recruitment and adhesion of inflammatory cells and attenuating the synthesis of inflammatory cytokines, which are associated with the suppression of the PI3K/Akt signaling pathway. 113 Furthermore, Xia et al demonstrated that emodin inhibited hypertrophic scar development and fibrosis by blocking macrophage recruitment and polarization, which is related to suppressing the TGF-β and Notch signaling pathways in macrophages. 114 Taken together, these findings suggest a potential positive effect of emodin on hypertrophic scars. However, whether emodin can be clinically used as a therapeutic agent for hypertrophic scar treatment remains uncertain. Thus, more research, especially clinical trials, is imperative to evaluate the therapeutic use of emodin.
Regarding the adverse effects of emodin, its toxicity, including reproductive toxicity, hepatotoxicity, and nephrotoxicity, has been reported among preclinical experiments, especially in high doses and prolonged use. 106 Another issue is that emodin has low absorption in the intestine, rapid elimination, and poor oral bioavailability in vivo. 106 Further research is thereby required to attenuate its toxicity and improve its pharmacokinetics.
Curcumin
Curcumin, a yellow polyphenolic compound derived from turmeric (Curcuma longa L.) rhizome and the most active constituent of turmeric, has been used extensively in Ayurvedic medicine. It has been shown to possess several biological activities, including anti-inflammatory, antioxidant, antimicrobial, anticarcinogenic, anticoagulant, and antithrombotic activities. Curcumin has also been demonstrated to possess wound healing activity by acting on various phases of wound healing.115-117 Furthermore, it has been shown to possess a potent antifibrotic effect.118-121 Even though there are many in vitro and in vivo studies reporting curcumin's role in fibrosis prevention and reduction, there are few reports on its effects on scarring.
Scharstuhl et al demonstrated that curcumin treatment in a high dose (>25 μM) induced apoptosis in human dermal fibroblasts and inhibited collagen gel contraction mediated by fibroblasts through a reactive oxygen species (ROS)-mediated mechanism. 122 Meanwhile, Phan et al demonstrated that curcumin inhibited KF and HSF proliferation by inducing cell cycle arrest and collagen lattice contraction in HSFs. 17 Curcumin has also been found to inhibit the TGF-βI/Smad signaling pathway and ECM synthesis in KFs. 123 In a rabbit ear hypertrophic scar model, curcumin suppressed hypertrophic scarring by reducing the release of inflammatory cytokines IL-1, IL-6, and IL-8. 124 The results of this series of studies led to the conclusion that curcumin may promise to be a potential therapeutic agent for pathological scarring. However, there is still a lack of documented clinical trials. Further clinical research is required to confirm its efficacy and safety on scarring.
Unfortunately, curcumin's clinical application is limited because of its poor water solubility, poor absorption, fast metabolism, fast elimination, poor bioavailability, photosensitivity, and yellow staining on the skin and clothes.115,116 Novel formulations should be explored to minimize these effects and maximize curcumin's therapeutic capability. In terms of the safety of curcumin, it has been approved by the USFDA as “GRAS”. 116 Curcumin is well tolerated and non or low toxic. The most commonly reported adverse events are nausea and diarrhea when used orally; however, they resolve spontaneously after 1 to 3 days. 125
Others
In recent years, there has been a steady global increase in the use of natural products, accompanied by a growing search for new phytochemicals that could potentially be developed as antihypertrophic scar agents.11,12,126 Nowadays, other emerging natural products, such as oleanolic acid (from the green leaf galls of Ligustrum lucidum W.T.Aiton and Prunella vulgaris L.),127-130 shikonin (from the dried root of Zicao or Lithospermum erythrorhizon Siebold & Zucc),131-134 glabridin (from Glycyrrhiza glabra L.),135,136 tripterine (from Thunder God Vine or Tripterygium wilfordii Hook.f.),137,138 and hyperforin (from Hypericum perforatum L.)139-141 have been shown to have anti-scarring properties in various in vitro and in vivo models. However, to the best of our knowledge, these compounds or herbal extracts have not been found for their efficacy and safety in RCTs. Further well-designed clinical trials are required to confirm their potential effects as a lead compound or active ingredient for anti-scar preparations. In another way, traditional medicines have been reinvestigated for their potential effects on scar management. For example, Wubeizi ointment (a traditional Chinese medicine), which consists of Chinese herbs Salvia miltiorrhiza Bunge, Clematis spp., black vinegar, Galla Chinensis (gall produced by the aphid Melaphis chinensis), Cortex Lycii (root of Lycium chinense Mill.), and alum, has been reported to diminish scar formation by downregulating type I and III procollagen expressions, inhibiting fibroblast proliferation, and promoting fibroblast apoptosis.142-145 Moreover, Saireito (a Japanese herbal (Kampo) medicine, a combined formulation of two herbal medicines, Shosaikoto and Goreisan) has shown beneficial effects in scar management by suppressing TGF-I-induced Smad2/3 phosphorylation.146-149 These natural products should be further investigated to ensure their beneficial effect on scars and they might become potential anti-scarring agents in the future.
Discussion
This review article gathered publications on the role and mechanism of some natural products in scar prevention and treatment, particularly hypertrophic scars. It also addressed whether scar management with these natural products is effective in humans, thus providing a reference for the further research and development of natural product-based drugs for scar therapy with greater efficacy and lower side effects. In this review, a number of natural products, including onion, vitamin E, Gotu kola, green tea, resveratrol, emodin, curcumin, and other emerging natural products, have displayed potential effects as effective anti-scarring agents in preclinical studies. The main mechanisms of these natural products on scar formation involve inhibiting scar fibroblast proliferation and collagen production and/or inducing cell apoptosis by regulating several pathways, such as the TGF-β/Smad, MAPK, and PI3K/Akt signaling pathways (Table 2). Although these natural products have been reported to have potential anti-scarring activity in preclinical trials, this is not enough to substantiate their efficacy and safety in humans due to their different physiological structures. Therefore, clinical trials are essential to ascertain these natural products’ efficacy and safety as anti-scarring agents.
A total of 23 clinical trials using natural products as interventions for scar management were identified in this review. Among these 23 trials, only 3 RCTs recruited patients during the wound healing process (surgical wound, burn wound, and diabetic foot ulcer).49,64,66 However, these 3 trials also evaluated the final outcome for scar management using the VSS, a 4-point scale for cosmetic appearance satisfaction, and scar formation. The natural products used in these 23 trials included 11 onion trials, 4 vitamin E trials, 6 Gotu kola trials, 1 green tea trial, and 1 resveratrol trial. Unfortunately, documented clinical trials are still lacking for both emodin and curcumin. Most clinical trials were categorized as level of evidence 2b (n = 16). Based on the existing evidence discussed in this review, even though some success has been shown in addressing specific aspects of scarring when these natural products are utilized as monotherapy or as combination therapy with other agents, there is limited evidence from clinical studies to support their efficacy due to the limited quality of the studies and their individual flaws, including small sample sizes, poor randomization and blinding, and short follow-up durations. These limitations imply that the existing evidence may not be sufficiently robust to support natural products’ efficacy and safety for clinical treatment and prevention of hypertrophic scars. The interpretation and clinical application of these results should be performed with caution. In terms of safety, while considered natural, some natural products have been associated with mild adverse events in human studies (such as onion, vitamin E, and Gotu kola; Table 3). However, limited data on toxicity is discussed in this review.
It is challenging to compare the natural product-based therapeutics among the studies owing to the various models and methodologies used to investigate the efficacy of these anti-scarring agents. Moreover, the variation in wound type and depth, anatomical location, age and type of scars, history of pathologic scarring, patient age, underlying health conditions, control groups (such as petrolatum ointment or no treatment), and follow-up and scar treatment periods among the clinical trials conducted make it difficult to directly compare agents between studies. Most studies identified in this review had follow-up periods less than or equal to 6 months. Ideally, the follow-up periods in scar studies should be at least 1 year to ascertain that the observed effect is a result of the treatment and not a spontaneous wound healing process that improves over time. The studies also varied in the scar assessment methods and the reported outcome measures. The scar assessment methods included objective scar assessments (noninvasive scar analysis techniques), such as LDI, and subjective scar assessment scales, such as VSS and POSAS. However, there is no standardized and universally accepted method to evaluate scars. Each scar scale assesses a different set of parameters with various criteria and frequently depends on individual investigators’ subjective interpretation, leading to problems in comparing multiple scar studies.
Among the 7 natural products for scar management reviewed, 5 demonstrated a positive impact on scar prevention and treatment, supported by RCTs. When compared in terms of the number and quality of trials, onion extract had the most RCTs (11) with the highest quality evidence (4 RCTs rated 1b and 7 RCTs rated 2b). This was followed by Gotu kola, which had 6 RCTs (2 rated 1b and 4 rated 2b); vitamin E, which had 4 RCTs (all rated 2b); green tea, which had 1 RCT (rated 1b); and resveratrol, which had 1 RCT (rated 2b). Each natural product exhibited different mechanisms of action for scar management, suggesting that a combination of various active ingredients or polyherbs may be the most suitable approach for developing anti-scar products.68,146,150 Moreover, using standardized herbal extract, such as onion extract (which contains flavonoids, namely quercetin and kaempferol), which consists of numerous bioactive compounds, may be more advantageous than single pure compounds (such as resveratrol or vitamin E), not only due to potential synergistic effects but also considering the cost of isolation and preparation.151-153
In conclusion, more robust, consistent, and well-designed RCTs with large-scale and prolonged follow-up durations are needed to establish a firm conclusion concerning these natural products’ long-term efficacy and safety profiles. These trials should also focus on immature and mature scars in order to provide clinically relevant insights to make phytotherapy a promising approach for scar prevention and treatment. Additionally, some of these agents have some limitations in clinical use because of their low bioavailability (EGCG, resveratrol, emodin, and curcumin; Table 4). Therefore, it is necessary to improve their bioavailability during systemic intake or to develop suitable formulations for local and topical applications to minimize these effects and maximize their therapeutic capability.
Conclusion
In this review, a number of natural products, including onion, vitamin E, Gotu kola, green tea, resveratrol, emodin, curcumin, and other emerging natural products, have shown beneficial effects as potential anti-scarring agents in preclinical studies. The main mechanisms of these natural products on scar formation involve inhibiting scar fibroblast proliferation and collagen production and/or inducing cell apoptosis by regulating several pathways, such as the TGF-β/Smad, MAPK, and PI3K/Akt signaling pathways. However, there is limited evidence from clinical studies to support their efficacy due to the limited quality of the studies. More robust and well-designed clinical trials with large-scale and prolonged follow-up durations are needed.
Footnotes
Acknowledgments
The authors are grateful to the Thailand Research Fund through the Research and Researcher for Industry Programme (RRi) and the 90th Anniversary of Chulalongkorn University Fund (Ratchadaphiseksomphot Endowment Fund) for their support.
Author Contributions
TM: Conceptualization, Methodology, Formal analysis, Investigation, Writing – Original Draft, Writing – Review and Editing, Visualization, Project administration.
AA: Validation, Writing – Review and Editing, Supervision.
PA: Conceptualization, Methodology, Validation, Formal analysis, Investigation, Writing – Review and Editing, Supervision, Funding acquisition.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Thailand Research Fund through the Research and Researcher for Industry Programme (RRi) [grant number PHD60I0068]; and the 90th Anniversary of Chulalongkorn University Fund (Ratchadaphiseksomphot Endowment Fund) [grant number GCUGR1125642013D].
