Abstract
Background
A systematic review and meta-analysis have been conducted to evaluate the efficacy of alkalinization for COVID-19 patients based on current evidence to determine the impact of alkalinization on COVID-19 outcomes.
Methods
We searched MEDLINE (Pubmed), Web of Science, Cochrane Library, and Clinicaltrials.gov for studies evaluating the efficacy of alkalinization up to 30 April 2023. Based on the PRISMA 2020 statement criteria a systematic review and meta-analysis of studies were performed.
Results
The results of our meta-analysis showed a significant reduction in mortality rate in the alkalinization group compared to controls (RR 0.73, 95% CI: 0.56-0.95; I2 = 0%). However, our subgroup analysis showed no significant improvement in RCT-only studies (RR 0.78, 95% CI: 0.59-1.05; I2 = 0%), the recovery rate was significantly higher in the alkalinization group (RR 2.13, 95% CI: 1.39-3.26; I2 = 0%), duration of recovery also has improved in alkalinization group (SMD 0.76, 95% CI: 0.33-1.18; I2 = 0%). The results of our meta-analysis showed a significant reduction in the duration of hospitalization in the alkalinization group compared to controls with very low certainty of evidence (SMD −0.66, 95% CI: −0.97 to −0.35; I2 = 36%).
Conclusion
With low certainty of evidence, alkalinization (by sodium bicarbonate) can be an efficient and safe adjuvant treatment for COVID-19 patients. Future randomized controlled trials are needed to strengthen the available evidence.
Introduction
Although the COVID-19 burden including mortality, hospitalization, and critical patients has been reduced, this pandemic still impacts public health in various ways. Therefore, several numbers of therapies have been investigated with various levels of effectiveness. To date, different types of drugs have been utilized including antivirals, monoclonal antibodies, immunomodulators, and immunoglobulins. 1 Also, several studies have released drugs that are not effective or safe enough to be used during treatment including ivermectin. 2 This evidence has thrown up many questions in need of more investigations to find the most effective, safest, and financially best therapies for COVID-19. 3
A study done by Yang N et al showed that treatment at pH 8.0 and 37 °C with a half-life of ∼30 min would inactivate the coronavirus. 4 Also, coronavirus load has been reduced in host cells under alkaline conditions and an acidic environment has improved the rate of successful infection.5,6 Taken together, a low pH environment allows the viruses to connect to the cell wall effortlessly. 7 The nose and mouth are the main portals for SARS-CoV-2 to infect the human body. 8 Therefore, alkalinizing the environment of the nose with different types of agents including solutions and inhalators can be a considerable treatment for COVID-19. Various agents are known to be used as alkaline agents including Sodium bicarbonate, which has been utilized as a treatment for nasopharyngeal diseases. 9
Several studies have been undertaken on the efficacy and safety of alkalinization in COVID-19 patients, leading to inconsistent results. Therefore, this study was conducted to address the gap by conducting a systematic review and meta-analysis of current evidence.
Methods
This study has been registered in PROSPERO (CRD42023422779) and conducted in accordance with the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA). 10
Search Strategy
Medline (Pubmed), Web of Sciences, the Cochrane Library, and ClinicalTrials.gov were searched up to 30 April 2023. The subsequent search string: (“Sodium Bicarbonate”[Mesh] OR Sodium Bicarbonate [tiab] OR Bicarbonate [tiab] OR alkalization[tiab] OR alkalinization[tiab]) AND ((((((“COVID-19”[Mesh]) OR “SARS-CoV-2”[Mesh]) OR (COVID-19[Title/Abstract])) OR (Coronavirus [Title/Abstract])) OR (vaccin*[Title/Abstract])) OR (SARS-Cov-2[Title/Abstract])) was used for including the studies that have been evaluating the impact of alkalinization on COVID-19 patients (Full search strategy available in supplementary table 2). No filters were implemented regarding language, time, or region.
Inclusion and Exclusion Criteria
Studies had been deemed qualified for inclusion if they used the following PICOT criterion: (A) population: COVID-19 cases, (B) Intervention: alkalinization with any common drugs, (C) Comparator: standard of care or placebo, (D) outcomes: mortality, hospitalization, and recovery, (E) type of study: observational studies and randomize control trials. Case reports, case series, review articles, and studies conducted on animals have been excluded.
Data Extraction and Outcomes
We extracted data from study main text, tables, figures, graphs, and Supplemental materials to excel extraction data sheet. Two reviewers (MJ and KJ) extracted the following data, separately: author, year, country, type of study, sample size, population, age, gender, Intervention, duration of study, and main findings. For outcomes of our meta-analysis, we took mortality rate as the primary outcome of this study. Hospitalization, recovery, duration of hospitalization, and duration of recovery were chosen as the secondary outcomes.
Quality Assessment and Certainty of Evidence
Two reviewers (AA and MR) individually graded the methodological quality of involved studies using the Newcastle-Ottawa Quality Assessment (NOS) for observational studies and Cochran risk of bias tool version 2 (RoB-2) for RCTs.11,12 The certainty of evidence regarding each outcome was evaluated based on Grading of Recommendations, Assessment, Development, and Evaluations (GRADE). 13 Any disagreements between two reviewers were solved by a discussion with the third reviewer.
Statistical Analysis
We used a random effect meta-analysis to pool the effect sizes of each study using the generic inverse variance method. Risk ratios and standardized mean differences with their 95% confidence intervals (CIs) were used to report dichotomous and continuous outcomes, respectively. We used Cochran Q statistic and I2 value to assess the heterogeneity. Funnel plot and Egger regression test were not conducted due to the limited number of included studies (n < 10). Subgroup analysis was done based on study design for the primary outcomes. All statistical operations and the production of graphs were conducted using R software and the meta package. 14
Results
Study Selection and Quality Assessment
After a through screening of 584 citations, a total of 7 studies deemed eligible to be include15–21 (Figure 1). There were 5 randomized controlled trials,15–17,19,21 and 2 observational cohorts.18,20 Studies were conducted between 2021 and 2023, with two performed in Egypt, one in Croatia, one in USA, one in India, one in Brazil, and one in China. Four studies were conducted on inpatients exclusively.16,19–21 The sample size of studies varied from 55 to 546. The detail characteristics of the included studies is available in Table 1.

PRISMA flow diagram.
Characteristics of the Included Studies.

Results of Meta-Analysis
Primary Outcomes
Mortality
Four studies comprising 923 patients were included.16–18,20 There were two RCTs16,17 and two cohorts.18,20 The results of our meta-analysis showed significant reduction in mortality rate in alkalinization group compared to controls (RR 0.73, 95% CI: 0.56-0.95; I2 = 0%; p = .02; Figure 2). However, our subgroup analysis showed no significant improvement in RCT only studies (RR 0.78, 95% CI: 0.59-1.05; I2 = 0%; Supplementary Figure 1). The GRADE assessment showed low certainty of evidence for the aforementioned outcome (Table 2).

Results of the meta-analysis for (a) mortality, (b) hospitalization, and (c) recovery.
GRADE Certainty of Evidence.

Hospitalization
Three studies comprising 685 patients were included.15,17,19 All were RCTs. The results of our meta-analysis showed no significant reduction in hospitalization rate in alkalinization group compared to controls with very low certainty of evidence (RR 0.61, 95% CI: 0.24-1.56; I2 = 80%; p = .30; Figure 2).
Recovery
Three studies comprising 262 patients were included.18–20 The results of our meta-analysis showed a significant improvement in recovery rate in alkalinization group compared to controls with low certainty of evidence (RR 2.13, 95% CI: 1.39-3.26; I2 = 0%; p = .0005; Figure 2). However, our subgroup analysis showed no significant improvement in cohorts (RR 1.72, 95% CI: 0.97-3.06; I2 = 0%; Supplementary Figure 2).
Secondary Outcomes
Duration of Hospitalization
Two studies comprising 682 patients were included.17,21 The results of our meta-analysis showed a significant reduction in the duration of hospitalization in alkalinization group compared to controls with very low certainty of evidence (SMD −0.66, 95% CI: −0.97 to −0.35; I2 = 36%; p < .0001; Figure 3).
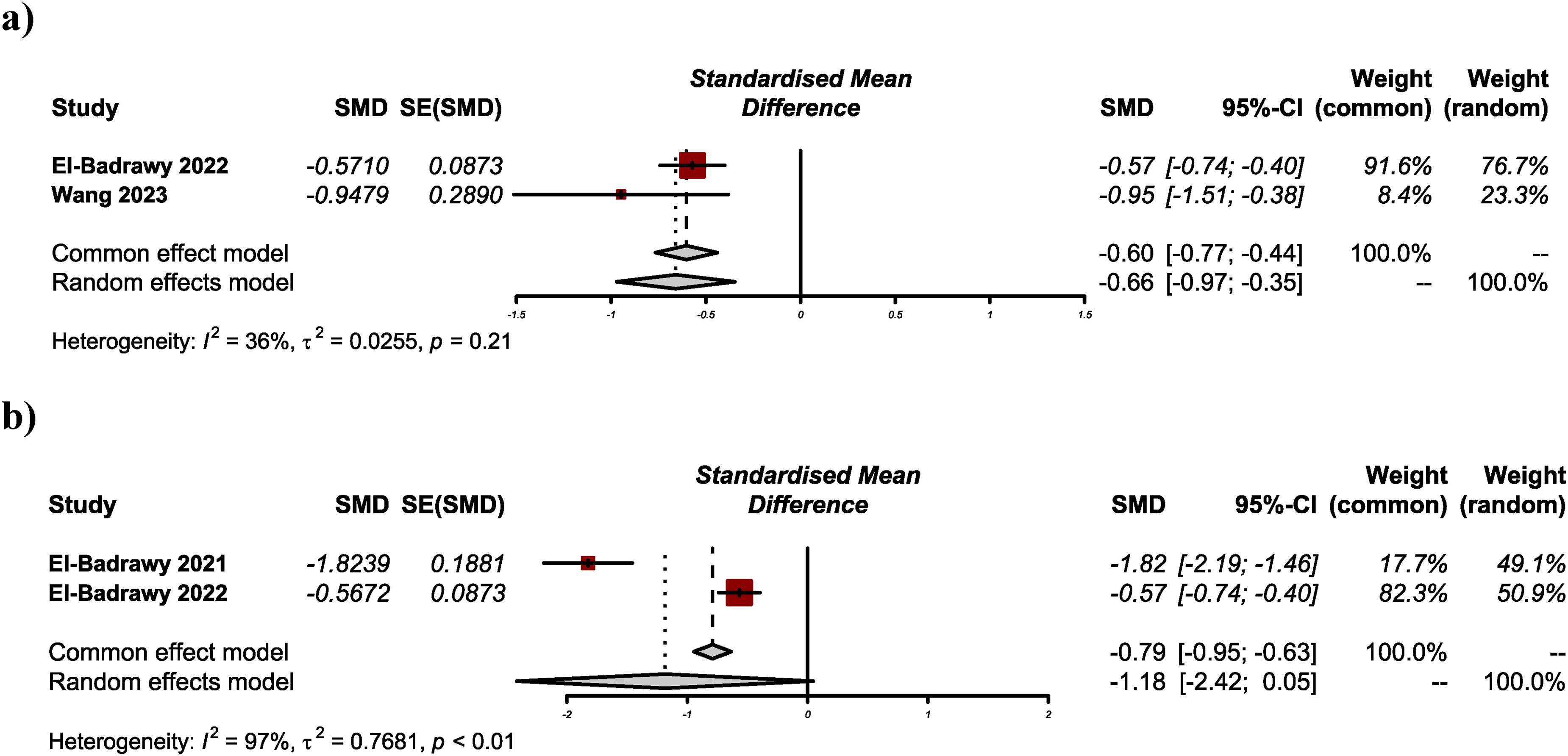
Results of the meta-analysis for (a) duration of hospitalization, and (b) duration of recovery.
Duration of Recovery
Two studies comprising 262 patients were included.17,18 The results of our meta-analysis showed a nonsignificant reduction in recovery time in alkalinization group compared to controls with low certainty of evidence (SMD −1.18, 95% CI: −2.42-0.05; I2 = 97%; p = .06; Figure 3).
Quality Assessment
The results of our quality assessment yielded most of the included studies to be subjected with moderate risk of bias (Supplementary Figure 3). Only one RCT was judged to have a high risk of bias. 21
Discussion
To the best of our knowledge, the present systematic review and meta-analysis is the first comprehensive study determining the efficacy and safety of alkalinization in COVID-19 outcome in patients including mortality, risk of hospitalization, rate of recovery and duration of hospitalization and recovery. Alkalinization in the present study is defined as utilizing alkaline agents including Sodium Bicarbonate, N-Acetylcysteine or Hypertonic NaCl, through nasal, oral irrigation and nasal sprays. Studies showed that alkalinization is an effective treatment for COVID-19. As a primary outcome, mortality has seen to be significantly improved. Duration of recovery and rate of recovery were significantly higher in alkalinization group in comparison with control group. Albeit risk of hospitalization and duration of hospitalization were not changed significantly between two groups.
SARS-CoV-2 infects human body cells by some early key steps, initially, the virion binds to the cell membrane, secondly, the S1 fragment releases upon cleavage by TMPRSS2 which leads to the viral membrane fusion, and finally penetration of the genome to the cytosol occurs. 5 The latter and membrane fusion requires an acidic environment with pH between 6.2 to 6.8. 5
Furin in producing cells and TMPRSS2 on target cells account for membrane fusion during the entrance of SARS-CoV-2.22,23 These are the essential steps for infecting the host cell, albeit Using real-time three-dimensional single-virion tracking has released that without an acidic environment, even when furin and TMPRSS2 cleavages have occurred, fusion and genome penetration would not happen. 5 Coronavirus has been found to be a pH-dependent virus, study done by Stuman et al showed that at 37 °C at pH 8 (half-life 30 min) the virus fusion with the cell membrane became unstable and the virus was inactivated. 6 In vitro studies showed that beside corona virus, influenza replication can be effected by pH. 24 Alkalinizing the virus environment by using the lysosomotropic alkalinizing agents (LAAs) has significantly inhibited the viral replication. 24 It is also reported that viruses including corona virus and influenza can be weakened or inactivated by alkalinizing the blood pH within the normal range by using the alkaline diet and drinks. 25 Besides TMPRSS2, the angiotensin-converting enzyme-2 (ACE2) receptor plays an essential role in SARS-CoV-2 cell entry and it is mostly expressed in the type-2 pneumocytes. 26 ACE2 expression in olfactory neuroepithelium is 700 times more than its expression in the lungs. 27 Due to SARS-CoV-2 cell-by-cell spreading and delayed activation of TMPRRS2 in the lungs, using agents with which virus particles that are lodged but not fused with the ACE2 receptor become debrided mechanically, would be useful to reduce the severity of infection.15,28 Salinated nasal irrigation has reduced symptoms and severity of COVID-19 infection by following mechanisms: bettering airways compliance, lowering viral loads and reducing the transmission, improving mucociliary clearance, moisturizing the respiratory epithelia and mucosa and reducing the viral ACE2/TMPRSS2 uptake and furin cleavage. 29 Taken together, evidence acknowledges that alkalinization by lowering the chance of membrane fusion, genome penetration, and deactivating the virus, and irrigation by mechanically preventing virus entrance to the host cell can be considered to lower the COVID-19 outcome and severity. Sodium bicarbonate irrigation is one of the effective treatments for rhinitis and nasopharyngeal diseases. 30 Hypertonic or normal saline nasal sprays or irrigations have been reported to be effective in treatment of Acute and Chronic Rhinosinusitis, Allergic Rhinitis and can reduce the illness days and need of antibiotic usage in children. 31 In addition, nasal irrigation by slightly alkaline pH can improve outcomes in flu and cold. 31 Common side effects of Sodium bicarbonate irrigation are dry mouth, polydipsia, and polyuria, which occur rarely. 32 In contrast, in a study done by Kraut et al, they have evaluated the effect of Sodium bicarbonate as a buffer on lysosomal pH, which showed that this agent was not able to change the patient's lysosomal pH and was not considered as a useful treatment for COVID-19. 33
There is no evidence that nasal irrigation by alkalizate factors can significantly impact blood pH. 34 Therefore, alkalization of nasal and oral cavities can be potent, cost-beneficial, safe, quick, and readily available for patients worldwide to combat COVID-19. Although some agents including chloroquine and hydroxychloroquine can affect lysosomal pH and are known as a weak base, but since their usage are mostly by oral administration, systematic side effects such as retinopathy and gastrointestinal adverse effects are expected.35,36 therefore, using substances that have less side effects and are used locally, can lead to safer treatments.
A number of limitations to the present study need to be acknowledged. differences in the studies performed including the type of agents used for alkalinization, their dosage, and variants of COVID-19 were various among included studies which confounded Comparison across treatments. Notably, some heterogenicity was reported in our study, therefore random-effects model was utilized to account for this heterogeneity. Subgroup analysis was defined based on the study population, and early/late treatment to explain heterogeneity. It should be noted that few numbers of studies are included in each subgroup analysis, therefore caution must be applied, as the findings might not be transferable.
Conclusion
Alkalinization using sodium bicarbonate in COVID-19 patients showed possible advantages through reducing the risk of mortality, and improving recovery rates. Although the results of the current study suggest the positive effect of alkalinization on COVID-19 outcomes, but higher- quality studies should concentrate on the investigation of alkalinization in COVID-19.
Supplemental Material
sj-docx-1-chp-10.1177_2515690X241258403 - Supplemental material for Alkalinization Using Sodium Bicarbonate for COVID-19 Treatment: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-chp-10.1177_2515690X241258403 for Alkalinization Using Sodium Bicarbonate for COVID-19 Treatment: A Systematic Review and Meta-Analysis by Arman Shafiee, Kyana Jafarabady, Hassan Moltazemi, Mohammad Javad Amini, Mohammad Ali Rafiei, Amirhossein Akhondi and Sayed-Hamidreza Mozhgani in Journal of Evidence-Based Integrative Medicine
Footnotes
CRediT Author Statement
Declaration of Conflicting Interests
The authors declare that they have no known competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publicationof this article: This study was financially supported by the Vice-Chancellor for Research and Technology, Alborz University of Medical Sciences, Karaj, Iran, under Grant number [ABZUMS 6033].
Trial Registration
Not applicable
Ethical Approval
This study was approved by the ethics committee of Alborz University of Medical Sciences, Alborz, Karaj, Iran (IR.ABZUMS.REC.1402.154).
Informed Consent
Not applicable
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
