Abstract
Diabetes mellitus is an oxidative stress-related disease characterized by hyperglycemia and a variety of complications, including nephropathy. Vitamin D has variable functions extending beyond the calcium metabolism to prevent oxidative tissue damage. We aimed to investigate whether vitamin D supplements could enhance Glibenclamide's effectiveness in treating diabetes and minimize the risk of associated pathology. Wistar rats were divided into normal control (n = 10) and diabetic (n = 30), where animals received two low doses of Streptozotocin 30 mg/kg/BW intraperitoneally to develop diabetes. The diabetic rats were then randomly divided into three equal groups: untreated, treated with Glibenclamide (0.6 mg/kg), and treated with Glibenclamide and Vitamin D3 (500 IU/kg). After eight weeks, the animals were sacrificed, and blood samples and kidney tissues were collected to evaluate biochemical, anti-oxidant, and pro-inflammatory cytokine levels and histological and immunohistochemical changes. Diabetic animals had significantly increased fasting blood glucose, lipid profile, blood urea, serum creatinine, and Malondialdehyde levels, whereas serum insulin, albumin, and the anti-oxidant enzymes superoxide dismutase and catalase were significantly decreased compared to normal control (p < 0.01). Furthermore, some renal histological changes were observed together with significantly increased immunoreactivity of anti-p53, anti-TNF-α, and anti-IL-6 antibodies when compared to the normal control. All abnormal parameters improved significantly with Glibenclamide therapy (p < 0.01), but combination therapy with vitamin D produced a much better result. In conclusion, vitamin D supplementation along with anti-diabetic medication can help prevent or reduce the severity of diabetic nephropathy due to its potent antioxidant, anti-inflammatory, and anti-apoptotic properties.
Introduction
Diabetes mellitus (DM) is a chronic inflammatory and metabolic disorder characterized by hyperglycemia. Diabetes affects more than 400 million people worldwide, with a marked rise in incidence. 1 On the other hand, chronic hyperglycemia induces oxidative stress, which can result in defective insulin secretion, impaired insulin gene expression, and increased tissue apoptosis. Increased oxidative stress in diabetics is responsible for the majority of diabetic complications including nephropathy. 2
Diabetic nephropathy (DN) is the leading cause of end-stage renal failure worldwide, as well as the leading risk factor for developing macrovascular disorders. 3 The prevalence of DN is approximately 30% for patients with type-I diabetes and ranges from 20% to 50% for patients with type-II diabetes. 4 However, many factors and pathways play a role in the pathogenesis of DN. Chronic hyperglycemia can result in a variety of cellular and molecular changes that affect the glomerular blood capillaries as well as tubular structure and function. 5 Hyperglycemia produces excess reactive oxygen species (ROS) and reduces the anti-oxidant potential resulting in oxidative damage to lipids, proteins, and DNA, with possible changes in the structure and function of these molecules. This will stimulate the immune system and help release inflammatory mediators and cytokines that promote the initiation and progression of tissue damage and apoptosis throughout the body, including the kidneys.6,7
Additionally, the accumulation of lipid peroxidation products, advanced glycation end products, and damaged DNA contributes to most diabetic complications. In the absence of an adequate compensatory response from the endogenous anti-oxidant network to glucotoxicity and lipotoxicity induced by hyperglycemia and hyperlipidemia, oxidative stress is exaggerated, resulting in activation of the stress-sensitive intracellular signaling pathways. 8 As a result, oxidative stress reduction and stress-sensitive signaling pathways were investigated to treat diabetes and diabetic complications. Malondialdehyde (MDA), on the other hand, is a commonly used lipid peroxidation marker for oxidative stress. At the same time, living organisms have evolved several protective anti-oxidant enzymes against ROS toxicity, such as superoxide dismutase (SOD), catalase (CAT), and many others. 9
Vitamin D is a fat-soluble secosteroid that acts as a prohormone to regulate bone mineralization. Vitamin D is found mainly in vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol). Vitamin D can be synthesized endogenously through ultraviolet solar radiation and obtained from a limited number of dietary sources. 10 Beyond calcium metabolism and bone mineralization, vitamin D regulates cell proliferation and differentiation, 11 protects multiple tissue organs from oxidative damage, 12 and acts as a transcriptional regulator factor for many genes. 13 Vitamin D first binds to the Vitamin D Receptors (VDR), a type of nuclear receptor family member expressed in nearly all body cells that regulate a variety of biological processes. 14 VDR is highly expressed in pancreatic B-cells, implying that vitamin D plays a vital role in regulating the risk of diabetes. 15
Diabetes management is increasingly focusing on preventing diabetes-related complications. However, managing diabetes without side effects remains a significant challenge. Anti-diabetes therapy aims to reduce near-normal blood glucose levels, but free radicals pose an additional challenge. 16 Diabetes medications, such as insulin, glibenclamide, and others, can effectively treat hyperglycemia but cannot prevent diabetic complications. 17 The current study aims to investigate whether vitamin D supplementation could enhance Glibenclamide's effectiveness in treating diabetes and minimize the risk of associated pathology such as dyslipidemia, oxidative stress tissue damage, and nephropathy in experimentally induced DN in rats.
Materials and Methods
Fasting blood glucose (FBG) was measured after 12 h of fasting using a One-Touch Ultra glucose meter (Life Scan, Milpitas, CA, USA). Serum insulin was measured using rat-specific Enzyme-linked immune sorbent assay (ELISA) commercial kits (DRG International Inc, USA). In addition, blood urea, serum creatinine, total serum cholesterol (TC), serum triglycerides (TG), and serum albumin were measured by using commercially available test kits.
Haematoxylin and Eosin (H&E) stain; Scytek Laboratories SDP Hematoxylin-Eosin Stain Kit (Catalog No. NC0510871). Periodic Acid Schiff (PAS); Diagnostic BioSystems CA, Stain Kit (Catalog No. 027) Masson's trichrome stain; Thermo ScientificTM Richard-Allan ScientificTM Masson Trichrome Kit (Catalog No. 22-110-648). Two independent observers examined the kidney tissues blindly to detect the histological changes. Immunohistochemical reactions to evaluate apoptosis and pro-inflammatory cytokines (IL-6 and TNF-α) in kidney tissue sections: The expression of anti-p53 antibody (for apoptosis), anti-IL-6 antibody, and anti-TNF-α antibody were investigated in formalin-fixed, paraffin-embedded 4 µm thick kidney tissue sections. Kidney tissue sections were deparaffenized, rehydrated. An antigen-retrieval procedure was then performed by heating the slides in 10 mM sodium citrate buffer solution (pH 6.0) in the microwave for 15 min. Sides were then incubated in hydrogen peroxide (H2O2) to block endogenous peroxidase, then the slides were incubated with primary antibodies [Anti-p53 antibody [(BP 53.12): sc-81168] (Santa Cruz Biotechnology Inc, Santa Cruz, CA, USA). Anti-IL-6 polyclonal antibody (ab6672), Abcam, USA. Anti-TNF-α monoclonal antibody (ab6671), Abcam USA] diluted in bovine serum albumin at 4 °C overnight. Slides were washed three times in PBS, then incubated with appropriate secondary antibodies for one hour, counterstained, and examined.
The stained sections were examined microscopically, and the results were interpreted accordingly. The expression pattern was blindly evaluated in five non-overlapping fields/sections in five serial sections/rat. The stained sections were graded based on staining intensity and number of stained cells, with 0 representing no staining/expression, 1 representing weak but detectable (>25%) staining, 2 representing moderate staining (25-50%), and 3 representing intense or high staining (>75%).
24
Results
Additionally, TNF-α and IL-6 levels in diabetic untreated animals were significantly higher (p < 0.01) than in normal controls. Moreover, GLB- and GLB/vitamin D-treated animals had significantly lower (p < 0.01) levels of TNF-α and IL-6 compared to diabetic untreated animals, with better results for GLB/vitamin D- than GLB-treated rats.
Discussion
Diabetes induced by STZ injection is associated with pancreatic β-cells dysfunction, which results in decreased insulin secretion and hyperglycemia. Chronic hyperglycemia contributes to carbohydrates, protein, and lipid metabolism defects and impaired kidney function and structure. 25 The current study showed that diabetic rats had significantly lower body weight, plasma insulin, and serum albumin levels but significantly higher blood glucose, TC, TG, blood urea, and serum creatinine levels compared to control rats. In kidney tissue homogenates, diabetic rats had significantly lower levels of oxidative stress markers and significantly higher levels of MAD and inflammatory cytokines. Furthermore, the diabetic kidney tissue sections had significant pathological changes associated with the overexpression of inflammatory cytokines (IL-6 and TNF-α) and p53 (for apoptosis). Our results were consistent with previously reported data.25–29

Photomicrographs of histologically stained kidney tissue sections showing. Haematoxylin and Eosin stained sections show the following: First, the normal histological structure of glomerulus and tubules, in normal control group. Second, glomerulosclerosis, tubular dilatation, and atrophy in the diabetic control group. Third, mild glomerulosclerosis, with mild tubular atrophic changes in the GLB-treated group. Finally, minimal glomerulosclerosis, and mild tubular dilatation in the Diabetic group treated with GLB/vitamin D-treated diabetic group (X 400). Periodic Acid-Schiff (PAS)-stained sections show the following: First, normal morphology of glomerulus and tubules in the control group. Second, in the diabetic control group, expanded mesangial matrix, and extended tubules in the diabetic control group. Third, the GLB-treated group experienced mild restoration of pathological changes. Finally, there was near normal renal tissue in the diabetic group treated with GLB/vitamin D. (X 400). Masson trichrome stained sections show the following: First, normal morphology of glomerulus and tubules in the control group. Second, the expanded mesangial matrix, and mild interstitial fibrosis in the Diabetic control group. Third, mild restoration of the expanded mesangial matrix in the glomerular area and minimal interstitial fibrosis in the GLB-treated diabetic group. Finally, the near-normal mesangial matrix in the glomerular area, and no interstitial fibrosis, in the diabetic group treated with GLB/vitamin D (X 400).
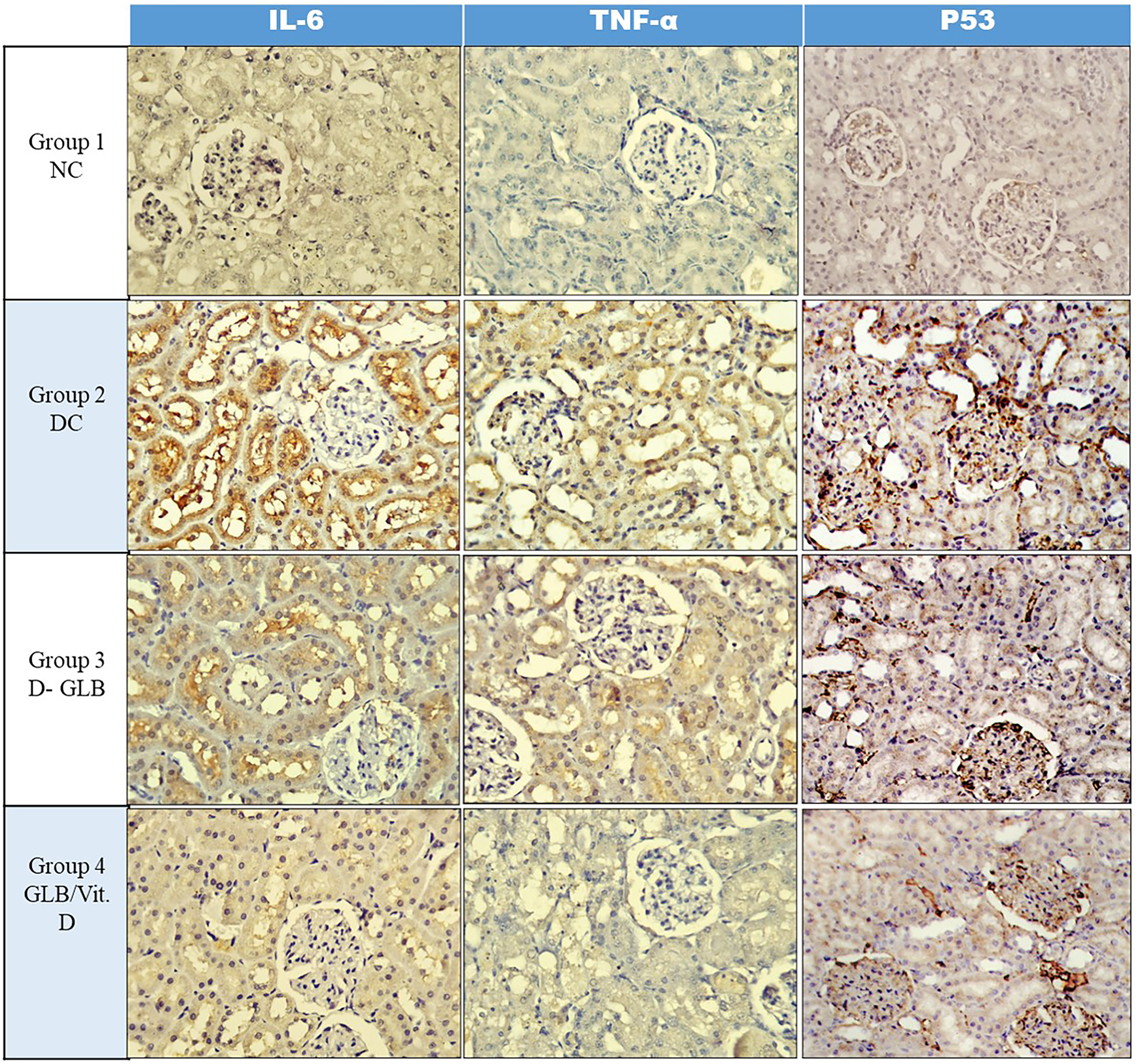
Photomicrographs of immunohistochemical reactivity in kidney tissue sections showing. Immunohistochemical expression of IL-6 and TNF-α showing a weak expression (∼25% of cells) in the normal group, marked expression (∼75% of cells) in the diabetic control group, mild expression (∼50% of cells) in Glibenclamide treated group, and finally a weak expression (∼25% of cells) in the diabetic group treated with Glibenclamide and Vit D3 (X 400). Immunohistochemical expression of P53 showing weak nuclear staining (∼25% of cells) in the normal group, marked nuclear staining (∼75% of cells) in the diabetic control group, mild nuclear staining (∼50% of cells) in the Glibenclamide group weak nuclear staining (∼25% of cells) in the diabetic group treated with Glibenclamide and Vit D3 (X 400).
Demonstrates the Body Weight and the Biochemical Parameters in all Study Groups. Values Indicate Mean ± Standard Deviation of the Mean (SDM) of ten Rats per Group.

NC: Normal Control, DC: Diabetic Control, D-GLB: Diabetic treated with Glibenclamide, D-GLB-VD3: Diabetic treated with Glibenclamide supplemented with Vitamin D3, FBG: Fasting Blood Glucose.
* P < 0.05: compared to the normal control values.
**P < 0.01: compared to the normal control values.
P < 0.05: compared to the diabetic control values.
P < 0.01: compared to the diabetic control values.
Demonstrates Oxidative Stress and pro-Inflammatory Biomarkers in the Kidney Tissue Homogenates of the Studied Groups. Values Indicate Mean ± Standard Deviation of the Mean (SDM) of ten Rats per Group.

NC: Normal Control, DC: Diabetic Control, D-GLB: Diabetic treated with Glibenclamide, D-GLB-VD3: Diabetic treated with Glibenclamide supplemented with Vitamin D3, FBG: Fasting Blood Glucose.
* P < 0.05: compared to the normal control values.
**P < 0.01: compared to the normal control values.
P < 0.05: compared to the diabetic control values.
P < 0.01: compared to the diabetic control values.
GLB-treated diabetic animals had a significant decrease in FBG compared to diabetic control values. GLB is an anti-diabetic medication that has long been used to lower blood glucose levels through various mechanisms; GLB stimulates pancreatic β-cells binding to the sulfonylurea receptor-1, blocks K+ ATP-sensitive channels that cause membrane depolarization, and opens the Ca++ voltage-dependent channels. This, in turn, increases the intracellular calcium influx and promotes insulin secretion, raising blood insulin levels while decreasing glucose levels. 30 GLB also reduces hepatocyte glucose synthesis, which lowers blood glucose levels. 31 On the other hand, the diabetic GLB-treated group supplemented with vitamin D showed more decreases in the FBG and increased insulin levels than the diabetic GLB-treated animals. This was consistent with the findings of Hassan et al, 32 who reported that vitamin D improves insulin secretion by stimulating VDR on pancreatic β-cells. Vitamin D also improves insulin sensitivity by increasing the expression of insulin receptors and promoting the expression of the peroxisome proliferator-activated receptor (PPAR), a nuclear receptor involved in lipid and glucose metabolism. 23 Furthermore, vitamin D can indirectly improve insulin secretion by normalizing extracellular calcium and maintaining normal intracellular calcium influx. 33
Compared to the normal control, diabetic rats had a significant increase in serum cholesterol and triglycerides. Furthermore, serum cholesterol and triglyceride levels decreased significantly in GLB- and GLB/vitamin D-treated diabetic groups than in the diabetic untreated animals, which was consistent with previous studies.34,35 GLB has a hypolipidemic effect by inhibiting or reducing cholesterol biosynthesis, reducing fatty acid synthesis, and/or promoting the formation of triglyceride precursors such as acetyl CoA and glycerol phosphate. 25 On the other hand, Vitamin D can improve hyperlipidemia either indirectly by stimulating VDR, which helps in reducing acetylated low-density lipoprotein cholesterol uptake, or directly by stimulating lipogenesis through the activation of lipoprotein lipase in adipocytes. 36 In addition, vitamin D stimulates calcium absorption from the small intestine to maintain serum calcium levels, increasing tissue fat oxidation. Furthermore, VDR overexpression reduces lipid catabolism and promotes lipogenesis pathways, and improves adipocyte differentiation and proliferation. 37
Blood urea and serum creatinine levels improved significantly in the GLB treated diabetic and GLB-treated with vitamin D supplemented diabetic groups compared to the diabetic untreated group in this study. Studies have shown that treating diabetic animals with GLB, 27 or vitaminD 38 significantly improved the kidney functions such as blood urea and serum creatinine levels. In addition, Gembillo et al 39 reported that Vitamin D supplementation has been shown to reduce proteinuria, improve glomerular filtration rate, and preserve the renal structure. Whereas, Wang et al 40 found that vitamin D supplementation significantly reduced blood urea but not serum creatinine in diabetic patients with chronic kidney diseases.
Diabetes is associated with inflammation, an increase in the production of free radicals and reactive oxygen species (ROS), as well as a decrease in anti-oxidants, and an increase in lipid peroxidation. All of which are involved in the pathogenesis and prognosis of most diabetic complications, including DN.32,41 Lipid peroxidation is a reaction between ROS and lipids that results in oxidative damage to cellular membrane lipids. Malondialdehyde, MDA, the end product of lipid peroxidation, is used to assess oxidative stress. 42 In our study, antioxidative enzymes such as SOD and CAT were significantly decreased in diabetic untreated rats compared to normal controls, whereas MDA was significantly increased. These findings were consistent with the findings of Chandirasegaran et al. 43 GLB treatment significantly increased anti-oxidant defense (SOD and CAT) while decreasing MDA in diabetic animals. Our findings agreed with previously published studies.17,44 We hypothesized that because GLB has low anti-oxidant activity, it cannot completely reverse kidney oxidative tissue damage. Furthermore, Vitamin D supplementation with GLB significantly improved the SOD and CAT and decreased lipid peroxidation (MDA), which agreed with the result of Hassan et al. 32 Vitamin D can reduce oxidative stress by reducing hyperglycemia, the leading cause of oxidative stress in diabetes. Vitamin D can also protect cell membranes from ROS-induced damage. 45
In the current study, the expression of inflammatory cytokines, IL-6 and TNF-α, was increased significantly in renal tissue homogenates as well as renal tissue sections, indicating the role of these cytokines in the pathogenesis and prognosis of DN. It has been reported that the level of IL-6 and TNF-α in DN patients’ serum and renal tissues were significantly higher than in diabetics without nephropathy. 46 Furthermore, there was a direct correlation between over-expression of IL-6 in renal tissue and the incidence of renal pathology and albuminuria, indicating that IL-6 is involved in the pathogenesis of DN. 22 TNF-α can also cause ROS to be produced in various renal cells, including mesangial cells, resulting in glomerular capillary permeability disturbances and albuminuria. 47 The findings confirm the role of inflammatory cytokines (IL-6 and TNF-α) in DN development.
Treatment of diabetic animals with GLB alone or in combination with Vit-D resulted in a significant reduction in the levels of inflammatory cytokines (IL-6 and TNF-α) in both renal tissue homogenates and renal tissue sections. Zakhary and colleagues 48 recently reported that short-term vitamin D3 supplementation could protect diabetic patients from diabetic complications and improve proteinuria. Several studies have also found that vitamin D supplementation reduces pro-inflammatory cytokines such as IL-1β, IL-2, IL-6, interferon-γ (IFN-γ), and TNF-α.49,50
Diabetic nephropathy is characterized by thickened basement membranes, tubule degeneration, necrosis, loss of podocytes, 51 mesangial enlargement, hypertrophy, interstitial cell infiltration, 52 inflammatory cell infiltration, dilation of tubules, as well as glomerular deterioration and atrophy. 53 Our results showed most of these findings. Hyperglycemia stimulates the expression of transforming growth factor-1 (TGF-1) in renal mesangial cells, which increases the synthesis and cross-linking of extracellular matrix, resulting in glomerulosclerosis and interstitial fibrosis. 54 In addition, ROS can activate the STAT kinases JAK2 and TYK2. JAK/STAT signaling activation promotes the growth and proliferation of renal mesangial cells, which contributes to DN. 55 On the other hand, insulin deficiency activates IGF-1 and IGF-1R expression in renal mesangial cells and increases cell proliferation, fibronectin, and collagen IV synthesis, all of which contribute to DN. 56 TNF-α, on the other hand, has been shown to have a cytotoxic effect on renal tissues, causing apoptosis, necrosis, and changes in intraglomerular blood flow and glomerular filtration rates. TNF-α is also involved in the development of renal hypertrophy and hyperfunction, which are early renal changes involved in the pathogenesis of DN. 47
GLB and GLB/vitamin D treatment resulted in a considerable reduction in glomerulosclerosis, cellular infiltration, and interstitial fibrosis, with better results in the case of the GLB/vitamin D combination. Tian et al 27 indicated that GLB could protect against diabetic complications by inhibiting the Notch/Snail1 signaling pathway, accelerating the anti-oxidant potential, and preventing renal fibrosis. In contrast, Ioannidis 57 indicated that the kidney metabolizes GLB inefficiently, resulting in an accumulation of non-metabolized products with impaired renal functions and/or incomplete recovery of diabetic renal pathological changes. Vitamin D supplementation has been shown to protect against DN through several mechanisms, including decreased oxidative stress, improved glucose tolerance, and insulin secretion regulation. Furthermore, vitamin D has been shown to protect against cytokine-induced apoptosis 50 and inhibit JAK/STAT signaling and reduce TGF-1, IGF-1, and IGF-1R expression in renal mesangial cells, preventing renal tissue fibrosis and podocytes` damage and preserving renal structure. Furthermore, vitamin D can activate anti-oxidant enzymes by stimulating the synthesis of metal ion protein carriers such as Zn and Cu, which act as cofactors to activate various enzymes, including anti-oxidant enzymes.55,58
Conclusion
Hyperglycemia, oxidative stress, and inflammatory cytokines were found to be responsible for pathological changes in renal tissues, abnormal biochemical parameters, and apoptosis in diabetic animals. Many studies, however, have found that hyperglycemia is a significant risk factor for the production of ROS, which stimulates the release of inflammatory cytokines and causes renal tissue damage and apoptosis. Ordinary anti-diabetes medication, such as Glibenclamide, had a significant effect in reducing blood glucose levels, improving the biochemical parameters, histological changes, and apoptosis, but at higher levels than normal. As a result, we hypothesized that controlling hyperglycemia would be ineffective in preventing diabetic complications. Furthermore, Glibenclamide has mild antioxidant and anti-inflammatory properties that cannot prevent diabetic complications.
However, several animal experiments and human case-control studies have suggested that vitamin D may play a protective role in diabetic complications. Because of its potent antioxidant and anti-inflammatory properties, vitamin D acts as a diabetes risk modifier. The current study found that combining Glibenclamide and vitamin D therapy was more effective in reducing apoptosis and improving the biochemical, histological, and immunohistochemical renal changes associated with diabetes to near-normal levels. As a result, combined therapy is critical in preventing diabetes-related inflammatory and oxidative stress-induced renal tissue injury.
Footnotes
Acknowledgments
The authors would like to thank the Deanship of Scientific Research at Prince Sattam bin Abdulaziz University, Alkharj, Saudi Arabia, for the assistance.
Authors’ Contributions
TA, HIS, MHA and MEE all contributed to the experimental design, data interpretation, and writing of the manuscript. MZI, HFA, DFM, MEA and MEE collaborated on the practical portion and data interpretation. MZI and HIS contributed to the statistical analysis. All authors contributed to the final revision.
Ethical Approval
The experimental protocol was established and approved by the University Bioethical Research Committee (BERC-001-09-20) at Prince Sattam University, KSA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
There was no fund for this work, the work was completely financially supported by the authors.
