Abstract
Chronic kidney disease (CKD) is debilitating, increasing in incidence worldwide, and a financial and social burden on health systems. Kidney failure, the final stage of CKD, is life-threatening if untreated with kidney replacement therapies. Current therapies using commercially-available drugs, such as angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers and calcium channel blockers, generally only delay the progression of CKD. This review article focuses on effective alternative therapies to improve the prevention and treatment of CKD, using plants or plant extracts. Three mechanistic processes that are well-documented in CKD pathogenesis are inflammation, fibrosis, and oxidative stress. Many plants and their extracts are already known to ameliorate kidney dysfunction through antioxidant action, with subsequent benefits on inflammation and fibrosis. In vitro and in vivo experiments using plant-based therapies for pre-clinical research demonstrate some robust therapeutic benefits. In the CKD clinic, combination treatments of plant extracts with conventional therapies that are seen as relatively successful currently may confer additive or synergistic renoprotective effects. Therefore, the aim of recent research is to identify, rigorously test pre-clinically and clinically, and avoid any toxic outcomes to obtain optimal therapeutic benefit from medicinal plants. This review may prove to be a filtering tool to researchers into complementary and alternative medicines to find out the current trends of using plant-based therapies for the treatment of kidney diseases, including CKD.
Keywords
Introduction
Chronic kidney disease
Interest in the health-benefiting properties of plant-based therapies is driven, in part, by the potential to prevent onset, or ameliorate progression, of certain diseases and reduce health care costs. The financial burden to health systems for CKD hospitalizations and treatments, worldwide, is immense and expected to increase.5,6 Complementary and alternative medicines have been used for CKD patients for many years, with reportedly disparate results for improving outcome. 8 In Western countries, these therapies are most likely used to complement conventional medicines, whereas in other countries such as China, other Asian, and African countries, sole use of alternative medicines is practiced for CKD patients. 9 Therefore, an important challenge to researchers is identifying low-cost and effective plant-based therapeutic options for CKD patients in poorer countries and disadvantaged populations. This review will describe the pathogenetic mechanisms of CKD that may be modulated by these therapies and emphasize and summarize information on some plants used for complementary and alternative medicine, using examples from pre-clinical and clinical studies.
Materials and Methods
PubMed, Medline, Google Scholar, Web of Science, Cochrane Library and China Journal Full-text Database for relevant articles published in English were used for this review of pre-clinical and clinical plant-based therapies for CKD. The contents of full articles were checked by Muhammad Ali Khan (MAK) and Glenda Gobe (GG) for quality of investigations. Only English articles were included.
Results
Pre-clinical research using plant-based therapies in in vitro and in vivo experiments demonstrates some robust therapeutic benefits. In the CKD clinic, combination treatments of plant extracts with conventional therapies that are seen as relatively successful currently may confer additive or synergistic renoprotective effects. These results are summarized in the following paragraphs.
Pathogenetic Mechanisms of CKD
The most common causes of CKD are aging, male gender, diabetes, hypertension and lifestyle. 10 Common pathogenetic mechanisms include tubulointerstitial fibrosis and chronic inflammation, tubular atrophy, glomerulosclerosis and proteinuria. 11 Chronic inflammation occurs through a series of biological signaling pathways involving the vasculature and the immune system, leading to the accumulation of inflammatory mediators in the tissue. 12 Fibrosis is a multifaceted cellular response primarily driven by various profibrotic and inflammatory cytokines such as transforming growth factor-β (TGF-β), tumor necrosis factor-α (TNF-α), platelet-derived growth factor, fibroblast growth factor-2, and some of the interleukins (ILs).13,14 Mechanistically, fibrosis and inflammation are exacerbated by oxidative stress. 15 Another key mechanism for kidney fibrosis is epithelial-mesenchymal transition (EMT) which is induced by various factors, including TGF-β, IL-1β and angiotensin II. 16
Hypoxia and inflammation coexist and have interactive roles in CKD. Hypoxia promotes inflammation by increasing vascular permeability, a central process in the movement of inflammatory cells that facilitate the production of inflammatory mediators. In contrast, inflamed tissues are often severely hypoxic because of increased metabolic demands. Some important inflammatory signaling pathways in CKD involve mitogen-activated protein kinase (MAPK), the transcription factor nuclear factor kappa B (NF-κB), p65 and some of the ILs. 17 The MAPK family consists of extracellular signal-regulated kinase, Jun N-terminal kinase and p38 MAPK. NF-κB is a key player in the production of pro-inflammatory cytokines and chemokines, such as TNF-α, IL-1β, IL-6, chemokine motif ligand 2 and macrophage inflammatory protein-2. 17 Hypoxia is closely regulated by hypoxia-inducible factor (HIF). HIF plays a critical role in inflammation and fibrosis during CKD through determining gene transcription, activation of multiple signaling pathways and epigenetic regulation, and contributes to the pathogenesis of comorbidities of CKD such as anemia, and aberrant angiogenesis. 18 Tissue hypoxia also causes mitochondrial dysfunction and oxidative stress, which lead to the generation of reactive oxygen species and reactive nitrogen species. Other leading causes of oxidative stress are proteinuria, uremic toxins, hyperglycemia and increased activity of the intra-renal angiotensin system.15,19
One of the recent research interests for pathogenesis of CKD is continuum of chronic injury after repair of acute kidney injury (AKI).20,21 AKI involves tubular epithelial cell apoptosis and/or necrosis. If the injury is mild, an adaptive repair process can lead to complete recovery. In contrast, if severe or prolonged injury occurs, this may be followed by maladaptive repair that progresses to chronic inflammation, vascular rarefaction, nephron loss, fibrosis and finally progression to CKD. One of the main causes of AKI is ischemia-reperfusion. Ischemia also causes release of damage-associated molecular pattern molecules (DAMPs) by damaged cells.22,23 The DAMPs promote and exacerbate the inflammatory response. 23 Inflammatory mediators such as inducible nitric oxide synthase, and pro- and anti-apoptotic signaling pathways also act in the continuum of disease progression from AKI to CKD. Ultimately, tubulointerstitial fibrosis develops, thereby impairing local oxygenation. Thus, a time-dependent, combined and cumulative interplay of fibrosis, inflammation and tubular cell loss leads finally to progression to CKD. Figure 1 shows relevant selected mechanisms for the pathogenesis of CKD.

Proposed mechanistic pathways to chronic kidney disease.
Multiple factors and many signaling pathways are responsible for chronic kidney disease (CKD). Ischemia-reperfusion injury (IRI) is one of the key causes of acute kidney injury (AKI) and apoptosis. Hypoxia produces oxidative stress and reactive oxygen species. Tubular inflammation stimulates immune cells to produce and accumulate extracellular matrix (ECM), causing malfunctioning of kidney structure. Chronic tubular inflammation causes vascular rarefaction and nephron loss. The transcription factor nuclear factor-κB (NF-κB) stimulates proinflammatory cytokines. Reactive oxygen species, proinflammatory cytokines and kidney structure malfunction cause kidney fibrosis and, ultimately, CKD.
Current CKD Treatment Strategies
As mentioned previously, there are limited therapies for CKD. Many current therapies rely on modulating the renin angiotensin system (RAS) which consists of angiotensinogen, renin, angiotensin-converting enzyme (ACE), angiotensin II, angiotensin II type 1 receptor and angiotensin II type 2 receptor. The renin-angiotensin-aldosterone system helps to regulate normal blood pressure. 24 Renin and ACE produce angiotensin II, which alone or in combination with other components of the RAS, upregulates TGF-β1 and causes renal fibrosis through extracellular matrix accumulation, via multiple signaling pathways. The RAS also regulates other profibrotic factors that lead to accumulation of extracellular matrix, causing podocyte loss in glomeruli and decrease in GFR. 25 ACE inhibitors (ACEi) and angiotensin receptor blockers (ARB), alone or in combination, are currently the most efficacious treatments for inhibition of the RAS and for maximal therapeutic effect on CKD, 26 although their use is not without controversy for kidney failure. 27 Recently, there has been an increasing interest in diet and the gut microbiome as modulators of CKD progression.28,29 Dietary supplements or natural foods are attractive, beneficial, and feasible complementary strategies to prevent or delay the progression of CKD. Natural products can also prevent kidney fibrosis through suppressing the RAS, inflammation and oxidative stress. 30 These products can also prevent kidney fibrosis by suppressing the Wnt/β-catenin and the TGF-β/small mother against decapentaplegic (Smad) profibrotic signaling pathways. 31 The inclusion of diet regulation in CKD therapies could be cost effective and possibly safer than combined regular drugs in reducing morbidity and mortality of CKD. Figure 2 summarizes the targets for current and alternative CKD therapies.
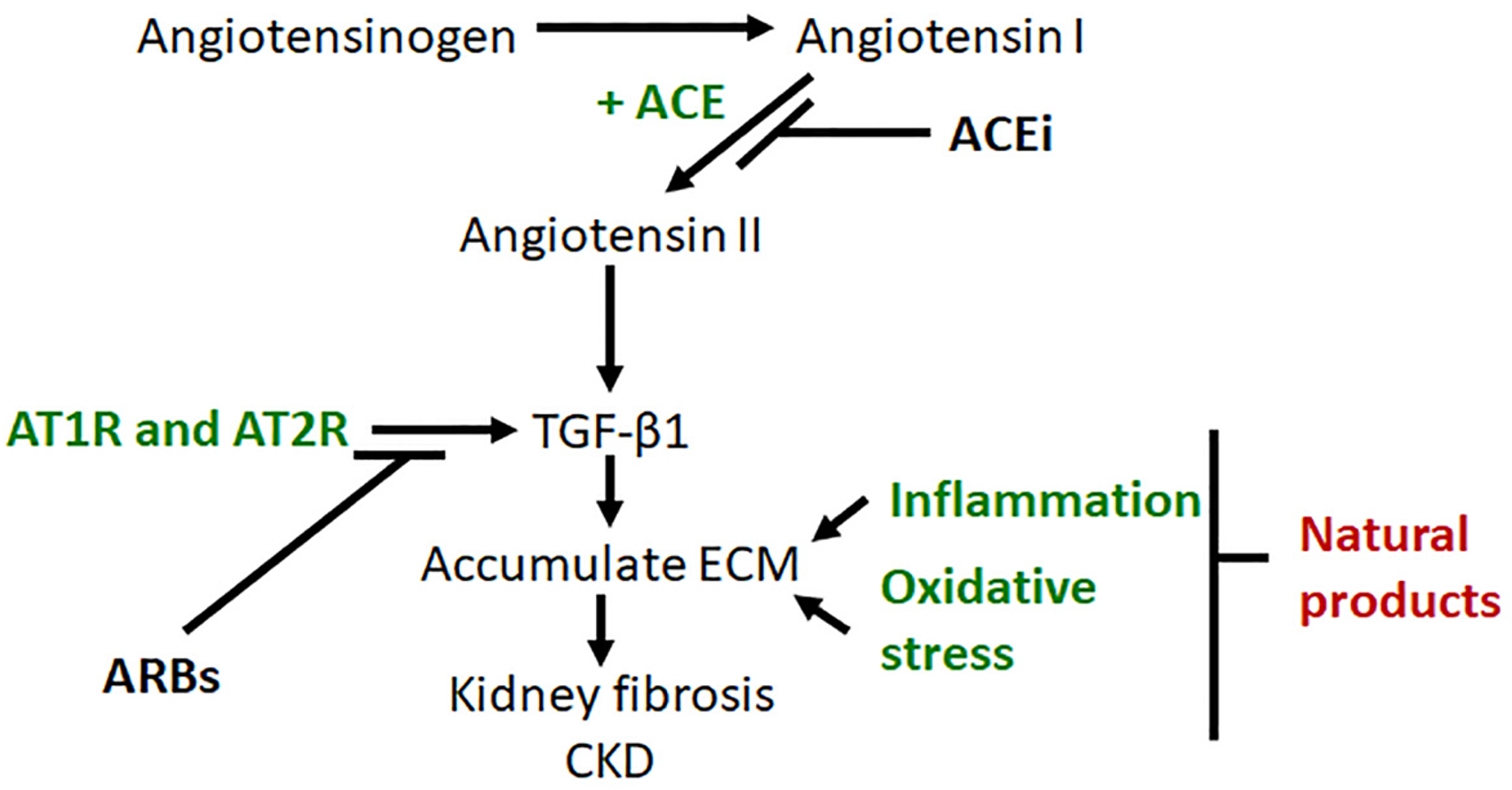
Pathways for current and alternative chronic kidney disease treatments.
After conversion of angiotensinogen to angiotensin by renin, angiotensin-converting enzyme (ACE) converts angiotensin I to angiotensin II. Angiotensin II together with angiotensin type 1 and type 2 receptors (AT1R and AT2R) stimulate pro-fibrotic transforming growth factor-β1 (TGF-β1) to produce and accumulate extracellular matrix, causing kidney fibrosis and finally chronic kidney disease (CKD). Inflammation and oxidative stress also stimulate ECM accumulation. The ACE inhibitor (ACEi) inhibits production of angiotensin II. Angiotensin receptor blockers (ARBs) block AT1R and AT2R and so TGF-β1 synthesis is blocked, ultimately inhibiting accumulation of extracellular matrix. Natural products (alternative therapies) may block the final common pathway of kidney fibrosis through multiple mechanisms, including those stimulated by oxidative stress and inflammation.
Plants as Natural Medicines
Plants, algae and fungi have been utilized as natural medicines throughout human history. 32 Medicinal plants are regarded as an acceptable, cheap, easily available and relatively safe source of many active compounds for pharmaceuticals. 9 In China, the use of traditional herbal medicine for kidney disease has shown some advantages over single conventional drug treatment.32,33 The beneficial effect of medicinal plants on kidney disease is often derived from their ability to boost the natural antioxidant defence mechanisms in the body. 34 Different types of phytochemicals such as flavonoids, vitamins, resveratrol, anthocyanin, curcumin and phenolic acid are often found in the plant-based medicines and may act as antioxidant.32,34,35 In this review, plants known to be beneficial in CKD, and to have minimal adverse outcomes, are discussed. Although by no means restricted to these plants, benefit has been demonstrated from Rheum spp. (Rhubarb), Astragalus membranaceus (Astragalus), Cordyceps sinensis (CS), Triptirygium wildfordii Hook F (TwHF), Abelmoschus manihot (L.) medic (AM), Salvia miltiorrhiza (SM), Vitis vinifera (Grape), and Zingiber officinale (Ginger). These plants and their extracts are sometimes used alone, but in many countries they are used in polyherbals for the treatment of kidney disease.36,37 The following section describes these selected plants and their extracts for treating CKD, their mechanisms of action, and their particular beneficial effects for CKD, using pre-clinical and clinical studies. Tables 1 and 2 give a broader list of plants used in pre-clinical and clinical studies for the treatment of CKD.
Medicinal Plants with Known Renoprotective Effects in Pre-clinical Trials.

Abbreviations: ALT, alanine amino transferase; BUN, blood urea nitrogen; CIC-5, chloride channel 5; CTGF, connective tissue growth factor; CKD, chronic kidney disease; Cr, creatinine; ER, endoplasmic reticulum; ECM, extracellular matrix; GSH, glutathione; Hb, hemoglobin; HO-1, heme-oxygenase-1; HDL, high density lipoprotein; IL-2, interleukin-2; IL-8, interleukin-8; MDA, malondialdehyde; MCP-1, monocyte chemoattractant protein-1; PPARα/γ, peroxisome proliferator-activated receptor α/γ; PLA-2, phospholipase A2; SCr, serum creatinine; SOD, superoxide dismutase; TGF-β1, transforming growth factor β1; VEGF, vascular endothelial growth factor; VLDL, very low density lipoprotein.
Medicinal Plants with Known Beneficial Effects in Human Clinical Trials.

Abbreviations: BUN, blood urea nitrogen; CKD, chronic kidney disease; CTGF, connective tissue growth factor; Cr, creatinine; Hb, hemoglobin; KIM-1, kidney injury molecule 1; OS, oxidative stress; PGE2, prostaglandin E2; SCr, serum creatinine; TGF-β1, transforming growth factor-β1; TCH, total cholesterol; UACR, urine albumin/creatinine ratio.
Rheum spp. (Rhubarb)
Rhubarb, derived from the root of Rheum spp indigenous to Asia, belongs to the Polygonaceae family. 91 Some species are cultivated for their potential to treat CKD.92,93 Rhubarb contains compounds such as saponins, flavonoids, volatile oils, polysaccharides, tannins, stilbene glycosides (resveratrol and piceatannol) and anthraquinone glycosides (physcion, aloe emodin, chrysophanol, emodin and rhein). The anthraquinone glycosides may have some inherent toxicities, but they can be removed from extracts 94 to produce an effective extract that is nephroprotective. 95 Many clinical and pre-clinical trials have consistently shown that extracts of rhubarb can reduce serum creatinine levels and offset other metabolic dysfunction related to kidney failure.
In CKD therapy, rhubarb increases the excretion of nitrogenous and other waste products through the intestine 75 and ameliorates uremic toxin accumulation, as demonstrated in various pre-clinical animal models of kidney failure. Using a model of diabetic nephropathy in mice, the proposed mechanism was thought to target the gut-kidney axis and trigger protective gut microbiota, rather than being directly nephroprotective. 96 The two primary active anthraquinones in rhubarb are rhein and emodin. Rhein prevents kidney damage by modulating various signaling pathways. It down-regulates the Wnt/β-catenin signaling pathway, up-regulates Sirtuin 1, decreases EMT that normally leads to fibrosis and ameliorates dyslipidemia.96–99 Rhein also improves cell metabolism through modulation of the glucose transporter I, decreasing mesangial cell hypertrophy and glomerulosclerosis. 100 Emodin reduces glycation of proteins, inhibits the lipopolysaccharide-induced expression of TLR4 and down-regulates pro-inflammatory TNF-α and IL-6 in the damaged kidney. 101 Emodin also decreases mesangial cell proliferation by inhibiting cellular FLICE-like inhibitor protein, 102 TGF-β1 and fibronectin, 103 p38 MAPK, 104 differentiation and maturation of dendritic cells, and increases the number of regulatory T cells. 105 These results indicate that the active ingredients in rhubarb have multiple mechanisms of action for the treatment of CKD, including regulation of inflammation and the immune response. Most of these reports have used pre-clinical models. 106 Clinically, rhubarb extract alone, or as a polyherbal, was beneficial in CKD patients, 75 delaying CKD progression and decreasing adverse effects of hemodialysis. The most common adverse side effects of rhubarb are nausea, vomiting, diarrhea, electrolyte disorders and liver toxicity.107,108
Astragalus membranaceus (Astragalus)
Astragalus membranaceus (synonymous name of Astragalus propinquus), belongs to the family Fabaceae and the genus Astragalus, which has more than 3000 species worldwide. 109 The plant is indigenous to the north and eastern regions of China but is grown worldwide and used widely in complementary and alternative medicines. Astragalus contains more than 60 bioactive compounds, including polysaccharides, saponins (astragalosides I–VII), flavonoids, amino acids and trace elements.110,111 In vitro and in vivo pre-clinical trials have revealed that Astragalus extract has potent antioxidant and anti-inflammatory effects.112,113 Other mechanisms include downregulating angiotensin receptors, inhibiting nitric oxide synthase and TNF-α production, and stimulating vascular endothelial growth factor and the immune system. 114 In addition, the plant extracts from Astragalus can rebalance profibrotic TGF-β/Smad signaling activity and inhibit endoplasmic reticulum stress-induced pathways.115,116
Several pre-clinical CKD models reported that the plant extract, either alone or as a polyherbal, showed anti-fibrotic effects, ameliorated proteinuria, attenuated kidney injury and promoted Reno protective effects.117,118 Wojcikowski et al. performed a pre-clinical trial in rats using the unilateral ureteral obstruction (UUO) model 119 of kidney fibrosis, with or without Astragalus membranaceus and Angelica sinensis (A and A). The ACEi Enalapril was administered in drinking water in some groups. The combined effect of A and A, with or without Enalapril, had anti-fibrotic benefits. Several clinical trials have reported that Astragalus significantly ameliorated CKD by decreasing proteinuria, with a corresponding increase in creatinine clearance, and hemoglobin levels.79,120 Astragalus is safe for most of the recipients. However, the plant is known to inhibit CYP3A4 hence it could affect the action of some other drugs that are metabolized by this enzyme. 121
Cordyceps sinensis (CS)
CS is a fungus that derives its nutrients from the larvae of Lepidoptera. 122 It is endemic to the Tibetan plateau but is produced commercially now. Various bioactive compounds, including amino acids, polysaccharides, organic acids, trace elements, nucleosides, peptides, steroids and other chemical components are present in CS extracts.123,124 The ability of CS to ameliorate urinary disorders and edema was described over 2000 years ago in traditional Chinese medicine. 9 In vitro studies with CS demonstrate potent antioxidant capacity. 125 In vivo, glomerular protection via inhibition of mesangial cell proliferation was demonstrated. 126 Treatment with CS also decreased apoptosis and kidney tubular atrophy, 127 and enhanced the immune system. 128
Extensive work has been carried out on CS alone, or in combination with other existing drugs, or other plant extracts. Different pre-clinical animal studies have been used to observe the beneficial effect on kidney disease, especially in CKD. In rat models of CKD, CS improved kidney function, reduced cardiovascular and endocrine comorbidities, inhibited oxidative stress, and reduced CKD-triggering metabolites.129–131 Clinically, CS extracts significantly reduced the levels of serum creatinine, proteinuria, hematuria, inflammatory cytokines, monocyte chemo attractant protein-1 and TNF-α. 132 Clinical trials of CS, alone or in combination with other medicinal plant extracts or existing drugs, showed significant improvement of kidney function, with reduced levels of serum creatinine, albuminuria, proteinuria, blood urea nitrogen (BUN) and profibrotic TGF-β1, and increased creatinine clearance.133–137 There are very limited reports of adverse effects of dietary supplements of CS in humans. The most common adverse side effects are nausea, dry mouth and stomach upset. 137
Tripterygium wilfordii Hook F (TwHF)
TwHF is a member of the Celastraceae family, and is native to Eastern and Southern China, Korea, Japan and Taiwan. 138 A stable glycoside named “multi-glycoside of TwHF” (GWT) 139 has been approved by the China State Food and Drug Administration (Z32021007) for the treatment of chronic diseases such as rheumatoid arthritis and glomerulonephritis. In China, GWT has been used extensively for the treatment of proliferative glomerulonephritis for more than three decades.140,141 The major active compound of GWT is triptolide. 142 GWT showed several beneficial therapeutic effects via its immunosuppressive, immunomodulatory, anti-inflammatory, antioxidant and antitumor activities.143,144
Pre-clinical studies of CKD have demonstrated that TwHF reduced tubular apoptosis and kidney fibrosis, as well as improving kidney function by inhibiting the TGF-β1/Smad3 signaling pathways. 144 Clinically, GWT in combination with irbesartan reduced excretion of urinary proteins, including urinary connective tissue growth factor and TGF-β1, in diabetic nephropathy patients. 145 In combination with an ARB, podocyte injury was reduced in a similar patient population. 80 However, the use of TwHF extract is limited due to the lack of high-quality clinical trial data. 146 TwHF causes some common adverse side effects, such as stomach upset, diarrhea, skin rash and changes in skin pigmentation. 147 The other concerning, although reversible, side effects are infertility in men and amenorrhea in women. 148 These adverse side effects limit its usefulness for the treatment of CKD.
Abelmoschus manihot (L.) medic (AM)
AM belongs to the Malvaceae family. It has been used as a traditional medicine for hundreds of years in China, notably for the treatment of CKD. 149 Due to its potential renoprotective properties, the State Food and Drug Administration of China approved the Huangki capsule (HKC, made from AM) as a Class III new drug (GUOYAOZHUNZI Z19990040) for the treatment of chronic glomerulonephritis. The main active components of HKC are total flavonoids extracted from the flowers of AM, sometimes known as Aibika flowers. Recent research considered HKC as an important adjuvant treatment for CKD, especially as a side effect of diabetes, and suggested that HKC is more effective than the ARB losartan in reducing proteinuria.150,151
The beneficial effect of AM or HKC on CKD and other kidney complications relate to potential anti-inflammatory and antioxidant activities, probably via the flavonoids. 152 Several pre-clinical studies in animal models demonstrated that HKC reduced urinary albumin excretion, serum triglycerides, cholesterol levels and podocyte apoptosis. 153 Kidney inflammation and glomerular injury were reduced as evidenced by fewer activated macrophages in glomeruli, reduced TNF-α protein expression in the kidney, and a dampened p38 MAPK signaling pathway.154,155 There are limited data on AM (or HKC) as an anti-fibrotic agent in clinical trials. AM alone, or together with other drugs, reduced levels of proteinuria, BUN, serum creatinine, and also decreased oxidative stress. 156 There were no serious adverse effects from taking AM, except mild to moderate gastrointestinal discomfort.
Salvia miltiorrhiza (SM)
SM, commonly known as Danshen, is a perennial plant of the genus Salvia, within the family Lamiaceae. It is native to most Asian countries, including China. The roots and rhizomes of the plant have been widely used in traditional Chinese medicine for thousands of years. 157 More than 100 bioactive compounds have been isolated from this plant and characterized for their structure. SM has been used for the treatment of numerous diseases, including CKD, because of its potent anti-inflammatory and antioxidant properties.158–161 The bioactivity of SM extract relates to the presence of phenolics, flavonoids, terpenoids, polysaccharides and other related quinone compounds. 160 For instance, Tanshinone IIA, a di-terpenoid isolated from SM, showed antioxidant benefit by activating the Nrf2 signaling pathway, improving the metabolic profile and exhibiting potent renoprotective effects, 160 and the lipophilic fraction attenuated oxidative stress in diabetic nephropathy. 161
Pre-clinical trials of SM alone, or in combination with other drugs, reduced albuminuria, improved kidney function, decreased fibrosis and TGF-β/Smad3 signaling, suppressed pro-inflammatory cytokines and adhesion molecules, and inhibited apoptosis.162,163 SM extracts also ameliorated proteinuria, serum and kidney tissue levels of TGF-β1 and collagen IV, reduced numbers of monocytes/macrophages and exhibited a significant renoprotective effect. Of note, a clinical trial of a polyherbal that contained SM for CKD stabilized the albumin/creatinine ratio, increased hemoglobin content and improved accumulative survival rate of the test group. 164 No serious adverse side effects of SM have been reported.
Vitis vinifera (grape)
Vitis vinifera, or grape, belongs to the Vitaceae family. 165 Grape seeds contain more than 1600 phytonutrients, including flavonoids, catechin, anthocyanins, flavonols, non-flavonoids and pro-anthocyanidins, and their extracts exhibit enormous biological activity. 166 For this review, one of the most important of the biological actions is inhibition of progress in CKD. 167 Grape seed extracts typically act by alleviating oxidative stress and endoplasmic reticulum stress-induced apoptosis.168–170 Pre-clinical studies of grape seed extracts in animal models of CKD have also reported improved kidney function, decreased proteinuria and reduced podocyte cell death. 171 Clinical trials have demonstrated that grape seed extracts increased GFR, decreased proteinuria and triglycerides, prevented anemia, and counteracted plasma low density lipoprotein and thrombocytopenia. 81 There are no known adverse side effects at normal doses of grape seed extracts.
Zingiber officinale (ginger)
Ginger is used widely as a spice but also often in folk medicine. It belongs to the Zingiberaceae family and has been cultivated for thousands of years especially in China and the South Asian countries. 172 Ginger contains many beneficial compounds, the most important of which are 6-, 8-, and 10-gingerol and 6-shogaol. 173 It displays diverse beneficial biological actions due to its potent antioxidant, anti-inflammatory, anti-tumour, anti-diabetic and neuroprotective activities. In pre-clinical animal models of kidney and cardiovascular diseases, ginger extracts lowered blood glucose levels, restored the total carbohydrates, pyruvate, glycogen and total protein in kidney tissue, promoted the regeneration of tubules and restored glomeruli, and reduced fatty infiltration.174–177 One clinical trial of ginger extract in CKD patients on peritoneal dialysis, demonstrated that daily administration of 1000 mg ginger reduced serum fasting glucose, a risk factor for diabetes, diabetic nephropathy and cardiovascular disease. 178 There have been no adverse side effects reported when doses are kept to a moderate level.
Some other nephroprotective medicinal plants
There are many other plants reported to have benefits for kidney failure and/or CKD. Rutin (or quercetin) is a flavonoid present in onions, apples, tea and red wine. This plant extract is readily available “over the counter” and has demonstrated strong antioxidant and anti-inflammatory properties in heart and liver in a high fat-high carbohydrate diet model of metabolic syndrome in rats. 179 Rutin also protected the kidney against ischemia-reperfusion injury, 180 cisplatin-induced nephrotoxicity, 181 and diabetic nephropathy. 182 It normalized BUN, thereby modulating a key factor in the pathogenesis of CKD. 73 However, some other studies did not show benefits. For example, rutin may induce protein-energy malnutrition in CKD. 183 Although there are some promising results, more analyzes are needed to confirm whether, or not, preclinical and clinical benefits exist for this plant extract, particularly in the context of CKD. Clinically, Glycyrrhiza glabra extracts consistently decreased pre-dialysis serum potassium concentrations in chronic hemodialysis patients. 83 Silybum marianum, known as “milk thistle” or silymarin, is a very safe herb that protects against kidney failure and end-stage diabetic nephropathy. 184 Significant benefits have been claimed for Lespedeza tincture for both AKI and CKD patients. 185 Clinically, beetroot juice decreased peripheral systolic and diastolic blood pressure, mean arterial pressure, improved kidney function, histological damage and kidney prognosis, and prevented cardiovascular events.86,186 In a recent study, a beetroot protease inhibitor was isolated and characterized. 187 The results showed the potential of such plant protease inhibitors for peptide-based drug discovery against targets involved in diseases such as cancers and immune system related diseases, such as is seen in CKD in some instances. Coptis rhizome extracts significantly reduced biomarkers of kidney damage, such as urinary albumin-to-creatine ratio, urinary osteopontin and KIM-1, and improved kidney hemodynamics. In addition, clinically, it decreased inflammation and oxidative stress. 87 Extracts of Urtica dioica, or “stinging nettle”, significantly attenuated kidney damage and tubular atrophy, loss of brush border, hydropic epithelial cell degeneration, glomerular shrinkage, and tubulointerstitial fibrosis, as well as demonstrating clinical benefits in patients undergoing partial nephrectomy or kidney transplantation. 188 The polyherbal named “Sairei-To” significantly decreased urinary protein excretion, hematuria and normalized proteinuria in CKD patients. 90 A dietary supplementation with curcumin (turmeric) reduced oxidative stress and proteinuria in CKD patients. 88 A combination of Curcuma longa and Boswellia serrata decreased the levels of inflammatory cytokines and ameliorated inflammatory markers in patients with CKD. 89
Concerns of Toxicities of Plant-Based Extracts to the Kidney
Natural products are often considered safer than traditional drugs, and many of our modern drugs are derived from herbs. Nonetheless, some researchers are concerned over their safe use.189–191 The possible nephrotoxicity and other chronic or insidious health disorders of using popular complementary and alternative medicine in different countries is not properly reported. The most well-known toxicity or side-effect is associated with traditional Chinese herbal medication that uses aristolochic acid and induces aristolochic acid nephropathy (AAN). The clinical features of AAN are characterized by extensive interstitial fibrosis and tubular atrophy in the kidney without obvious glomerular injury. Prolonged use causes uroepithelial malignancies. 189 In addition, traditional herbal remedies have been implicated in 35% of all cases of AKI in Africa. 190 The Therapeutic Goods Administration restricted the use of ephedra in Australia due to its potential toxic effects (quickened heartbeat and elevated blood pressure, heart palpitations, nausea and vomiting). 189 Some herbal drugs that contain specific herbs, nuts and mushrooms may also be associated with inherent nephrotoxicity. Interestingly, the inherent properties of the herbs are not the only source of herb-associated kidney disorders. Herb-drug interactions, mistakes in dosage and identification, contaminants within mixtures, adulteration with heavy metals, and even deliberate adulteration with non-labelled plant extracts are all issues of concern. 191 However, with proper identification, rigorous pre-clinical and clinical trials, strict controls on the presence of adulterants within herbal medicines, labeling of dosages and contraindications, and robust manufacturing techniques, the safety of those consuming herbal medicines should be maintained.
Conclusion
There is no doubt that medicinal plants present a largely untapped source of new CKD therapies. Clinical and pre-clinical trials of plant extracts sometimes demonstrate benefit but some research has also demonstrated that plant extracts may produce chronic organ dysfunction when used for long term due to the presence of harmful chemicals. Therefore, the aim of recent research is to identify, rigorously test pre-clinically and clinically, and avoid such toxic outcomes in order to obtain optimal therapeutic benefit from medicinal plants. This review may prove to be a filtering tool to researchers into complementary and alternative medicines to find out the current trends of using medicinal plants and plant extracts for the treatment of kidney diseases, including CKD.
Footnotes
Abbreviations List
Acknowledgments
Muhammad Ali Khan is supported by a University of Queensland Research Training Scholarship (RTP) and a Top-up Scholarship from the Kidney Disease Research Collaborative, Translational Research Institute, Brisbane Australia. This group received National Health and Medical Research Council of Australia support from its Centre of Research Excellence grant, 2017-2020.
Author Contributions (Roles)
Each author has participated sufficiently in the work to take public responsibility for the content. M.A.K. and G.G. conceptualised the review article. All authors contributed in writing, reviewing and editing of the original draft. M.A.K. compiled the manuscript and G.G. supervised the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Health and Medical Research Council of Australia.
