Abstract
VAMP-associated proteins (VAPs) are highly conserved, endoplasmic reticulum (ER)-resident receptors that tether the ER to various membrane compartments in eukaryotic cells. Each VAP contains a transmembrane helix at its extreme C-terminus and a conserved N-terminal major sperm protein (MSP) domain that mediates various cytosolic interactions via both protein and lipid binding. Here, I question the fundamental difference between protein- and lipid-based associations in VAP-driven membrane contact site (MCS) formation and function – could the lipid affinity of VAPs be an overlooked factor in MCS dynamic regulation?
Keywords
Introduction
In eukaryotes, membrane-bound organelles are interconnected at MCSs where respective activities can be jointly regulated. Among various characterized membrane tethering machineries, VAPs (Scs2 and Scs22 in yeast; VAP27, VAP27-1, and VAP27-3 in plant; VAPA and VAPB in human) are unique versatile ER tethers that couple the ER to multiple organelles. They interact with a diverse array of proteins containing well-conserved FFAT (two phenylalanines/F in an acidic tract) or related motifs at multiple MCSs, while FFAT-independent VAP-protein interactions are also common (Murphy and Levine, 2016). Additionally, VAPs exhibit general binding affinity towards anionic phospholipids, a feature that appears to be conserved among homologs across eukaryotic kingdoms (Hoh et al., 2024; Kagiwada and Hashimoto, 2007; Stefano et al., 2018).
In this article, I focus on VAP-mediated associations occurring at MCSs and summarize these interacting partners identified thus far across various organisms. I further discuss how the distinct properties of protein- or lipid-based VAP interactions may differentially contribute to the dynamic formation and function of MCSs. By emphasizing the importance of MCS plasticity for assembly and disassembly, I propose that membrane coupling via weak interactions may play a crucial role in dynamic MCS remodeling.
VAP-mediated Interactions at MCSs: Via Proteins and Anionic Phospholipids
FFAT or related motifs are commonly found in VAP-interacting proteins at various MCSs, many of which are lipid transfer proteins (LTPs) or sensors (Table 1). The classic FFAT motif binds VAPs at a highly conserved, positively charged region in the MSP domain. This association is primarily driven by hydrophobic interactions, particularly between the first aromatic residue of the FFAT motif and its binding cavity on VAP (Kaiser et al., 2005). Non-specific electrostatic interactions of VAPs with flanking residues before the FFAT motif are also thought to aid their initial association (Furuita et al., 2010; Mikitova and Levine, 2012). Variations in FFAT motif sequences can create a range of binding affinities for VAPs. For example, the canonical FFAT motifs – originally identified in the LTP oxysterol binding protein (OSBP)-related protein (ORPs, also named as Osh in yeast) family and the budding yeast transcriptional repressor Opi1 as efficient ER-targeting signals (Loewen et al., 2003) – exhibit much stronger binding to VAPs than the expanded FFAT-like motif found in proteins such as the fission yeast plasma membrane (PM) proton pump Pma1 (Hoh et al., 2024). Moreover, phosphorylation of serine or threonine residues at the fourth position of some poorly conserved non-conventional FFAT motifs, referred to as phospho-FFAT (#, Table 1), acts as phospho-mimicry of acidic residues and has been shown to promote VAP binding (Di Mattia et al., 2020; Johnson et al., 2018). The MSP-binding mode of this phospho-FFAT motif differs from that of the canonical FFAT motif, exhibiting generally weaker affinity which also depends on additional phosphorylation in the surrounding region (Di Mattia et al., 2020).
VAP-Interacting Proteins at Various MCSs Across Organisms.

Proteins that bind VAP via a phospho-FFAT motif are marked with #. VAP interactors that lack a FFAT motif are marked with *; those lacking a FFAT motif within their recognized VAP-binding domain are marked with **; those that bind via a noncanonical FFAT variant are marked with ***.
Notably, several major PM-localized VAP interactors at ER-PM contact sites across yeast, plant, and human either lack a verified FFAT-related motif entirely (*, Table 1), lack one in the identified VAP-binding domain (**, Table 1), or carry a noncanonical variant (***, Table 1). This may suggest broader VAP-protein interaction modes at MCSs beyond FFAT-based binding and raises the question of whether VAP-FFAT interactions are the dominant mechanism for VAP-mediated MCS formation, especially given the high abundance of ER-PM contacts in yeast and plant. It is conceivable that the strength of cross-membrane interactions collectively determines membrane tethering and further influences the abundance and dynamics of MCSs. However, little is known quantitatively about the threshold of membrane tethering forces required to establish bona fide MCSs. Some VAP-protein interactions at MCSs, regardless of their dependence on a FFAT-related motif, may not represent effective cross-membrane associations. Instead, they might mainly reflect protein recruitment to the MCS (also discuss further below). Indeed, a recent study in fission yeast suggests that direct protein interactions are likely not the primary drivers for VAP-mediated ER-PM coupling. Rather, VAP interactions with major anionic phospholipids (PLs) in the PM (∼ 20% PI, 10% PS and 1% PI4P + PI(4,5)P2 in yeast (McLaughlin and Murray, 2005; van ’t Klooster et al., 2020)) may provide alternative membrane tethering forces (Hoh et al., 2024). These weak and nonspecific electrostatic VAP-PLs interactions could sustain constitutive ER-PM contacts while allowing their highly coordinated local and global remodeling during fission yeast growth and division (Ng et al., 2018; Zhang and See, 2022; Zhang et al., 2012).
VAP interactions with abundant phosphatidylinositol (PI), phosphatidylserine (PS) and highly charged phosphoinositides (PIPs) appear to be conserved across the family (Table 2), although binding affinities can vary among homologs (Hoh et al., 2024). As organelles in some cell types may contain sufficient levels of anionic PLs in their cytosolic leaflets – for instance, the Golgi complex (enriched in PI and PI4P), endosomes (enriched in PS, PI3P and PI(3,5)P2), mitochondria (enriched in PI), peroxisomes (enriched in PI), and autophagosomes (enriched in PI3P) (Gillooly et al., 2000; Leventis and Grinstein, 2010; Pemberton et al., 2020; Petiot et al., 2000; Uchida et al., 2011; van Meer et al., 2008; Zewe et al., 2020) – VAP-PLs tethering could also promote ER contacts with these organelles. It is an enthralling idea that the distribution of anionic PLs among organelles might inherently shape the scale and types of VAP-mediated MCSs under varying conditions.
VAP Interactions with Anionic Phospholipids Across Organisms.
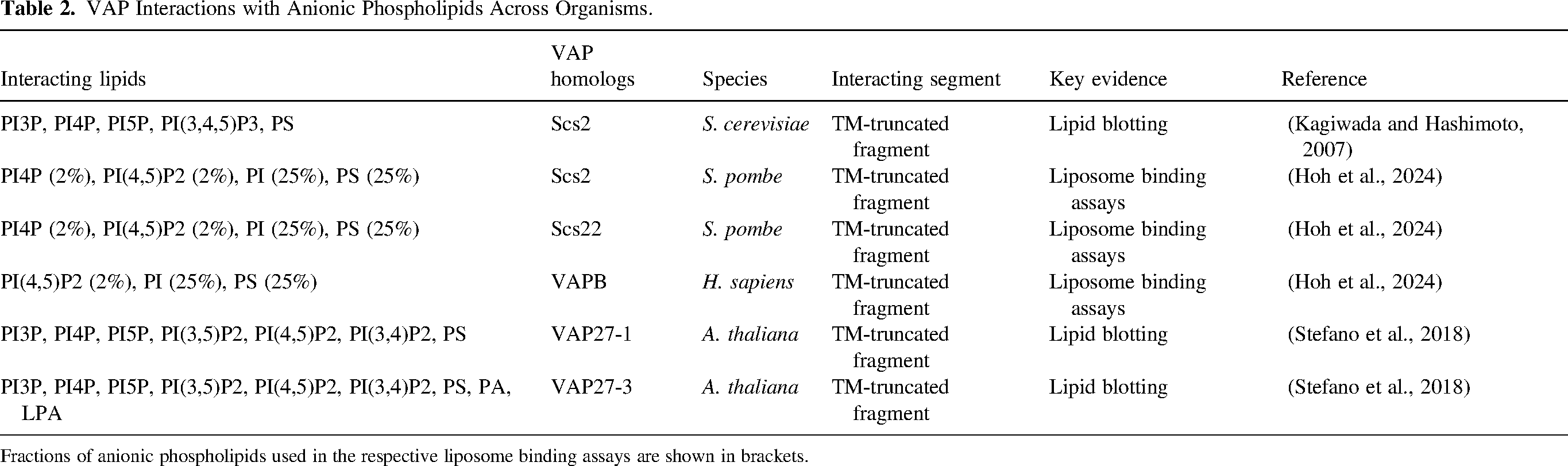
Fractions of anionic phospholipids used in the respective liposome binding assays are shown in brackets.
Membrane Contact Plasticity: Balancing Membrane Coupling and Uncoupling
Membrane tethering and molecular assemblies at MCSs are often dynamic. For example, transient contacts between the ER and organelles such as mitochondria or endosomes frequently form to promote organellar fission. Moreover, machineries for store-operated Ca2+ entry rapidly assemble at induced ER-PM contacts to restore Ca2+ when its ER luminal level drops (Zhang, 2020). Beyond their roles in organelle and calcium homeostasis, MCSs also facilitate non-vesicular lipid transport via LTPs, making them hotspots for lipid regulation. These findings highlight the importance of MCS formation in diverse cellular functions. However, this does not imply that MCS disassembly is always unfavorable for cells.
In fact, MCSs are confined regions, typically 10–80 nm apart, where two membranes are closely apposed without fusion. This limited spacing can hinder space-demanding processes that involve large protein complexes or vesicles. For instance, ER-PM contact sites can impose steric constraints on contractile ring assembly (Zhang and See, 2022; Zhang et al., 2012, 2016), septin collar formation (Sugiyama and Kono, 2024) and the exocytic territory (Ng et al., 2018, 2020) in yeast. These physical effects depend on the abundance and plasticity of ER-PM contacts, making their removal a crucial step in finetuning this barrier effect. Thus, suitable MCS plasticity – i.e., a delicate balance between membrane coupling and uncoupling – is likely fundamental to cellular homeostasis, although it remains to be seen how MCS plasticity affects the global state of lipid regulation.
While much attention has been given to unraveling the molecular interactions involved in MCS assembly, far less is known about how tethering forces adapt to permit MCS disassembly. From a biophysical perspective, membrane association at MCSs may be sustained by numerous weak interactions, or relatively fewer strong ones, or a mixture of both. Membrane tethering strength could further depend on the number and spatial distribution of these interactions, the size and geometry of individual contact points, and the dynamic nature of the involved organelles. Small MCSs, albeit not well-defined, such as dynamic ER-endosome interfaces (∼130 nm in length along a single cross-section (Eden, 2016; Friedman et al., 2013)), may accommodate a limited number and variety of tethering machineries, possibly favoring stronger interactions to support contact formation while retaining the disassembly potential. In contrast, larger and more stable MCSs, like the extensive ER-PM junctions in yeast, may afford a broader set of tethering players and weaker interactions to preserve MCS flexibility while maintaining continuous presence.
Although the molecular details of MCS disassembly remain underexplored, membrane decoupling at smaller MCSs may involve relatively simple processes, such as reducing the levels or tethering activity of a few key proteins. In contrast, modulating the structural plasticity of larger and more constitutive MCSs could be more complex. Taking yeast ER-PM contacts as an example, reticulation of the cortical ER by various shaping proteins generates a network of regularly spaced, interconnected small contact sites, likely enabling localized membrane contact remodeling (Ng et al., 2020; Zhang and See, 2022). Weak interactions between anionic PM PLs and the major ER tethers – VAPs, tricalbins and Ist2 (Hoh et al., 2024; Manford et al., 2012; Zhang et al., 2012) – may be relatively vulnerable to disruption by shearing forces from the cytoskeleton or motor proteins. This could allow for local remodeling of ER-PM contacts in response to dynamic cortical events, such as actomyosin ring assembly (Zhang and See, 2022). Moreover, these electrostatic interactions can be weakened by cytosolic acidification (Hoh et al., 2024), linking ER-PM contact plasticity to the cellular metabolic state. Furthermore, a reduction in the levels of tethering counterparts (both lipid and proteins) in the PM, due to exocytic membrane flow and associated changes in PM tension, can also promote ER-PM decoupling near exocytic regions, as observed in both budding and fission yeast (Ng et al., 2018, 2020; Sugiyama and Kono, 2024; Zhang and See, 2022). Together, these possible mechanisms highlight the advantage of membrane coupling via weak interactions, particularly for constitutive MCSs, which allows flexible and responsive remodeling without compromising overall stability.
How do VAP-Based Interactions fit into Dynamic MCS Regulation?
VAPs engage in both strong and weak protein interactions at MCSs, ranging from high-affinity interactions via classic FFAT motifs (e.g., Osh1 (Loewen et al., 2003)), to weaker but specific ones involving FFAT variants (e.g., Pma1 (Hoh et al., 2024)) or FFAT-independent mechanisms, and even to non-specific electrostatic associations (e.g., the eisosomal protein Pil1 (Ng et al., 2020)). Many VAP-interacting LTPs contain FFAT-related motifs (Table 1) and can associate both membranes at MCSs by binding to lipids or resident proteins. For example, some ORP/Osh proteins possess a pleckstrin homology (PH) domain that binds PIPs enriched in the PM, Golgi, or endosomes, and are thus often considered as part of the VAP-based tethering complex. These proteins mediate non-vesicular PI4P transfer at corresponding ER MCSs, enabling ER-resident phosphoinositide phosphatase Sac1-mediated PI4P metabolism and sustaining PI4P levels in the respective membranes (Mesmin et al., 2013; Stefan et al., 2011). However, despite their essential role in maintaining PI4P levels in yeast PM implying a tethering potential at ER-PM contacts, ORP/Osh proteins appear to contribute little to membrane coupling. This is supported by the observation that the elimination of all ORP/Osh proteins does not reduce VAP-dependent ER-PM contact sites in either budding (Quon et al., 2022) or fission yeast (our unpublished data). It is thus plausible that such VAP-FFAT interactions may primarily serve to recruit ORP/Osh or other LTPs to function at MCSs, rather than to mainly form contact sites themselves. Indeed, a recent work showed that ORP/Osh-dependent PI4P metabolism remains functional even when VAPs are artificially anchored to the PM to mediate ER-PM contacts in fission yeast (Hoh et al., 2024). However, this does not exclude the possibility that strong VAP interactions with ORP/Osh or other LTPs could still be major forces to drive the formation of other perhaps more transient ER MCSs. This would also rely on the strength of these LTPs’ associations with the opposing membranes.
Could FFAT-mediated VAP interactions play a broader role in regulating protein activities, beyond just recruiting proteins to MCSs or facilitating MCS formation? For example, in the case of LTPs, it remains unclear whether VAP-FFAT interactions may directly modulate their lipid transfer functions through conformational regulation. It is intriguing to consider if the unbound form of the FFAT motif could structurally differ from the extended loop observed in purified, stable VAP-FFAT complexes (Di Mattia et al., 2020; Furuita et al., 2010; Kaiser et al., 2005; Kim et al., 2022). Conformational adaptations of both the FFAT motif and VAPs may occur during complex formation or dissociation (Di Mattia et al., 2020; Furuita et al., 2010). Thus, resolving the ultrastructure of an intact VAP-LTP complex, rather than a minimal complex containing just the motif, could be highly informative, albeit technically challenging. Alternatively, complex modelling using Alphafold 3 may offer preliminary insights (Abramson et al., 2024).
Moreover, a recent study suggests that a conserved FFAT-like motif located at the autoinhibitory site of the PM proton pump Pma1 may mediate competitive binding with VAPs. This interaction may modulate Pma1 activity by preventing self-inhibition, potentially prolonging proton pumping under low-glucose conditions to delay cytoplasmic acidification before ATP exhaustion. Interestingly, unlike electrostatic VAP-PLs interactions that render ER-PM contacts sensitive to cytosolic pH, the FFAT-mediated VAP-Pma1 association remains stable under mild acidification (Hoh et al., 2024). This may indicate that non-specific VAP-lipid interactions might primarily support the structural flexibility of MCSs, whereas specific VAP-FFAT associations are likely to play more specialized roles in functional regulation at MCSs. Thus, employing both types of VAP interactions at MCSs could enable coordinated structural and functional remodeling.
Alternatively, the emergence of phospho-FFAT motifs may enable similar coordination through phosphorylation-dependent regulation. Notably, many proteins that possess a phospho-FFAT motif, such as STARD3, MIGA2, PTPIP51, Kv2.1, and Kv2.2, are membrane-anchored via transmembrane domains. These phosphorylation-regulated FFAT-VAP coupling and uncoupling could be an ideal adaptation for maintaining the structural flexibility of MCSs.
Concluding Remarks and Perspectives
With our growing understanding of major tethering machineries that define various MCSs, one of the next challenges is to unravel how cells spatiotemporally regulate the dynamic assembly and disassembly of these structures, and how activities of distinct MCSs are integrated and coordinated across different physiological and stress-related cellular states (Zhang et al., 2021). While VAP-based MCSs represent some of the most well-characterized examples, some fundamental questions remain unanswered, e.g., how do cells decouple the tight association of VAPs with classic FFAT motifs? In parallel, intriguing new functions continue to emerge, such as roles of VAP-FFAT interactions in host-pathogen interplays during viral and bacterial infections (Angara et al., 2024; Barajas et al., 2014; Furuita et al., 2021; Roulin et al., 2014), which are not discussed here.
Most structural studies conducted so far suggest that non-specific electrostatic interactions between VAPs and surrounding residues of various FFAT motifs are crucial for overall binding affinity and the ultimate formation of a stabilized complex. Based on this, several speculative questions may be worth considering. Could the similar charge-based affinity between VAP and anionic PLs provide a general background for initiating interactions at membrane-rich niches to establish nascent contacts, where more specific associations would then evolve and prevail at MCSs? Could lipid affinity of VAPs be an overlooked factor that intertwines with the functioning of LTPs, such as facilitating lipid extraction and deposition?
Footnotes
Acknowledgments
This work has been funded by the Temasek life sciences laboratory.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Temasek Life Sciences Laboratory (grant number NA).
