Abstract
Apicomplexan parasites are a group of protists that cause disease in humans and include pathogens like Plasmodium spp., the causative agent of malaria, and Toxoplasma gondii, the etiological agent of toxoplasmosis and one of the most ubiquitous human parasites in the world. Membrane contact sites (MCSs) are widespread structures within eukaryotic cells but their characterization in apicomplexan parasites is only in its very beginnings. Basic biological features of the T. gondii parasitic cycle support numerous organellar interactions, including the transfer of Ca2+ and metabolites between different compartments. In T. gondii, Ca2+ signals precede a series of interrelated molecular processes occurring in a coordinated manner that culminate in the stimulation of key steps of the parasite life cycle. Calcium transfer from the endoplasmic reticulum to other organelles via MCSs would explain the precision, speed, and efficiency that is needed during the lytic cycle of T. gondii. In this short review, we discuss the implications of these structures in cellular signaling, with an emphasis on their potential role in Ca2+ signaling.
Introduction
Eukaryotic cells are defined by the presence of membrane-bound organelles, which allow the compartmentalization of numerous biochemical processes. To ensure their interaction, organelles can exchange materials via vesicular trafficking or cytoplasmic diffusion. In addition, inter-organellar communication can take place via specialized structures known as membrane contact sites (MCSs). In yeast, plants and mammalian cells, MCSs are ubiquitous (Scorrano et al., 2019; Shai et al., 2018; Valm et al., 2017) and are maintained by protein tethers, which physically link membranes from two different organelles (Gatta and Levine, 2017). These interactions create a contact area where the exchange of molecules such as metabolites and lipids is facilitated. MCSs are important for organellar trafficking and division, for cellular signaling, and for autophagy. In humans, their dysregulation is associated with various diseases, underscoring their role in cellular homeostasis (Janer et al., 2016; Vance, 2020).
Ca2+ signaling plays essential role in the regulation of many cellular functions (Clapham, 2007). However, to enable proper signaling, the concentration of cytosolic Ca2+ is kept low by the activity of pumps and transporters that extrude Ca2+ through the plasma membrane (PM) or sequesters it into organelles like the endoplasmic reticulum (ER), the Golgi, lysosomes or mitochondria (Bootman and Bultynck, 2020). In most cells, the largest calcium store is undoubtedly the ER. This organelle is known to form a dynamic membranous network (Cremer et al., 2020; Grigoriev et al., 2008) that extends far into the cell periphery, which facilitates its interactions with other organelles (Phillips and Voeltz, 2016; Westrate et al. 2015) including the Golgi apparatus, lysosomes, lipid droplets, the PM and the mitochondria (Wu et al., 2018). These interactions, established through MCSs and localized to specific areas within the organellar membranes (King et al., 2020), facilitate Ca2+ tunneling through the ER lumen and delivery to targeted sites without involving global Ca2+ increases which may activate unsuitable processes in the cell cytosol (Petersen et al., 2017). In this way, Ca2+ is released on demand, delivering bursts of local concentration gradients (Filadi and Pozzan, 2015).
Apicomplexans are a group of single-celled parasites that cause disease in humans. Some of the most well-known apicomplexan parasites include Plasmodium spp., which causes malaria, and Toxoplasma gondii, which infects a third of the world's population and causes toxoplasmosis (Tenter et al., 2000; Cowman et al., 2016). As an obligate intracellular parasite, T. gondii replicates inside cells and the clinical manifestations of toxoplasmosis are the result of its replication and dissemination to all tissues. In T. gondii, Ca2+ signaling triggers a cascade of molecular events that culminate in the stimulation and regulation of several essential parasitic functions including host cell invasion, motility, differentiation, and egress from the infected cell (Lourido and Moreno, 2015). The cytosolic concentration of Ca2+ is likely maintained at ∼70 nM through the concerted operation of transporters at the PM and intracellular stores (Moreno and Zhong, 1996). The ER, the plant-like vacuolar compartment (PLVAC) and the acidocalcisomes have been identified as major Ca2+ stores (Hortua Triana et al., 2018; Lourido and Moreno, 2015).
The ER of T. gondii is an extension of the nuclear envelope and it was seen continuous with the Golgi stacks and the apical secretory organelles—the micronemes and rhoptries—which are unique to the apicomplexan phylum (Hager et al., 1999). These organelles perform a plethora of functions ranging from host cell attachment, invasion, and establishment of the parasitophorous vacuole. As the cytoplasmic Ca2+ concentration increases, it initiates responses like microneme secretion (Carruthers and Sibley, 1999; Nagamune et al., 2007), conoid extrusion (Del Carmen et al., 2009), invasion (Lovett and Sibley, 2003; Vieira and Moreno, 2000) and egress (Arrizabalaga et al., 2004). These biological events require precise spatiotemporal control of diverse effectors and suggest the presence of distinct systems to deliver Ca2+ to specific locations rather than allowing global increases, which would activate unnecessary and potentially detrimental signaling events for the parasite.
In order to store Ca2+ ions, T. gondii expresses an ER-localized SERCA Ca2+-ATPase (TgSERCA), which was functionally validated by rescue experiments in yeast lacking Ca2+-ATPases and by its specific inhibition by thapsigargin (TG) (Sagara et al., 1992). TgSERCA showed a distinct distribution in extracellular parasites, where the protein was partially found in ER vesicles in the apical region near micronemes (Nagamune et al., 2007). This distribution pattern was different to the one obtained with other ER markers, which were mainly seen near the nuclear envelope, suggesting an expansion of the ER toward the apical end. This expansion could play a role in the rapid release and effective recovery of cytosolic Ca2+, events that likely govern both motility and microneme secretion (Nagamune et al., 2007). The observed distribution and expansion of the ER in extracellular parasites most likely allow the interaction of the ER membranes with other organelles in the apical region of the parasite.
While MCSs have been extensively investigated in model organisms like yeast and in mammalian cells, our understanding about the function and composition of these structures in apicomplexan parasites is only becoming apparent (Santos and Nozaki, 2021) In apicomplexans, MCSs have been identified in Sarcocystis spp. (Tomova et al., 2006), Plasmodium spp (Burda et al., 2017). and T. gondii (Tomova et al., 2009) as areas of close contact between two organelles. These pathogens possess a single mitochondrion and phylum-specific organelles such as the apicoplast, a relict plastid acquired during a secondary endosymbiotic event (Lim and McFadden, 2010; Roos et al., 1999). Consequently, it is possible that apicomplexans would possess MCSs mediating associations between canonical organelles—such as the ER/mitochondrion association—but also additional and divergent MCSs to ensure communication between their phylum-specific organelles. We will discuss the implications of these structures in cellular signaling, with an emphasis on their potential role in Ca2+ signaling.
Endoplasmic Reticulum–Mitochondrion
At the mammalian and yeast ER-mitochondria interface, different types of MCSs—with different protein compositions—are recruited to perform a variety of functions such as lipid trafficking, mitochondrial fission, mitophagy and Ca2+ transfer. In yeast, a protein complex named the ER–mitochondria encounter structure (ERMES) was shown to tether the two organelles (Kornmann et al., 2009). While ERMES plays a crucial role during mitophagy in yeast (Böckler and Westermann, 2014), most of its subunits seem to be absent from mammalian genomes (Aoyama-Ishiwatari et al., 2021; Kornmann and Walter, 2010). On the ER side of ER-mitochondria MCSs in mammalian cells, vesicle-associated membrane protein-associated proteins (VAPs) may have replaced the functions of ERMES. For instance, VPS13A localizes to ER–mitochondria contacts, where it seems to be involved in lipid transport between the two organelles (Kumar et al., 2018), and VAPB is involved in calcium homeostasis via its interaction with the mitochondrial outer protein PTPIP51 (De Vos et al., 2012).
In apicomplexans, there is no genomic evidence for the presence of the ERMES subunits, but complexome profiling in Plasmodium falciparum demonstrated the presence of an ER membrane complex (EMC) (Evers et al., 2021). Tethering and transfer of phospholipids from the ER to the mitochondria were proposed to be some of the functions of the yeast EMC (Lahiri et al., 2014). However, their potential involvement in the establishment of MCSs in apicomplexans has not been determined.
Tethers between the ER and mitochondria also involve interactions between the inositol 1,4,5-trisphosphate receptor (IP3R), the mitochondrial voltage-dependent anion-selective channel (VDAC) protein and the glucose-regulated protein GRP75 (Prinz et al., 2019; Szabadkai et al., 2006) in yeast and mammals. Establishment of these MCSs creates regions enriched with IP3Rs on the ER side. The Ca2+ concentration is higher in those domains, allowing the rapid channeling of Ca2+ ions from the ER to the mitochondria. Although apicomplexans lack an IP3R homolog, they seem to possess several GRP75 homologs (Table 1). Recently, a characterization of the T. gondii VDAC showed that the channel may mediate contacts between the ER and the mitochondrion. Depletion of the VDAC homolog in T. gondii resulted in significant morphological changes in the mitochondrion and ER, suggesting a role in mediating contacts between these two organelles (Mallo et al., 2021). However, the work did not include direct measurements of mitochondrial Ca2+, so the role played of VDAC in the transfer of Ca2+ from the ER into the mitochondria in apicomplexans requires further exploration, particularly using genetically encoded indicators in the mitochondrion for direct measurements of Ca2+ transfer.
Known and potential Toxoplasma gondii genes involved in MCSs between various organelles.
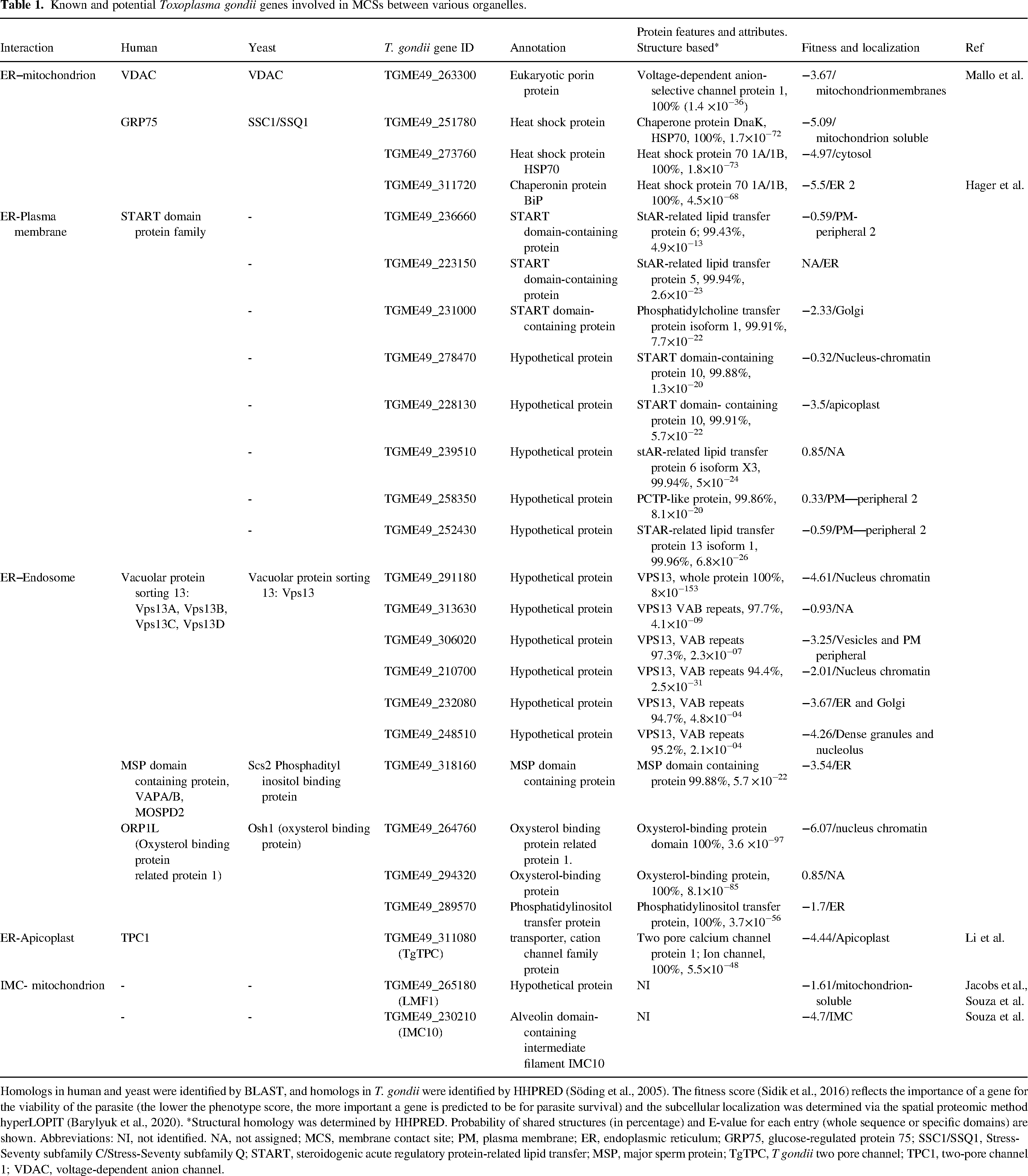
Homologs in human and yeast were identified by BLAST, and homologs in T. gondii were identified by HHPRED (Söding et al., 2005). The fitness score (Sidik et al., 2016) reflects the importance of a gene for the viability of the parasite (the lower the phenotype score, the more important a gene is predicted to be for parasite survival) and the subcellular localization was determined via the spatial proteomic method hyperLOPIT (Barylyuk et al., 2020). *Structural homology was determined by HHPRED. Probability of shared structures (in percentage) and E-value for each entry (whole sequence or specific domains) are shown. Abbreviations: NI, not identified. NA, not assigned; MCS, membrane contact site; PM, plasma membrane; ER, endoplasmic reticulum; GRP75, glucose-regulated protein 75; SSC1/SSQ1, Stress-Seventy subfamily C/Stress-Seventy subfamily Q; START, steroidogenic acute regulatory protein-related lipid transfer; MSP, major sperm protein; TgTPC, T gondii two pore channel; TPC1, two-pore channel 1; VDAC, voltage-dependent anion channel.
Another class of proteins mediating ER-mitochondria interactions are the dynamin family GTPases mitofusins 1 and 2 (Mfn1/2). These proteins localize to the ER-mitochondria interface, where Mfn2 can form homotypic or heterotypic complexes. In mammalian cells, depletion of the protein disrupts the interaction between the two organelles, leading to an aberrant ER morphology and a reduction in mitochondrial Ca2+ uptake (De Brito and Scorrano, 2008). While homologs of Mfn1 and 2 are absent in apicomplexans, their genomes contain three dynamin-related proteins (Drps) (Breinich et al., 2009). Only the T. gondii DrpC has been reported to have a mitochondrial localization (Melatti et al., 2019) but its role mediating ER-mitochondrial interactions has not been assessed.
ER–Plasma Membrane
At the ER–PM interface, MCSs carry out functions mainly in lipid and Ca2+ transfer. In yeast and mammals, primary ER–PM tethers stem from different protein families: VAPs (which are also involved in MCSs between the ER and the mitochondrion), Anoctamin/TMEM16/Ist2p homologs and extended synaptotagmins (E-Syts) (Zaman et al., 2020). In addition, ER-PM MCSs contain different protein families involved in lipid trafficking. In mammals, the oxysterol binding protein (OSBP)-related proteins (ORPs), the phosphatidylinositol-transfer protein Nir2 and the steroidogenic acute regulatory protein-related lipid transfer (START) domain-containing proteins are involved in this process, whereas yeast possesses OSBP homologue (Osh) proteins (Im et al., 2005; Kim et al., 2013; Clark, 2020).
In apicomplexans, evidence of MCSs between the ER and the PM comes from observations of PM biogenesis and dynamics in the rodent malaria parasite P berghei (Burda et al., 2017). Using serial block face scanning electron microscopy, the authors observed ER extensions in proximity with the surrounding PM during the development of mosquito and liver forms of the parasite. Those results were then confirmed with PM and ER fluorescent reporters combined with live-cell imaging and super-resolution microscopy. The presence of MCSs at the interface of these two organelles could be important for lipid transport during the extensive membrane remodeling taking place in this part of the parasite's life cycle. In T. gondii, the existence of MCSs between the PM and the ER remains to be determined. Although the molecules mediating interactions between the two organelles are still unknown in apicomplexans, ER-PM MCSs homologs seem to exist in these organisms. While ORP and Osh homologs may be present, three START domain-containing proteins can be readily detected in the genome of T. gondii. HHpred analysis of the three sequences support the presence of the START domain with high probability (Table 1). Additionally, five other T. gondii proteins share structural similarities to the START domain protein family (Table 1).
Cytosolic Ca2+ influx from the extracellular milieu has been demonstrated in T. gondii (Pace et al., 2014). Proximity of the ER with the periphery would most likely result in the ER filling with extracellular Ca2+. This proximity has not been demonstrated yet. Moreover, apicomplexans possess an inner membrane complex (IMC), a unique organelle composed of flattened vesicles located right beneath the PM (Morrissette and Sibley, 2002). It is not known if the presence of the IMC could preclude the formation of MCSs between the ER and PM, or if MCSs could form between the IMC and the ER instead.
ER and Acidic Compartments
Endosome-ER contacts have been implicated in several cellular functions including lipid transfer, calcium exchange, and receptor tyrosine kinase signaling (Eden et al., 2016). The first studies of ER-endosome contacts were done with yeast and identified the junction between the nucleus and vacuole (NvJ) formed by direct interaction between the ER protein NvJ1 and the vacuole protein Vac8 (Pan et al., 2000). No orthologs for NvJ1 or Vac8 could be found in the T. gondii database.
In mammalian cells, the establishment of contacts between the ER and endosomes consist of an ER-associated protein interacting with an endosomal protein or lipid. Most of the different tethering complexes involve VAP-A, VAP-B and motile sperm domain-containing protein 2 (MOSPD2), a major sperm protein (MSP) in the ER. VAP-A, VAP-B, and MOSPD2 are anchored to the membrane of the ER by their carboxyl transmembrane domain while the cytoplasmic MSP domain interacts with proteins through the FFAT domains (two phenylalanine in an acidic track) forming tethers and allowing the formation of MCSs between the ER and endosomes (Loewen et al., 2003; Loewen and Levine, 2005). Other tethering complexes are found with specific modes of formation and function, and the nature of contacts would vary according to the cell type (Di Mattia et al., 2020).
The T. gondii database revealed a gene (TGME49_318160) predicted to localize to the ER (Barylyuk et al., 2020) and annotated as a MSP domain-containing protein. HHpred analysis of the TgME49_318160 protein sequence supported the database predictions with 99.8% probability as MOSPD, VAP-A, SCS2 (Table 1). As with the rest of the potential MCSs homologs in T. gondii, the involvement of this protein in MCS formation requires experimental validation.
The large evolutionary conserved VPS13 (vacuolar protein sorting 13) proteins are lipid transport proteins that localize to MCSs (Park et al., 2016). These large proteins are suspected to bridge membranes and form channels that allow lipid transport between organelles (Dziurdzik and Conibear, 2021). We searched the T. gondii proteome database for Saccharomyces VPS13 protein orthologs and we found six genes (Table 1). These genes are annotated as hypothetical proteins and appear to possess the most conserved parts of Vps13 (Chorein-N, VAB repeats, ATG_C). The most identifiable domain of VPS13 is the VAB repeats as they are unique to VPS13 (Levine, 2022). The potential localization and function of these proteins to MCSs in T. gondii remains to be determined.
In eukaryotic cells, lipids are unevenly distributed between organelles, and they are transported using vesicular or non-vesicular transport pathways. Non-vesicular transport involves lipid transport proteins (LTPs) and the most studied lipid transport in ER-endosome contacts is cholesterol transport (Di Mattia et al., 2020). The ORP 1L (ORPL1) plays a role in sensing cholesterol levels at the endosome membrane. This protein is part of a family of LTPs. ORP1L possesses a pleckstrin homology (PH) domain, an FFAT domain and a carboxyl-terminal OSBP-related ligand binding domain (ORD). The domain organization of ORPL1 is such that the PH and ORD domains allow the protein to connect two distinct organelles. Moreover, the presence of an FFAT domain is important for its interaction with VPAs and MOSPD2 (Loewen et al., 2003; Loewen and Levine, 2005). We searched the T. gondii proteome database for orthologs of the mammalian oxysterol binding protein sequence and found three genes predicted as OBPs (Table 1). TgME49_264760 appears as the most likely to be involved in the ER-endosomes interaction. This is supported by HHpred and Pfam (ToxoDB) (Table 1). In addition, both the database and HHpred predict the presence of a PH domain (amino acids 283–391) and an FFAT domain (amino acids 767–781).
Several populations of MCSs were identified between the ER and different endocytic organelles (Eden et al., 2016) in mammalian cells, and Ca2+ fluxes appeared to regulate contact (Kilpatrick et al., 2017). VAPs may function in tethering either by direct interaction with the FFAT motifs of ORPL1 and STARD3, or indirectly, by interacting with another integral ER membrane protein, protrudin, which associates with Rab7 and phosphatidylinositol 3-phosphate (PI3P) at the endosome (Raiborg et al., 2015). We did not find genes with significant homology to protrudin in the T. gondii genome. In addition, we searched for homologs to the yeast Mdm1, which has been shown to localize to MCS (ER side) (Henne et al., 2015) but the important features to suggest a MCS protein were not found.
In T. gondii, the organelles that form part of the endolysosomal system are not well defined. A mixture of endolysosomal organelles and vesicles were named the endosomal-like compartments (ELCs) (Jackson et al., 2013), which likely include early and late endosomes and a series of intermediary vesicles. The ELCs were proposed as a precursor in the biogenesis of the lysosome-like organelle termed PLVAC (Stasic et al., 2022), an organelle that shares many features of the lysosome, such as storage of acidic hydrolytic enzymes and an acidic environment that facilitates degradation of proteins (Miranda et al., 2010; Parussini et al., 2010). The PLVAC stores Ca2+ and its transport was demonstrated in PLVAC-enriched fractions energized with pyrophosphate (PPi) (to activate a vacuolar H + -pyrophosphatase), by the release of protons, upon addition of Ca2+ (Miranda et al., 2010). Tachyzoites loaded with the Ca2+ indicator Fura2 showed a cytosolic Ca2+ increase in response to glycyl-L-phenylalanine-naphthylamide (GPN) (Miranda et al., 2010), a reagent known to cause osmotic swelling of lysosomes in mammalian cells and Ca2+ release, a phenomenon that was used to demonstrate the presence of Ca2+ in lysosomes (Haller et al., 1996; Morgan et al., 2020). Although contacts between the PLVAC and the ER have not been characterized, exchange of Ca2+ between both organelles is highly likely and awaits to be demonstrated. In addition, it is likely that both organelles interact for other roles as several ortholog genes found in the T. gondii genome (Table 1) possess the essential domains required for contact.
Two pore channels (TPCs) mediate release of Ca2+ from the endolysosomal system in response to nicotinic acid adenine dinucleotide phosphate (NAADP), a Ca2+-mobilizing messenger (Galione, 2015; Patel, 2015). The essential role for NAADP and TPC1 in the regulation of MCS formation between late endosome and the ER was demonstrated by disruption of the interaction by chemical inhibition of TPC1 or by downregulating the expression of the channel (Kilpatrick et al., 2017).
In T. gondii, Ca2+ exchange between the PLVAC and the ER most likely occurs, but the channels and transporters involved—likely apicomplexan-specific—remain to be discovered. Interestingly, a TPC homologue was characterized in T. gondii. While it is not expressed in the membrane of the PLVAC, it was localized to the surface of the apicoplast, a phylum-specific organelle (Li et al., 2021).
Apicoplast-ER
The apicoplast is a nonphotosynthetic plastid found in most apicomplexan parasites and it has been proposed to originate from the secondary endosymbiosis of an ancestor that engulfed a red alga, thereby explaining the presence of four membranes (Striepen, 2011). This event occurred at least 600 million years ago and the mitochondrion and apicoplast co-evolved, becoming intimately connected both physically and metabolically. The apicoplast houses pathways for fatty acid (FASII pathway) and isoprenoid (deoxy-xylulose phosphate pathway) synthesis, iron-sulfur cluster assembly, and a portion of the heme synthesis pathway (Nair and Striepen, 2011). It is therefore an essential, phylum-specific organelle with considerable potential for tackling parasitic infection. Cryoelectron tomography observations suggested the presence of MCSs between the apicoplast and the ER (Tomova et al., 2009), and more recent work demonstrated their existence and their potential function in Ca2+ exchange. Transfer of Ca2+ from the ER into the apicoplast was detected in T. gondii tachyzoites expressing a genetically encoded calcium indicator specifically localized to the apicoplast (Li et al., 2021). This was observed upon inhibition of SERCA with TG (Sagara et al., 1992) as this would allow the buildup of local Ca2+ at the cytoplasmic side of the ER most likely reaching the high Ca2+ concentration needed to activate transport into the apicoplast. This transfer activity was dependent on the presence of an active two-pore channel (TgTPC) at the membrane of the apicoplast, as null mutants of TgTPC showed a significantly reduced Ca2+ transfer activity. Deletion of the TgTPC gene not only caused limited apicoplast Ca2+ uptake but in addition, caused a reduction in the formation of MCSs between the apicoplast and the ER as monitored by electron microscopy. These analyses were the first demonstration of the functional role of MCSs between two organelles in T. gondii and identified for the first time the apicoplast as a novel Ca2+ store (Li et al., 2021).
TPCs are known for their role in Ca2+ release from acidic stores, so it is peculiar for the T. gondii TPC to be present in the apicoplast and perform essential functions for the parasite. TgTPC appears to regulate the formation of MCSs like the role reported for TPC1 at ER-endosome junctions in mammalian cells (Kilpatrick et al., 2017). It is possible that TgTPC may facilitate Ca2+ uptake through a second transporter at the contact site or alternatively, TgTPC might directly mediate Ca2+ uptake akin to the mitochondrial Ca2+ uniporter at ER-mitochondria junctions. This model would necessitate a lumen-negative membrane potential. How uptake of Ca2+ into the apicoplast multimembrane system is transmitted to its innermost matrix requires investigation. Regardless of the exact mechanism, communication between the apicoplast and the ER is essential for apicoplast function and cell growth and TgTPC plays a central role in this communication.
Mitochondrion–Apicoplast Association
The mitochondrion and the apicoplast harbor numerous biosynthetic and metabolic pathways. They depend on each other for optimal functioning and are essential during the apicomplexan life cycle. For instance, heme-biosynthesis enzymes localize to the mitochondrion, the apicoplast, and the cytosol, and de novo heme synthesis is required for T gondii growth and pathogenesis (Bergmann et al., 2020; Sheiner et al., 2013). Similarly, the apicoplast synthesizes ubiquinone precursors that are required for the mitochondrial electron transport chain (Lim and McFadden, 2010). In T. gondii, a citrate shunt between the two organelles further illustrates their metabolic crosstalk (MacRae et al., 2012). The presence of MCSs at the mitochondrion–apicoplast interface would likely facilitate nonvesicular transport of metabolites and other molecules. Even though no such contacts have been identified to date, the proximity between the mitochondrion and the apicoplast has been observed for more than 50 years. One of the earlier instances comes from the avian malaria parasite P fallax, where electron microscopy observations found the mitochondrion associated with the spherical body, the structure that years later was identified as the apicoplast (Hepler et al., 1966; McFadden et al., 1996). This association has also been documented in P falciparum, where both organelles remain associated with each other during the asexual cycle (Van Dooren et al., 2005). In T. gondii, the apicoplast-mitochondrion association is prevalent during the G1 phase and the early stages of apicoplast elongation but was not observed during the rest of the cell cycle (Nishi et al., 2008). These results suggest that the mitochondrion–apicoplast association could be more transient in T. gondii compared to P falciparum. However, mitochondrial purification methods in both organisms recovered mitochondria and apicoplasts in the same fractions, suggesting the presence of a physical association—perhaps a tether—between the two compartments (Kobayashi et al., 2007; Maclean et al., 2021). The mitochondrion–apicoplast association has been observed by different research groups using multiple techniques, hinting at the presence of MCSs linking the two organelles. However, the function and molecular identity of this association remains unknown, and whether MCSs are present at the mitochondrion-apicoplast interface awaits further research.
Mitochondrion-Inner Membrane Complex
As members of the Alveolata supergroup, apicomplexans possess an IMC, a cortical, cytoskeleton-like organelle located underneath the PM. It is composed of flattened vesicles and a network of intermediate filament-like proteins called alveolins (Anderson-White et al., 2011). In addition to fulfilling a structural role, the IMC is important for parasite motility, cell division and host cell invasion (Chen et al., 2016; Ferreira et al., 2020). Recently, several studies in T. gondii have shown that the IMC is also involved in the mitochondrial morphology of the parasite (Ovciarikova et al., 2017; Souza et al., 2022).
When T. gondii is intracellular, microscopy observations show that its mitochondrion surrounds the periphery of the cell in a lasso-shape conformation. On the other hand, the mitochondrion of extracellular parasites drastically changes its morphology: upon egress from the host cell, the organelle adopts a sperm-like or collapsed conformation (Ovciarikova et al., 2017). These observations suggest that the transition between intracellular and extracellular stages in T. gondii triggers changes in mitochondrial morphology. The authors also observed areas where the IMC and the mitochondrion are in proximity and proposed that an association between these two compartments—likely mediated via MCSs—contributes to the maintenance of the lasso-shaped mitochondrion observed in intracellular parasites. In extracellular parasites, the authors suggested that the mitochondrion retracts from the IMC.
These findings were subsequently confirmed first with the identification of lasso maintenance factor 1 (LMF1), a T. gondii outer mitochondrial protein (Jacobs et al., 2020). LMF1 depletion causes the mitochondrion of intracellular parasites to adopt a sperm-like or a collapsed conformation, reminiscent of the mitochondrial morphology of extracellular parasites. Thus, LMF1 plays a crucial role in preserving the lasso-shaped morphology observed in intracellular parasites, raising the possibility that depletion of LMF1 abolished the IMC-mitochondrion MCSs responsible of maintaining the mitochondrion at the cell periphery during the intracellular stage of T. gondii.
Recently, the same research team performed a yeast two-hybrid screen to identify LMF1 interactors (Souza et al., 2022). Among the candidates they identified IMC10, an alveolin-containing protein of the IMC, as an LMF1 interacting partner. After confirming the interaction between the two proteins at the IMC–mitochondrion interface, they demonstrated that a conditional knockdown of IMC10 disrupts mitochondrial morphology in intracellular parasites, phenocopying LMF1 depletion. Using expansion microscopy, the presence of LMF1/IMC10 complexes was also followed during cell division, hinting at a role of the LMF1/IMC10 interaction for mitochondrial distribution between the daughter cells. Taken together, these observations show that LMF1 and IMC10 define a novel MCS between the mitochondrion and the IMC in T. gondii. Intriguingly, homologs of LMF1 are absent in P falciparum, bringing into question whether or how the IMC and mitochondrion interact in this parasite.
Concluding Remarks
It has now been established that MCSs are widespread structures within eukaryotic cells. Although most of what we know about MCSs comes from a handful of well-studied eukaryotes, contacts should also be present in early-branching eukaryotic organisms, such as apicomplexans. The study of MCSs in apicomplexan parasites is only in its very beginnings, but basic biological features in T. gondii support numerous organellar interactions potentially mediated by MCSs (Figure 1). For example, Ca2+ signals precede a series of interrelated molecular processes occurring in a coordinated manner that culminate in the stimulation of each step of the parasite lytic cycle. Calcium transfer from the ER to other organelles via MCSs would explain/support the precision, speed, and efficiency that is needed for each lytic cycle step (Hortua Triana et al., 2018; Lourido and Moreno, 2015; Tomova et al., 2006). The precise spatiotemporal distribution of specific effectors suggests the presence of a strategic delivery of Ca2+ at specific locations like the apical end of the parasite, rather than having global Ca2+ increases which would be detrimental to the cell and could stimulate unwanted signaling events.
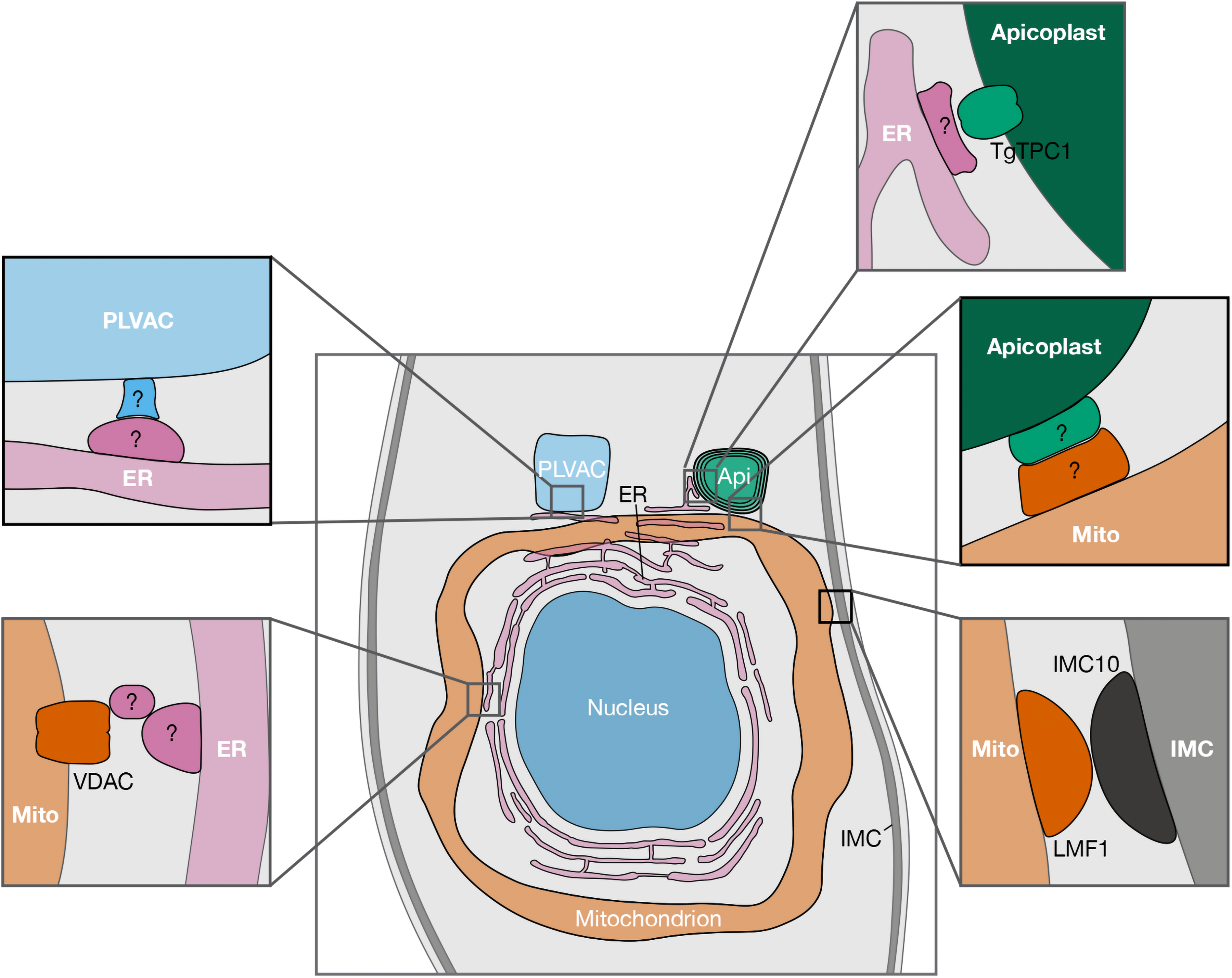
Reported and potential MCSs between organelles of Toxoplasma gondii. Schematic representation showing proteins recently reported to be involved in MCSs, along with putative MCS candidates (indicated with “?”). For clarity purposes, only the central part of the parasite is shown. Abbreviations: AP, apicoplast; ER, endoplasmic reticulum; PLVAC, plant-like vacuolar compartment; IMC, inner membrane complex; TgTPC, T. gondii two pore channel; VDAC, voltage-dependent anion channel; LMF1, lasso maintenance factor 1.; MCS, membrane contact site.
The interaction and Ca2+ transfer between the ER and the apicoplast suggest that the ER most likely transfers Ca2+ to other organelles like the mitochondrion and the PLVAC. These experiments would require expression of genetically encoded indicators in the mitochondrion and the PLVAC for direct measurements of Ca2+ transfer. As new molecular and genetic tools become available, experiments demonstrating the direct role of MCSs in Ca2+ transfer appear to be feasible. Identifying the proteins involved will not only shed light on apicomplexan biology, but their divergent nature could make them attractive targets against these pathogens.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Allergy and Infectious Diseases (grant numbers: AI128356, AI137218, AI154931, AI174600).
