Abstract
Autophagy of the cortical ER in budding yeast was unexpectedly found to require End3, a component of the endocytic machinery that promotes the assembly of actin at endocytic pits on the plasma membrane. The cortical ER transiently interacts with invaginating endocytic pits through a linkage consisting of VAP proteins, oxysterol binding proteins and type I myosins. These proteins are required for actin assembly and for autophagy of the ER. Assembly of actin at these contact sites may direct the movement of ER away from the cortex towards sites of autophagosome assembly.
Domains of the endoplasmic reticulum (ER) undergo autophagic degradation in response to starvation or ER stress. This process, called ER-phagy, uses selective autophagy receptors that connect the ER to the autophagy machinery via their ability to bind Atg8 in yeast or LC3 or GABARAP in mammals. In budding yeast, Atg40 serves as the autophagy receptor for the cortical ER (Mochida et al., 2015). The yeast cortical ER consists of interconnected ER sheets and tubules directly beneath the plasma membrane and is linked by cytoplasmic tubules to the ER that surrounds the nucleus. Atg40 packages cortical ER into autophagosomes in conjunction with Lst1-Sec23, a cytosolic complex that binds to Atg40 (Cui et al., 2019). The mechanisms by which cortical ER domains are selected for degradation, severed from the ER network, and sequestered within a phagophore (autophagic precursor) at the
A screen of the library of yeast deletion strains for mutants defective in ER-phagy, but not bulk autophagy, identified a number of strains including end3Δ (Chen et al., 2020). End3 is known to act with its stable partner Pan1 at endocytic sites on the plasma membrane to recruit the Arp2/3 complex. The End3-Pan1 complex thus links an endocytic site to a branching actin network that helps to drive invagination of the endocytic pit, leading to the formation of an endocytic vesicle (Sun et al., 2015). A recent study demonstrated that invaginating endocytic pits undergo a close but transient interaction with the edge of a cortical ER sheet before progressing to a free endocytic vesicle (Encinar Del Dedo et al., 2017). These observations offered a clue to a possible connection between End3 function in endocytosis and the seemingly unrelated process of ER-phagy. A protein linkage was established between the cortical ER and the endocytic pit consisting of Scs2/Scs22, Osh2/Osh3 and Myo3/Myo5 (Figure 1A) (Encinar Del Dedo et al., 2017). Scs2 and Scs22 are VAMP associated protein (VAP) family members. These are integral ER membrane proteins that recruit cytosolic proteins containing FFAT motifs (phenylalanines in an acidic tract) to the surface of the ER. Osh2 and Osh3 are OSBP-related proteins (ORPs) containing FFAT motifs that serve to transport ergosterol and phospholipids between cellular membranes (Murphy and Levine, 2016). Osh2 and Osh3 also interact with Myo3 and Myo5, type I myosins, which sit at the base of endocytic invaginations. Osh2 and type I myosins act to deliver ergosterol from the ER to the plasma membrane at endocytic sites, promoting localized actin assembly by Arp2/3 and driving membrane invagination (Figure 1A) (Encinar Del Dedo et al., 2017) (Encinar del Dedo et al., 2021).
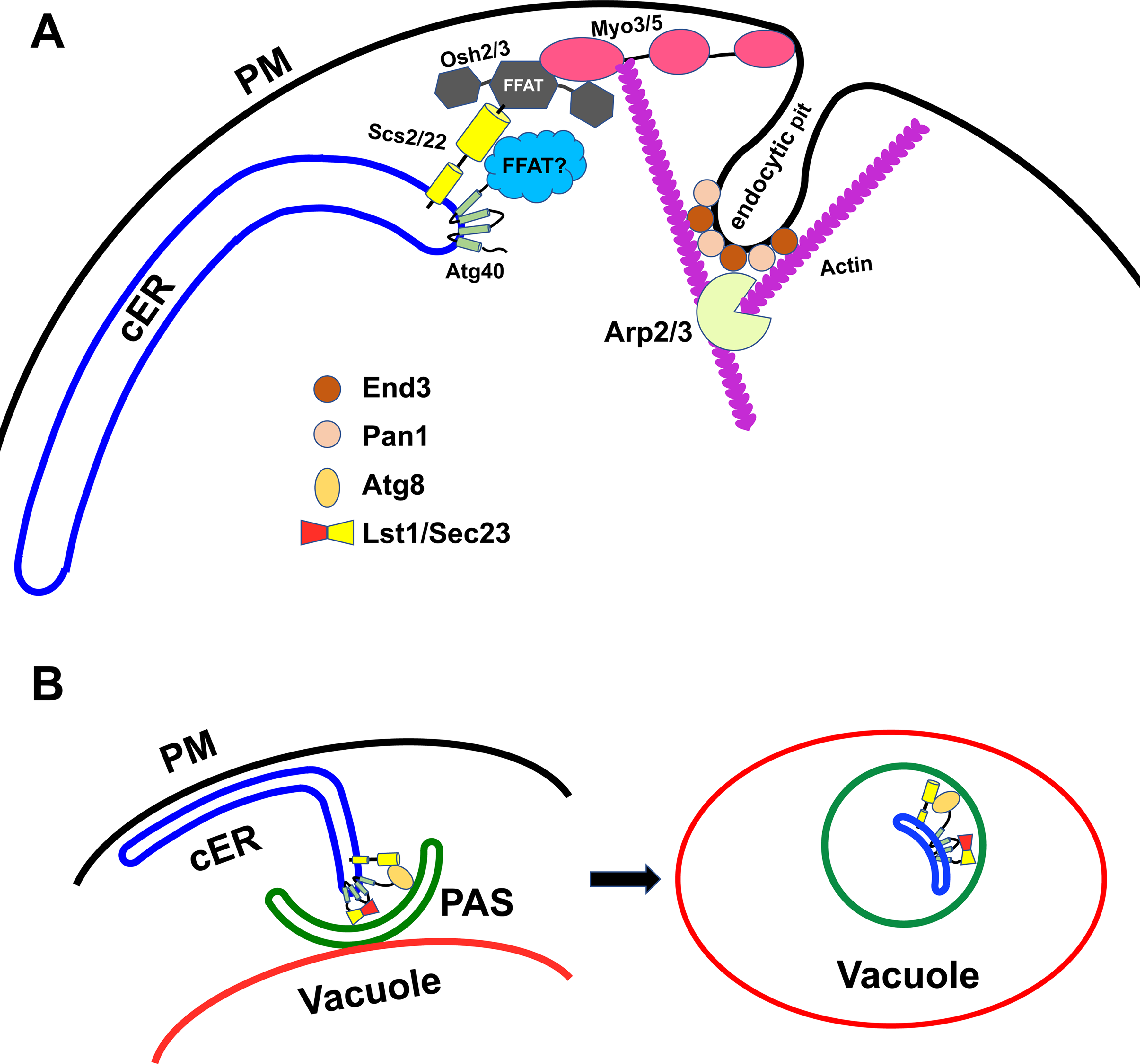
A model for the role of ER-endocytic pit contacts in ER-phagy.
In support of the relevance of these endocytic components to ER-phagy, loss of Scs2/Scs22, Osh2/Osh3 or Myo5 was found to inhibit autophagy of resident cortical ER proteins (Liu et al., 2022). Furthermore, inhibition of Arp2/3 or loss of End3/Pan1 or Myo3/Myo5 was found to inhibit the association of Atg40 with the core autophagosome assembly machinery. In principle, these results could reflect a dependence of ER-phagy on endocytosis, however loss of Rvs167, an amphiphysin homolog implicated in severing endocytic pits after actin-driven invagination, had no effect on ER-phagy, despite the block in endocytic function (Liu et al., 2022). Thus it appears that ER-phagy is dependent on actin assembly at ER-endocytic contact sites rather than on endocytosis.
Scs2 and Atg40 are both concentrated at the edges of cortical ER sheets (Figure 1A and 1B) where they transiently colocalize with assembled actin at invaginating endocytic pits (Liu et al., 2022). Scs2 coprecipitates with Atg40, but only after chemical crosslinking, suggesting an indirect or unstable interaction. Mutation of the FFAT-motif binding domain of Scs2 blocks the interaction with Atg40, yet Atg40 has no identifiable FFAT motifs, consistent with the possibility of an intermediary protein that links Atg40 to Scs2. Mutation of the FFAT-motif binding domain of Scs2 impairs ER-phagy suggesting that the Scs2-Atg40 association is of functional importance (Liu et al., 2022). Going forward it will be important to identify this linker protein and evaluate its role in ER-phagy.
It is interesting to compare the requirements for ER-phagy in budding yeast with those for stress-induced ER-phagy in fission yeast. Here, as in budding yeast, there is a requirement for Scs2, however in fission yeast the autophagy receptor is a soluble protein (Epr1) that is recruited to the surface of the ER by binding directly to Scs2 through a FFAT motif (Zhao et al., 2020). The role of Epr1 can be replaced with an artificial construct containing both a FFAT motif and an Atg8 binding site. However, the role of Scs2 is not limited to recruiting Epr1. It is also needed to form a bridge between the cortical ER and the plasma membrane in a fashion that might be analogous to its role in budding yeast. It will be instructive to explore the parallels between these two systems as more details are uncovered.
Still unresolved is exactly how actin assembly at ER-endocytic pit contact sites serves to promote ER-phagy. A simple model is that actin assembly pushes on the edge of an ER sheet, driving it away from the cell cortex towards the PAS where Atg40 engages autophagic membranes (Figure 1A-1B). While no data currently argues against this model, it is very challenging to directly visualize such an event. ER-phagy is a slow process, taking hours for a significant degree of turnover, while endocytosis is much faster, occurring within minutes. Therefore, we anticipate that actin-driven movement of an ER segment away from the cortex (Figure 1B) would be much less frequent than the invagination of endocytic pits. Other selective autophagy pathways, such as mitophagy and pexophagy, also require actin assembly, although it is not yet clear if these pathways similarly require assembly at ER-endocytic contact sites or if they use alternative components to promote the assembly of actin at other sites.
Footnotes
Acknowledgments
This study was supported by NIH grants GM35370 to P.N. and 5R35GM131681 to S. F-N.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health, (grant number 5R35GM131681, GM35370).
