Abstract
Lipid transfer proteins are crucial for intracellular cholesterol trafficking at sites of membrane contact. In the OSBP/ORPs (oxysterol binding protein and OSBP-related proteins) family of lipid transfer proteins, ORP1L, ORP1S and ORP2 play important roles in cholesterol transport. ORP1L is an endosome/lysosome-anchored cholesterol sensor which may also move cholesterol bidirectionally at the interface between the endoplasmic reticulum and the endosome/lysosome. ORP2 delivers cholesterol to the plasma membrane, driven by PI(4,5)P2 hydrolysis. ORP1S may also transport cholesterol to the plasma membrane, although it is unclear if phosphoinositides are involved. The source of cholesterol delivered to the plasma membrane by ORP1S and ORP2 remains unclear. This review summarises the roles of these proteins in maintaining cellular cholesterol homeostasis and in human disease.
Keywords
Introduction
The oxysterol binding protein (OSBP) and OSBP-related proteins (ORPs) comprise a family of 15 lipid transfer proteins. These proteins are divided into subfamilies I to VI based on amino acid homology, as illustrated previously (Raychaudhuri and Prinz, 2010). This family is well conserved between various organisms including yeast, C. elegans, Drosophila, and zebrafish (Raychaudhuri and Prinz, 2010; Du et al., 2015; Olkkonen, 2015). Deletion of all ORP homologous genes in yeast or C. elegans is lethal, but deletion of any individual genes is not, suggesting compensatory functions between family members in these organisms (Beh et al., 2001; Kobuna et al., 2010). In humans, however, ORP proteins exhibit less redundancy. For instance, the viability of ORP2 knockout HEK293 cells is very low (Wang et al., 2019b), and OSBP is essential for embryogenesis (Mesmin et al., 2017).
All OSBP/ORPs contain a C-terminal conserved OSBP-related domain (ORD), structured as a β-barrel core and an N-terminal lid, which can accommodate a single lipid molecule at one time (Im et al., 2005; Tong et al., 2013; Dong et al., 2019; Wang et al., 2019b). Ligands of the ORD include cholesterol, oxysterols, and phospholipids. Transport of these ligands appears to be the main function of the OSBP/ORPs; however, ligand binding may also enable OSBP/ORPs to serve as sensors. Transport often occurs at sites of membrane contact, where pre-existing domains and motifs of OSBP/ORPs bind membrane-anchored proteins or membrane lipids to associate with specific organelles, including the endoplasmic reticulum (ER) and plasma membrane (PM). The ORPs likely facilitate a “counter-transport” mechanism of bi-directional lipid transfer, employing a phosphoinositide (PIP) gradient to drive the transfer of common lipids such as cholesterol (de Saint-Jean et al., 2011; Moser von Filseck et al., 2015; Wong et al., 2019). This was best illustrated in the case of yeast Osh4p, which transfers PI(4)P and ergosterol between the ER and Golgi. PI(4)P synthesised at the Golgi by specific PI-4-kinases is transported by Osh4p to the ER, where it is hydrolysed by Sac1, a PI(4)P phosphatase in the ER. Hydrolysis of PI(4)P by Sac1 in the ER enables Osh4p to transfer ergosterol from ER to the Golgi against a concentration gradient. Two such examples of this mechanism have also been identified in mammalian cells: cholesterol/PI(4)P transport at ER-Golgi contacts by OSBP and phosphatidylserine/PI(4)P transport at ER-PM contacts by ORP5 and ORP8 (Mesmin et al., 2013; Chung et al., 2015; Ghai et al., 2017). Expanding this concept, recent studies demonstrate that PI(4,5)P2/PI(3,4)P2, in addition to PI(4)P, can also serve as a PIP ligand to drive the counter-transport by ORP2, ORP5 and ORP8 (Ghai et al., 2017; Wang et al., 2019b). In fact, the ORP8-ORD and ORP2-ORD can transfer PI(4,5)P2 much more efficiently than PI(4)P in vitro (Ghai et al., 2017; Wang et al., 2019b).
Research in the past decade has provided insight into the lipid trafficking functions of ORP1 and ORP2. ORP1 (spliced into ORP1L and ORP1S) and ORP2 are closely related and belong to Subfamily II (Raychaudhuri and Prinz, 2010). Similar to OSBP, their ORD can also transfer cholesterol (Dong et al., 2019; Wang et al., 2019b). These proteins also have affinity for phosphatidylinositol-bisphosphates, and appear to transport these PIPs more efficiently than the prototypical PI(4)P. This review summarises the reported roles of ORP1L, ORP1S and ORP2 in cholesterol transport and sensing, and explores the effects of their functions in cellular homeostasis and disease.
Oxysterol Binding Protein-Related Protein 1 (ORP1)
ORP1 transcribes two alternatively spliced proteins: a long isoform ORP1L and an ORD-only short isoform, ORP1S. Though both are ubiquitously expressed, ORP1L is more abundant in macrophages, brain, and lung tissues while ORP1S is abundant in skeletal muscle and the heart (Johansson et al., 2005). Different targeting motifs also contribute to different cellular phenotypes between isoforms (Figure 1A). ORP1L contains ankyrin repeats, a FFAT (two phenylalanine in acidic tract) motif and a PH (pleckstrin homology) domain. ORP1L is found on late endosome/lysosomes (LELs), arising from the interaction between the ankyrin repeats and LEL-anchored Rab7 in addition to phosphoinositide binding by the PH domain (Johansson et al., 2003; Johansson et al., 2005; Ma et al., 2018; Tong et al., 2019). ORP1L exists in the same LEL subclass as NPC1 (NPC1 intracellular cholesterol transporter 1), a key protein in cholesterol egress from LELs (van der Kant et al., 2013b). ORP1L can also exist at LEL-ER contacts through FFAT motif binding to ER-localised VAPs (Rocha et al., 2009). ORP1S, devoid of all targeting domains, exists diffusely in the cytosol and nucleus (Johansson et al., 2003; Lee et al., 2012). The ORP1-ORD binds a variety of ligands including cholesterol, oxysterols and various phosphoinositides (Suchanek et al., 2007; Yan et al., 2007; Zhao and Ridgway, 2017). The ORD also transports cholesterol and orientates on membranes by PIP binding (i.e. PI(4,5)P2, PI(3,5)P2) via basic patches (Dong et al., 2019). Thus, both ORP1L and ORP1S may mediate cholesterol transport at PI(4,5)P2- and/or PI(3,5)P2-rich membranes.

Cellular functions of ORP1. A: Structure of ORP1L and ORP1S. B: The ORP1L-Rab7-RILP tethering model. i) In high cholesterol conditions, the ORD binds cholesterol and inhibits FFAT binding. Dyneindynactin subunit p150Glued interacts with RILP, which also recruits HOPS proteins to stabilise the interaction. The dynein-dynactin motor complex associates with p150Glued and attaches to microtubules, promoting retrograde transport towards the nucleus. ii) In low cholesterol conditions, the ORP1L ORD does not bind cholesterol, enabling FFAT motif to interact with VAPs and tethering LELs to the ER. The tether inhibits formation of the dynein-dynactin motor complex. C: Roles of ORP1L in cholesterol transport between endosomes and ER. i) ORP1L transports cholesterol from the ER to the limiting membrane of LELs and into intraluminal vesicles, forming MVBs. ii) ORP1L transports LEL cholesterol, delivered by NPC1 and NPC2, to the ER. iii) ORP1L tethers LELs to the ER as GRAMD1B transports LEL cholesterol to the ER. D: ORP1S transports cholesterol to the PM, likely from the LELs, and may deliver oxysterols to nuclear-localised LXRs to upregulate apolipoprotein E expression. PH = pleckstrin homology; FFAT = two phenylalanine (FF) in an acidic tract (AT); VAP = VAMP-associated protein; ORD = OSBP-related domain; RILP = Rab-interacting lysosomal protein; LEL = late endosome/lysosome; ER = endoplasmic reticulum; HOPS = homotypic fusion and vacuole protein sorting; MVB = multivesicular endosome/body; NPC1/2 = NPC intracellular cholesterol transporter 1/2; GRAMD1B = GRAM domain containing 1B; PM = plasma membrane; LXR = liver X receptor; RXR = retinoid X receptor; LXRE = LXR response element; ApoE = apolipoprotein E.
ORP1L Facilitates LEL-ER Contact Formation
ORP1L forms LEL-ER contacts as a dimeric tripartite complex with Rab7 and its effector RILP (Johansson et al., 2007; Ma et al., 2018; Tong et al., 2019). The complex is anchored to LELs through phosphoinositide binding by the PH domain of ORP1L and through Rab7, which interacts with the ankyrin repeats of ORP1L and dimeric RILP. ORP1L tethers LELs to the ER by binding ER-localised VAP proteins with the FFAT motif (Weber-Boyvat et al., 2015). Cellular cholesterol levels regulate ER tethering of LELs in tandem with microtubule associated LEL motility (Rocha et al., 2009). In high cholesterol conditions, microtubules associate with LELs through the dynein-dynactin motor complex, which is recruited by initial binding of subunit p150Glued to RILP (Figure 1B-i) (Rocha et al., 2009). This interaction is stabilised by HOPS (homotypic fusion and vacuole protein sorting) proteins (van der Kant et al., 2013a). The dynein intermediate chain and other motor complex proteins assemble and latch onto microtubules, thereby facilitating retrograde transport of LELs (Rocha et al., 2009). In low cholesterol conditions, ORP1L does not bind cholesterol, instead, it interacts with VAPs and forms LEL-ER tethers (Figure 1B-ii). Tethering causes ORP1L to undergo a conformational change which blocks p150Glued recruitment to RILP, thereby inhibiting dynein-dynactin motor assembly and microtubule association. Thus, ORP1L tightly regulates LEL positioning in response to cellular cholesterol conditions.
The ORP1L tethering system has been implicated in lysosomal dynamics. To promote autophagy and degradation of cellular compartments, LELs fuse with autophagosomes, a process requiring ORP1L recruitment of Rab7 effector PLEKHM1 and HOPS proteins to the LEL surface (Wijdeven et al., 2016). ER tethering inhibits maturation of the autophagosome and fusion with LELs. In macrophages, ORP1L is recruited to the phagosome, tethering them to the ER for resolution, the final stage of phagocytosis (Levin-Konigsberg et al., 2019). ORP1L expression also regulates LEL motility by inhibiting kinesin-mediated anterograde transport (Vihervaara et al., 2011). ORP1L-mediated transport of PI(4)P from LELs may reduce kinesin association to LELs (Levin-Konigsberg et al., 2019). As such, ORP1L tethering may be coupled with PI(4)P/lipid transport during phagocytosis.
Models of Cholesterol Transport at the LEL-ER Interface
ORP1L is an ideal candidate as a cholesterol transporter because it can tether LELs and ER, and because its ORD can transport cholesterol. Several models of cholesterol transport have been proposed in both directions between LELs and the ER involving ORP1L.
ORP1L-mediated cholesterol transport was first reported to occur from the ER to multivesicular endosomes/bodies (MVBs) (Eden et al., 2016), which form cholesterol-rich intraluminal vesicles that are degraded in lysosomes (Piper and Katzmann, 2007). Formation of MVBs was previously linked to ORP1L expression in C. elegans (Kobuna et al., 2010). ORP1L establishes ER-MVB contacts via VAPA and Rab7 binding and facilitates cholesterol transport to MVBs for intraluminal vesicle formation in low cholesterol conditions (Figure 1C-i) (Eden et al., 2016). It is not understood whether this process is due to direct cholesterol transport by ORP1L or indirect mechanisms such as ER-MVB tethering. Recent work in macrophages suggests that ORP1L moves PI(4)P from phagosomes to the ER, which may be coupled with cholesterol transport from the ER to phagosomes (Levin-Konigsberg et al., 2019). If cholesterol transport to MVBs is direct, it may also be coupled to PI(4)P removal, suggesting a typical ORP counter-transport mechanism for ORP1L function.
Cholesterol transport from LELs to the ER has also been proposed. These models involve the movement of cholesterol from the dense LEL lumen to the limiting membrane by NPC2 and NPC1 (Winkler et al., 2019; Qian et al., 2020). Defective cholesterol egress phenotypes in ORP1L-deficient cells lead to suggestions that ORP1L transports NPC1-derived cholesterol to the ER (Figure 1C-ii) (Zhao and Ridgway, 2017). This model is somewhat at odds with the established understanding of the ORP1L tether, which suggests cholesterol binding of the ORD is blocked by the FFAT interaction with VAPs in low LEL cholesterol conditions (Rocha et al., 2009). Moreover, the cholesterol transfer activity of the ORD is moderate without the activation by PI(3,4)P2/PI(4,5)P2, whose level at steady state is low on LELs (Dong et al., 2019). It is therefore possible that cholesterol accumulation in the absence of ORP1L is due to a loss of its tethering function, not its cholesterol transfer function. Indeed, a recent study suggested that ORP1L acts as an LEL-ER tether while LEL cholesterol egress to the ER is executed by NPC1 and GRAMD1B (Figure 1C-iii) (Höglinger et al., 2019). GRAMD1B is an ER-anchored protein containing a StART-like lipid transfer domain which transports cholesterol at the ER-PM interface (Sandhu et al., 2018; Naito et al., 2019). Increased LEL-ER contacts promoted by ORP1L tethering facilitated greater cholesterol efflux from NPC1-deficient cells (Höglinger et al., 2019). Thus, the increased LEL-ER tethering by ORP1L can promote an NPC1-independent mechanism for LEL cholesterol to reach the ER. Although the molecular details remain to be elucidated, ORP1L appears capable of facilitating bidirectional transfer of cholesterol at the LEL-ER interface. The direction of the cholesterol transfer may well depend on the nutritional state and/or growth stages of the cell.
ORP1L in Disease and Infection
Dysregulation of ORP1L can affect cellular cholesterol homeostasis, leading to systemic effects. A heterozygous nonsense mutation in human ORP1L is linked to low plasma HDL dyslipidaemia (Motazacker et al., 2013). This mutation results in a heavily truncated protein that lacks all targeting motifs and the ability to bind Rab7 (Motazacker et al., 2016). Subsequently, cholesterol efflux to the major HDL protein, apolipoprotein A1 is reduced in cells of patients with this mutation, resulting in decreased levels of plasma apolipoprotein A1 and HDL. In atherosclerotic mice, ORP1L overexpression exacerbated atherosclerotic lesion growth, likely a product of downregulated ABCG1-mediated cholesterol efflux to HDL (Yan et al., 2007). Thus, ORP1L expression and activity appears to be important for regulation of cholesterol efflux.
Exploitation of ORP1L function occurs in viral and bacterial infections. In Ebola infection, the ORP1L-regulated microtubule mechanism is exploited by the viral hijacking of HOPS proteins, which can be prevented by inducing conditions where ORP1L forms LEL-ER tethers (van der Kant et al., 2013a). In flavivirus infections, such as West Nile fever, ORP1L expression may contribute to the efficiency of replication and infection through LEL motility (Courtney et al., 2012). Additionally, ORP1L localises to NPC1-positive LELs (van der Kant et al., 2013b), which are required for cellular infection by some viruses, including coronaviruses such as COVID-19 (Ballout et al., 2020). Hijacking of ORP1L can also occur during infection. Coxiella burnetti, an intracellular bacterium that causes Q fever, recruits ORP1L via the Type 4 secretion system to a parasitophorous vacuole which is required for bacterial replication and growth (Justis et al., 2017). ORP1L facilitates extended ER-vacuole contacts which rapidly expand the vacuole. Although unnecessary for bacterial growth, these contacts may enable fusion events with the vacuole, similar to autophagosome fusion with LELs in uninfected cells (Wijdeven et al., 2016). Similarly, adenoviral protein RIDα mimics Rab7 and binds the ORP1L-ORD to form ER-endosomal contacts (Shah et al., 2007). Remarkably, this interaction enables NPC1-independent, NPC2-dependent egress of cholesterol from late endosomes and increased production of lipid droplets (Cianciola et al., 2013). Normally, NPC2 accepts cholesterol from LDL in the LEL lumen and delivers it to NPC1, which transports it to the limiting membrane of LELs (Winkler et al., 2019; Qian et al., 2020). RIDα, interacting with early endosome maturation machinery such as β-COP, may bring hijacked ORP1L to maturing early endosomes for ER tethering, thereby bypassing later NPC1-mediated LEL-egress mechanisms and producing cholesterol ester-rich lipid droplets (Cianciola et al., 2017). Thus, elucidating the details of ORP1L-mediated endosome-ER tethering and lipid transfer will contribute to pharmaceutical and diagnostic approaches in treating pathogen infections and outbreaks.
Where and How Does ORP1S Operate?
Compared to ORP1L, the molecular function of ORP1S is poorly understood. It is unclear if ORP1S operates at sites of membrane contact, given the lack of targeting motifs and a relatively cytosolic distribution. However, transient association with PI(3,4)P2 and PI(4,5)P2 using basic patches on the ORD surface may facilitate membrane association (Dong et al., 2019). PI(4,5)P2 is abundant in the PM and PI(3,4)P2 can be synthesised on LELs under starvation conditions (Marat et al., 2017). Therefore, it is possible that ORP1S may transfer cholesterol between LELs and the PM.
Tentative models for cholesterol transport have been proposed for ORP1S. Initial reports suggested ORP1S transports cholesterol from the PM to ER, affecting cholesterol esterification rates and production of LDs (Jansen et al., 2011). This process may occur at ER-PM contacts, perhaps utilising other tethering proteins. However, recent studies suggest that ORP1S likely delivers cholesterol to the PM instead. PM cholesterol levels, visualised by the sensor D4H, increased when ORP1S was overexpressed (Wang et al., 2019b). This was further confirmed by another study which proposed that ORP1S transports cholesterol from LELs to the PM, thereby increasing cholesterol efflux by ABCA1 (Figure 1D) (Zhao et al., 2020). This model complements an ORP1L LEL-ER transport model (Figure 1C-ii) (Zhao and Ridgway, 2017). In ORP1-deficient cells, cholesterol accumulated in LELs, some of which could be redistributed to the PM when ORP1S was overexpressed. Surprisingly, a PIP-binding mutant of ORP1S could still transport cholesterol to the PM, suggesting that this transfer mechanism is independent of PIP binding and distinct from the counter-transport mechanism (Zhao et al., 2020). While the detailed mechanisms remain to be elucidated, evidence to date suggests that ORP1S can deliver cholesterol to the PM.
ORP1S has also been reported to regulate cholesterol metabolism via oxysterol binding. ORP1S can bind multiple oxysterol species, including the liver X receptor (LXR) activators 24(S)-hydroxycholesterol and 22(R)-hydroxycholesterol. Oxysterol binding is suggested to enhance nuclear translocation via pre-existing nuclear localisation sequences and binding of LXRs to enhancer elements, thus increasing expression of LXR target genes such as apolipoprotein E (Figure 1D) (Lee et al., 2012). However, the affinity and stoichiometry of the ORP1S-LXR interaction requires further investigation.
Oxysterol Binding Protein-Related Protein 2 (ORP2)
ORP2 is the only short ORP produced from a full-length transcript, consisting only of an FFAT motif and an ORD (Figure 2A). It is highly expressed in the brain and the lung (Laitinen et al., 2002). In cells, ORP2 is mainly cytosolic with some aggregation on LD surfaces and at the PM (Hynynen et al., 2009). The ORP2-ORD can bind cholesterol, most phosphoinositides and various oxysterols, including 22(R)-hydroxycholesterol with high affinity (Suchanek et al., 2007; Hynynen et al., 2009; Wang et al., 2019b). The FFAT motif associates with ER-localised VAPs (Weber-Boyvat et al., 2015). ORP2, like ORP1S, has nuclear localisation sequences and may also function in transcriptional regulation (Lee et al., 2012).
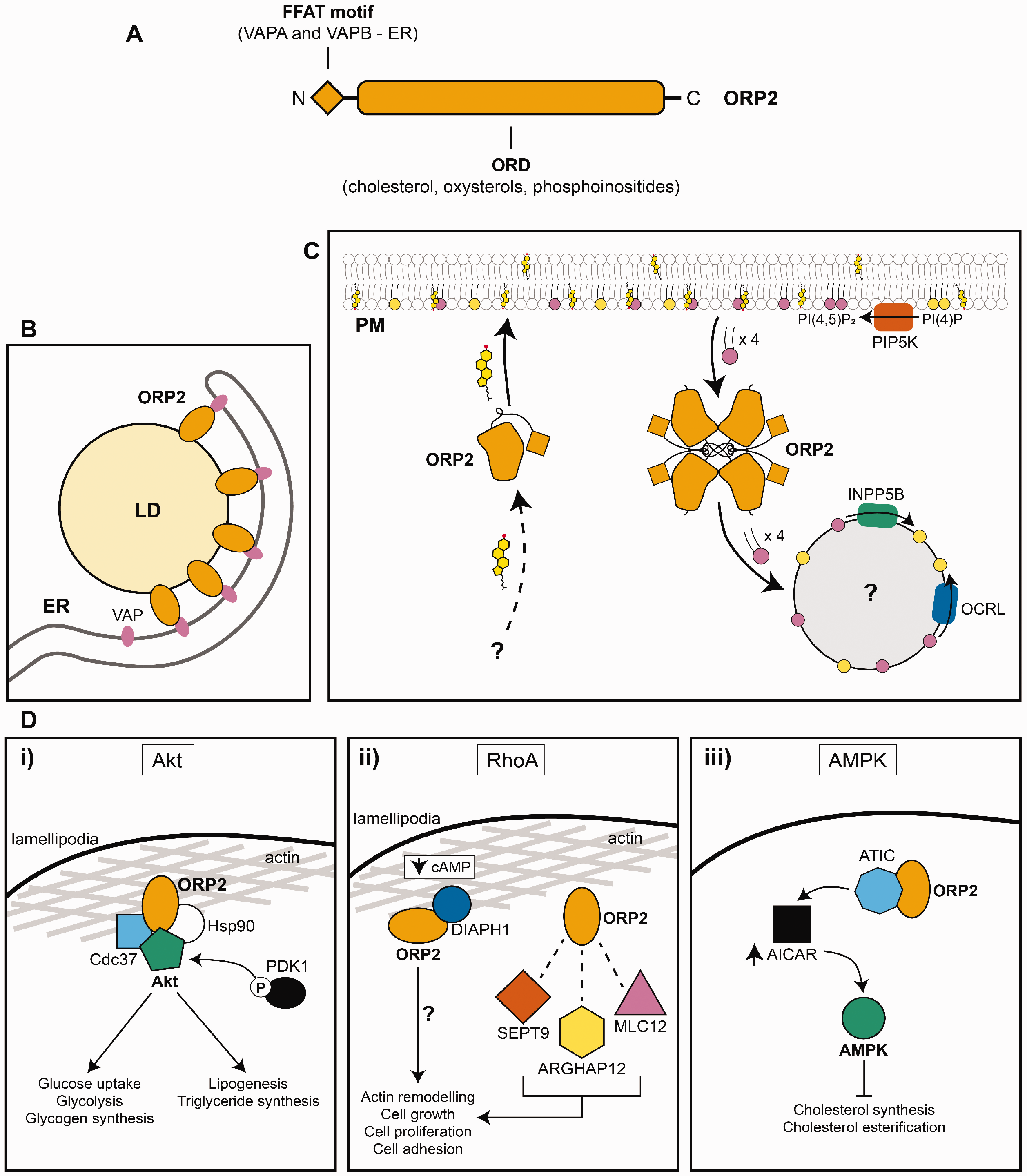
Cellular functions of ORP2. A: Structure of ORP2. B: ORP2 exists at ER-LD contacts by binding VAPs. C: ORP2 transports cholesterol to the plasma membrane as a monomer and removes PM PI(4,5)P2 as a tetramer. PM PI(4,5)P2 originates from phosphorylation of PI(4)P by PIP5K. PI(4,5)P2 is dephosphorylated to PI(4)P by 5-phosphatases INPP5B and OCRL. D. Roles of ORP2 in signalling pathways. i) ORP2 forms a complex with Akt, Cdc37 and Hsp90 to facilitate Akt phosphorylation by PDK1 at lamellipodia. ii) ORP2 interacts with DIAPH1 when cyclic AMP levels are low, which may promote actin remodelling. ORP2 interacts with RhoA downstream proteins SEPT9, ARGHAP12 and MLC12. iii) ORP2 can interact with AMPK effector ATIC, which increases the levels of AMP analogue AICAR that activates AMPK. FFAT = two phenylalanine (FF) in an acidic tract (AT); VAP = VAMP-associated protein; ORD = OSBP-related domain; LD = lipid droplet; ER = endoplasmic reticulum; PM = plasma membrane; PIP5K = phosphatidylinositol-4-phosphate 5-kinase; INPP5B = type II inositol 1,4,5-trisphosphate 5-phosphatase; OCRL = Lowe oculocerebrorenal syndrome protein; Hsp90 = heat shock protein 90; PDK1 = phosphoinositide-dependent kinase-1; cAMP = cyclic adenosine monophosphate; DIAPH1 = diaphanous homologue 1; SEPT9 = septin-9; MLC12 = myosin regulatory light chain 12; ARGHAP12 = Rho GTPase activating protein 12; ATIC = 5-aminoimidazole-4carboxamide ribonucleotide formyltransferase/inosine monophosphate cyclohydrolase; AICAR = aminoimidazole carboxamide ribonucleotide; AMPK = adenosine monophosphate-activated protein kinase.
ORP2 Associates With Lipid Droplets
ORP2 can be found on lipid droplets in various cell types (Hynynen et al., 2009; Koponen et al., 2019; Du et al., 2020). Lipid droplets are organelles bound by a phospholipid monolayer that bud from the ER and are filled with cholesterol esters and triglycerides (Gao et al., 2019). In recent years, their role has expanded from inert lipid storage deposits to organelles involved in stress response, signalling and cellular homeostasis (Welte and Gould, 2017; Henne, 2020). Largely cytosolic, ORP2 can also exist at ER-LD contacts through FFAT motif interactions with VAPA/B (Figure 2B) (Weber-Boyvat et al., 2015). How ORP2 targets the LD surface is unknown. Mutating key residues in the ligand binding tunnel that mediate both PIP binding and sterol binding of the ORP2-ORD increased LD targeting (Weber-Boyvat et al., 2015; Koponen et al., 2019). However, transient associations with negatively charged lipids may occur via basic patches on the surface of the ORP2-ORD (Wang et al., 2019b; Du et al., 2020). LD localisation can be abolished by 22(R)-hydroxycholesterol, which reduces the association between ORP2 and VAPA and increases PM association (Hynynen et al., 2009; Kentala et al., 2015; Weber-Boyvat et al., 2015; Wang et al., 2019b). Current work suggests ORP2 controls triglyceride hydrolysis and synthesis, likely at specific ER domains where it forms contacts with LDs (Weber-Boyvat et al., 2015). ORP2 may also modulate cholesterol esterification as knocking-down ORP2 increases the levels of esterified cholesterol (Hynynen et al., 2009).
ORP2 Transports Cholesterol
Originally, ORP2 was suggested to transport cholesterol from the PM to the ER (Jansen et al., 2011). This conclusion was at odds with previous findings, which indicated ORP2 overexpression enhanced cholesterol efflux (Hynynen et al., 2005). More recently, data from in vitro and in vivo experiments established ORP2 as a bona fide cholesterol transporter: ORP2 delivers cholesterol to the PM (Wang et al., 2019b) (Figure 2C). This delivery is coupled with removal of PM PI(4,5)P2 and driven by PI(4,5)P2 hydrolysis by PIP2 5-phosphatases OCRL and INPP5B. Biochemical and structural analyses indicated that cholesterol transport to the PM occurs in a monomeric form, while PI(4,5)P2 is transferred by homotetrameric ORP2. Through this transport cycle, ORP2 may influence cholesterol efflux and PI(4,5)P2-dependent cell signalling. Cholesterol delivery to the PM by ORP2 is likely coupled with increased cholesterol efflux by ABCA1 (Escajadillo et al., 2016). Removal of PI(4,5)P2 by ORP2 may regulate phospholipase C and PI3K signalling pathways.
ORP2 Regulates Cellular Metabolism
ORP2 regulates cellular metabolism through interactions with proteins from several signalling pathways (Figure 2D). ORP2 deficiency causes dysregulation in Akt signalling, subsequently reducing synthesis of triglyceride and glycogen (Kentala et al., 2018b). Complex formation by ORP2 with Hsp90, Cdc37 and Akt at lamellipodia induces Akt phosphorylation by PDK1, resulting in downstream upregulation of glucose metabolism and SREBP1-mediated lipogenesis. ORP2 overexpression also causes formation of cellular protrusions, a result of actin remodelling and lamellipodia formation (Kentala et al., 2018a). ORP2 can interact with downstream RhoA signalling proteins SEPT9, MLC12 and ARGHAP12, leading to remodelling of actin, increased cell growth, proliferation, and adhesion. In adrenocortical cells when cyclic AMP levels are low, ORP2 can also interact with RhoA effector DIAPH1, which associates with cytoskeletal proteins (Li et al., 2013). This interaction may be a mechanism for ORP2-mediated actin cytoskeletal modification. ORP2 deficiency also increases cholesterol ester formation and total cholesterol levels. The lost interaction between ORP2 and AMPK effector ATIC is speculated to be the cause of this phenotype. As a result, AMPK activator analogue AICAR and activated AMPK levels are reduced, thus upregulating cholesterol synthesis and esterification genes (Zhang et al., 2019; Wang et al., 2019a). Additionally, it may be affected by decreased expression of transcription factor steroidogenic factor 1, which increases cholesterol and 25-hydroxycholesterol levels (Escajadillo et al., 2016). 25-hydroxycholesterol also regulates ORP2 expression, most likely through upregulation of the p53 signalling pathway (Wang et al., 2019c). Finally, it should be noted that most, if not all the observations above may be mediated by PI(4,5)P2, a well-established molecule that regulates actin dynamics and cell signalling (Balla, 2013). ORP2 has profound effects on the level and distribution of PI(4,5)P2 (Wang et al., 2019b).
ORP2 Mutations Can Cause Cholesterol Dysregulation in Late-Onset Hearing Loss
ORP2 is highly expressed in the ear, especially within the cochlea in the spiral ganglion and inner and outer hair cells (Thoenes et al., 2015; Xing et al., 2015). Several mutations in the ORP2 gene OSBPL2 have been linked to autosomal dominant late-onset hearing loss in humans (Thoenes et al., 2015; Xing et al., 2015; Wu et al., 2019). These mutations are functionally deleterious because ORP2 is expressed with a truncated or absent ORD. Recently, an ORP2 knockout (KO) pig model provided insight into the mechanism of hearing loss (Yao et al., 2019). Inner and outer hair cells of ORP2 KO pigs had degenerated or absent stereocilia morphology and increased levels of apoptosis. This may be an effect of dysregulated extracellular matrix receptor interaction and focal adhesion, leading to disrupted ciliogenesis in inner and outer hair cells (Shi et al., 2020). Apoptosis may also result from dysregulated signalling pathways, increased cellular sterol levels, and increased mitochondrial damage (Wang et al., 2019a; Wang et al., 2019c). Systemic cholesterol metabolism was also assessed in the pig model (Yao et al., 2019). ORP2 KO pigs exhibited moderate hypercholesterolaemia even on a basic chow diet. High-fat diet exacerbated this condition, and mildly increased hearing loss by increasing cellular apoptosis levels, suggesting that cholesterol regulation by ORP2 contributes to the maintenance of normal hearing and impacts whole body metabolism. However, blood lipid levels of human patients appeared normal in one mutation study (Wu et al., 2019). This may be attributed to truncated ORP2 in patients, different from the complete deficiency of ORP2 in the pig model. Testing of a greater sample size is required to determine a relationship between late-onset hearing loss and high-fat diet. Sensorineural hearing loss has been linked to cholesterol dysregulation including in hypercholesterolaemia and trafficking disorders such as Niemann-Pick type C disease (Malgrange et al., 2015). There is also some evidence that diet can affect severity of hearing loss (Rosen et al., 1970; Yamasoba et al., 2013; Yang et al., 2015; Huang et al., 2020). Reducing cholesterol levels through diet and drug interventions may therefore be beneficial for reducing severity of ORP2-mediated hearing loss in patients.
ORP1 and ORP2 Crosstalk
Recently, the interaction between ORP1 and ORP2 was reported (Kentala et al., 2018a; Koponen et al., 2019). Various ORPs can homo- and heterodimerise, thus enabling localisation and function to otherwise unpredicted compartments (Ridgway et al., 1992; Wyles et al., 2007; Zhou et al., 2010). ORP5 and ORP8, two closely related ORPs of Subfamily IV, are also known to interact (Chung et al., 2015; Ghai et al., 2017). As such, ORP2 may localise to the LELs by interacting with ORP1L or ORP1S, both of which could help orientate the ORP2-ORD onto the membrane to extract cholesterol, thereby enabling LEL cholesterol delivery to the PM by ORP2 (Figure 3). Cholesterol efflux mechanisms may also be regulated through oxysterol delivery to the nucleus by ORP1S and ORP2 (Lee et al., 2012; Escajadillo et al., 2016). Therefore, ORP1 and ORP2 may work in tandem to regulate cholesterol distribution between LELs and the PM. These new hypotheses will guide future investigation into the exact roles of ORP1 and ORP2 in cholesterol metabolism.

Hypothetical models for functional ORP1 and ORP2 interaction. Cholesterol is delivered to the LEL limiting membrane by NPC1 and NPC2. A: ORP2 may transport LEL cholesterol to the PM by interacting with ORP1L, which may facilitate cholesterol movement to ORP2. B: ORP1S and ORP2 may interact to facilitate cholesterol transport from LELs to the PM. LEL = late endosome/lysosome; PM = plasma membrane; NPC1/2 = NPC intracellular cholesterol transporter 1/2.
Conclusions and Future Directions
Recent studies have provided critical insights into the functions of the ORP1/ORP2 subfamily of OSBP/ORPs. The structures of ORP1-ORD and ORP2-ORD are the first characterised mammalian ORPs at atomic level. Both ORPs can transfer cholesterol and appear able to transfer PIPs. While ORP1S and ORP2 can deliver cholesterol to the PM, ORP1L is an important LEL-ER tether in both normal and infectious contexts with limited cholesterol transfer activity. Although mechanistic progress has been made towards understanding the function of these proteins, many questions remain. For instance, how does ORP1L facilitate cholesterol egress in NPC1 deficiency? Where do ORP1S and ORP2 transport cholesterol from? What is the destination of ORP2-transported PI(4,5)P2 since ORP2 has yet to be convincingly detected in internal organelles besides LDs? How does ORP2 regulate triglyceride hydrolysis from lipid droplets? How does ORP2 interact with ORP1L and ORP1S? Finally, what are the physiological functions of ORP1L, ORP1S and ORP2 in vivo? Further work is required to answer these important questions.
List of Abbreviations
OSBP: oxysterol binding protein; ORP: oxysterol binding protein-related protein; ORD: OSBP-related domain; PH: pleckstrin homology; FFAT: two phenylalanine (FF) in an acidic tract; PM: plasma membrane; LEL: late endosome/lysosome; MVB: multivesicular endosome/body; LD: lipid droplet; PIP: phosphoinositide; PIP2: phosphatidylinositol-biphosphate; PI(4)P: phosphatidylinositol-(4)-phosphate; PI(4,5)P2: phosphatidylinositol-(4,5)-bisphosphate; PI(3,4)P2: phosphatidylinositol-(3,4)-bisphosphate; RILP: Rab-interacting lysosomal protein; VAP(A/B): VAMP (vesicle-associated membrane protein)-associated protein (A/B); HOPS: homotypic fusion and vacuole protein sorting; PLEKHM1: pleckstrin homology domain-containing family M member 1; NPC1: NPC intracellular cholesterol transporter 1; NPC2: NPC intracellular cholesterol transporter 2; GRAMD1B: GRAM domain containing 1B; StART: steroidogenic acute regulatory protein (StAR)-related lipid transfer; ABCG1: ATP-binding cassette subfamily G member 1; HDL: high-density lipoprotein; COVID-19: coronavirus disease 2019; β-COP: coatomer subunit β; LXR: liver X receptor; RXR: retinoid X receptor; LXRE: liver X receptor response element; ApoE: apolipoprotein E; ABCA1: ATP-binding cassette subfamily A member 1; PIP5K: phosphatidylinositol-4-phosphate 5-kinase; OCRL: Lowe oculocerebrorenal syndrome protein; INPP5B: type II inositol 1,4,5-trisphosphate 5-phosphatase; PI3K: phosphoinositide 3-kinase; Hsp90: heat shock protein 90; Cdc37: cell division cycle 37; PDK1: phosphoinositide-dependent kinase-1; SREBP1: sterol regulatory element-binding protein 1; RhoA: Ras homologue family member 1; SEPT9: septin-9; MLC12: myosin regulatory light chain 12; ARGHAP12: Rho GTPase activating protein 12; DIAPH1: diaphanous homologue 1; cAMP: cyclic adenosine monophosphate; AMPK: adenosine monophosphate-activated protein kinase; ATIC: 5-aminoimidazole-4carboxamide ribonucleotide formyltransferase/inosine monophosphate cyclohydrolase; AICAR: aminoimidazole carboxamide ribonucleotide
Footnotes
Acknowledgments
The authors thank members of the Yang and Brown labs for helpful feedback and suggestions and apologise to those whose work we could not discuss or cite due to space limitations.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants 1141939 and 1144726 from the National Health and Medical Research Council (NHMRC) of Australia, and by a grant from the Ara Parseghian Medical Research Fund.
