Abstract
Dendritic cells (DC) are postulated to play a role in autoimmune diseases such as Systemic Lupus Erythematosus (SLE). We reported a 13-year-old female SLE patient who presents with chronic arthritis accompanied by persistent fever, dyspnea, sleep disturbance, headache, stomatitis, rash, and muscle weakness. The supporting examinations showed abnormal blood cell counts, positive antinuclear antibody profile, serositis, and neuropathy. Immunosuppressants failed to improve the condition. DC-based vaccine derived from autologous peripheral blood which was introduced with SARS-CoV-2 protein was given to this patient. There was a significant improvement in clinical and laboratory findings. Thus, DC immunotherapy appears to be a potential novel therapy for SLE that needs to be studied.
Introduction
Systemic Lupus Erythematosus (SLE) is an autoimmune disease with diverse clinical manifestations. Therefore, the diagnosis of this disease is difficult to establish. 1 Currently, the primary treatment of SLE is by administration of immunosuppressant agents, but the administration of these drugs often causes side effects that cannot be tolerated. 2 SLE occurs due to self-antibodies caused by innate and adaptive immune system dysfunction. 3 Dendritic cells (DC) play an essential role in the innate and adaptive immune system. Therefore, DC dysfunction becomes a factor in the development of the disease. 4 Furthermore, DC plays a role in SLE and other autoimmune diseases. 5 Therefore, DC therapy, including in SLE, needs to be researched.
In this article, we discuss the case of a girl who has diverse clinical manifestations and is diagnosed with SLE. The patient’s condition did not improve after being given various immunosuppressant therapies. However, after the administration of DC immunotherapy, there was a significant improvement in the clinical conditions.
Case presentation
A girl aged 13 years came with complaints of pain in all body joints, especially both pelvis and left shoulder, for 3.5 months. The pain worsened with movement. Initially, the pain was recurrent, but it got heavier and did not improve with nonsteroid anti-inflammatory drug administration. There was no history of trauma.
For 2 months, patients complained of persistent fever, with the highest reported temperature being 39°C, accompanied by shortness of breath, sleep disturbances, headaches, and sores on the lips. Routine blood tests at the time showed abnormalities in blood cell counts (Hemoglobin 10.9 g/dL, Platelets 131,000/µL, Leukocytes 2630/µL), decreased hematocrit (HCT 32.9%), relative increase of lymphocytes (Lymphocytes 40.7%), increased coagulation time (87 mm/h), and increased C-reactive protein (CRP) titers (227.50 mg/L). On X-ray thorax and CT-scan, there was right pleural effusion. The patient was suspected of having dengue fever, but the NS1 (Nonstructural protein-1) antigen rapid test result was negative. After 4 days, platelet counts were improved (233,000/µL), but the patient still had anemia, leukopenia, and increased CRP. Because there was no improvement, the patient was referred to a neurologist and psychiatrist. The head CT-scan showed no abnormalities.
Two weeks prior, the patient had persistent fever and led to hospitalization in Pediatric Intensive Care Unit. The complaints were accompanied by joint pain throughout the body, weakness, sometimes coughing and swallowing pain, and rashes on both forearms and lower limbs. The patient appears limp with hypotension (70/40 mmHg). Physical examination revealed that the patient appeared anemic. Bilateral subconjunctival bleeding, right neck lymphadenopathy, and petechiae on both forearms and lower legs were found [Figure 1(a) and (b)]. Motor functions were decreased in the lower limbs (right motor function scale: four out of five and left: three out of five), while the upper limbs were within normal limits. Laboratory examination showed pancytopenia (Hemoglobin 6.5 g/dL; Leukocytes 1420/µL, Platelets 54,000/µL), coagulation system disruption (elongated prothrombin time, fibrinogen and D-Dimer increased), increased SGPT (64 µ/L), increased Lactate Dehydrogenase (361 µ/L), hypoalbuminemia (3 g/dL), mild hyponatremia (132 mEq/L), increased procalcitonin (5.92 ng/mL), and increased CRP (316 mg/L). In blood cultures, Staphylococcus aureus were found.
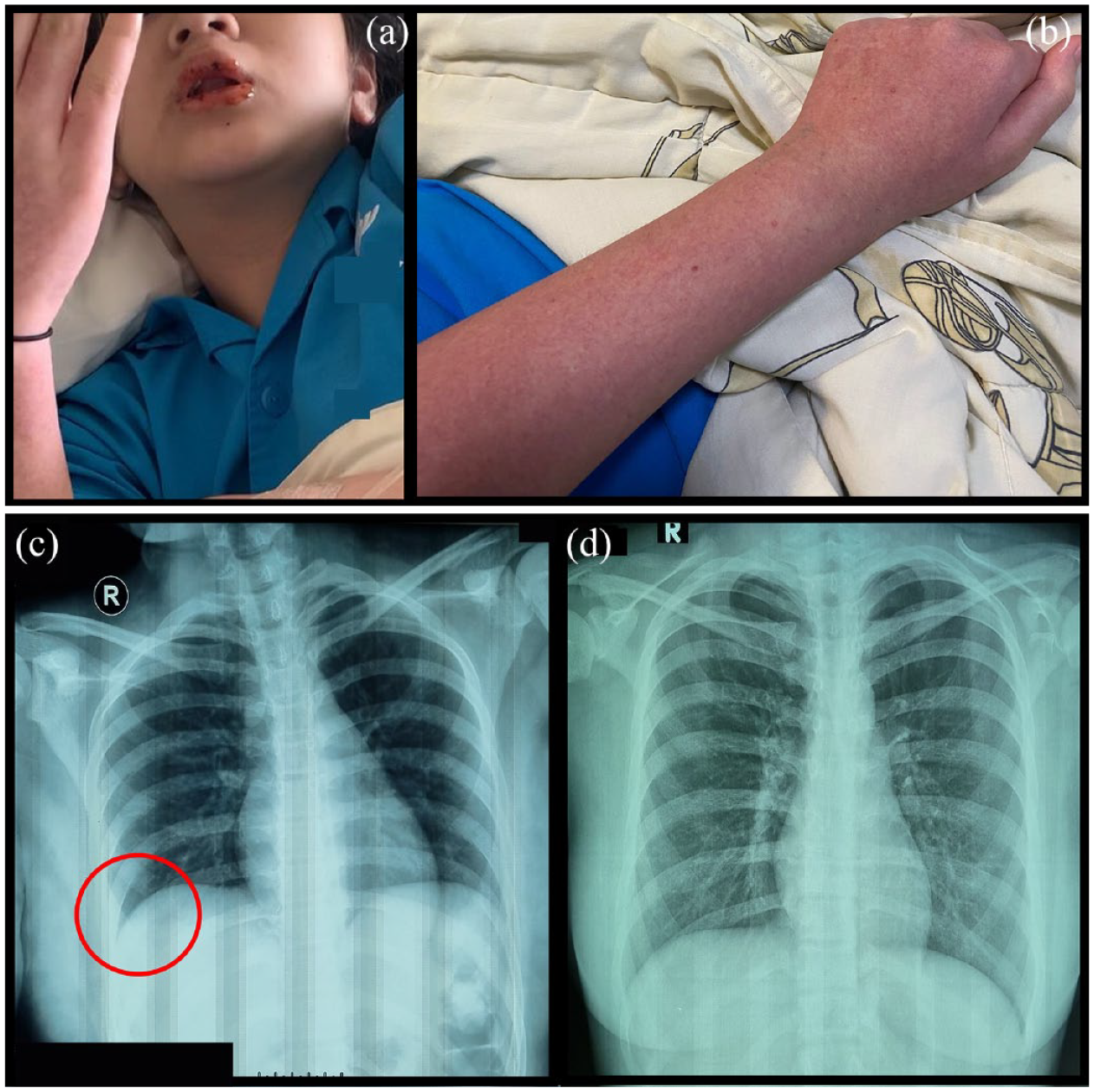
(a) Stomatitis at the upper and lower lips. (b) Rash and petechiae in the upper extremity. (c) Thorax X-ray radiography before vaccination shows thickening of the right pleura (circle) indicating a pleural effusion. (d) Postvaccination thorax X-ray shows a normal condition.
Peripheral blood smears showed erythrocytes that were microcytic, hypochromic, anisopoikilocytosis and fragmented, with the presence of pencil cells and teardrop cells. Leukocytes appeared to have toxic granulation with atypical lymphocytes. In addition, giant thrombocytes were found.
There was a bilateral paracardial infiltration on the X-ray examination of the thorax [Figure 1(c) and (d)]. Electromyography (EMG) examination suggested chronic denervation lesions with a picture of giant potential, in accordance with spinal muscular atrophy. Furthermore, examination of the nerve conduction velocity showed the presence of motor axonal neuropathy in the left ulnar and posterior tibial nerve. Antinuclear antibody (ANA) serological examination was conducted with a positive result of titer 1/100 with a rough speckled picture, positive anti-U1-Ribonucleoprotein, and positive anti-Smith. Anti-double strain Deoxyribose Nucleic Acid (dsDNA) and complement were within the normal limits.
The patient was given conservative treatment as well as intravenous antibiotics. During treatment, the patient experienced epistaxis that led to a transfusion-packed red cell and thrombocyte concentrate of 10 cc/kg. Methylprednisolone was also given intravenously at 50 mg/12 h in two doses, followed by an Intravenous Immunoglobulin (IVIG) drip of 94,000 mg. After administering such therapy, the clinical condition was stable, but complaints of fever loss, joint pain, and weakness of the limbs remained. The patient went home against medical advice and did not take any drugs.
The patient came to our hospital 3 days post-hospitalization. We diagnosed SLE with a score of 24 based on the 2019 EULAR/ACR criteria. The findings included a positive ANA test, persistent fever, serositis (pleural effusion), leukopenia, thrombocytopenia, hemolysis anemia, joint pain, and neurological disorders. Meanwhile, based on the Systemic Lupus International Collaborating Clinics (SLICC) criteria, we found one immunological criterion and six clinical criteria that met the SLE diagnosis. The total of SELENA-SLEDAI score was 29.
Dendritic cells are obtained from incubation of autologous peripheral blood monocytes with Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF) and Interleukin-4 (IL-4). To obtain a maturing dendritic cell, incubation was carried out with the SARS-CoV-2 S-protein. DC was made under standard procedures, and as much as 0.5 cc (estimated 0.5–8 million autologous dendritic cells per dose) was subcutaneously injected into the left arm. One day after the vaccination, pain gradually improved. There was an improvement in motor function, evidenced by the patient’s ability to walk with aids. One week after the injection, the patient could move independently and walk without any aids. Clinical improvement was also supported by laboratory findings, where routine blood examination and peripheral blood smear test were within the normal limits.
Supporting examination finding before and after vaccination.

The bold values are abnormal findings (out of reference value).
At 1-month post-injection evaluation, the patient had no complaints. Motor examination of upper and lower limbs was within the normal limits (left and right motor function: five out of five). Laboratory examination and X-ray of the thorax were within the normal limits. There was a significant change in the SELENA-SLEDAI score with a total of 0. The patient had a mild relapse (swelling and pain of the joints) after 3-month post-injection and was treated with Ibuprofen 200 mg two times daily.
Discussion
In this case, an SLE is obtained in a 13-year-old child. The manifestations of SLE are very diverse, so it is a complicated diagnosis. 6 Nevertheless, clinical manifestations in the patient support the diagnosis of SLE, that is, ANA positive test accompanied by persistent fever, serositis (pleural effusion), stomatitis, leukopenia, thrombocytopenia, hemolysis anemia, arthritis, and neurological disorders.
The challenge in developing dendritic cell transfer for therapy of SLE is that to make sure that these cells will not revert to the immunogenic state under inflammatory conditions, thus exacerbating the symptoms. 7 On the contrary, we found that significant clinical improvement occurred immediately after the autologous DC was being administered. Physical, laboratory, and radiology examinations were obtained normally after 1-month postvaccination. Patients did not receive any therapy for SLE 10 days before and still did not get any therapy after vaccination. This proves the safety of autologous DC transfer in children with SLE. In addition, significant improvement of symptoms shows that there is a therapeutic potential in this approach.
Dendritic cells (DC) are the most potential Antigen Presenting Cells in the immune system that also act as immune tolerance. 8 The outcome of DC interactions with T cells is determined by maturation, subset type, microenvironment, and location. Although the conditions that cause DC to have tolerogenic functions are well known, the process of tolerance induction by this DC is still unclear.8,9,10 In general, DC can control the tolerance through the formation of regulatory T (Treg) cells by the surface molecules (Cluster of Differentiation (CD) 70, CD80/86, Inducible costimulator-ligand (ICOS-L), Programmed Death Ligand-1 (PD-L1), and Programmed Death Ligand-2 (PDL1)) and secretion of tolerance-inducing factors (TGFβ, IL-10, and IL-27). 8 One method that is proposed to induce tolerogenic DC is by using an immature DC (not yet exposed to antigens) that will interact with T cells suboptimally causing anergy. 11 However, there is evidence that mature DC is more efficient at inducing tolerance through the differentiation of Treg cells. 10 This process is also possible to control autoimmune states [Figure 2(a)].

(a) Postulated dendritic cell role in controlling autoimmunity in SLE. Tol-DC presents antigen to naïve DC which causes differentiation of naïve T cell to Treg cell. Otherwise, immature DC differentiates naïve T cells which leads to T cell anergy. These conditions control the autoimmunity and activation of anti-inflammation states in SLE. (b) The immunopathogenesis of SLE. Apoptotic or necrotic cells release their self-DNA/RNA which is captured by DC. This process induced increasing antigen presentation to B and T cells and leads to their differentiation into autoreactive cells. B cell will secrete autoantibodies otherwise Th-17 will be produced with a decrease in T regulatory count. This condition leads to autoimmunity and hyper inflammation state in SLE.
Inducing tolerogenic DC can be done in a variety of ways. Recent developments suggest that the addition of part of the pathogen may also elicit a tolerance response. DC isolated from mice spleen showed suppressive phenotypes after being introduced with lipopolysaccharide (LPS) or lipoteichoic acid (LTA). 12 Other studies have shown that semi-mature DC of SLE patients incubated with a combination of vitamin D3 and dexamethasone, then matured with LPS, elicits a better tolerogenic response compared to immature DC. 13 Recently, there is also strong evidence showing that mature DC can induce tolerogenic response by inducing Foxp3+ T reg. 7 In vitro DC culture with probiotic bacteria (Lactobacillus salivarius, Bifidobacterium bifidum, Bacillus coagulans, and Bacillus subtilis) can also give rise to tolerogenic DC. 14 Tolerogenic DC can also be obtained through differentiation of monocyte-derived dendritic cells with cytokines IL-10 and TGF-β, inhibiting in vitro differentiation of DC with immunosuppressive agents (glucocorticoids, vitamin D3, mofetil mycophenolate, and rapamycin), and manipulation of immunosuppressive genetic elements in DC [cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4), indoleamine 2,3-dioxygenase 1 (IDO), Fas Ligand (FasL), and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)]. 11
In this case, DC was introduced to the SARS-CoV-2 S-protein. This approach was initially developed as a vaccine to prevent COVID-19. The previous study has shown that ex vivo DC incubation with peptides from the S-protein SARS-CoV-2 can elicit an immunogenic response in the body through the response of T cells and B cells.15,16 However, this case suggests the possibility that the DC vaccine can also control autoimmune through the tolerogenic response of the DC. Several things that need to be considered in future research are the possibility of special conditions or factors that can cause a tolerogenic response from DC introduced to the SARS-CoV-2 S-protein, such as autoimmune conditions triggered after SARS-CoV-2 infection, genetic variations of DC obtained from people with autoimmune diseases, or the use of immunosuppressive agents such as vitamin D3 and glucocorticoid.
Immunopathologically, SLE occurs due to impaired tolerance of the central and peripheral immune system where there are abnormalities in the elimination of body cells undergoing apoptosis and necrosis. 17 This can lead to the self-release of DNA/RNA which will stimulate plasmacytoid DC (pDC) resulting in the production of interferon type I (IFN-I). 3 This increase in IFN-I production leads to DC differentiation so that there will be an increase in pro-inflammatory cytokines. DC will present self-antigens so that autoreactive lymphocytes are formed. 4 In SLE patients, there is an increase in the number and activity of Th-17 cells, and a decrease in Treg that will lead to autoimmunity. In addition, there is Th-1 cell dysfunction and Th-2 cell hyperfunction that will cause activation of various types of B cells that produce autoantibodies. 18 The immune complexes of antigens and autoantibodies formed cause damage to various tissues. 19 This process of disruption of the natural and innate immune system takes place continuously and forms the chain of the pathogenesis of SLE [Figure 2(b)].
DC immunotherapy in autoimmune patients has been studied before. Preclinical trials of tolerogenic DC administration in mice with rheumatoid arthritis showed that tolerogenic DC may decrease CD4+ activation, decrease pro-inflammatory cytokines (IL-6), and increase anti-inflammatory cytokines (IL-10), and may improve clinical manifestation. 20 In another similar study, the administration of autologous tolerogenic DC derived from the bone marrow of mice suffering from SLE showed clinical improvement. This is characterized by a decrease in IL-6, IL-12, the number of anti-DNA antibodies, and anti-histone IgG, as well as an increase in the number of Treg cells. However, the administration of tolerogenic DC was not accompanied by improvements in proteinuria and glomerulonephritis. 21
To date, several clinical trials of DC immunotherapy have been conducted for autoimmune diseases. 22 Administration of DC introduced with autologous synovial fluid intra-articularly in subjects with rheumatoid arthritis showed good safety, tolerability, and efficacy. This is indicated by the absence of flares during the observation period and no increase in the severity of the disease. 23 Phase I clinical trials with type I Diabetes Mellitus (DM) subjects showed that DC immunotherapy was tolerable and there were no serious adverse events. However, the results of the study did not show any clinical changes in the subjects given the therapy. 24 Other research on the subject of type 1 DM is still ongoing and cannot be concluded. 25 Phase I clinical trials of DC immunotherapy in Multiple Sclerosis showed that patients given DC did not experience worsening during the follow-up phase. In addition, an increase in IL-10 was found to be associated with an increase in the number of Treg cells. 26 However, no results of clinical trials of DC immunotherapy on SLE have been published. From the results of the clinical trial, it can be seen that DC immunotherapies are potentially effective for autoimmune patients and have good safety. However, the efficacy of the use of this therapy in SLE still requires further research.
Conclusion
Herein, we report the first case of a patient with SLE showing significant clinical improvement after administration of the DC vaccine. It can be seen that DC immunotherapies are potentially effective in treating autoimmune diseases and have good safety. However, the efficacy of this approach to treat SLE still requires further research.
