Abstract
Highly pathogenic avian influenza viruses (HPAIVs), originating from the A/goose/Guangdong/1/1996 H5 subtype, naturally circulate in wild-bird populations, particularly waterfowl, and often spill over to infect domestic poultry. Occasionally, humans are infected with HPAVI H5N1 resulting in high mortality, but no sustained human-to-human transmission. In this review, the replication cycle, pathogenicity, evolution, spread, and transmission of HPAIVs of H5Nx subtypes, along with the host immune responses to Highly Pathogenic Avian Influenza Virus (HPAIV) infection and potential vaccination, are discussed. In addition, the potential mechanisms for Highly Pathogenic Avian Influenza Virus (HPAIV) H5 Reassorted Viruses H5N1, H5N2, H5N6, H5N8 (H5Nx) viruses to transmit, infect, and adapt to the human host are reviewed.
Keywords
Introduction
Avian influenza viruses (AIVs) naturally circulate in wild aquatic birds, such as ducks, geese, swans, gulls, shorebirds, and terns. 1 AIVs of the subtype H5N1 are often highly pathogenic avian influenza viruses (HPAIVs), which was originally discovered in geese in China’s Guangdong province in 1996. Several outbreaks occurred in farmed geese in Sanshui, a small town 50 miles outside the capital of Guangdong with a mortality rate of more than 40%. 2 By 1997, the A/goose/Guangdong/1/1996-like viruses spilled over into the live poultry markets in Hong Kong with high rates of mortality. Simultaneously, there were 18 confirmed human cases of HPAIV infection, 6 of whom died. 3 There was a large degree of homology between the avian isolates and the viral isolates collected from these human infections indicating that these viruses were being transmitted from birds to human hosts. 3
The 1997 outbreak was contained through the culling or ‘stamping out’ of all poultry in Hong Kong. 4 However, AIVs continued to circulate in healthy duck populations in surrounding areas. Its re-emergence in 2003 resulted in the infection of 2 human cases caused by novel H5N1 genetic variants that continued to circulate and evolve into 10 phylogenetic clades (0–9). 5 At the end of 2017, 860 laboratory confirmed cases of H5N1 influenza virus infection from 16 different countries, resulting in 454 deaths had been reported to the World Health Organization (WHO). 1 Infection of humans with AIVs are rare, but sporadic infections can occur due to direct contact with infected birds or through contaminated environments. 6 According to the Food and Agriculture Organization of the United Nations (FAO), China has around 64% of the world’s domesticated ducks and 95% of the domesticated goose population breeding in live poultry markets alongside other poultry and swine. These conditions allow these markets to become breeding grounds for H5Nx influenza virus circulation. 7 Outbreaks caused by AIVs have devastated live poultry markets in Asia and have had a substantial negative impact on the US economy. 7 In this review, H5Nx viruses will be discussed for their replication, infection, evolution, and threat to the poultry industry, with emphasis on the need for a broadly reactive vaccine to protect the human population.
Influenza virus replication cycle
Influenza viruses fall into the Orthomyxoviridae family, which consists of six genera, Influenzavirus A, Influenzavirus B, Influenzavirus C, Thogotovirus, Isavirus, and Quarajavirus, classified by serological cross reactivity to the nucleoprotein and matrix proteins. Of the three types of influenza viruses, Influenza A has the most genetic variation and the broadest host range. 8 Influenza viruses are further categorized into subtypes by their hemagglutinin (HA) and neuraminidase (NA) genes: as of now, there are 18 HA and 11 NA types. Phylogenetic analyses of viral genes have indicated that these viruses have a long-established history of infections in avian hosts. The nomenclature system for AIVs was established by WHO, the World Organisation for Animal Health, and the FAO. 9 Phylogenetic analysis is performed on HA sequences that have evolved from A/goose/Guangdong/1996 H5N1 virus. Viruses are grouped into virus clades based on their phylogenetic characterization and sequence homology of the HA gene. 9 The average percentage pairwise nucleotide distances between clades is greater than 1.5% and is less than 1.5% within clades. 9 As these viruses evolve through time, new subclades emerge. Influenza A viruses contain negative-sense, single-stranded segmented genomes that encode for 10 viral proteins: HA, NA, M1, M2, NP, NS1, NEP, PB1, PB2, and PA. 10 The 10 viral proteins are encoded by 8 segmented genomic strands, 11 which are coated with NP, have a double-helical hairpin structure, and carry one polymerase heterotrimer consisting of PB1, PB2, and PA (viral ribonucleoprotein particles [vRNPs]).12,13
Upon entry into a cell, the HA protein on the surface of the virion recognizes and binds to sialic acid on the surface of host cells (Figure 1, step 1). After binding, the virus enters the cell through receptor-mediated endocytosis. 14 The exact mechanism of endocytosis is not known, but it has been speculated that influenza can use both clathrin-dependent and clathrin-independent mechanisms to enter the cell. 14 Upon entry, the endosome travels into the cell and undergoes a change in pH, progressively becoming more acidic. 15 This acidification process causes an irreversible conformational change in the HA molecule on the influenza virus and exposes the hydrophobic fusion peptide. 16 The fusion peptide inserts into the endosomal membrane that causes the fusion of the viral and endosomal membrane. 17 The viral M2 protein forms a tetramer in the virion, where its transmembrane domain acts as a pore for the M2 channel.18–20 The M2 protein functions as an ion channel that modulates the intra-virion pH, pumping free hydrogen atoms into the viral core, causing the dissociation of the vRNPs from the M1 matrix proteins (Figure 1, step 2).20,21 This mechanism allows the inner contents of the viral core to be released into the cytoplasm and subsequently enter the nucleus (Figure 1, step 3). 20 After dissociation from M1, vRNPs are translocated to the host nucleus where viral replication and transcription occurs (Figure 1, steps 3 and 4).13,20 Influenza viruses are one of the few RNA viruses that can replicate in a host nucleus, due to the need for a cap sequence in order for the RNA polymerase to perform transcription (Figure 1, step 4).22,23 Influenza viral RNA segments do not contain a 5’ cap in order for the RNA-dependent RNA polymerase to perform transcription, so the PB1, PB2, and PA components perform ‘cap-snatching’ of host DNA in order to complete this process.22–25 Cap-containing viral mRNA is released into the cytoplasm to be translated by the host ribosome machinery. Surface proteins such as HA and NA are translated into the rough endoplasmic reticulum and are then translocated into the Golgi apparatus for post-translational modifications. 26 (Figure 1, steps 5 and 6).

Cartoon depiction of the replication cycle of influenza viruses. (Step 1) Viral entry into host cell. (Step 2) Virus endocytosis into host endosome and acidification, leading to conformational change of the HA molecule exposing fusion peptide and fusion of viral and host membrane. M2 protein pumps H+ atoms into the viral core, causing the dissociation of M1 and the release of vRNP. (Step 3) Release of vRNP into the cytoplasm and translocation into the nucleus. (Step 4) vRNP replication and transcription, and cap-snatching mechanisms occur in the nucleus. Viral proteins such as M1 and NS2 chaperone vRNP out of the nucleus and into the cytoplasm to be packaged into viral particles. (Step 5) Structural proteins are translated by host ribosomes and are transported to the endoplasmic reticulum for proper folding. (Step 6) Properly folded viral proteins are released from the endoplasmic reticulum and are directed towards the plasma membrane or to the Golgi for modifications prior to release. (Step 7) Movement of modified proteins from the Golgi network to the plasma membrane for viral budding. (Step 8) Release of infectious viral progeny.
The viral nuclear export protein NS2 is critical to nuclear export of vRNPs, viruses that lack NS2 resulted in reduced viral growth. 27 After genomic replication, transcription, and protein synthesis, NS2 and M1 help escort the new viral proteins to the host cell membrane, where they assemble and bud newly synthesized virions (Figure 1, step 7).28,29 As the nascent virions bud from the host cell, NA cleaves the sialic acid residues on the host cell membrane, 30 which allows the viruses to escape the host membrane (Figure 1, step 8). 31 Antiviral drugs that block NA activity result in influenza viruses accumulating at the membrane and cannot further disseminate to cause infection of neighboring cells. 32
HPAIV HA
HA is located on the surface of the influenza virus and it facilitates viral entry into the host cell by binding to sialic acid on the host cell surface. 33 Avian-adapted strains of influenza virus preferentially bind to N-acetylneuraminic acid with α-2,3-sialic acids. 34 These sialic acids are located in the gut and the digestive tract of avian species and in the lower respiratory tract of humans.35,36 HA is synthesized as polypeptide chain-encoded domains HA1 and HA2, co-translationally translocated into the lumen of the endoplasmic reticulum and eventually to the surface. 37 The HA protein contains a cleavage site between the HA1 and HA2 domains, cleavage is essential for infectivity and allows the HA molecule to undergo an irreversible conformation change in acidic endosomes. This cleavage is performed by cellular proteases to create two subunit HA1 and HA2 domains linked by disulfide bonds. 38 The cleavage nature of H5 HA proteins is achieved when virions are incubated with trypsin. This results in the conversion of HA to HA1 and HA2. The cleavage of HA can be blocked by a protease inhibitor. 37 HA is expressed on the virion as a trimeric protein that is stabilized by residues on the HA2 region. The HA ectodomain is composed of two regions, a stem region and a globular head region. 39 Neutralizing antibodies directed to the globular head of HA are critical for reducing viral infection and disease. 40 An important factor affecting viral pathogenicity depends upon the sequence of the amino acids in the HA0 cleavage site.41,42 HA proteins from HPAIVs contain a multibasic cleavage site that is cleaved by the ubiquitous furin cellular protease.42,43 In contrast, in low pathogenic avian influenza viruses (LPAIVs), that contain only one basic amino acid, the cleavage of HA is tissue specific, which results in a lower clinical manifestation in poultry.
The polybasic cleavage site on HA is a strong determinant for high pathogenicity of H5 viruses, however insertion of polybasic sequences into an LPAIV HA does not always result in a lethal phenotype as tested in chickens. 44 Other influenza proteins such as PB2, PB1, and NP may increase pathogenicity of an influenza virus. The pathogenic phenotype of H5 viruses is not HA dependent. Pathogenicity and efficiency of replication can also be dependent on PB2, NP, NA, and M genes. 45 The deleted stalk region of NA found in HPAIVs also confers pathogenicity, where rescue of the NA stalk region leads to a decreased pathogenesis in chickens. 45 Deletion of the stalk region increases lethality and transmission compared with the wild-type viruses that display a lower lethality. In addition to H5 viruses, this same NA stalk deletion abrogates H2N2 virus replication in ducks, but shifts the virus tropism from the intestinal tract to the respiratory tract in chickens. 46 The presence of the polybasic HA cleavage site was sufficient enough to induce viral neurotropism. 44
The dominant circulating AIV strains that have arisen since 2015 comprise viruses in the clade 2.3.4.4, which includes reassortant viruses in the H5N6, H5N8, and H5N2 subtypes. However, the strains within this clade that have crossed over into the human population are limited to the H5N6 subtype. The ability of H5N6 viruses to spill over into the human population may be associated with mutations in the HA molecule that affect the specific RBS binding preference of HA. These H5N6 viruses preferentially bind to different sialic acids depending on the host from which each virus was isolated. 47
NA in H5Nx viruses
Characterization of LPAIV or HPAIV infections in poultry usually refers to the pathogenicity of the virus during infection and whether the virus contains a polybasic cleavage site in its HA molecule (as reviewed above). However other proteins, such as NA, can add to the pathogenic nature of the virus. To date, the newly circulating strains of AIV in China are H5N6, H5N8, and H5N2 of type H5Nx viruses. 48 These viral reassortments can result in a dominant NA molecule that increases the pathogenicity and release of viral particles. Overall, this can increase viral transmission between hosts. The role of non-HA viral gene products and how these proteins contribute to viral tissue tropism and virulence are still not well understood. Multiple passages of H5N3 viruses in poultry result in a mutation in the catalytic site of NA, which increases the virulence of these viruses in poultry. 49 Mutations in internal genes, such as PB2, have also been linked to increased viral pathogenicity in H5Nx viruses. Multiple passages of H5N5 viruses in mice resulted in a substitution in amino-acid position 627 from glutamic acid to lysine (E627K) in the HA protein. 50 This adaptive mutation increased the pathogenicity of these viruses in mice by 1000× and enhanced viral replication in vivo and in vitro. 50 There were significant structural and functional differences in the NA proteins (N6, N8, and N2) from several viruses associated with the clade 2.3.4.4. 49 The HA/NA interplay may be age dependent: whereas nonfunctional H5 viruses result in the death of day-old chickens, infection with the same virus in week-old chickens showed no signs of clinical illness at all. 51 This seems to be an H5-specific phenomenon, whereas H7 viruses were less dependent on a functional NA to cause illness. The dominant AIVs that infected humans have been associated with H5N6 viruses from clade 2.3.4.4. Out of the 17 human infections with H5N6 virus, 16 of the viruses contain a NA stalk deletion. Recombinant H5N6 viruses containing a 10 amino acid NA stalk deletion (amino acids 58–68) had an increase of viral replication in mammalian cell lines compared with the intact NA of H5N6 viruses. These viruses containing the NA stalk deletion also showed an increased viral replication in avian CEF cells, whereas H5N2 virus had lower titers in these cells. 52 This recombinant virus with the NA deletion (∆H5N6) did not infect neural tissue in mice, whereas the full length H5N6 recombinant virus was neurotropic. 52 Wild-type H5N6 viruses had higher rates of viral transmission and were more lethal to poultry compared with the ∆H5N6 virus. Wild-type H5N6 viruses were 100% lethal to chickens. All birds died within 10 days postinfection, whereas, only 85% of the ∆H5N6 challenged chickens died by day 14 postinfection. These data suggest that the NA stalk region in H5N6 viruses plays an important role in pathogenicity in mammalian hosts and displayed a decreased pathogenicity in chicken cells.
AVI infection in birds
Domesticated birds, such as chickens and turkeys, may become infected with AIVs through direct contact with infected waterfowl or infected poultry. AVis infect over 105 bird species across the globe, but the natural reservoirs for this virus reside in aquatic fowl such as gulls, terns, and shorebirds. 53 Waterfowl can transmit AIV to other avian species such as terrestrial poultry. Infection of poultry with LPAIV can result in little to no disease. Clinical signs of LPAIV infection are ruffled feathers and a drop in egg production. 54 Infection of birds with LPAIV can result in the virus mutating and adapting to the unsusceptible bird, possibly creating an HPAIV in these birds. 53 Adaptation of the virus to increase replication efficacy can result in a LPAIV transforming into an HPAIV with the addition of basic amino acids inserted into the cleavage site on HA. 54 Natural LPAIV infections in wild birds do not present with clinical signs of infection or tissue lesions.55,56 The H5N1-associated response in chickens includes a massive influx of cytokines, antiviral cytokines, and interferons, which should inhibit viral replication. 57 However, some cytokines that are activated like IFN TNF-α, IL-8 and IL-6 may be responsible for influenza-induced pathology. 58
Wild birds, such as ducks, are more resistant to H5N1 HPAIV infection compared with gallinaceous poultry. 59 The rapid disease progression seen in infected chickens is not observed in ducks. 60 Ducks are able to maintain H5 infections without developing severe disease and continue to spread the H5 virus into susceptible chicken populations. 61 Infection with H5N1 in susceptible birds results in systemic infection, leading to multiple organ failure, damage to the cardiovascular and nervous systems, and ultimately death.53,54 HPAIV H5 viruses replicate in the respiratory and gastrointestinal tracts of birds.59–61 Clinical signs of infection include loss of appetite, lack of energy, loss of coordination, discoloration and swelling of body parts, diarrhea, nasal discharge, coughing, sneezing, and misshapen eggs. 54 Diagnosis of AIV in birds is carried out by taking throat swabs of birds; in wild birds, a fecal sample is taken instead and is tested through PCR analysis. Positive PCR results then leads to virus isolation and growth of the virus in an embryonated chicken egg. 62
The evolution and spread of H5 viruses
The A/goose/Guangdong/1/96 virus was initially detected in wild birds in Southeast Asia, but shortly thereafter was detected in several areas in Asia, Europe, Africa, and recently North America. 63 Sequence analysis of the H5N1 viral gene segments isolated from poultry in Hong Kong distinguished two groups of viruses circulating in domestic poultry. 64 During infection, the HA segment did not undergo mutation to adapt to the human hosts. However, the PA, PB2, NP, and M2 gene products had multiple amino-acid changes after replicating in human hosts. 64 Those mutated amino acids were similar to those in viruses that commonly infect the human population. 64 There are nine different types of H5 viruses in wild-bird populations (H5N1, H5N2, H5N3, H5N4, H5N5, H5N6, H5N7, H5N8, and H5N9). These H5 subtypes can present considerable risk to the human population.
This rapid expansion of the A/goose/Guangdong/1/1996-like viruses was driven by reassortment with other AIVs. 65 Following the goose/Guangdong virus emergence, H5N1 viruses continued to circulate in China with a seasonal pattern peaking from October to March when the temperature is below 20°C. 65 In 2003, the H5N1 outbreaks in humans revealed that the goose/Guangdong-like viruses had diverged into eight genotypes. Several of these genotypes survived, while others went extinct. 65 Specific adaptations to the viral genome led to an increased fitness of select strains that continued to circulate in southern China. 45 A series of genetic reassortment events led to the initial human outbreak in Hong Kong in 1997, which can be directly traced back to a viral genotype in chickens and ducks named ‘Z’. Strains of the genotype Z replaced the genotypes A-E, X, and Y in 1997 and then became dominant in aquatic fowl and terrestrial poultry. The overall prevalence of H5 viruses has increased since 2010. The most prevalent AIVs are viruses in the H5N6 and H5N8 subtype circulating in domestic waterfowl in China. 66
Since 2008, HPAIV subtypes H5N5, H5N2, and H5N8 viruses have caused outbreaks in poultry across Asia, Europe, and North American.67–70 By 2014, outbreaks of novel reassortant viruses such as H5N6 and H5N8 were reported in Asia.48,70 Viral reassortants of H5N1 and H5N2 variants have also been isolated from chickens and waterfowl and have caused outbreaks in chickens in Hebei Province in China during December 2013. 66 The H5N2 viruses expressing the HA gene from clade 2.3.2, 2.3.4 or 7.2 have been isolated with internal genes from both clade 7.2 H5N1 viruses and H9N2 viruses. H5N2 viruses contain gene segments from multiple viral clades including HA, PA, M1, PB2, NS1 from 2.3.4.4, and three American wild birds lineage genes NA, NP, and PB1.71,72 These H5N2 viruses were the source of an outbreak in Hubei, Shandong, and Henan Provinces in China. Low pathogenic H5N2 influenza viruses naturally infect wild birds. 73 However, these LPAI H5N2 viruses can transform into an HPAIV isolate by either adding basic amino acids to its cleavage site or by loss of a N-glycosylation at amino acid site 11. 74 These viruses rapidly adapted to replicate in terrestrial poultry by adding an additional glycosylation site on the HA molecule, as well as deleting 19 amino acids in the NA stalk region. 73
The A/goose/Guangdong/1/1996-like viruses are still being detected in poultry and wild birds in many countries. The majority of these isolates are classified as clade 2, including the subclades 2.2.1.2 in Egypt and 2.3.2.1a in Bangladesh, Bhutan, and India. 75 H5 viruses designated in the clade 2.3.2.1c have been detected in wild-bird populations in China, Southeast Asia, and Africa. 75 H5 influenza viruses of the clade 2.3.4.4 have been detected in wild birds around the world. The clade 2.3.4.4 viruses from Africa and Europe are primarily of the H5N8 subtype, whereas those viruses isolated in Asia are of the H5N6 subtype, and those strains isolated in the USA are classified as H5N2 isolates. 75 Clade 2.3.4.4 H5 influenza viruses have caused 1537 outbreaks in birds in 14 countries over the past decade. 76 These outbreaks have been associated with H5N6, H5N8, H5N2, and H5N3. These viruses emerged through multiple reassortment events with the H5N1 subtype and now routinely circulate in domestic poultry and waterfowl. 76 The first 2.3.4.4 virus that emerged was classified as a H5N6 subtype and it circulated throughout China and traveled to Southeast Asia causing the death of 457 birds in Laos in March 2014. 77 The H5N6 subtype arose from a reassortment event with HA genes from H5N1 and NA genes from the H6N6 virus. 77 The H5N8 subtype caused an outbreak in 2014 in South Korea leading to a distinction of two different H5N8 virus subgroups, group A and group B. 78 Group A comprises a set of H5N8 isolates and is referred to as the intercontinental group A (icA) group. The icA H5 viruses further evolved into three different subgroups, icA1, icA2, and icA3. The icA1 subtype group contains viruses that were isolated from Europe, Russia, and Japan. The icA2 subgroup is composed of H5N8 and HPAIV reassortants H5N2 and H5N1 from North America in 2014. The icA3 subgroup is composed of H5N8 viruses isolated in Japan and Korea. 78 In 2014, H5N8 Eurasian subtypes emerged in Canada, Germany, The Netherlands, UK, and East Asia and was concurrently detected in the US state of Washington in captive falcons, wild birds, and poultry. It spread across US mid-western and north-central states causing devastation to the poultry industry. Over 48 million chickens and turkeys were culled, which led to a loss of US$1.6 billion. 79 The spread of these intercontinental-like viruses coincided with bird migration out of Russia and most likely were spread by migratory birds. 78 Clade 2.3.4.4 viruses are less pathogenic in wild waterfowl, which increases the chances of being spread through migratory birds.
Currently, organizations such as the WHO have taken global surveillance efforts in order to monitor the occurance of influenza H5 infections in humans. The widespread epidemic of H5Nx strains in poultry poses a considerable threat to human public health. The specific objectives of the WHO include (a) monitoring the global occurrence of influenza H5 viral infection in humans; (b) identifying and characterizing emergen inflluenza strains, (c) monitoring changes in transmission patterns of influenza H5 viruses and detecting potential human-to-human transmission of influenza H5 viruses; (d) monitoring unusual morbidity and mortality due to acute respiratory illness; (e) contributing to the monitoring of outbreaks of HPAIV in animal populations. 80 Surveillance efforts of H5 viruses will increase researchers knowledge on the nature of H5Nx reassortant events and can help government organizations predict new reference strains for pandemic vaccine development.
Transmission of HPAIV into swine
The ability of an AIV to transmit amongst humans may rely on the ability of the virus to replicate efficiently in swine cells. Swine are often the host for viral reassortment and generating novel viruses in the ecosystem (Figure 2). 81 Cells found in the respiratory tract of swine species have both -2,3 and -2,6 sialic acid receptors, making them a ‘mixing pot’ for the generation of novel pathogenic viruses. 82 Reassorted H3N2 swine viruses cause epidemics in the swine population, as seen in the 1998 outbreak that occurred in the USA in North Carolina, Texas, Minnestota, and Iowa pig farm outbreaks.83,84 Genetic analysis of the swine viral isolates revealed viral genes from human, swine, and AIVs. 84 Swine viruses are able to spill over into the human population and cause severe disease in people. 85 There have been a total of three documentated swine infections in the human population recorded in the USA. 83 Although H3N2 and H1N1 viruses are known to replicate and reassort in swine, the ability of H5Nx viruses to infect these species is still under investigation. As of October 2018, there have been 50 submissions of H5N1 swine infection uploaded to the GISAID (https://www.gisaid.org) website between 1968 and 2017, with 36 of the HA sequences being unique. Few pigs are naturally infected with H5 viruses. Surveillance of pig farms in Vietnam in 2004 showed that only 0.25% of 3175 pig tested were positive for H5N1 viruses. 86 These H5N1 viruses were found to replicate in naïve swine, but did not cause severe disease or weight loss, and did not transmit to naïve contact pigs. 86 The potential for reassortment of AIVs and mammalian influenza viruses in pigs is still unknown, but swine are permissive hosts for viral replication of AIVs.87–89 Newly characterized clade 2.3.4.4 H5Nx viruses do not readily infect trachea cell extracts collected from pigs. 89 There are multiple studies showing that HPAIVs and LPAIVs do not cause clinical symptoms in naïve swine.87–91 Microscopic pathological analysis does reveal some cellular infiltrates, along with minor macroscopic lesions, but replication in these hosts does not permit transmission to naïve pigs. 87 In addition, co-housing poultry that were infected with HPAIVs were not able to transmit the virus to naïve pigs. 92 In pigs infected with H5Nx influenza viruses the virus replicated in the lower respiratory tract, since nasal swabs were negative in reverse transcriptase polymerase chain reaction (RT-PCR) assays, but bronchoaveolar lavage fluid contained viral titers postinfection 87 demonstrating that AIVs replicate in the lower respiratory tract in pigs. This site of replication in the lungs is consistent with the location of the α-2,3 alpha receptors found in the lower respiratory tract of pigs and not present in the upper respiratory tract. 82 A survelliance study performed in China monitored and tested 16 swine farms in southern China, and serum samples collected from the swine determined that no pigs were serologically positive for H5N1 virus. 93 These studies showed that pigs are not easily susceptible to H5Nx infection, and that those that do become infected do not easily transmit to naïve companions. However, there is always the concern that due to the lack of H5N1 virus-induced clinical manifestations, H5Nx and seasonal H1N1/H3N2 influenza viruses could co-infect the same animal resulting in a reassortment event that leads to a human transmissible H5 influenza virus.
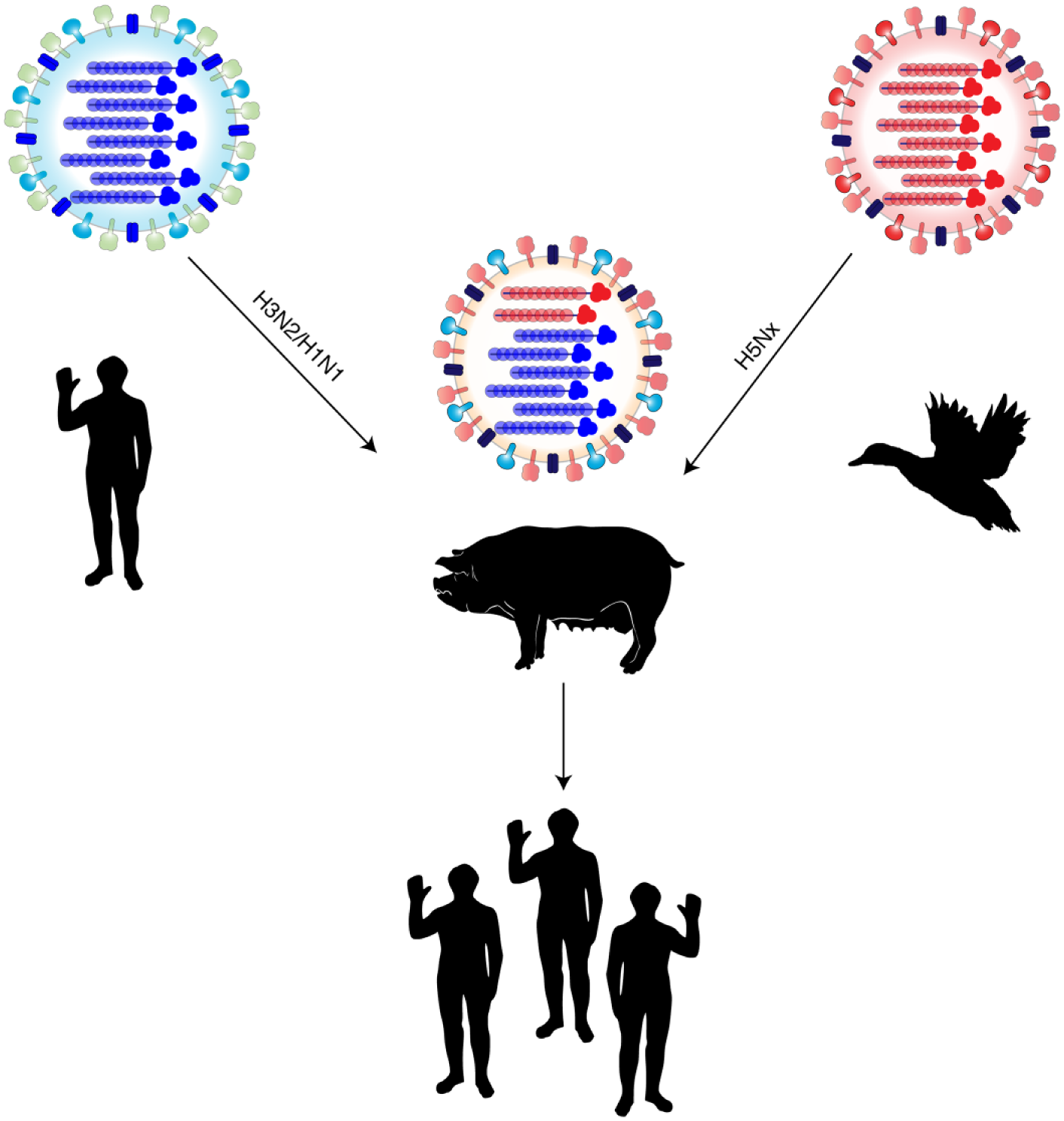
Depiction of swine reassortants. Seasonal influenza from human hosts (left) can be transmitted into susceptible swine. Concurrent infection by avian strains (H5Nx) with seasonal influenza strains can lead to reassortants, novel influenza viruses that contain genetic segments from both humans and avian viruses. These novel viruses can then be transmitted into susceptible human populations, possibily leading to a pandemic outbreak.
AIV: human transmissible agent
HPAIV infection in humans occurs sporadically in Asia, Africa, Europe, and the Middle East. 1 As of October 2018, there have not been any reported cases of human H5N1 infection in the USA, but one case was reported in Canada in 2014 from a person who had recently traveled from China. 94 HPAIV H5 virus infection in humans can initially present as an uncomplicated seasonal influenza infection with clinical signs of fever, body aches, and upper respiratory tract symptoms.1,95–97 However, the infection eventually progresses into a lower respiratory tract infection. The infection can progress to become severe pneumonia, multi-organ failure, encephalitis, and septic shock.1,95,96 The incubation period for H5N1 virus infection is estimated to be 7 days, but is more commonly 2–5 days after exposure. In the rare cases where human-to-human transmission occurred the incubation period varied from 3–4 days to 2–10 days. Variability in this incubation period can possibly reflect the level of viral shedding, exposure, and immunological factors. 98 A reported pediatric case with H5N1 virus infection presented with fever, diarrhea, and seizures, which progressed to a coma and eventually developed into encephalitis. The patient had contracted the virus from his sister 2 weeks earlier who also suffered from encephalitis and died. Neither patient presented with any respiratory symptoms. 99 The severity of the illness varies with the virus clade, age of the patient, and other unknown genetic factors. Rapid disease progression following AIV infection occurs in the majority of human cases. Extrapulmonary complications that arise from H5 viral infections include cardiac failure, kidney failure, encephalitis, muti-organ failure, and intravascular coagulation.97,100–102 Human-to-human transmission of avian H5N1 virus has been recorded in several households, but is often limited.95,96 Transmission usually results from close intimate contact and/or care of a family member infected with H5N1 viruses. Surveillance by RT-PCR of contact patients led to the detection of mild cases of infection in older adults and an increased number of seropositive families in northern Vietnam. 103
During influenza infection, a large infiltration of cytokines, inflammatory cells, and tissue damage in infected individuals increases the morbidity and mortality rate of influenza disesase.104,105 Elevated levels of proinflammatory cytokines, also known as a ‘cytokine storm’ significantly impact the recovery of infected individuals.104,105 Increased cytokine levels are often found in clinical specimens from patients infected with H5N1 influenza viruses.105–107 Autopsy studies revealed alveolar damage and high levels of circulating chemokines and cytokines in the peripheral blood of the patient. 107 H5N1 viral infections in humans induce a higher transcription of proinflammatory cytokines than seasonal H3N2 or H1N1 influenza viruses. 108 The cytokines TNF-α and IL-β are significantly elevated following H5N1 viral infection. 108 This is partially due to the nonstructural gene segment of the H5N1 viruses, 109 and may contribute to the overall severity of H5N1 influenza virus infections in humans. 108
As of July 2018, HPAIVs have caused a total of 860 confirmed cases and resulted in 454 deaths worldwide. 110 The reported number of human infections has been decreasing since 2003. Although there have been a few cases of documented human-to-human transmission, these viruses are not easily transmitted in humans. This may reduce the threat of H5Nx viruses causing widespread infections in people and becoming a pandemic issue in the human population. However, the virus only needs to accumulate minor mutations in order for viruses with H5 HA proteins to transmit easily between mammalian hosts. 111 One factor that may increase the pandemic potential of AIVs is the ability of these viruses to be transmitted through aerosolized particles. 112 This is largely dependent on the ability of the virus to replicate in the upper respiratory tract of mammalian cells. Distribution of sialic acid receptors between human and avian hosts differ, with α-2,6 sialic acids being predominant in the upper respiratory tract of humans, and α-2,3 sialic acids being more predominant in the lower respiratory track of humans, whereas avian species have α-2,3 sialic acid receptors predominantly expressed in both the respiratory tract and digestive tract. Therefore, sustained human-to-human transmission of AIVs may also include the switch from α-2,3 sialic acid binding to preferentially α-2,6 sialic acid binding to ensure dissemination through aerosolized droplets from the upper respiratory tract. 112 Sialic acid preference is not solely responsible for the pathogenicity of AIV in mammalian cells. 113 AIV mutants that preferentially bind to α-2,6 sialic acid receptors have lower viral replication rates compared with wild-type viruses. 113 Efficient transition for AIV to replicate in human hosts is dependent on more than receptor binding preference. Replication and release of these infectious particles are also critical when discussing the switch of infection from birds to mammals. A specific mutation in the polymerase protein PB2 in site 627 from a glutamic acid to a lysine (E627K) increases the pathogenicity of AIV infection in mammalian models.114–116 Influenza viruses that readily infect humans hosts predominantly display a lysine in site 627 in the PB2 protein. In avian species, the 627 site in the PB2 protein is glutamic acid. The function of PB2 is restricted in mammalian cells when these AIVs contain glutamic acid (E) in site 627. These viruses do not assemble into ribonucleoprotein complexes and have decreased genome transcription and virus production. 114 The lysine (K) amino-acid substitution in the PB2 protein allows the virus to replicate in the lower temperatures of the upper respiratory tract of mammals (33°C), as opposed to the higher temperature (41°C) in the gastrointestinal tracts of avian species (117). H5N1 viruses that contain the PB2 E627K substitution have increased viral replication in the nasal passages of mice compared with viruses with the E627 wild-type virus. 117 Other changes in PB2, such as the D701N mutation, have been associated with an increased animal tropism, increased polymerase activity, and enhanced pathogenicity in mammals. The SR polymorphism in PB2 allows the polymerase to escape species-specific restriction factors that target polymerases from AIVs. 117 These mutations increase the overall viral replication of AIVs in mammalian hosts cells and ensure a high replicative titer and infection. Viral reassortments containing replicative proteins from multiple species can help AIVs to expand their tropism and adapt to new hosts. 118
PB1-F2 is a small protein that is encoded from the same vRNA segment as PB2 from a +1 reading frame. PB1-F2 protein is an important virulence factor in influenza viruses and can have three roles in viral infection, that is (a) regulate antiviral innate immunity; (b) enhance viral polymerase activity; (c) induce cellular apoptosis. 119 Viruses isolated from the 1997 H5N1 pandemic contained an amino acid at position N66S that correlated with high pathogenicity in mice. 120 When an S66N mutation is introduced into the PB1-F2 protein of an H5N1 virus, there is an attenuation of disease in mice. Viruses that contain a N66S mutation have increased disease severity and production. 120 The truncated version of PB1-F2 does not increase virulence in mice or their predisposition to co-infection by bacterial strains. 121 However, the PB1-F2 protein that lacks an ATG start site was 1000-fold more pathogenic than the full length PB1-F2 containing an ATG site. 122 The ability of H5Nx viruses to replicate successfully in human hosts is dependent on the adaptation of multiple viral RNA segments and are not restricted to HA and NA activity.
Future perspective
In order to combat the evolution and divergence of H5Nx avian influenza, a universal avian influenza vaccine (UAIV) for the human population should be developed. A UAIV would be broadly protective against multiple viruses that span throughout several clades and against multiple mismatched NA (H5N1, H5N2, H5N6, H5N8). This vaccine should provide protective antibody- or cell-mediated response against viral challenge with one vaccine dose. Multiple or boost vaccination has been attempted with heterologous prime-boost strategy in AIV vaccination studies. 123–125 Although these types of vaccine protocols have proven successful, in the event of an epidemic, single-dose adjuvanted vaccination would be less time consuming and more cost effective. In the event that a single dose is sufficient, a greater portion of the population would have access to the vaccine compared with a two-dose strategy where perhaps only half the population would receive the dose. 126 Protective efficacy would also span multiple age groups, taking into account pre-immunity to seasonal influenza strains such as H1N1 and H3N2. Although these qualifications for a UAIV may seem too far fetched, there has been progress in recent years. Our group has reported on a computationally optimized broadly reactive antigen adjuvanted vaccine that provides protection against heterologous challenges in mice, ferrets, and nonhuman primate models,127–131 and is being analyzed for potential vaccine candidacy.
Conclusion
The molecular basis of HPAI H5Nx viruses to induce pathogenicity, as well as the potential for these viruses to adapt to humans was reviewed. The segmented genome of the influenza virus facilitates reassortment in hosts that are infected with more than one strain of influenza virus. These reassortment events in birds and swine have generated new and novel strains of AIVs, including the divergent 2.3.4.4 clade viruses that contain avian NA proteins. Influenza viruses with genomic RNA segments coding for internal and nonstructural proteins such as PB1, PB2, PA, and NS1 have increased viral replication and overall enhanced pathogenicity. Reassortment of AIV RNA segments with seasonal human influenza virus strains in swine by co-infection may not factor into the evolution of novel avian viruses. Swine that are infected with AIVs have no clinical signs of infection and do not transmit nascent virions to immunologically naïve hosts. However, these animals may have a subclinical infection that could in fact promote reassortment events upon co-infection.
The next influenza pandemic is inevitable and most likely will emerge from a novel subtype to which the human population has no pre-existing immunity. The ability of an HPAIV to accumulate the appropriate mutations or reassortment with an efficient human or swine virus resulting in a human transmissible virus is possible and could initiate a new pandemic. Therefore, continued scientific studies and surveillance of AIVs are essential to develop vaccines and therapeutics, not only for the poultry industry, but also to prepare for the next AIV emergence into the human population.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article. This work was supported by the Center of Vaccines and Immunology at the University of Georgia, Athens, Georgia, USA.
Conflict of interest statement
The authors have no competing interests with the funding sources and the manuscript is solely the opinion and thoughts of the authors.
