Abstract
Background:
Many countries rely upon subdural grid electroencephalography in the planning of epilepsy surgeries. However, craniotomy for subdural grid implantation is known to result in a variety of complications and requires diligence from the surgical team. We describe a minimally invasive method of subdural grid insertion, termed the linear oblique craniectomy, designed to mitigate complications and increase ease of subdural grid insertion.
Objective:
To demonstrate feasibility of minimally invasive subdural grid insertion utilizing skull anatomy.
Methods:
Three fresh frozen and embalmed human cadavers underwent surface landmarking and craniectomy to introduce a 4 × 5 cm2 subdural grid over the Sylvian fissure. Anteroposterior lens-shaped craniectomy measured 5 cm in length with 1 cm maximal width. The dura mater was longitudinally incised, and subdural grids were introduced over the Sylvian fissure.
Results:
The total area of the craniectomy created by the linear oblique approach consists of only approximately 20% of the total area removed by the traditional approach to access the Sylvian fissure for mesial temporal epilepsy monitoring/preoperative planning. The locations of the grids were evaluated by MRI and computed tomography scans postoperatively to ensure accurate alignment with the Sylvian fissure.
Conclusion:
In this cadaveric study, we demonstrate the linear oblique craniectomy procedure that provides an alternative approach to subdural grid implantation with significantly decreased invasiveness. This surgical approach has the potential of reducing complication rates of subdural grid insertion for surface monitoring of the brain activity and/or neuromachine interface analysis and is associated with significant reduction of surgical time.
Introduction
The World Health Organization recognizes epilepsy as a major public health concern, with more than 70 million people worldwide living with the condition. 1 In cases refractory to antiepileptic drugs, roughly 30% of cases, surgical resection and invasive electroencephalography are indicated. 2– 4
Subdural grid is globally the most common technique for seizure localization. 5 Classical craniotomy to implant subdural grids is associated with significant mortality rates and morbidity compared to depth electrode. 5– 7 Centers lacking access to depth electrodes rely upon subdural grids in their surgical planning, as do centers looking to maximize localization via combined subdural grid–depth electrode recording. 8
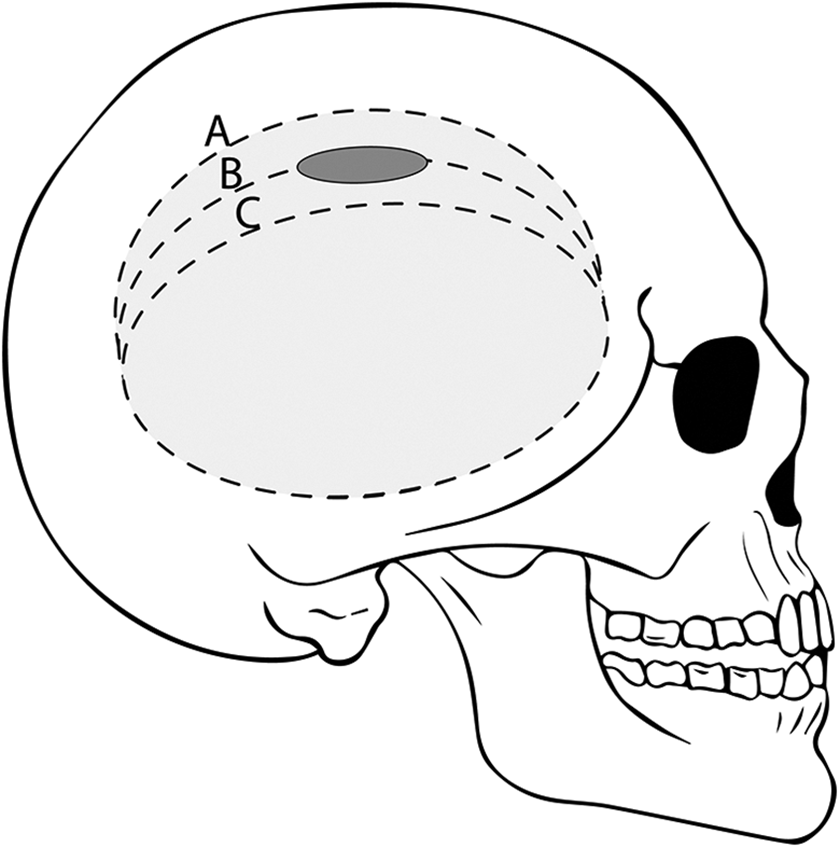
Recognizing the importance of subdural grids and complications associated with craniotomy implantation, 5–7 we describe a novel minimally invasive procedure—the linear oblique craniectomy (Figure 1). A similar craniectomy, termed the “letter-box” which utilizes a linear perpendicular craniectomy, reduced craniotomy complications but introduced new complications. 9 Our linear oblique craniectomy seeks to eliminate these complications and improve the overall approach. Linear oblique craniectomy is quick, may mitigate complications, allows simple grid removal, and is more aesthetically pleasing than craniotomy. Implantation of the subdural grid over the Sylvian fissure was chosen as proof-of-concept, given it is a common target and may be well targeted using craniometric measurements. This does not seek to provide clinical data but to provide the technical note for future clinical studies.

Illustration of linear oblique craniectomy. By utilizing the curvature of the superior temporal line and making an incision into the skull at 30–50° (θ) to the tangent of the surface of the calvaria, a trajectory may be fashioned that is in the same plane as the subdural space overlaying the Sylvian fissure. Dashed lines represent the boundaries and trajectory of the craniectomy.
Methods
Specimens
Three linear oblique craniectomies were performed on two cadavers attained from the Cumming School of Medicine Body Donation Program in accordance with the current Alberta legislation consent for donation. All procedures were carried out in accordance with Human Tissue and Organ Donation Act and approved by the ethics board of University of Calgary Conjoint Health Research Ethic Board.
Procedure
The cadaveric head was positioned supine and slightly flexed via Mayfield head clamp. The Sylvian fissure’s location was estimated by connecting a line from the lateral canthus to the posterior ¾ of the Taylor–Haughton line. Posterior edge of the Sylvian fissure was approximated by measuring 8 cm posterior from the supraorbital fissure and translating this to the estimated trajectory of the Sylvian fissure. This point was translated medially 1.5 cm from the superior temporal line, defining the access point for the linear oblique craniectomy. Neuronavigation was not used to target the Sylvian fissure, given the strong ability of craniometric measurements to target the fissure.
An anteroposterior linear incision was created from this point directly to the periosteum (Figure 2(a)). Periosteum was then reflected. A 1-cm diameter burr hole was created at 30–50° to the tangent of the calvaria (Figures 1 and 2(b)), resulting in a hole roughly parallel with the inner table of the calvaria (Figure 1 and 2(c)). Approximation of this angle could be attained by drilling in parallel with the temporalis. As the subdural grid used was 4 cm, extension of the burr hole of 2.5 cm in each direction was planned. Penfield #1 was used to separate dura from the inner table of the burr hole, to mitigate the risk of unintentional durotomy due to the footplate of the craniotome. Curvilinear extension of the access point was achieved by craniotome, resulting in a lens-shaped craniectomy at an oblique angle (Figure 2(d)). Craniotome incision was maintained at 30–50° in order to maintain parallel nature with the inner table of the calvaria.
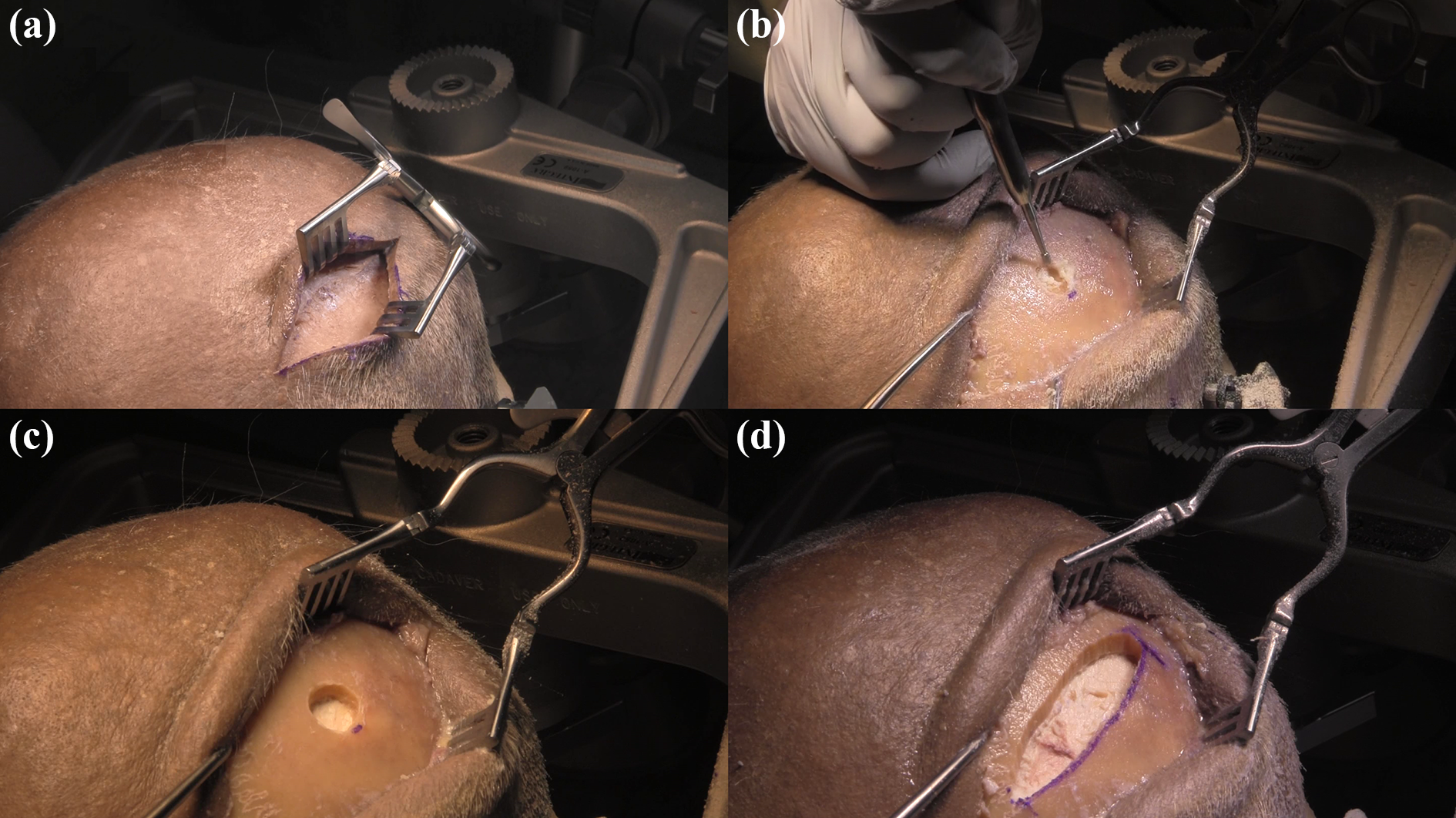
Linear oblique craniectomy. (a) Ideal incision to begin linear oblique craniectomy, prior to further expansion due to rigidity of cadaveric skin. (b) Burr drill at 30–50° to the calvaria. (c) Demonstration of burr hole angled at 30–50°. (d) Demonstration of the completed linear oblique craniectomy exposing the dura.
The dura was then raised with a 3-0 Prolene suture needle to incise it with a #15 scalpel blade. The incision was made along the medial edge to increase visualization of the lateral subdural space. Toothed forceps were used to raise the inferior dural flap, allowing visualization of potential bridging veins and other obstacles.
With the lateral dural flap retracted, bayonets were used to manually introduce a saline-wetted 5 × 4 cm2 subdural grid until it reached the approximated location of the Sylvian fissure. The subdural grid was then sutured to the dura to prevent potential migration of the grid from the desired implantation site. The lateral dural flap was larger and more accessible due to medial dural incision which enabled easier anchoring of the subdural grid. Dura was closed with 4-0 Nurolon simple interrupted suture while approximating the lateral dural flap using toothed forceps, while the medial dural edge was anchored by the calvaria. The medial dural incision approach also permits utilization of surgical adhesives such as fibrin glue or other dural sealants. Bone pieces were not replaced. Should the bone pieces be replaced, the subdural grid wires may exit via the craniectomy. Subdural grid wires were tunneled and anchored to the skin 4 cm medial to the incision and the scalp closed via 4-0 Prolene simple interrupted sutures. X-Ray demonstrated left linear oblique craniectomy at the appropriate location resulting in a trajectory tangential to the brain surface (Figure 3(a)). X-Ray, computed tomography, and MRI confirmed location of the subdural grid over the Sylvian fissure (Figure 3(b), (c), and (d), respectively).

Neuroimaging of completed linear oblique craniectomy. (a) X-Ray depicts implanted subdural grid over left Sylvian fissure. (b) Lateral view of subdural grid location (head is malaligned with X-ray). (c) Computed tomography depicting electrodes overlaying the left Sylvian fissure. (d) T1-weighted MRI depicting the left Sylvian fissure at the same level in greater detail.
Involvement of the craniectomy into subsequent craniotomy for surgery is possible. Depending upon the preference, craniotomy may simply be encapsulated by the bone flap (Figure 4(a)), craniectomy may utilize the craniotomy (Figure 4(b)), or craniectomy may be uninvolved in the subsequent craniotomy entirely (Figure 4(c)).
Depiction of linear oblique craniectomy involvement in subsequent epilepsy surgery craniotomy. (a) Craniotomy encapsulates the craniectomy entirely. (b) Craniectomy is used similarly to an additional burr hole, incorporating the craniectomy in the craniotomy. (c) Craniotomy is unrelated to the craniotomy.
Discussion
Improvements upon the preceding nonoblique linear craniectomy
The oblique nature of this craniectomy ensures a linear trajectory of subdural grid during introduction (Figures 1 and 3(a) and (b)). This procedure may be utilized at any location on the calvaria; however, as we have demonstrated, it is well suited to overlie an apex of the calvaria. Utilizing a calvarial apex allows maximal subdural space visualization and ease of insertion by bringing the trajectory more parallel to the subdural space.
This procedure is comparable to a similar procedure, the “letter-box” by Raftopoulos and colleagues. 9 The letter-box procedure for subdural grid implantation was conducted on 61 consecutive patients with medically refractory epilepsy. They found that a nonoblique linear craniectomy approach was superior to traditional craniotomy for subdural grid insertion by increasing simplicity, decreasing operating period, resulting in significantly lower levels of intra/postoperative complications, and in reducing the need for mannitol or dexamethasone.
We compare several aspects of these two procedures here. The first comparison is complications. Letter-box technique caused rupture of bridging veins and associated subdural hematomas. The linear oblique craniectomy minimizes the risk of this by allowing more tactile feedback due to the oblique nature, avoiding the significant complication of bridging vein rupture, with no bridging veins damaged (Figure 5(a)). This is not possible with the letter-box technique. However, our technique did have an unintentional durotomy in one-third of trials due to the craniotome footplate. Furthermore, the angle of the linear oblique craniectomy reduces friction and force placed upon the cortex by implantation of the subdural grid compared to perpendicular approach. 9
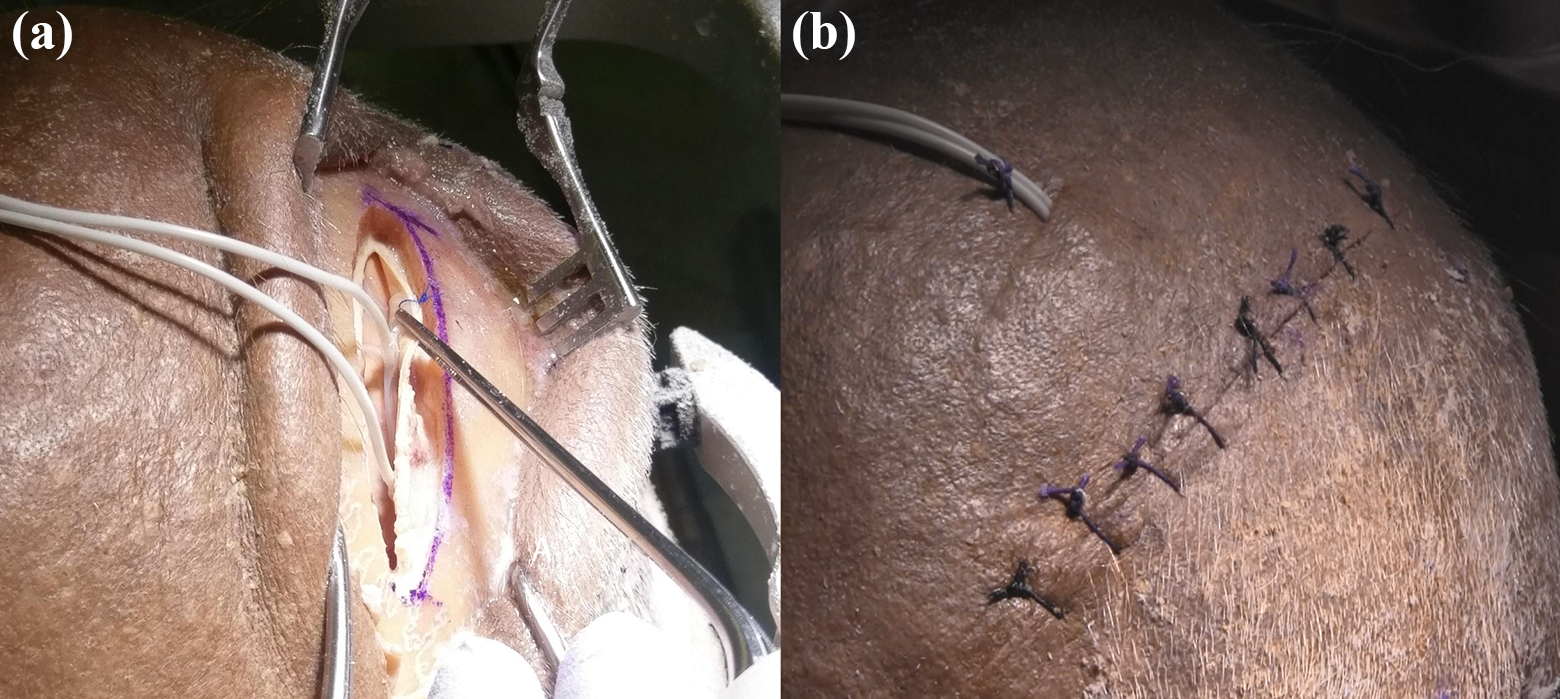
Completed linear oblique craniectomy. (a) Oblique view of the linear oblique craniectomy with the subdural grid implanted. (b) Closed linear oblique craniectomy with leads tunneled.
The second comparison is of the letter-box paper’s proposed burr angling as compared to craniotome angling. The authors of the letter-box technique suggested burring the craniectomy to create an oblique approach. Burring the lateral edge of the skull would require sliding the burr under the calvaria beside the dura, increasing risk of dural damage and difficulty. Our procedure achieves the angle with relative ease, although unintentional durotomy occurred. Further, burring the craniectomy will spread bone dust under the calvaria. Excess intracranial bone dust will increase the risk of postoperative aseptic meningitis. This is not a concern for the craniotome approach. Finally, burring bone creates a permanent craniectomy, whereas our craniotome approach allows replacement of the bone, ideally leaving only a burr hole behind.
The third comparison is the duration of procedure. The lens-shaped linear oblique craniectomy requires two craniotome passes. The letter-box requires four craniotomy passes with associated angled burring of the width of the calvaria along the perimeter of the craniectomy. The premade angulation also eases expedited removal of the grid in case of swelling or bleeding by opening of the sutures and placing traction on the grid (Figure 5(b)).
Future improvements upon the linear oblique craniectomy
While our oblique angle was moderate at 30–50°, decreasing this angle will increase the ability to visualize the subdural space and improve ease of subdural grid introduction. Depending upon the proximity to apex of curvature such as the superior temporal line, the angle of incision may be altered to increase the parallel nature of subdural grid introduction. It is notable that during three introductions, the dura was damaged once by the craniotome footplate, which is of concern. This may be avoided by slanting the craniotome footplate or by removing the craniotome footplate entirely during craniectomy with a Penfield held under the inner table to prevent plunging.
Utility of the linear oblique craniectomy
This procedure will be best utilized when the cortical region of interest is known as the oblique nature restricting implantation to one direction. 5,9 However, use away from an apex is possible. Importantly, this minimally invasive procedure may have aesthetic benefits along with associated reduction in craniotomy complications. 9 This procedure is a candidate for improved subdural grid insertion in centers without access to depth electrodes. Centers in North America that elect for concurrent utilization of subdural grids and depth electrodes may utilize this procedure. 10
Conclusion
We present here an improved minimally invasive method of introducing subdural grids to overlay the Sylvian fissure. The linear oblique craniectomy improves visualization, increases ease of introduction, decreases pressure on the cerebral cortex during instrumentation, and improves ability to sense bridging veins via tactile feedback. However, given that this is a cadaveric study technical note, clinical validation with extensive safety analysis is required before general utilization.
Footnotes
Acknowledgments
Authors thank Dr Walter Hader and Dr Stefan Lang for providing mentorship and surgical education; Yves Pauchard and Stephanie Kwong and the MoJo Center for Mobility and Joint Health at the University of Calgary for providing the CT scans and images; and Dr Bradley Goodyear and Paul Romo and the Seaman Family MR Research Centre for providing the MRI scans and images.
Author contributions
Calvin W Howard and Nader S Aboelnazar contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
