Abstract
A 90-year-old woman developed a severe stroke (National Institutes of Health Stroke Scale Score (NIHSS) 24) due to an occlusion of the left carotid T. Computed tomography 60 min after symptom onset showed a large area of tissue at risk with a relatively large area of reduced cerebral blood volume indicating a relative large infarct core. After successful reperfusion, she recovered very well with only residual facial asymmetry (NIHSS 1). Up to now, therapeutic decisions for intravenous thrombolysis and endovascular treatment were mainly based on these time windows, although it was well known that the individual time window may vary widely due to a large variability of the quality of collateral circulation. The recently presented results of the DAWN trial have demonstrated that patients can be successfully treated in a time window up to 24 h when selected by imaging and clinical criteria. The described case impressively demonstrates that this positive selection of patients beyond established treatment time windows does not implicate in reverse that patient selection for therapy within the established time windows can be based on the same imaging criteria. In conclusion, patient selection beyond the established time windows may be based on the available techniques, but imaging-based decisions against therapy in patients within the established time windows should be made only with extreme caution.
Case report
A 90-year-old woman living at an old people’s home suddenly developed a severe right-sided hemiparesis and difficulty to speak. She was immediately transferred to the emergency department of the University Hospital of Bern and examined by the neurologist on duty 50 min after symptom onset. The patient was hemodynamically stable with a blood pressure of 185/55 mmHg at admission. Clinically she presented with the National Institutes of Health Stroke Scale Score (NIHSS) of 24/42 with a severe paresis on her right side, global aphasia, a hemispatial neglect, and sensory deficits.
Naive computed tomography (CT) 60 min after symptom onset excluded any bleeding or early infarct signs (Figure 1). CT angiography demonstrated a left carotid T occlusion with occlusion of the distal internal carotid artery and the proximal segments of the middle and anterior cerebral artery (Figure 2). Time to maximum (T max)perfusion imaging maps showed a large area of severe hypoperfusion mainly within the territory of the middle cerebral artery (Figure 1). A larger proportion of the hypoperfused brain tissue showed reduced cerebral blood volume (CBV) indices as indicated on the CBV maps (Figure 1). The interdisciplinary stroke team decided to perform a direct mechanical endovascular treatment (EVT), because the patient was treated with Rivaroxaban for atrial fibrillation and factor Xa activity was 184 ng/ml (specific factor Xa activity for Rivaroxaban). Reperfusion was then delayed due to an iatrogenic dissection of the femoral artery and could finally be successfully achieved only 191 min after symptom onset (Thrombolysis in cerebral infarction (TICI) 3 reperfusion) with a Solitaire Stent Retriever (Medtronic, USA). The patient was transferred to the stroke unit and clinical examination on the following day revealed only a residual slight facial asymmetry (NIHSS 1/42). A control CT 40 h after reperfusion showed only very small areas of infarction (Figure 3). She developed a hemodynamically stable non-ST-segment elevation myocardial infarction during the first 24 h which was treated conservatively. After 3 days, she could be transferred to a geriatric rehabilitation unit in a good general condition.

T max CT perfusion maps showing a large area of severe hypoperfusion mainly within the territory of the middle cerebral artery. A greater proportion of the hypoperfused brain tissue showed reduced CBV indices as indicated on the CBV maps. CBV: cerebral blood volume; CT: computed tomography; T max: time to maximum.
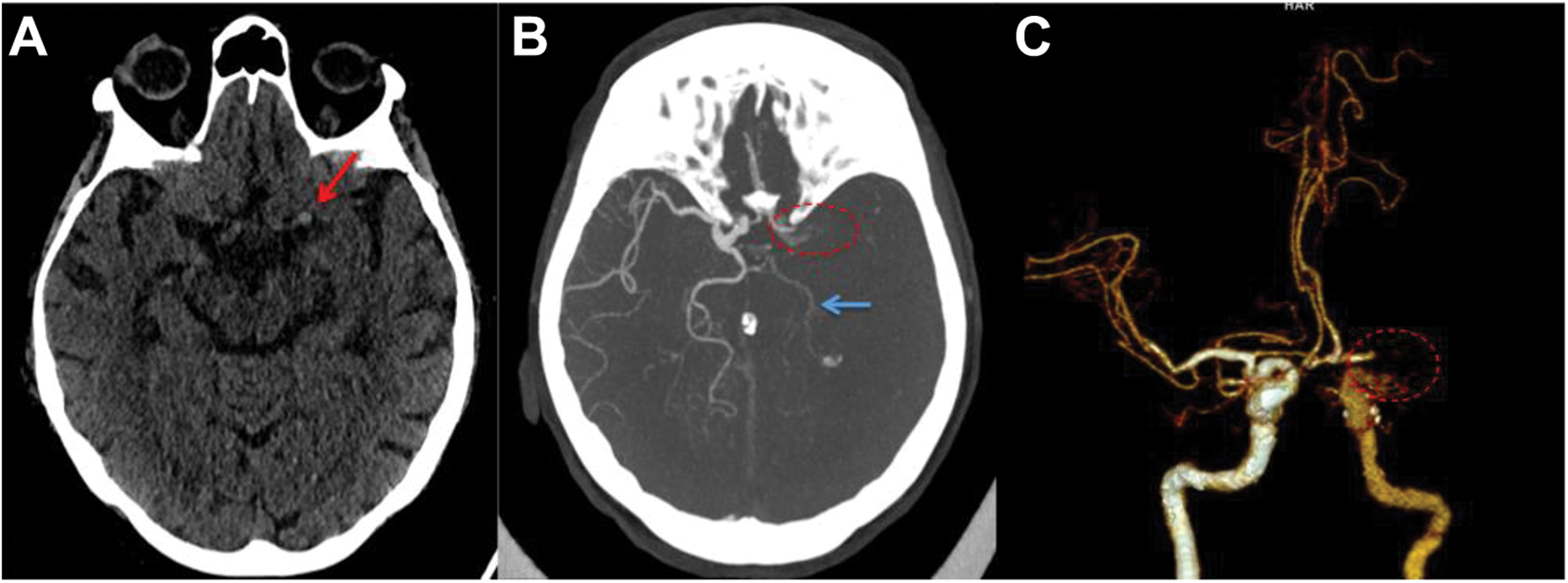
Axial non-enhanced CT at admission (a) showing hyperdense artery sign (red arrow) of the left distal ICA. On the axial maximum intensity projection CT angiography (b) the distal ICA, the middle cerebral artery, as well as the lateral part of the A1 segment of the anterior cerebral artery are not opacified, representing carotid T occlusion (dotted circle), which is also demonstrated in the anterior view of the volume rendered 3-D CT angiography (c). In this early arterial phase, the left side ICA proximal to the occlusion site is less opacified than the right side. There is a reduced opacification of the left posterior cerebral artery (blue arrow) as it is supplied by the anterior circulation (fetal origin of the posterior cerebral artery (PCA)). There is also a retrograde filling of the medial part of the A1 segment anterior cerebral artery (ACA) via Acom. CT: computed tomography; ICA: internal carotid artery.
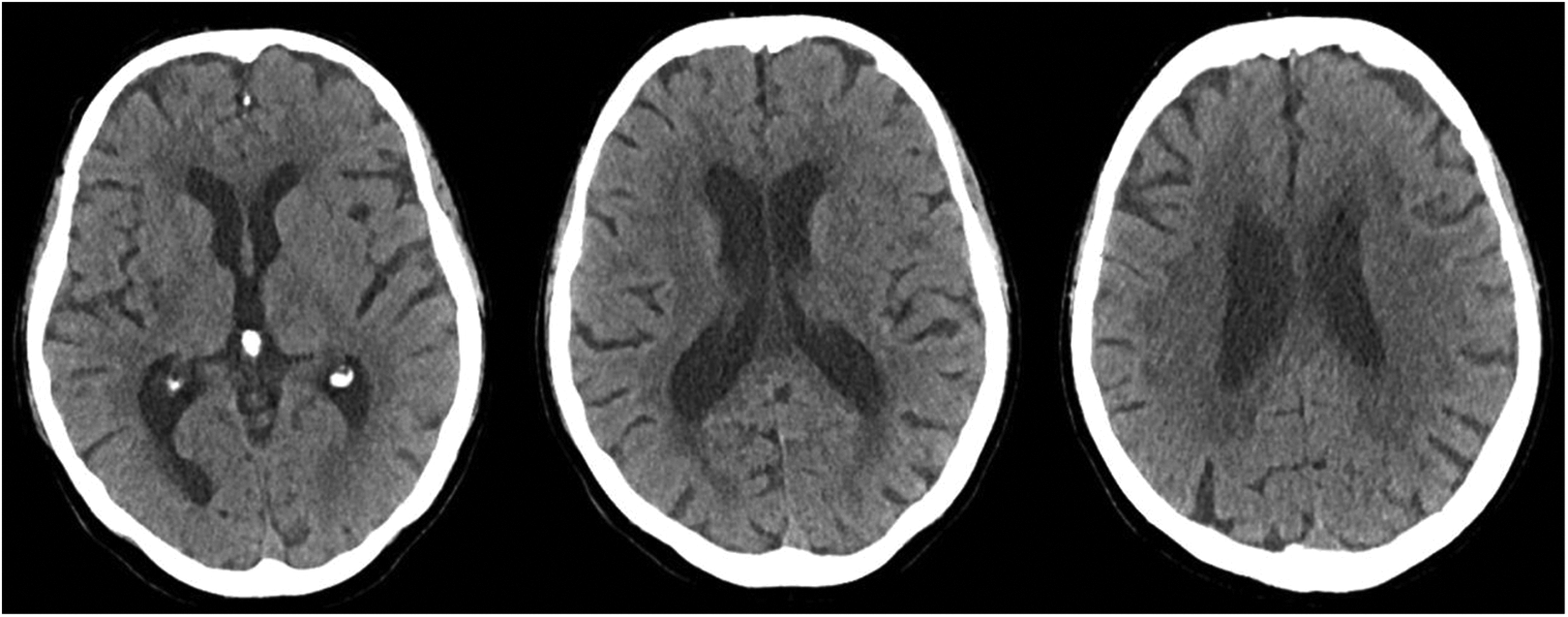
Control CT 40 h after reperfusion therapy showing only small hypodense areas of infarction. CT: computed tomography.
Discussion
Reperfusion therapy by intravenous thrombolysis (IVT) and EVT is highly effective and only 5–14 patients had to be treated with IVT and 3–7 patients with EVT in the large randomized trials to prevent one patient from death or dependency. 1 –9 Therapeutic success relies on the already established infarct core and the size and localization of the salvageable penumbral tissue. Because the size of the penumbra continuously declines with the elapsed time, the therapeutic effect is highly time-dependent and treatment time windows of 4.5 h for IVT and 7.3 h for EVT have been defined by pooled analysis of the patients treated in the large trials. 10,11 Up to now, therapeutic decisions for IVT and EVT were mainly based on these time windows, although it was well known that the individual time window may vary widely due to a large variability of the quality of collateral circulation. The recently published results of the DAWN trial have demonstrated that patients can be successfully treated in a time window up to 24 h when selected by imaging and clinical criteria. 12 The DAWN trial included patients with large artery occlusion between 6 h and 24 h after symptom onset for EVT, if patients presented with a so-called clinical-image mismatch: Patients had to be clinically severely affected (NIHSS ≥ 10) but infarct core had to be restricted (<21 cc if patients were ≥80 years, <31 cc if patients were <80 years, and NIHSS ≥ 10 or <51 cc if patients were <80 years and NIHSS ≥ 20). The trial revealed a clear effect of reperfusion therapy in these selected patients: The number needed to treat was reported to be 2.8 for 90-day functional independency. The results of this trial will probably lead to a paradigm change in therapeutic decisions in acute stroke treatment. Future patient selection will be based more and more on imaging criteria. The imaging-based selection of patients beyond the established treatment time windows may offer a treatment option for patients which had been excluded from therapy so far.
Our above-described case impressively demonstrates that this positive selection of patients beyond established treatment time windows does not implicate in reverse that patient selection for therapy within the established time windows can be based on the same imaging criteria.
The decision for therapy in our patient was on the clinical evaluation, the time window, and native CT not showing any infarct signs. CT perfusion showed already large areas of reduced CBV within the hypoperfused tissue (Figure 1). Because areas of reduced CBV approximate the infarct core, one may have decided against reperfusion therapy in this elderly patient. The rapid clinical recovery after reperfusion and the small areas of infarction on follow-up imaging demonstrate the possible inaccuracy of the imaging-based approximation of the penumbra.
The problem still to be resolved for an optimized patient selection is the accurate imaging of the core and penumbra: Although positron emission tomography (PET) imaging was able to accurately visualize the infarct core and the hypoperfused tissue, it is not practicable in the acute setting of stroke. 13 Magnetic resonance (MR) and CT allow the approximation of the penumbra but show a relevant inaccuracy of the imaging techniques used up to now. 14–16 The core is estimated using MR by the lesion in diffusion-weighted imaging (DWI) and the hypoperfused area by perfusion imaging. With CT, the core is visualized by CBV and the hypoperfused area by perfusion imaging. Visualization of the infarct core by MR imaging is more accurate than by CT, but diffusion lesion reversal can occur because the DWI lesion may overestimate the infarct core and contain up to 25% false positive, that is, surviving tissue. 17 More accurate is the definition of the core by quantitative analysis of the calculated apparent diffusion coefficient maps. 17 Imagination of the hypoperfused tissue at risk by MR and CT perfusion imaging relies on surrogates of perfusion parameters calculated from nondeconvolved (e.g. time to peak) or deconvolved (e.g. CBF, mean transit time, and T max) tissue residue function (time contrast curve) of the intravascular contrast bolus. 15 Different maps computed from MR and CT imaging have been developed and compared to PET measurements. Recent studies indicate that T max thresholds of >6 s improve the prediction of salvageable tissue compared to previous maps. 18 Nevertheless, estimation of the tissue at risk based on maps derived from simple thresholding is prone to errors in about 25% of the patients with variations of the predicted penumbra of up to 100 ml. 19 Newer studies applying machine learning techniques instead of only using somehow arbitrary thresholds of surrogate parameters have shown first promising results with improved accuracy of the penumbra imaging. 20 Nevertheless, the accuracy of the CT or MR imaging-based visualization of the infarct core and penumbra still has to be improved for an optimized patient selection. Until then, patient selection beyond the established time windows may be based on the available techniques, but imaging-based decisions against therapy in patients within the established time windows should be made only with extreme caution.
In addition, this case shows though patients aged 90 years or older despite being at risk for a poorer clinical outcome compared to younger patients may still benefit from EVT especially if they had a good pre-stroke functional status.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SJ received Scientific advisory board honoraria from Bayer, Boehringer Ingelheim, Pfizer, Daichy Sankyo; and research grants from the Swiss Heart Foundation and the Swiss National Science Foundation. MA received Speaker honoraria from Bayer, Boehringer Ingelheim, and Covidien; scientific advisory board honoraria from Amgen, Bayer, Boehringer Ingelheim, BMS, Pfizer, Covidien, Daichy Sankyo and Nestlé Health Science; and research grants from the Swiss Heart Foundation and the Swiss National Science Foundation.
