Abstract
Aquaporins (AQPs) are water channel molecules that allow the passage of water through the lipid bilayers of cell membranes. AQP1, AQP4 and AQP9 are expressed in the central nervous system and AQP4 is well known as the target of auto-antibodies in Devic’s neuromyelitis optica. The role of AQPs in facilitating water movements suggests a link with oedema formation and resolution in the brain. Furthermore, AQPs are also involved in process formation in glial cells with a close link to neuroinflammation. This mini-review gives an overview of what is currently known about the role of AQPs in different neurological disorders.
Introduction
Aquaporins came to the attention of neurologists mainly due to Devic’s syndrome, an autoimmune condition associated with anti-AQP4 antibodies found in serum. Aquaporins, as indicated by their name, are water channel molecules that allow water to cross the hydrophobic lipid bilayers of cellular membranes. AQPs were first described by Peter Agre, an American physician and molecular biologist, while studying red blood cell membranes and the Rhesus group. He discovered a red cell membrane protein, CHIP28, with homologies to proteins expressed in various tissues such as renal tubules, fruit fly brains and plant roots. 1 He went on to show this protein was a water channel, now termed Aquaporin-1 (AQP1), providing an explanation for the rapid water movement through hydrophobic red cell membranes. 2 This led to the discovery of the AQP family of water channel proteins. Peter Agre was awarded the Nobel Prize in Chemistry in 2003, which he shared with Roderick MacKinnon, for their ‘discoveries concerning structure and function of channels in cell membranes’. The finding that AQPs were expressed in the brain prompted research into possible links between AQPs and neurological conditions associated with disturbed water movements such as cerebral oedema, with the hope of using AQPs as therapeutic targets to modulate cerebral water exchange.
Aquaporins
AQPs are proteins of approximately 30kD located in cell membranes. They contain six transmembrane domains, two intracellular loops and three extracellular loops that form a channel that allows passive water movement following osmotic or oncotic gradients between the extra- and intracellular spaces. The AQP family shares a common consensus motif, asparagine-proline-alanine (NPA), which contributes to the specificity of the pore. Most AQPs form tetramers, although each AQP molecule constitutes an individual channel by itself. Twelve AQPs have been identified so far. The majority are permeable to only water but some are also permeable to small solutes such as glycerol or lactate (aquaglyceroporins). Currently three main AQPs have been observed and studied in the brain: AQP1, AQP4 and AQP9.
AQP1, permeable to only water, has been observed in epithelial cells of the choroid plexus at the site of production of the cerebrospinal fluid (CSF). Interestingly, although endothelial cells commonly express AQP1, cerebral endothelial cells do not and it appears that astrocytes are responsible for the downregulation of AQP1 in the central nervous system (CNS). In cultures of brain endothelial cells, cells regain the expression AQP1 progressively after a few passages and this expression is downregulated in co-cultures of brain endothelial cells with astrocytes. 3 This indicates that an interaction between elements of the neurovascular unit contributes to the phenotype of endothelial cells in the blood–brain barrier (BBB). 3 AQP1 was also observed in primary sensory neurons, such as those in the dorsal root ganglion, and is proposed to be involved in pain perception. 4,5
AQP4, another water-only channel, is the most abundant AQP in the brain. It is expressed mostly in astrocytes and ependymal cells. In astrocytes, AQP4 localizes in the end feet in contact with microvessels and synapses (Figure 1). Astrocyte processes and their end feet surround the brain microvessels and also constitute the glia limitans, a glial layer at the outer boundary of the CNS. The strategic location of AQP4 on astrocytic end feet covering the microvasculature positions it at the site of water exchange in the BBB. On the other hand, astrocyte end feet of the glia limitans bordering the outer surface of the brain are in contact with the pia mater and in the vicinity of the CSF in the subarachnoid space (Figure 1). Therefore, expression of AQP4 in both ependymal cells lining the ventricles and astrocytes of the glia limitans places it at the interface of the CSF and brain parenchyma, at the sites of water movement between the CSF and the brain. Recently, AQP4 found on astrocyte end feet, which are in contact with cerebral blood vessels, has been linked with CSF and solute movements in the perivascular space 6 and to the clearance of metabolites from the brain tissue such as ß-amyloid. 7 It has been proposed that CSF enters the perivascular space of brain tissue around penetrating arterioles and exchanges with insterstitial fluid. Insterstitial fluid and solutes leave the brain in the perivascular spaces of venules and veins. This phenomenon is facilitated by astroglial water movements through AQP4. 6 By analogy to the lymphatic system found outside the brain, the perivascular space system was named the glymphatic system. Recently, the glymphatic system was proposed to contribute to lactate concentration changes in the brain, in correlation with sleep–wake cycles and sleep homeostasis. 8 AQP4 is also associated with cell movement, the size of the extracellular space and connectivity between neurons.
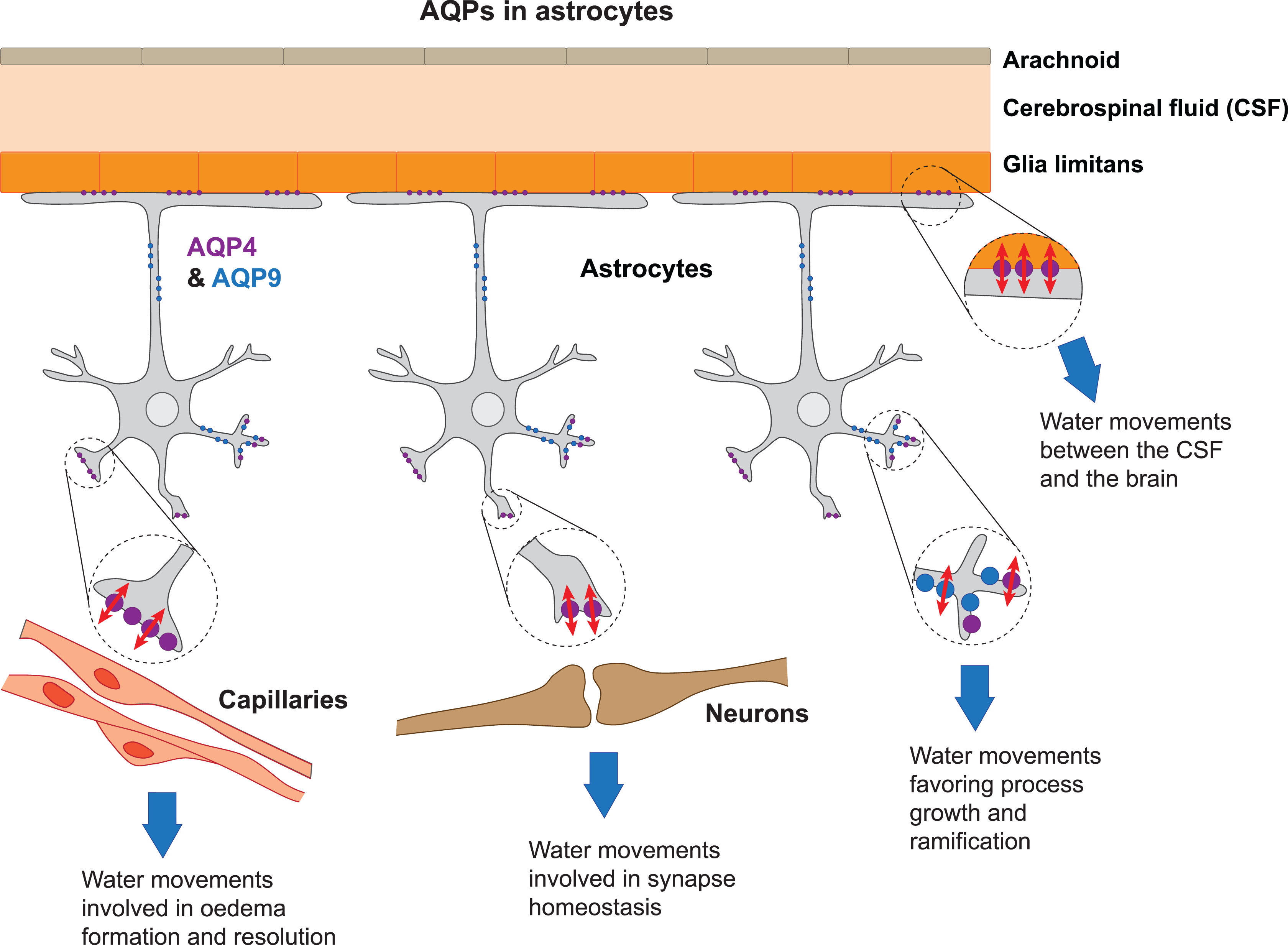
The most abundant AQP expressed in the brain (in astrocytes) is AQP4 (purple). AQP4 is expressed mainly on astrocytic endfeet. Astrocytes extend their endfeet to the glia limitans (top) where AQP4 is involved in water movements between the CSF and the brain, to capillaries (left), where it plays a role in water movements in oedema formation and resolution and to synapses (center), where it is involved in synapse homeostasis. AQP9 (blue) in the brain is expressed in the cytoplasmic membrane and mitochondria (not illustrated) of astrocytes. Both AQP4 and 9 play a role in water movements involved in process formation and elongation (right).
AQP9 is an aquaglyceroporin that is permeable to both water and small solutes such as glycerol, monocarboxylate and urea. It is present as two isoforms of 30 and 26 kD. The 30 kD isoform is expressed in the cytoplasmic membrane and on the 26 kD isoform on the inner mitochondrial membrane. AQP9 was shown to play an important role in cell morphology in leucocytes, allowing the outgrowth of processes, 9 while the presence of AQP9 in the liver is associated with energy balance. 10 In the brain, AQP9 is preferentially expressed in catecholaminergic neurons, for example, in tanycytes of the medio-basal hypothalamus. 11,12 It is also expressed in astrocytes (Figure 1) and our groups have shown that AQP9 expression is induced in astrocytes 13 after cerebral ischemia. However, currently the expression of AQP9 in the CNS is debated as other authors have described expression in hepatocytes, epididymis, vas deferens and epidermis but not in brain. 14,15 Discrepancies between observations have been attributed to differences between species, different splice variants or antibody specificity. 14 Also, AQP9 mRNA has been detected in the brains of both rodents and non-human primates, arguing in favour of AQP9 expression in the CNS. 11,16 To date, the role of AQP9 in brain is thought to be associated with brain energy metabolism 17 and astrogliosis after stroke. 18
Involvement of AQPs in specific neurological disorders
Role of AQPs in oedema formation and resolution
After the description of the location and expression of AQP4 in the brain, 19 various studies focused on its potential role in the oedema process. The field benefited from the generation of AQP4 knockout (AQP4−/−) mice by Verkman and colleagues, who showed the importance of AQP4 in early oedema formation after water intoxication and in experimental stroke. 20 So far, the available data strongly suggest that AQP4 promotes the formation of cytotoxic oedema by facilitating the entry of water into astrocytes. However, the absence of AQP4 decreases the water clearance after infusion of saline solution into the brain parenchyma leading to a significantly higher increase in intracranial pressure. 21 So far, most immunohistological studies showed that changes in AQP4 expression seem to parallel changes in the oedema process (for a detailed review, see Badaut et al. 22 ). However, the pattern of AQP4 expression changes differs between brain disorders. Most of the work on AQP4 and oedema has been carried out in stroke models and we discuss this in more detail below.
Devic’s disease
Neuromyelitis optica (NMO) or Devic’s disease is an autoimmune demyelinating disease of the CNS affecting mostly the optic nerve and spinal cord (for a detailed review, see Yang et al.
23
). It is rare in the European population (1:100,000/year) but more frequent in West Indian and Asian populations where it represents up to 50% of demyelinating CNS disorders. A significant number of Devic’s patients, although not all, have antibodies against AQP4 (AQP4-Ab or NMO-IgG). Initially questioned, the pathogenic role of AQP4-Ab is becoming increasingly clear as it has been shown in mice that intracerebral injection of AQP4-Ab isolated from NMO patients, co-injected with human complement, leads to inflammation and demyelination reproducing the features of NMO.
24,25
AQP4-Ab appear to contribute to the disturbance in the immunoregulatory function and barrier capacity of astrocytes. The term
Cerebral ischemia/stroke
Ischemic stroke can be a devastating disease and has high incidence, leaving many patients with a handicap. Cerebral oedema is an important aspect of ischemia-induced cerebral tissue alterations. The cytotoxic oedema, demonstrated by a reduced apparent diffusion coefficient (ADC) in the early hours after an ischemic insult, is a hallmark of ischemic stroke in magnetic resonance imaging. A second later phase of oedema, termed vasogenic oedema, reaches a peak between 24 h and 72 h after ischemic stroke onset. The swelling of ischemic brain tissue can cause compression and damage of other structures such as the brain stem, with further neurological worsening or even death. This phenomenon, termed mass effect, is a feared complication of ischemic stroke. Possible roles of AQP4 in stroke outcome have been investigated in AQP4-deficient mice. Mice lacking AQP4 develop less pronounced cerebral oedema after stroke in a mouse model of severe permanent cerebral ischemia. 20 A more recent study from our labs, using the milder transient cerebral ischemia stroke model in AQP4 knockout mice, showed decreased mortality and increased motor recovery up to 14 days after stroke. In this model, behavioural improvement was associated with a smaller lesion volume and decreased neuronal cell death and neuroinflammation, without a direct effect on oedema formation. 27 In wild-type (WT) mice, we observed an early increase (1 h after ischemia onset) in AQP4 expression on astrocytic end feet lining the capillaries of the lesion border, coinciding with an early phase of hemispheric swelling. 28 Using a preconditioning paradigm of intracerebroventricular thrombin injection 24 h before ischemia to trigger endogenous neuroprotection mechanisms, 29,30 we demonstrated increased tolerance to ischemia and reduced hemispheric swelling coinciding with a further enhancement of perivascular AQP4 expression at 1 h after ischemia. 29,31 This observation suggests that the early AQP4 upregulation on astrocytic end feet lining capillaries is part of an endogenous protective mechanism to attenuate cerebral oedema. These different observations in distinct models illustrate the particular functions of astrocytes as part of the BBB and in regulation of inflammatory processes.
AQP9 expression is induced in astrocytes after cerebral ischemia around the lesion, but also at a distance from the ischemia in the cell bodies and on processes, but not on perivascular end feet. 13 Using the thrombin preconditioning paradigm, we observed that thrombin addition before stroke further enhanced the AQP9 expression observed in stroke with associated morphological changes suggesting AQP9 expression may have a role in astrocyte activation and the astroglial response to ischemic brain injury. Importantly in this respect, we recently showed that AQP9 overexpression influences astrocyte morphology in cultures of astrocytes, with an increase in cellular process complexity, 18 as was shown earlier in leucocytes. Another hypothesis for increased expression of AQP9 after stroke is that AQP9 could facilitate clearance of the accumulated lactate and glycerol from the extracellular space following the stroke. 18
Glioblastoma
Expression of AQP1 and AQP4 is also reported in human samples from glial cell tumours. 32,33 The role of AQPs is at present unclear in malignant glial cells. An increase in AQP9 mRNA and protein expression was measured in human astrocytic tumours, reaching higher values in high-grade tumours compared to normal brain tissue. 34,35 As AQPs are involved in filopodia formation by facilitating water entry into the cell process, it has been proposed that the presence of AQPs on tumour cells could be linked to cell migration and invasion of the surrounding nervous tissue by tumour cells. Furthermore, the presence of AQP1 promotes tumour metastasis. 25,36
Traumatic brain injury
In traumatic brain injury (TBI), AQP4 plays a role in astrogliosis after the injury. Adult AQP4−/− mice developed a stronger microglial reaction and attenuated astrogliosis compared to WT mice subjected to TBI. One month after injury, the AQP4−/− mice were more vulnerable to epileptic seizures than WT mice. 37 Similar results were obtained when juvenile mice were subjected to TBI (jTBI). 38 AQP4 knock-down using siRNA resulted in attenuated astrogliosis and enhanced microglial activation 3 days post-injury in jTBI. This suggests that the astroglial reaction plays a key role in regulating microglia and underscores the role of AQP4 in neuroinflammation (for a detailed review, see Badaut et al. 22 ).
Hydrocephalus
Hydrocephalus presents as dilated ventricles. While obstructive hydrocephalus is due to a compression of the CSF flow path, for example, at the level of the aqueduct of Sylvius, communicating hydrocephalus results from an imbalance in CSF production and elimination. AQP1 and AQP4 are expressed at critical locations for CSF production and drainage. Interestingly, the expression of AQP4 in brain samples from patients suffering from hydrocephalus is increased and less polarized to astrocytic end feet than in controls. 39 The role of AQP1 and AQP4 in hydrocephalus pathogenesis has been investigated using mutant mice. AQP1 is expressed on the epithelium of the choroid plexus at the site of CSF production. In AQP1−/− mice, the intracranial pressure is lower than in WT mice and the production of CSF is reduced by 25%. A mouse hydrocephalus model induced by Kaolin injection into the cisterna magna that leads to an obstruction of the outflow from the fourth ventricle was tested in AQP4−/− mice: Mutant mice had a higher intracranial pressure and more dilated ventricles than WT mice. 40 This can be explained by a reduced drainage of CSF in the absence of AQP4.
Epilepsy
Astrocytes express AQPs and extend their processes to both capillaries and synapses. They play an important role in neuronal metabolism and modulation of neuronal excitability and synaptic transmission. In mice deficient in AQP4, the threshold for seizures is reduced while the seizure duration is extended, showing a link between AQPs and epilepsy (for a detailed review, see Binder et al. 41 ). The underlying mechanism may be an impairment of K+ homeostasis in the absence of AQP4. 42 Furthermore, in hippocampal atrophy, a feature of mesio-temporal lobe epilepsy, AQP4 expression is increased in samples from atrophic hippocampi from epileptic patients. 43
Neurodegenerative diseases
Presence in blood of antibodies against AQP4 has been identified as a potential biomarker in Devic’s disease as discussed above. The role of the AQPs in multiple sclerosis (MS) has not been studied extensively so far. In a model of MS, experimental autoimmune encephalomyelitis (EAE), an upregulation of AQP4 was observed in correlation with BBB disruption characterized by a decrease in tight junction proteins such as occludin. 44 Accordingly, the absence of AQP4−/− is associated with a less severe clinical deficit score and tissue inflammation after EAE and lipopolysaccharide injection compared to WT animals, suggesting a deleterious role of AQP4 in MS pathophysiology. 45 The benefits due to AQP4 absence could be linked to a decreased production of the proinflammatory cytokines, tumour necrosis factor-alpha and interleukin (IL)-6 as seen in AQP4−/− compared to astrocyte cultures from WT mice. 46
In Alzheimer’s disease (AD), the levels of AQP1 and AQP4 are altered, with an increased number of AQP1 expressing astrocytes in the cortex in AD brains. Interestingly, AQP1 expression was not high in the vicinity of senile plaques. 47 The level of expression of AQP1 differs depending on the intensity of the Aβ immunoreactivity, with high expression of AQP1 in astrocytes close to low Aβ immunoreactivity and the absence of AQP1 in the astrocytes adjacent to high Aβ immunoreactivity. On the other hand, AQP4 expression in astrocytes is increased in association with Aβ in patients in the vicinity of senile plaques. 47 The role of these changes is still debated. In rodent models of AD, vascular alterations have been observed with loss of AQP4 expression in astrocyte end feet. 48 Changes in AQP4 expression in astrocyte end feet could affect the clearance of Aβ from the perivascular space. 7 It is important to point out that the changes in AQP4 in rodent preclinical models do not seem to reproduce the alterations observed in AD patients.
In inflammation, AQP4 has been related to the production of cluster of differenciation (CD)4+ and CD25+ regulatory T cells. 46 The absence of AQP4 possibly interrupts the immunosuppressive regulators in Parkinson’s disease, leading to increased microglial activation and a worse outcome due to enhanced loss of dopaminergic neurons after 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine administration. 46 In summary, it is likely that AQPs contribute to various pathophysiological mechanisms leading to neurodegeneration.
Molecular mechanisms for AQPs in mediating disease pathology
AQPs are water channels and their presence facilitates water movements through cell membranes. Thereby, AQPs influence oedema development and resolution as well as volume and shape changes of cells. In particular, they are involved in cell process formation such as is the case for AQP9 in astrocytes involved in astrogliosis, which may be both beneficial and deleterious. Astrocytic swelling favoured by AQP4 was shown to lead to Ca++ signalling via the release of ATP and activation of purinergic receptors. 49 Disturbed AQP4 expression could influence the glymphatic system and clearance of potentially toxic solutes such as Aβ from the perivascular space. 7
As indicated above, many of the brain disorders are associated with a neuroinflammatory process, which encompasses activation of astrocytes as well as secretion of cytokines and chemokines. AQP4 is commonly upregulated in neuroinflammation. 38 Indeed, IL-1 is increased in several brain disorders and has been shown to induce AQP4 expression in a concentration-dependent manner. 50,51 Regarding the mechanism of action, IL-1β has been shown to upregulate AQP4 at the transcriptional level: Downstream of the IL-1 receptor, NFĸB is translocated to the nucleus and binds the AQP4 gene promoter. 50,52 Similarly, the high mobility group box protein-1 (HMGB1) released by necrotic neurons induces a neuroinflammatory response together with an increase in AQP4 possibly triggered, at least in part by the pro-inflammatory cytokine IL-6. 53 This, in turn, could lead to astrocyte volume changes and Ca++ signalling. 49 It is interesting to outline the bidirectional role on cytokine regulation. Reduced production of the proinflammatory cytokines, tumour necrosis factor-alpha and IL-6 was observed in AQP4−/− compared to WT mice astrocyte cultures, whereas, in these same cultures, adenovirus-mediated increases in the expression of AQP4 or AQP1 lead to increased release of the same cytokines. 46 Additionally, it has been shown that water transport mediated by AQP1 induces IL-1 release from macrophages, 54 although this phenomenon has not been studied in the CNS.
Activation of the protein kinase C pathway in astrocyte cultures induced a decrease in AQP4 and AQP9 mRNA and protein. 55 V1a receptor stimulation is known to induce activation of protein kinase C, and application of vasopressin on cortical slices induced changes in water flux through the astrocytic syncytium. In addition to protein kinase C, inhibition of one of the Mitogen activated protein kinases (MAP-kinases), JNK, in a subarachnoid haemorrhage model has been associated with increased AQP4 expression and decreased oedema formation. In summary, the regulation of AQP expression is tightly linked with neuroinflammatory mechanisms with bidirectional flow: With, for example, inflammatory signals promoting the expression of AQP4, AQP4 facilitates astrocytic activation but seems to limit microglial activation.
Conclusions
The study of AQPs in the brain reveals new and unexpected roles for these channels in CNS cell physiology and physiopathology (Figure 1). Apart from Devic’s disease, evidence linking AQPs to the pathogenesis of specific neurological disorders is weak. There are been multiple reports and very heterogeneous findings and effects that depend on the studied pathologies and models used. It should be pointed out that the timing of observations after injury is critical. AQPs are passive channels allowing the flow of water (and in some cases small solutes) through lipophilic membranes and the roles of such channels may be diagonally opposed at different time points in development and resolution of a neurological disorder. Importantly, there is still a lack of selective pharmacological compounds to study the effect of specific blockage of AQPs to allow the analysis of their physiological and pathological roles. Many studies are based on transgenic mice, where developmental adjustments and redundancy may mask the true role of the specific endogenous AQP. Knock-down strategies, which have the advantage of inducing an acute change in gene expression, are however providing assessment of AQP function. As water and solute movements are part of lesion development and resolution in many neurological disorders, a fine characterization and better understanding of these phenomena could allow targeted interventions to attenuate lesion progression and enhance recovery after injury.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research grants from the Swiss Science Foundation (FNS 31003A_163465), the Biaggi Foundation and the Juchum Foundation (LH); eraNET neuron CNSaflame, eraNET neuron TRAINS and, ANR-TRAIL (JB).
