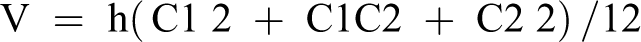Abstract
Immediate lymphatic reconstruction (ILR), performed concurrently with nodal dissection, has shown promise in reducing the rates of lymphedema in patients with various types of cancers. Herein, we detail the case of a 42-year-old patient who underwent nodal dissection in the management of their melanoma. This patient underwent ILR at the time of lymph node dissection of the right axilla and was followed for 24 months. Circumferential measurements of both the operative and non-operative limbs, as well as lymphedema-specific quality of life questionnaire (LYMQOL) data, were collected at each appointment. Our patient developed lymphedema transiently at 3 months which had resolved by the 6-month follow-up and maintained favorable measures of quality of life over the course of 2 years. This novel approach has yet to be implemented as a standard of care in Canada. Such an outcome would be overwhelmingly positive for our cancer population, and on our health-care system overall.
Keywords
Background
According to the Canadian Cancer Statistic Advisory Committee, melanoma is one of the fastest rising cancers in Canada, accounting for 3.8% and 3.3% of new cancer cases in men and women, respectively. Melanoma cases also represent a significant proportion of cancer-related mortality, implicated in 2.0% of all cancer-related deaths in men, and 1.1% in women. In 2020 alone, approximately 8,000 Canadians will have received a diagnosis of melanoma, with 1,300 associated deaths projected over the calendar year. 1
Although the majority of melanoma cases are amenable to surgical management via wide local excision (WLE) alone with or without sentinel lymph node biopsy (SLNB), approximately 20% of patients will present with advanced disease, and thus require lymph node dissection to manage pain symptoms, improve quality of life and provide significant disease control with a small chance of even providing a cure in 25% of cases. Despite advancements in surgical techniques, the incidence of lymphedema in this population is markedly elevated as compared to the rates observed in the general populous, ranging from 5-10% in cases requiring upper extremity dissection, and 28- 40% in cases with analogous lower extremity involvement. Consequently, many such patients are left with chronic and debilitating swelling. 2 -4
Presently, the state of consensus regarding the management of lymphedema remains nebulous, as the current body of evidence detailing treatment modalities is limited, with no definitive therapy described in the literature. Moreover, existing strategies, including conservative management, complex decongestive therapy, use of compression garments, and excisional interventions have all been shown to be ineffective, time-consuming or expensive.5 Retroactive surgical interventions such as node transfers and excisional therapy amongst others, have been described as effective alternatives to non-surgical management; however, these procedures are not curative, and some have been shown to contribute further to morbidity or disfigurement in some patients. 5 -9
Anecdotally, a notable number of patients refuse highly recommended nodal dissection, which allows for significant disease control, in fear of developing the symptomologic sequelae of lymphedema. Commonly these symptoms are associated with the progression of the disease itself, and the subsequent impact on patients’ self-esteem and quality of life. 7,10 Discomfort, described as heaviness and tightness of the affected limb, as well as pain and decreased range of motion can limit patients’ functionality, and predispose them to body image sensitivity. 11 Further, symptoms of lymphedema can impact patients’ socio-economic status; many patients describe a decrease in their capacity to be financially productive, superimposed on continuous out-of-pocket treatment expenses. 12 In light of these psychosocial and physical morbidities, patients, tend to require frequent access to care, leading to high direct and indirect costs to health-care systems. 13
Immediate lymphatic reconstruction is an innovative microsurgical technique, first described in 2009, where lymphatic vessels are connected to the venous circulation via microsurgical anastomosis. This allows for the creation of lymphatico-venous shunts, bypassing the local lymphatics thereby minimizing the risk of secondary lymphedema. 14 This has many benefits as the operation occurs on a freshly cut lymphatic and vein of reasonable caliber making lymphedema surgery potentially more successful, thus preventing lymphedema as opposed to trying to correct it after it has already installed with significant post-operative fibrosis in the tissues when doing retroactive surgical interventions. Since its inception, published studies have demonstrated the benefits of immediate lymphatic reconstruction, as a preventative procedure in patients with various types of cancers who undergo nodal dissection. 15 -17 Given that this intervention is not yet commonly performed in Canada, and quality of life of this procedure has not been described as of yet, we sought to explore the impact of immediate lymphatic reconstruction on quality of life (QOL) in patients with skin cancer, undergoing axillary or groin dissection. In this paper, we focus on the experience of one of our patients, who underwent immediate lymphatic reconstruction at the time of axillary node dissection.
All procedures that follow were performed per the ethical standards of the responsible committee on human experimentation (institutional and national), as well as with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was obtained from the patient prior to inclusion.
Case Report
Written informed consent for patient information and images to be published was provided by the patient.
Case Presentation
A 42-year-old male was referred to surgical oncology for surgical management of a suspicious lesion located on his right upper back. Biopsy of the lesion revealed a 2.2 mm malignant melanoma without ulceration, a mitotic rate of 3, and involvement of the deep margin. Given the Breslow depth, the patient underwent a WLE with 2 cm margins and a SLNB. The final pathology report provided evidence of nodular melanoma, staged IIIB (T3a N2a). This case was prior to the era of MSLT2 and so given his young age, the histologic features of his primary cancer, and having 2/3 nodes positive, a completion axillary lymph node dissection (ALND) with immediate lymphatic reconstruction was recommended. 18 At the time of presentation, the patient was a nonsmoker with well-controlled hypertension and had a BMI of 35.9.
ALND and Lympho-Venous Bypass Procedure
In accordance with the standard local operative guidelines for ALND, the patient received prophylactic antibiotics before skin incision. With the patient in the dorsal decubitus position, patent blue dye was injected into the arm. This was done approximately 10 minutes before the incision, at the level of the wrist, and vigorously massaged for 5 minutes. This was done to identify the lymphatics of the arm. These lymphatics specifically belong to the arm and are not related to the melanoma from the back. This is the concept of reverse axillary mapping which has previously been described in Breast Cancer by Dr. Suzanne Klimberg et al. 19 These lymphatics do not drain the tumor and thus there is no risk for spread of cancer or “leaving cancer nodes behind”. Using the SPY Fluorescence Imaging System, lymphography was then performed to better delineate the lymphatic anatomy.
An incision was then made at the anterior axillary hairline, at the level of the previous scar. Advancement through the clavi-pectoral fascia, using electrocautery, was then performed. Skin flaps were raised on both sides of the incision; following which, 2 well defined blue lymphatics were identified and dissected out, in preparation for the lymphatic reconstruction. Axillary lymph node dissection was then performed by the surgical oncology team where careful attention was placed to not disrupt the 2 blue lymphatics. They also dissected out the anterior venous branch off of the axillary vein to preserve as much length of it as possible for this vein to be used in the bypass, prior to clipping and ligating it. Following the removal of all of the lymph nodes, an immediate lymphatic reconstruction was performed, at the level of the right upper arm.
The larger of the 2 blue lymphatics previously identified from the upper arm was used for the reconstruction. As previously mentioned, the anterior branch from the axillary vein that was dissected out was used as the vein for the bypass. It is of reasonable caliber and drains directly into the axillary vein, which is ideal for the prevention of lymphedema. The clip was removed from the ligated end of the vein and it was prepped. Using an operative microscope, an end-to-end lymphatico-venous anastomosis was performed, using 10-0 ethilon sutures. Following bypass, blue dye from the lymphatic could be seen flowing across the anastomosed venule (Figure 1). Placement of surgical spear beneath the anastomosis also did not reveal any leakage of blue stained lymphatic fluid from the anastomotic site and these 2 elements were used as a proxy through which LVA patency was inferred. The procedure was very well tolerated by the patient, and the estimated blood loss was estimated at 10 cc. The total duration of surgery was 4 hours and 12 minutes. No intraoperative complications were noted.
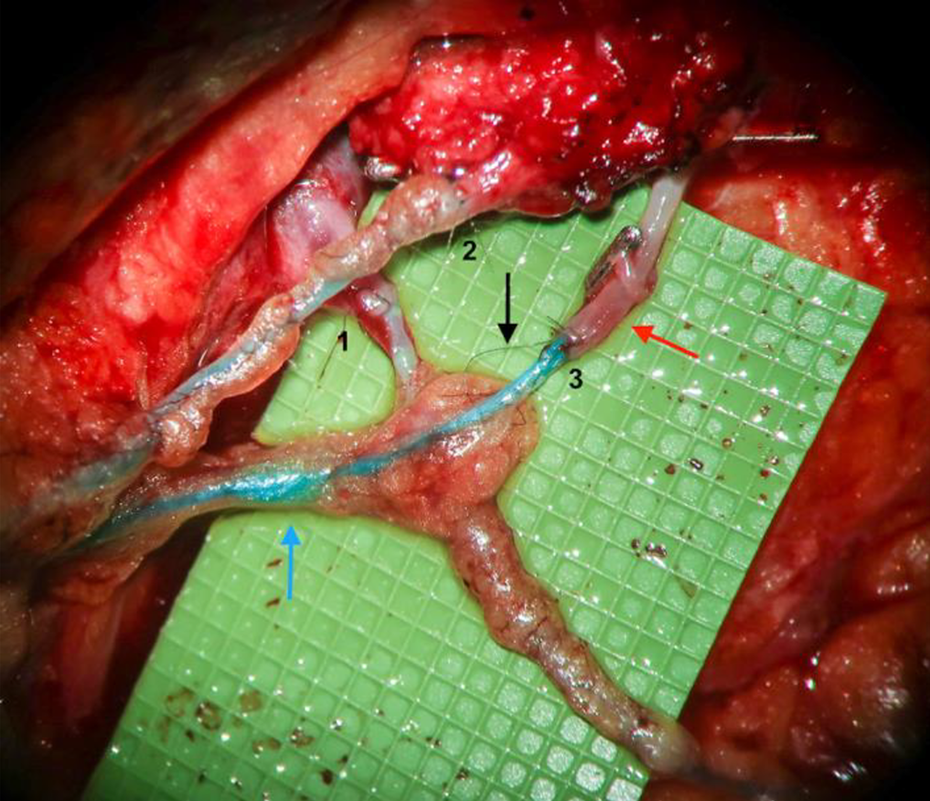
During a lympho-venous bypass procedure, a lymphatic vessel (blue arrow) is connected to a vein (red arrow). In this patient, 3 separate lympho-venous bypasses (numbered 1, 2, 3) have been performed. Each bypass is handsewn using an operating microscope with nylon microsutures (black arrow). The green background is used to provide contrast when using the microscope. Each small green is box 1 x 1 mm.
Follow-Ups and Measurements
The patient was seen in clinic for follow-up at 2 weeks, 3, 6, 9, 12, and 24 months postoperatively. No protocol for limb elevation or compression were employed post-operatively, as there is no data to support its use in the prophylactic setting. To evaluate for the presence of postoperative lymphedema, our patient first underwent baseline measurements of both upper limbs prior to surgery. These were performed circumferentially, at 4 cm increments, from the wrist to the axilla. Measurements were then repeated in an identical fashion at each follow-up visit. Given that volume, estimated by the truncated cone formula, has been demonstrated to correlate well with limb volume measured by plethysmography, Brorson’s formula was used to estimate limb volume in our study. 20 Volumes were calculated using the following based on limb circumference measurements:
Here, V is the volume of the segment, C1 and C2 represent the circumferences of the proximal and distal ends of that segment, and h is the distance between these ends (segment height). 20
With regard to diagnosing lymphedema, the literature reports many methods. The most frequently described criteria are defined by either a 2 cm circumferential difference between the affected and unaffected limb, or a 10% volume difference between limbs. 21 Since the latter method better accounts for variations in body and limb sizes, it is the most salient diagnostic measure used in recent studies. 22 -25 As such, we employed this diagnostic metric as our operation definition of lymphedema in our patient.
Limb Volumes
At baseline, our patient presented with an inter-limb volume difference of 3.1%. At his 3-month follow up, he presented with a difference of 22.9%. At 6-months, he presented with a difference of 2.0%, a measurement similar to his baseline, and below the 10% threshold. A low interlimb volume difference was maintained at the 9-month time-point (4.5%), 12-month time-point (1.7%), and 24-month time-point (0.9%).
Overall, this patient developed transient lymphedema 3 months postoperatively. The differences between affected and non-affected limb volume overtime are outlined in Figure 2.
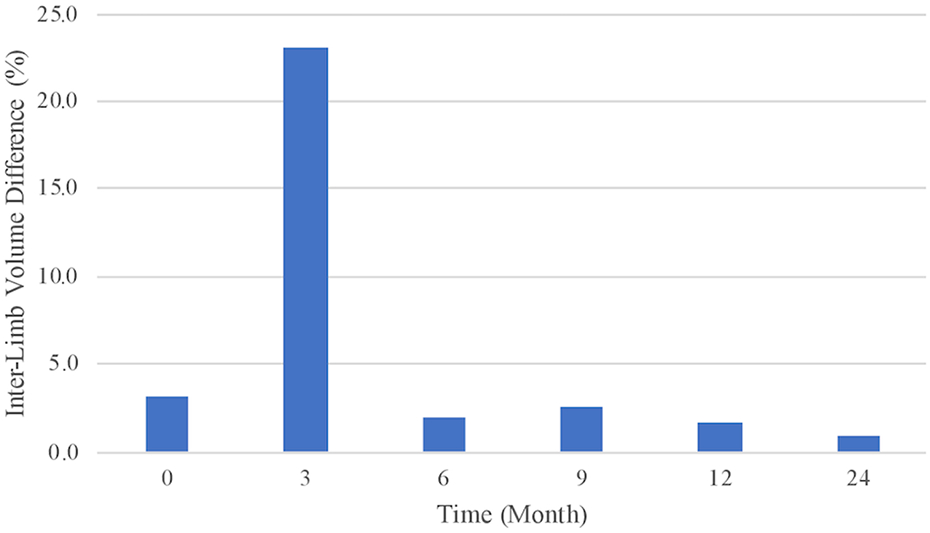
Patient’s volume difference in percentage between affected and non-affected limb over time.
Quality of Life
At each follow-up visit, quality of life (QOL) was measured using the Lymphedema Quality of Life Questionnaire (LYMQOL); a validated quality of life assessment tool for patients with upper and lower extremity lymphedema. 26 Permission to this questionnaire was granted to us by Dr. Vaughan Keeley.
Our patient answered questions falling into the 4 following domains: symptoms, body image/appearance, function, and mood. 26 For each domain, our patient responded to items on a 5- point Likert scale where 0 = not applicable, 1 = not at all, 2 = a little, 3 = quite a bit, and 4 = a lot. A total score for each domain was calculated as a fraction, with the numerator represented by the sum of all individual scores in a given domain, and denominator the total number of items in said domain. Thus, the maximum total score is 4, with a lower score indicating a better QOL. 26 The final part of the LYMQOL questionnaire measures global quality of life, where our patient reported their overall quality of life on a scale from 0 – Poor to 10 – Excellent. This questionnaire is illustrated in Figure 3.
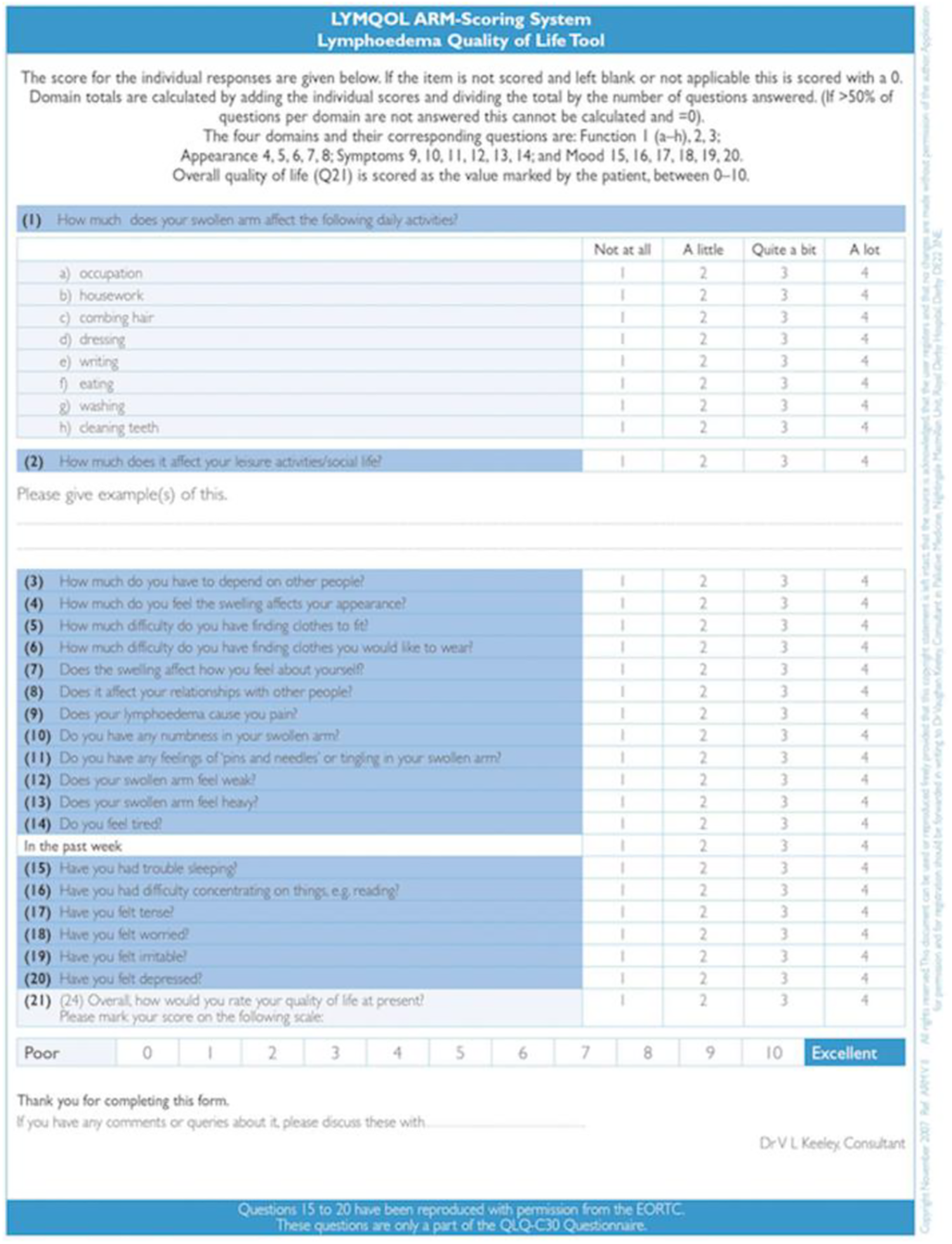
LYMQOL ARM questionnaire with scoring system.
The global QOL score in this patient was 7 at the 3-month time-point, 8 at 6 and 9 months, 7 at 12 months, and 8 at 24 months (Figure 4). The domain with the least-inter-visit variation was the patient’s symptoms (variance of 0.1039) and the domain with the most inter-visit variation was the patient’s function (variance of 0.5850) (Figure 5).
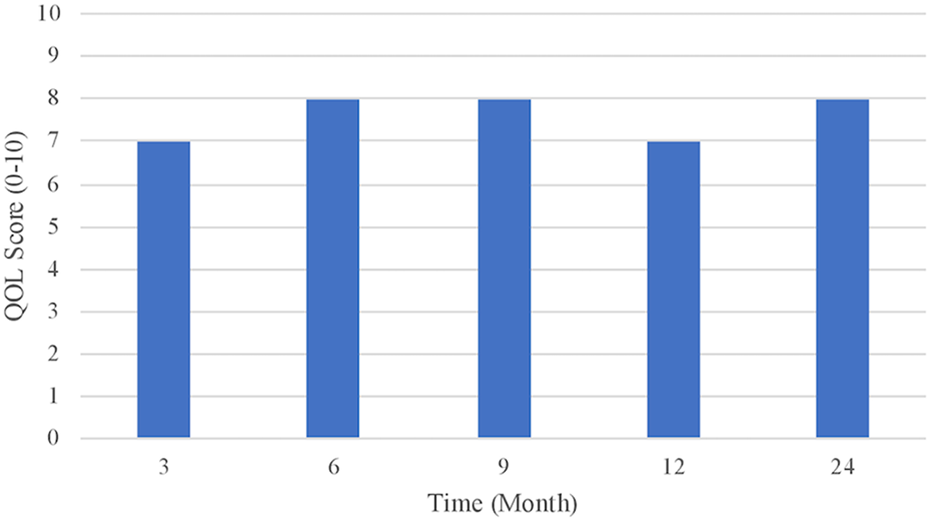
Patient’s global quality of life over time (LYMQOL ARM Q.21 – Overall, how would you rate your quality of life at present).
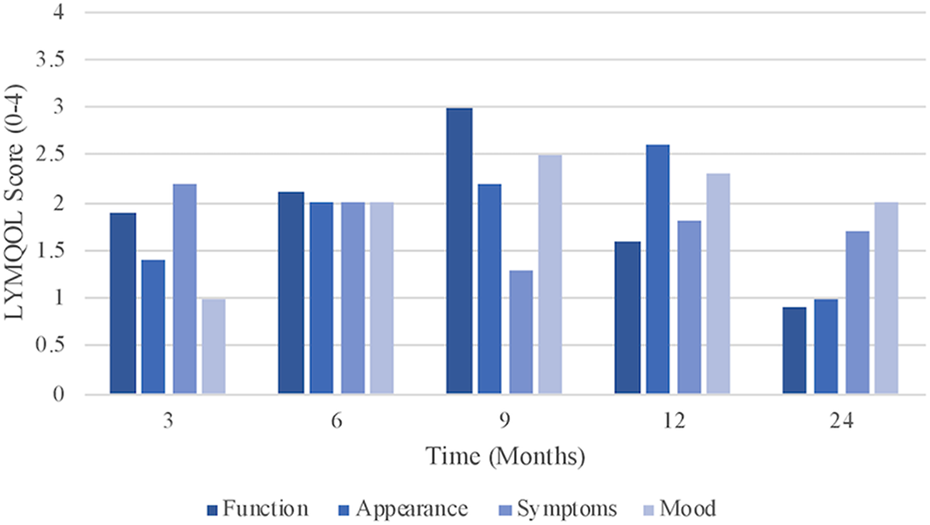
Patient’s LYMQOL ARM domain scores over time.
Discussion
This case report describes the outcomes of a patient who underwent immediate lymphatic reconstruction, at the time of axillary lymph node dissection, for the primary prevention of lymphedema. Our case study demonstrated consistency in perceived QOL at various time points following immediate lymphatic reconstruction. Moreover, the patient did not develop chronic lymphedema postoperatively.
The decision to pursue ILR in our patient was informed by the existing literature and was balanced with the operative morbidity associated with the selected technique of LVA, as compared to those associated with alternative techniques. As in all surgical procedures, performing LVA at the time of nodal dissection adds risks for the patient. The additional dissection to identify nearby veins, results in a modest increased risk of hematoma and or bleeding. Additionally, there is an increase in added OR time both during the ALND since any encountered lymphatics must be safely dissected free by the general surgeon before the node dissection can proceed. Finally, the need to prepare the lymphatics and veins and the completion of the anastomosis under the microscope also contribute to increased operative time.
With respect to the specific implications of immediate lymphatic reconstruction on limb volume, several studies have been published showing its benefit. In a study performed on patients with melanoma, Boccardo et al. (2013) demonstrated a potential prevention of increased limb volume in 93.75% of their patients. 27 Only one of the 16 patients developed lower-extremity lymphedema; however, this was only transient and resolved after 2 months. Additionally, reports from Nacchiero et. al and Cakmakoglu et. al, have described LVA as an effective approach for the primary prevention of postoperative upper extremity lymphedema - particularly in the context of axillary lymphadenectomy for locally advanced melanoma. Respectively, these studies have reported lymphedema rates of 4.3% and 4.5% in those undergoing ILR, representing a significant rate reduction when compared to the lymphedema rates in these patients as reported in the literature. 28,29 The results of several other studies investigating ILR at the time of axillary node dissection are outlined in Table 1. They have all shown favorable outcomes. Comparatively, there is a paucity of evidence supporting alternative physiologic techniques, namely VLNT, in the setting of lymphedema prophylaxis. Further, from a morbidity standpoint, LVA does not require a donor site or free tissue transfer, thus negating the risks of flap failure, donor-site lymphedema, or other donor-site specific complications.
Summary of the Literature on Patients Undergoing Axillary ALND and Immediate Lymphatic Reconstruction.
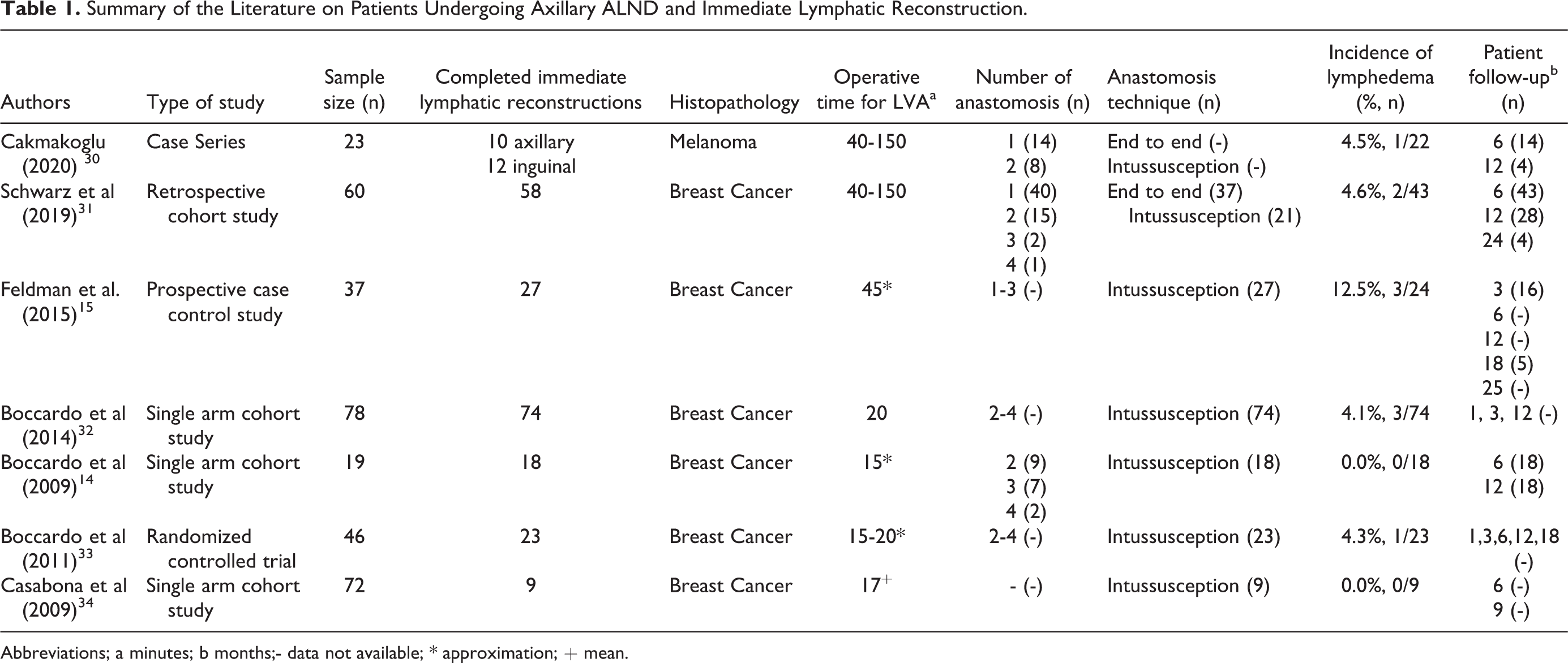
Abbreviations; a minutes; b months;- data not available; * approximation; + mean.
With these elements in mind, the multifactorial and lifelong implications of lymphedema, coupled with the patient’s risk of developing lymphedema were juxtaposed with the additional surgical time and risk-complication profile of performing an LVA at the time of nodal dissection. Following a meaningful discussion and voluntary consent, the care team decided that LVA was the most appropriate intervention for this patient.
At present, there are no reports in the literature that describe QOL in patients who have undergone prophylactic procedures to limit lymphedema (i.e. immediate lymphatic reconstruction). That said, studies have measured QOL in patients who have received treatment for established lymphedema. For instance, Coriddi et al. (2020) reviewed QOL outcomes after lymphatico-venous bypass (LVB) or a vascularized lymph node transplant (VLNT) and indeed reported QOL improvements following surgical treatment. 35 However, since the goal of our intervention is primary prevention, rather than treatment of established lymphedema, we cannot compare our findings to those of the aforementioned study. Quantitative studies in the literature show that lymphedema patients present with negative perceptions of themselves, thus contributing factors that limit sociability and contribute to a state of decreased well-being. 36,37 Over time, our patient maintained high QOL scores and only experienced transient lymphedema. This result shows promise in primarily preventing the debilitating aforementioned psychosocial sequelae of lymphedema, especially for those with complications in their lower extremities.
Interestingly, research also suggests that overall QOL may be more closely associated with level of physical function, rather than limb excess. This may explain why our patient continued to report such high levels of QOL, seemingly independent of changes in limb volumes. 37 -39 Our patient’s affected limb, the right arm, is his dominant arm. We theorize that the development of lymphedema in the dominant arm may have greater implications on patients’ daily functioning, precipitously diminishing QOL. Given this, despite dominant limb involvement, our patient maintained stable measures of QOL throughout. When reviewing the literature on the topic, several studies reported natural asymmetry of the arms, secondary to handedness. Looking at the baseline volume measurements, our patient had an inter-limb volume difference of 3.1%, in favor of the right arm. This initial difference can be explained by arm dominance, contributing to the anatomy in this patient. 30,31,40 The majority of recent studies accounting QOL and lymphedema do not account for hand or arm dominance. Future studies should account for limb dominance, to yield more accurate results and better interpretation of their findings.
Our patient developed transient lymphedema (TLE) at the 3-month post-op timepoint, with an inter-limb volume difference of 23%. TLE is defined in the literature as lymphedema that resolves spontaneously by the next follow-up appointment, and usually occurs in the first year after surgical treatment. 41 -44 Research has previously illustrated that the development of transient lymphedema is a common finding postoperatively after an extremity lymph node dissection. This may occur with or without a lymphatico-venous anastomosis, with respective occurrences reported between 3-12.5% and 40-50%. 16,42,45,46 TLE may also be explained by a transient reaction to chemotherapy or radiotherapy. 43 Thus, the occurrence of spontaneous lymphedema in our patient does not necessarily imply that the immediate lymphatic reconstruction was unsuccessful, as interlimb volume difference at 6-months was measured at 2.0%, well below the 10% threshold. This remained consistent, through the remainder of the 24-month follow-up duration (Figure 6).
Post-operative photos at the first follow up appointment (A, B) and at the 24-month appointment (C).
Another interesting implication of immediate lymphatic reconstruction is its potential for long-term financial benefits. In a Canadian international study, 20.6% of research subjects indicated that lymphedema affected their work-life, and 26.8% reported that family income was adversely affected by their swelling. 9 Loss of productivity secondary to lymphedema has been described as multifactorial, including lost workdays, missed educational opportunities, unemployment, and early retirement. A recent study published by Head et al. found the cost of immediate lymphatic reconstruction appears to be comparable to combined decongestive therapy (CDT), the current mainstay of management in established lymphedema. This article also raises a potential economic benefit, through decreased healthcare costs, and improved productivity, and increase in QOL which could be interesting to elucidate further in future studies. 47
The limitations of this study include firstly that this is a report on a singular patient outcome, with no comparative arm. Another important limitation pertains to the method of LVA patency confirmation. A review of the literature consistently identifies ICG lymphangiography as the gold standard for confirming patency post-operatively, with potential applications in intraoperative decision-making 48 -53 A 2010 retrospective study by Holm and colleagues investigating the utility of ICG in the context of identifying microvascular thrombosis reported a sensitivity and specificity of 100% and 86% respectively. 54 Additionally ICG benefits from minimal invasiveness, while presenting no radiation exposure and real-time functional imaging. 55 Unfortunately, due to lack of funding the imaging technology to assess LVA patency such as SPY-PHI and Hamamastu are not available at our institution. As such LVA patency was not confirmed with ICG lymphangiography. Notwithstanding, passage of blue dye was observed flowing across the anastomosis under the operative microscope prior to closure. Further, placement of the surgical spear beneath the anastomotic site did not reveal any leakage of blue stained lymphatic fluid from the anastomotic site. These clinical findings were felt to be sufficient in deducing LVA patency intra-operatively as a resource-limited alternative to ICG. Lastly, we acknowledge that it is impossible to remark on the trajectory of our patient’s clinical course, had he not undergone ILR. In spite of this, given the current body of evidence in supporting reduced rates of postoperative lymphedema pursuant to immediate ILR, along with the minimal morbidity and maintenance in quality of life reported by our patient, we maintain ILR was an appropriate choice for this case. However, this remains the only report on quality of life after prophylactic lymphatico-venous bypass, and higher quality evidence is needed to evaluate the benefits of ILR in both reduction in lymphedema rate and quality of life in patients requiring lymph node dissections.
Conclusion
This case report shows promising results. Our patient only developed lymphedema transiently and maintained favorable measures of quality of life over the course of 2 years. Given these encouraging outcomes, corroborating data in the literature, and markedly heightened rates of secondary lymphedema and associated symptomologic sequelae in high-risk patients, routine implementation of immediate lymphatic reconstruction should be explored in such patients. This novel approach has yet to be implemented as a standard of care in our Canadian health-care system, and a more thorough examination of these outcomes is needed. Specifically, a randomized controlled trial, involving a larger patient cohort, should be conducted, in an effort to replicate our findings. Such an outcome would be overwhelmingly positive for our cancer population, and on our health-care system overall.
Footnotes
Authors’ Note
We also express our gratitude to Dr. Vaughan Keeley for allowing us to use his validated tool the LYMQOL, to monitor the impact of our intervention in our patient.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5). Informed consent was obtained by all patients before being included in the study.
Statement of Informed Consent
Informed consent was obtained from all individual participants included in the study.