Abstract
We present a pediatric patient with an ASMT of the wrist with the unique approach of a plastic and reconstructive surgeon to surgical oncologic resection and reconstruction: WLE, ICG and NIRF imaging-assisted SLNB followed by reconstruction with a keystone perforator flap.
Introduction
Spitz tumors occasionally exhibit histologic features of both Spitz nevus and melanoma. These are called ‘Atypical Spitzoid Melanocytic Tumors’ (ASMT). 1 Diagnostically problematic, the metastatic potential of these Spitzoid melanocytic proliferations generates uncertainty with regards to diagnosis and management. There is significant controversy around sentinel lymph node biopsy (SLNB) with recent data discouraging the procedure in children given favorable outcomes despite a high rate of node deposits. 1,2 The approach to surgical oncologic resection and reconstruction through the unique lens of a plastic and reconstructive surgeon is presented in this case report.
Case Report
A 13-year-old otherwise healthy Asian female presented to her family physician with a growing 1-cm nodule on her right wrist dorsum, present for one month and neither itching nor bleeding (Figure 1). Biopsy of the lesion was performed and showed an ASMT measuring 2.9 mm in thickness. Ulceration was present. There were a lack of cytologic maturation with depth and a brisk mitotic index (10 mitoses/mm2) including deep mitoses (Figure 2a and b). In addition, a focus suspicious for satellitosis was present.
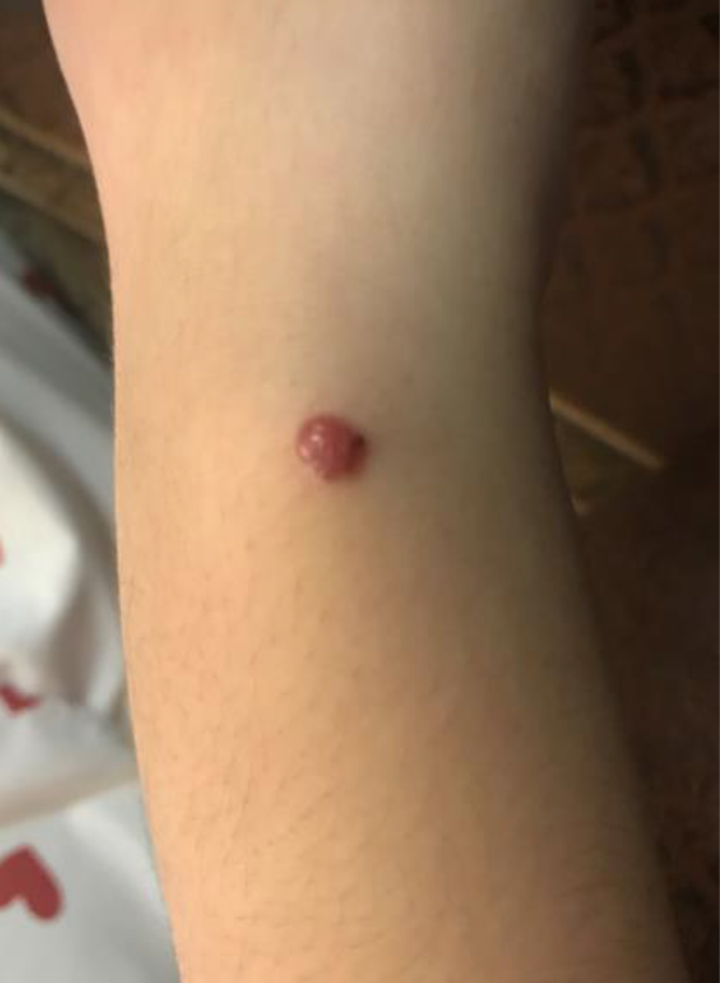
Atypical Spitzoid melanocytic tumor of the right wrist dorsum.
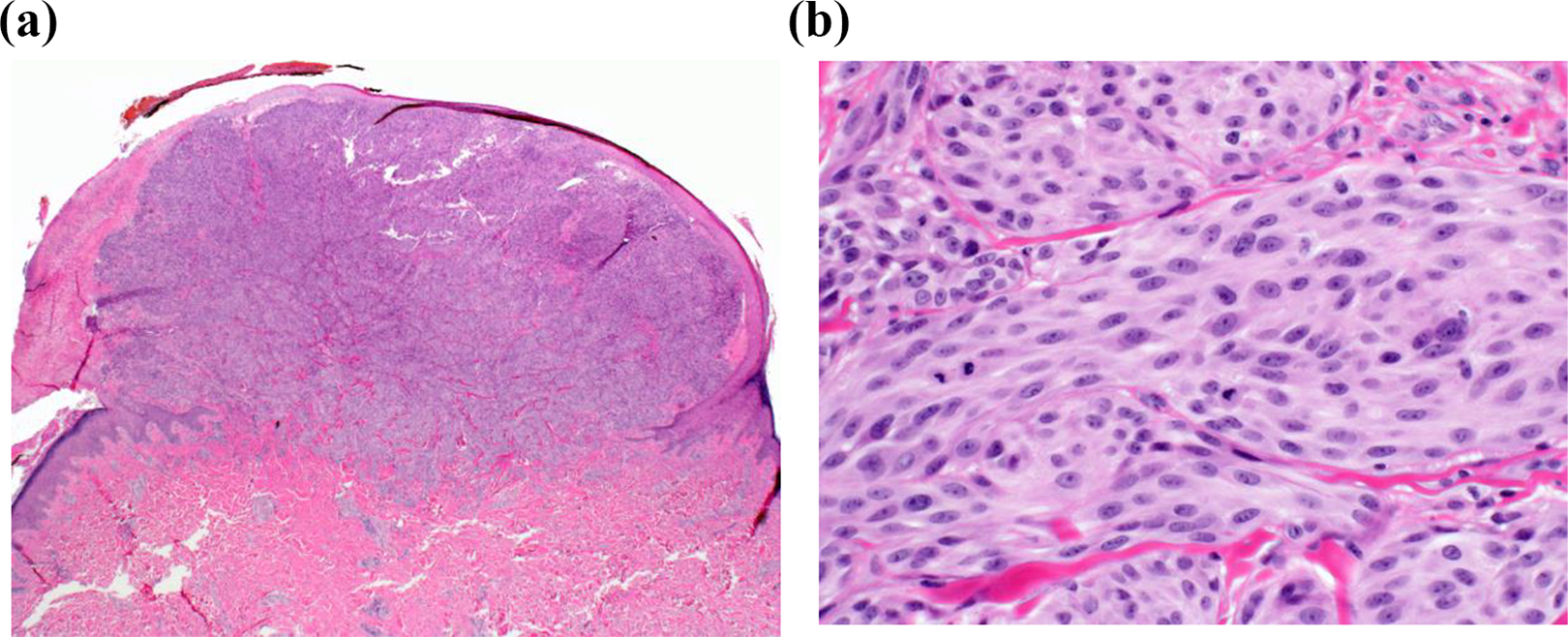
Histopathology showed an exophytic and ulcerated dermal based tumor (a) composed of cellular fascicles of spindled melanocytes with Spitzoid cytomorphology, cytologic atypia and mitotic activity (b).
She was referred to our multidisciplinary cutaneous oncology team. On further history, she denied prior tanning bed use and blistering sunburns. She had no metastatic symptoms on functional inquiry. There was no family history of skin cancer. Physical examination demonstrated a healing scar on her wrist, with no residual lesion and no palpable lymphadenopathy. The biopsy specimen was reviewed by 5 dermatopathologists and classified as severely atypical Spitzoid neoplasm concerning for Spitzoid melanoma. The margins were narrowly clear by 1.8 mm peripherally and 1.3 mm deeply. Applying the Memorial Sloan-Kettering sentinel node prediction nomogram (designed for adult patients), her node positivity risk was 36% if this were unequivocal melanoma. She was planned for wide local excision (WLE) with reconstruction and SLNB.
Pre-operative lymphoscintigraphy identified one epitrochlear and two axillary sentinel lymph nodes. WLE of the primary lesion included a 1-cm margin around the scar. Dissection was carried to the extensor retinaculum, with preservation of the dorsal sensory branch of the ulnar nerve. The resulting defect measured 3 x 3 cm. A type IA keystone perforator flap was designed to close the resulting defect (Figure 3a and b). Three sentinel nodes were removed from the right axilla, two of which were detected with both the gamma probe (TruNode counts ex-vivo were 32 and 17) and near-infrared fluorescence (NIRF), while the third was identifiable with glowing indocyanine green (ICG) (TruNode count ex-vivo was 8) (Figures 4a and b). One epitrochlear node was removed using combined modalities (TruNode count ex-vivo was 12).
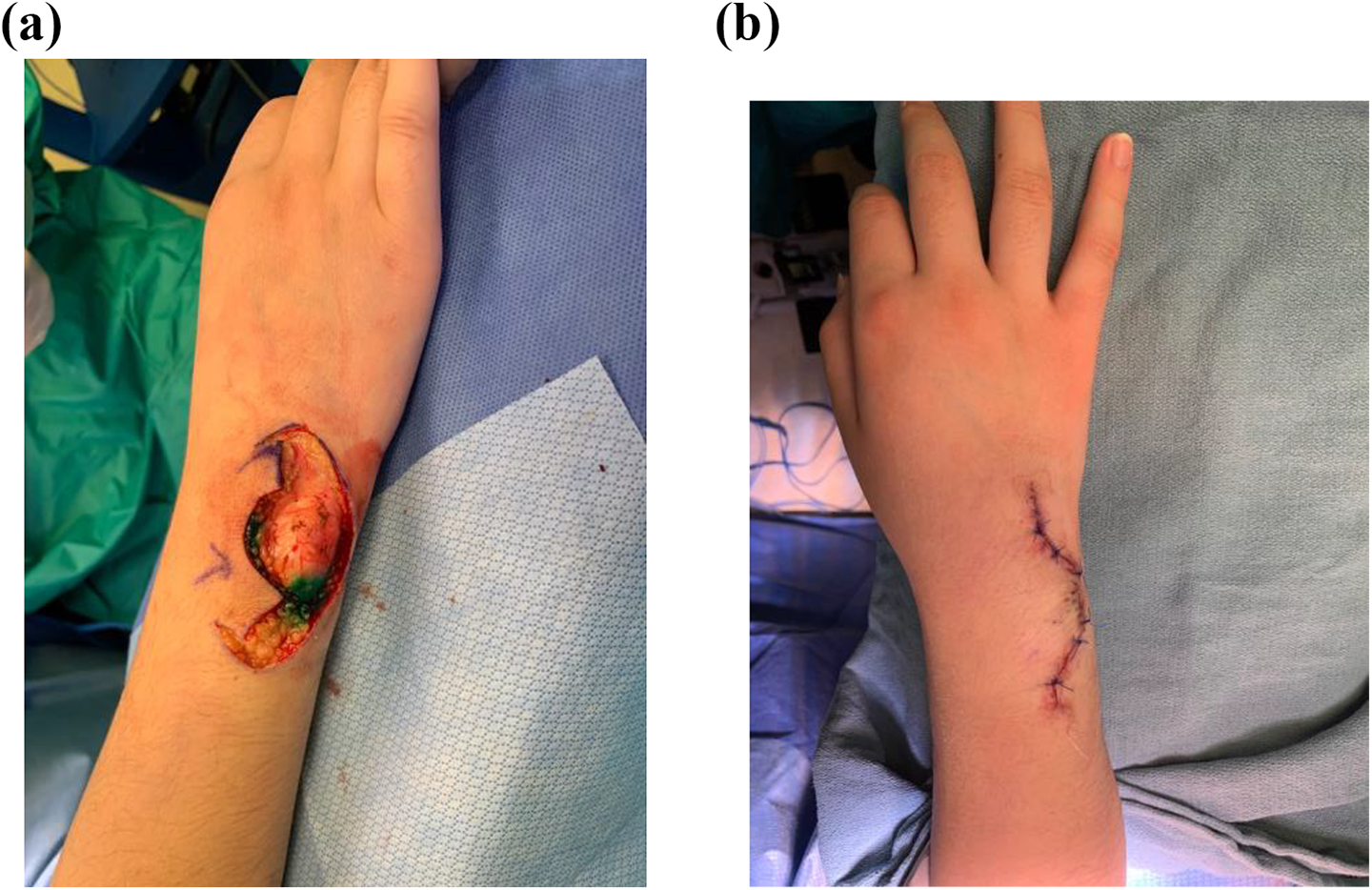
Following WLE, (a) a keystone perforator flap was designed and incised (b) to reconstruct the right dorso-ulnar wrist defect.

(a) Right axillary sentinel lymph node identified with fluorescence imaging and demonstration of fluorescent lymphatic channels and (b) ex-vivo capture of the three axillary nodes.
Pathology demonstrated a 1-mm subcapsular deposit of the ASMT in one axillary node (Figure 5a and b). The other nodes did not demonstrate evidence of tumor, nor did the wrist wide excision specimen.
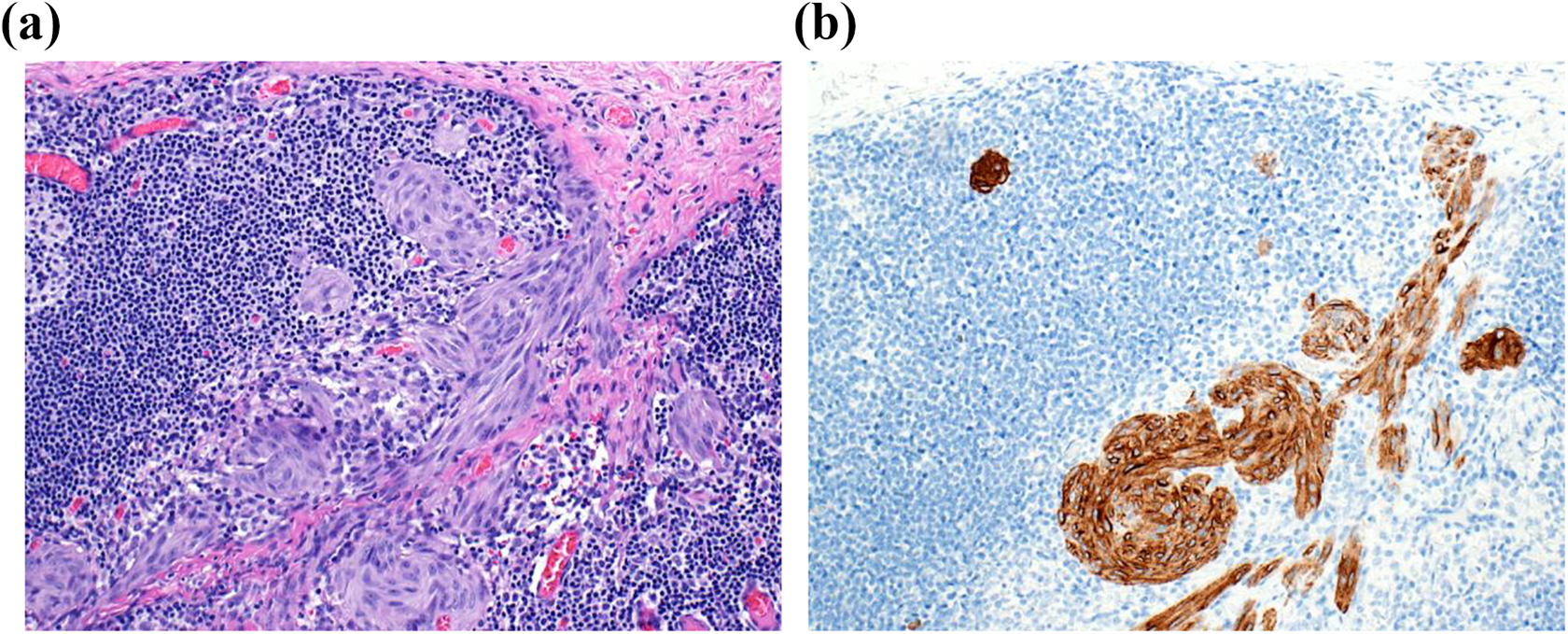
Subcapsular lymph node deposit (a) highlighted by Melan A immunohistochemistry (b).
She was seen in clinic for follow-up 2 weeks after surgery. Her keystone flap and primary skin closures at the elbow and axilla healed well. Given the positive SLNB result, the patient underwent BRAF mutation testing, which was negative, and a CT-PET scan, which demonstrated the expected post-operative metabolic activity in the right wrist, axilla and epitrochlear regions, but no evidence of metastatic disease. Medical oncology elected to omit adjuvant therapy, but to closely follow-up clinically with ultrasound imaging of the right axilla and epitrochlear regions 6-monthly.
Discussion
The surgical approach to ASMTs consists of 1-cm marginal excision. 2 Performing SLNB is controversial: despite commonly resulting in positive SLNB, the natural history of ASMTs is somewhat indolent. The largest series of patients with ASMT reported a survival rate of 100% despite a positive SLNB rate of 47%. 2 The parenchymal and subscapular lymph node deposits are thought to represent a benign process. 2 In patients with a positive SLNB, even with unequivocal melanoma, CLND has been discouraged, as it does not improve melanoma-specific survival. 3 Ultrasound follow-up of the nodal basin is a safe alternative.
To increase the yield of nodes harvested and to minimize the risks associated with the procedure, we used ICG and NIRF imaging as an adjunct to SLNB. Injected closely to the primary malignancy, ICG binds albumin, an integral component of lymph. 4 Infrared light has high tissue penetration and excites ICG molecules: glowing lymph nodes and channels can be visualized 10 mm beneath the skin. 4 This real-time visualization of lymphatic structures permits a selective and focused dissection, limiting the disruption of channels 5-7 and reducing the risk of lymphocele and lymphedema. 8 ICG and NIRF do not require radiation and have a low risk of anaphylaxis compared to blue dye (0.05% versus 1.1%). 8 Multiple studies have demonstrated the usefulness of ICG in support of SLNB 5,6,8 with a sentinel lymph node detection rate oscillating between 86 and 100% 9 . ICG can lower false-negative rates, improve regional control, limit mistaging of patients and give them opportunities for adjuvant therapy. In this case, the SPY Elite System identified one additional occult axillary node not identified by the lymphoscintigraphy, although the occult node did not contain Spitzoid cells after pathologic analysis. Plastic surgeons familiarity with the ICG and NIRF, frequently used intra-operatively to assess flap perfusion, was a natural segue into imaging of SLN, minimizing the risk of a false-negative result.
WLE and SLNB are typically performed by general surgeons. We believe that as plastic surgeons we have a unique skill set which was useful and efficient in this patient’s care. During the oncologic resection, our training with the upper extremity anatomy facilitated protection of important nerves (i.e. dorsal sensory branch of the ulnar nerve) as did our routine use of magnifying loupes. Our training of the anatomy of the elbow from peripheral nerve release surgery and trauma was useful for the epitrochlear node biopsy. Our familiarity with flap design for complex wound closure, another intrinsic feature of plastic surgery, allowed a single team to do both the resection and the reconstruction. A keystone perforator flap was chosen to avoid skin grafting for dorsal wrist coverage, an important functional and aesthetic zone. 10
Our comfort with talking to families and putting children at ease was nurtured by broad exposure in training and clinical practice to pediatric brachial plexus, cleft lip and palate and hand clinics. This child was seen at an adult cancer center which lacks some of the comfort and security of a pediatric facility, but hopefully our demeanor and approach was a natural fit to increase this child’s and family’s comfort.
We hope this case encourages plastic surgeons to gain additional oncologic training to add value to multidisciplinary melanoma teams.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statements
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was obtained from the participant included in the study. Additional informed consent was obtained from the participant for whom identifying information is included in this article.’.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
