Abstract
Introduction:
Skin graft reconstruction is a common method of providing wound coverage. Rarely, skin grafting can be associated with the development of squamous cell carcinoma (SCC) in the graft donor site.
Case Report:
The patient is a 72-year old male with a 15-year history of bilateral hip wounds. He underwent a multitude of treatments previously with failed reconstructive efforts. After presenting to us, he underwent multiple debridements and eventual skin grafting. Within 4 weeks of the final skin graft, a mass developed at the skin graft donor site at the right thigh. Excisional biopsy returned a well differentiated keratinizing SCC.
Discussion/Conclusion:
This case demonstrates the acute presentation of SCC in a patient following a skin graft without known risk factors. The purpose of this unique case report is to highlight a very rare occurrence of SCC at a skin graft donor site.
Introduction
Skin graft reconstruction is a common method of providing wound coverage. 1 The technique relies upon harvesting skin from a donor site and transplanting it to a recipient site. Postoperative complications can include infection and graft failure. 2 Rarely, skin grafting can be associated with the development of squamous cell carcinoma (SCC) a nonmelanoma skin cancer of cutaneous keratinocytes, at the graft donor site. The leading risk factor for SCC is ultraviolet radiation, though non-solar risk factors including immunosuppression, ionizing radiation, manufacturing chemicals, and chronic wounds have been also described. 3 The pathogenesis of disease is thought to be through mutations in the tumor suppressor gene, TP53, which encodes the p53 protein. 4
The development of squamous cell carcinoma in a split thickness skin graft (STSG) donor site has been described in only 16 previously reported cases in the literature. In this study, we present a case of de novo squamous cell carcinoma arising from a thigh STSG donor site in a patient with chronic wounds who did not carry a history of malignancy or burns.
Case Report
The patient is a 72-year old male with a 15-year history of bilateral hip wounds. The original wounds arose after the patient developed infections from medication injections that necessitated drainage and debridement. The wounds were initially treated with serial debridements and local wound care. Within the 15-year time frame, the patient underwent multiple debridements, local wound care with a variety of different wound care products, biopsies, and eventual attempted reconstruction. Attempts at skin graft reconstruction were completed on 3 separate occasions, all of which failed. The wounds were subsequently managed with local wound care without improvement.
The patient presented to our service with large draining bilateral hip wounds (Figure 1). He desired further attempts at debridement and reconstruction in order to improve the quality of his life. On examination, large bilateral hip wounds were noted. There was necrotic tissue throughout the wounds with concern for chronic infection and possible Marjolin’s ulceration. Old skin graft harvest sites were noted. The patient was counseled that attempts at debridement and reconstruction may be futile but attempts to eliminate what was likely a chronic infection would be necessary prior to any form of reconstruction. Also, the patient was informed that larger biopsies would be necessary to ensure a lack of skin cancer.
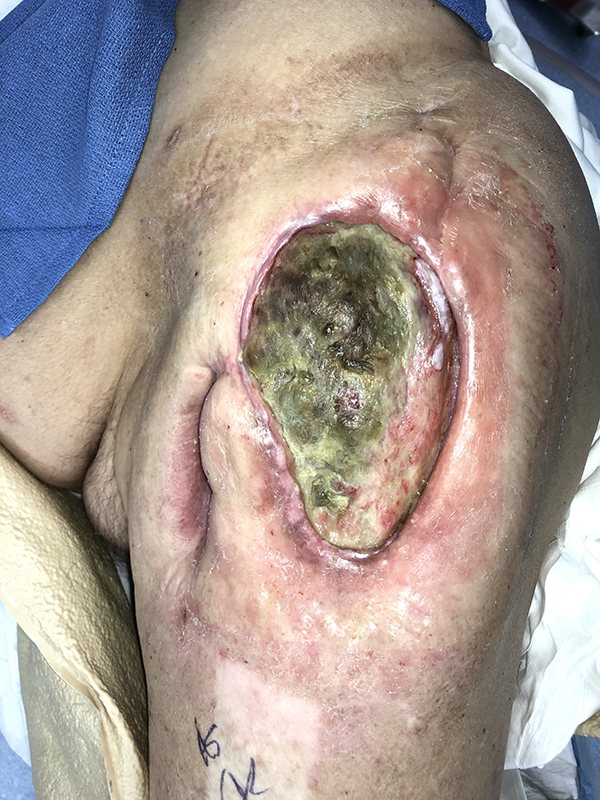
Chronic left hip wound with evidence of necrotic infected tissue throughout.
The patient was brought to the operating room and large scale bilateral debridements with excisional biopsies were performed. Tissue cultures returned positive for pseudomonas aeruginosa bilaterally, and all biopsies returned negative for malignancy. These biopsies included the entire periphery of the wound, multiple samples from within the wound itself, and the surrounding normal tissue. VeraFlo wound VAC therapy with CleanseChoice wound debridements (KCI) were performed until the wound was deemed ready for reconstruction (Figure 2). In order to augment the native wound bed, integra bilayer wound matrix dermal replacement was used to rebuild the deeper layers of lost tissue. After allowing the integra time to incorporate, the bilateral wounds were deemed ready for skin grafting (Figure 3). The patient completed his antibiotic course and repeat wound cultures were taken – these wound cultures returned negative. The patient underwent split thickness skin graft reconstruction from the right thigh to the right hip, which healed after graft incorporation and a small period of local wound care (Figure 4). Four weeks later, the patient then underwent split thickness skin graft reconstruction for the left hip from the previous left thigh donor site. The majority of this skin graft healed uneventfully (Figure 5).
Left hip wound after multiple debridements and cleansechoice wound VAC debridement therapy.
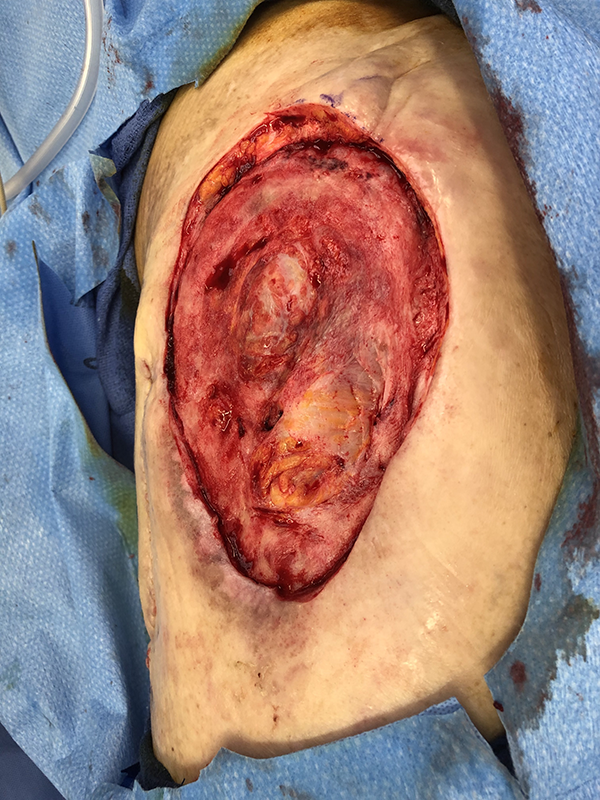
Left hip wound after multiple debridements and application of integra bilayer wound matrix.
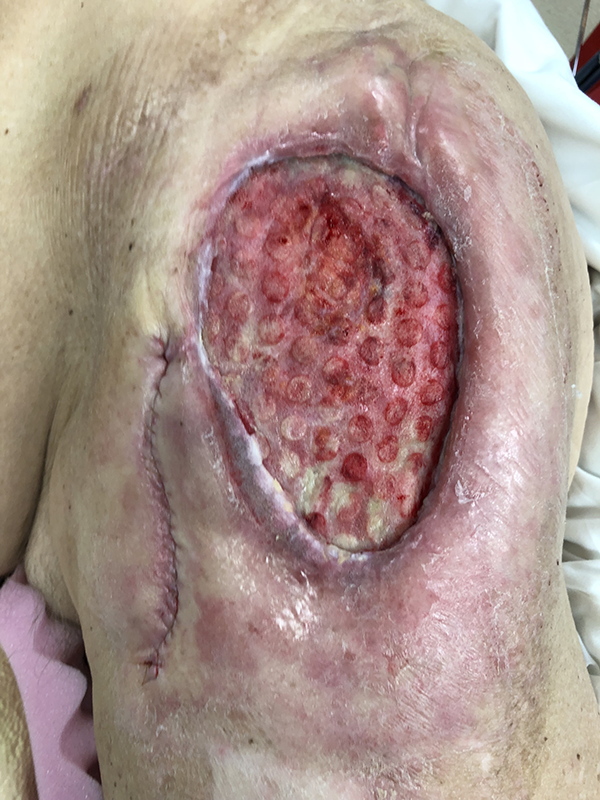
Early healing of right hip wound after split thickness skin graft.
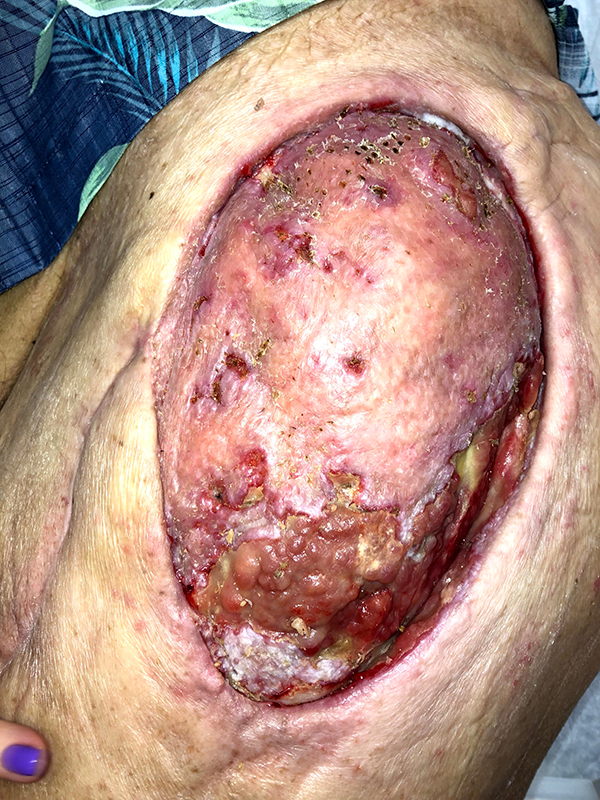
Early healing of left hip wound after split thickness skin graft.
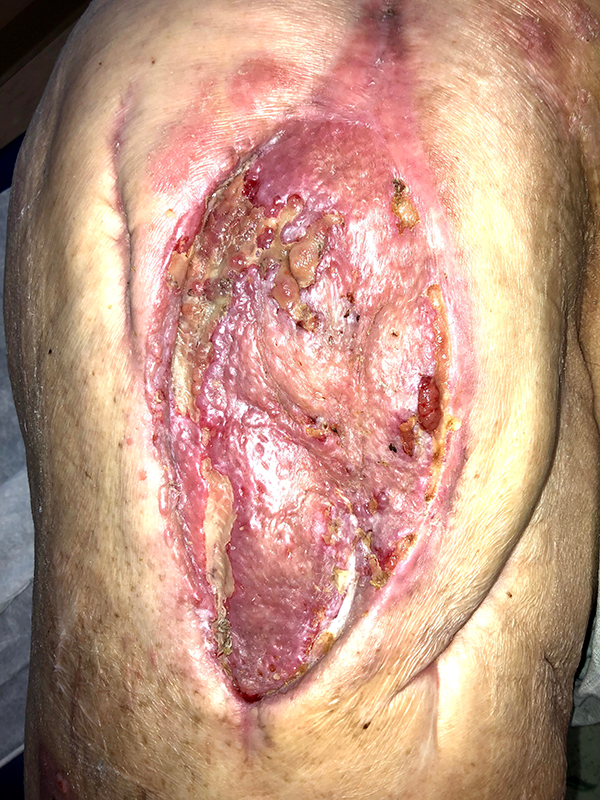
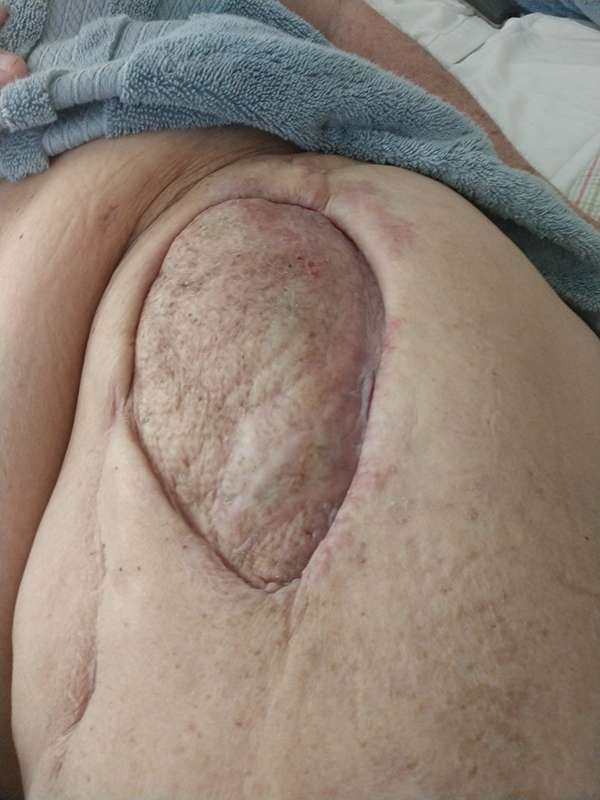
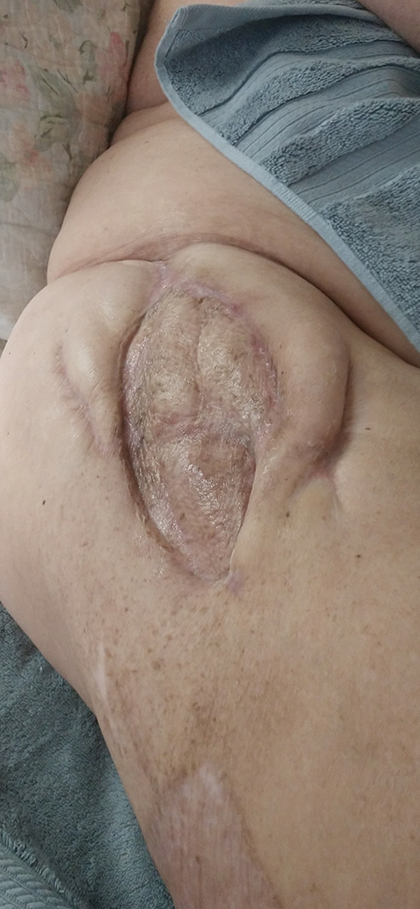
During the healing of the left hip skin graft, a mass developed at the skin graft donor site at the right thigh. This mass was exophytic, rubbery, and fixed to the skin. The decision was made to simultaneously perform an excisional biopsy of this mass during a repeat skin graft to the left hip for a small persistent open wound. This excisional biopsy returned a well differentiated keratinizing squamous cell carcinoma, completely excised with negative margins (Figure 6). The left hip skin graft healed without complication, and both wounds fully healed at the completion of the patient’s treatment without return of the SCC at 1 year follow up (Figure 7,8).
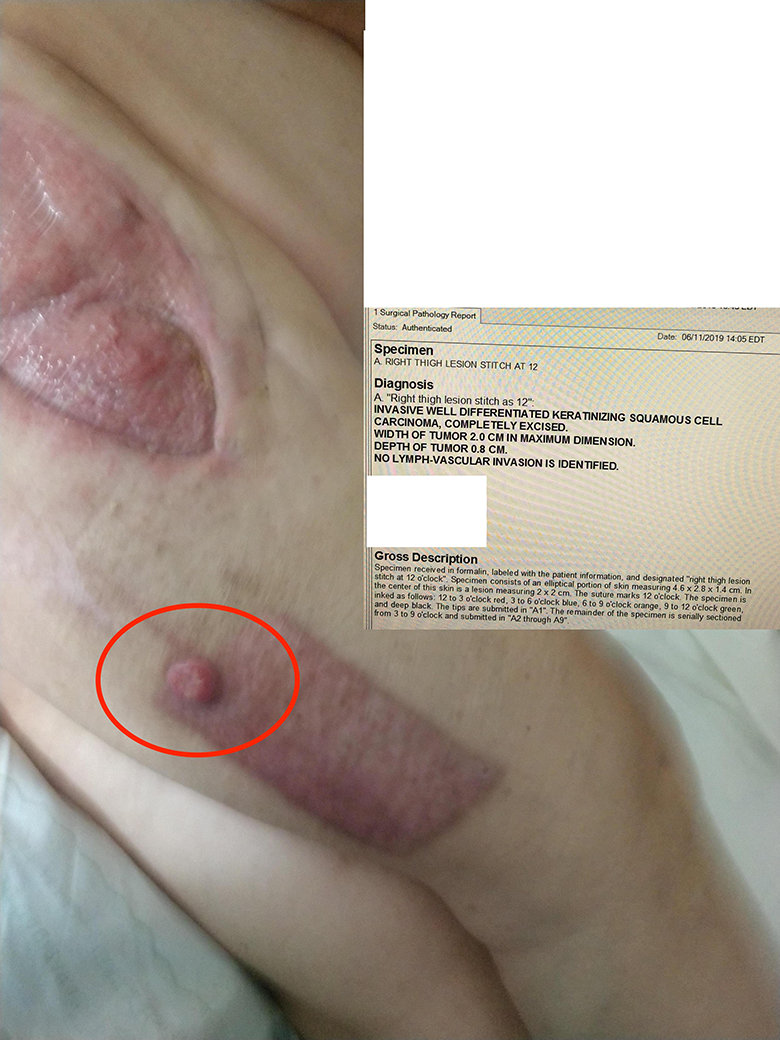
Exophytic, rubbery, fixed mass whose pathology results returned for well differentiated SCC after excision.
Well healed left sided reconstruction.
Well healed right sided reconstruction.
Discussion
The development of squamous cell carcinoma in skin graft donor sites is rarely encountered and less frequently reported on. In our review of the literature, we found a total of 16 documented cases. 5-10 Our case report represents a de novo occurrence of a well differentiated SCC at a skin graft donor site for reconstruction of chronically infected bilateral hip wounds. It can be inferred that our patient’s chronic wounds were a factor for the development of SCC at the donor site.
The presentation of SCC in skin graft donor sites has been reported in the setting of existing skin cancer or burns. Five of the previous case reports found SCC in skin graft donor sites in patients who were undergoing reconstruction with skin grafting for resected skin cancers, which indicates a potential relationship between the original cancer and the SCC arising at the donor site. 5 -9 One case report demonstrated the implantation of SCC after using the same hollow bore needle for injection in both the area around the skin cancer and the donor site, indicating the cancer may have seeded into the donor site. 6 Other case reports describe patients who developed SCC at the skin graft donor sites who had undergone reconstruction after burns. 10 Of 3 patients with donor site SCC after skin grafts for burns were completed, 1 patient developed numerous nodules both at the donor site and the burn sites.
Our case is one of the few reported occurrences in a patient without evidence of skin cancer or acute burns at time of reconstruction. We consider seeding or metastasis an unlikely cause of de novo malignancy in our patient given his negative intraoperative biopsy results. Our patient also did not report any burns or acute trauma to the skin prior to surgery, making this an unlikely etiology as well. However, despite the absence of these potential risk factors, SCC developed at the STSG donor site in the acute post-operative period.
The predisposing factors that contribute to this development of SCC at the donor site remain unclear. Seeding of the donor site with cancer cells in one postulated theory for the rise of SCC at skin graft donor sites. 6 Another case study theorizes that surgery itself induces a degree of immunosuppression. The consequent decrease in immune surveillance could allow metastasis into the healing donor site. 9 Research also suggests a link between inflammation and carcinogenesis. The increased cell proliferation, growth factors, and local blood flow associated with chronic inflammation may create a microenvironment that supports cancer growth. 11 In the setting of acute burns, a similar setting is provided for cancer cells to develop; evidence suggests that prolonged and repeated epithelization, in conjunction with immunosuppression, makes epidermal cells highly susceptible to carcinogenesis. 10
This rationale is potentially applicable to our patient. Despite the absence of malignancy or burn wounds, our patient had large sites of chronic inflammation that undoubtedly created an environment that could support growth of cancerous cells. 11 The combination of a chronically infected wound could have led to rampant cell proliferation, which may have promoted neoplasm formation.
Keratoacanthoma, a slow-growing benign skin tumor that appears most frequently in sun-exposed areas, should also be on any clinicians’ list of differential diagnoses when considering SCC of the skin graft donor site. Although new lesions after skin grafting may be exceedingly unlikely to be SCC, our experience with this rare case suggests prudence may be warranted. We propose that an acute lesion at the skin graft donor site in a patient who recently underwent reconstruction should undergo prompt biopsy and excision.
Conclusion
Squamous cell carcinoma of the donor site is a rare complication of skin grafting. Cases have most frequently been described in the setting of existing malignancy or burn reconstruction. We present a case of de novo SCC in a patient with chronically inflamed hip wounds to highlight another potential risk factor for the development of this condition in the setting of skin graft reconstruction. We emphasize the importance of early biopsy and removal of donor site SCC lesions in the prevention of disease evolution.
Footnotes
Authors’ Note
Written consent for the use of images has been provided by the patient.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statements
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants involved in the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies involving animals performed by any of the authors.
