Abstract
The potentially curative treatment for soft tissue sarcomas is wide resection. When located on the scalp, this can require removal of significant volumes of adjacent soft tissues as well as the skull periosteum. Consequently, reconstruction of the surgical defect is challenging. For patients receiving preoperative radiation or chemotherapy, gradual tissue expansion through the placement of a preoperative tissue expander can allow for primary closure of the wide resection defect, typically with hair-bearing scalp.
Keywords
Introduction
Soft tissue sarcomas (STS) are rare, representing only 1% of adult cancers. 1 The potential curative treatment for STS is wide resection, including a margin of grossly uninvolved tissue adjacent to the tumor. When there is a high risk of local recurrence, radiation therapy is often utilized. When compared to post-operative radiation, preoperative radiation therapy is associated with a smaller field size and lower total dose. Patients with STS at increased risk of occult distant metastases may also be considered for preoperative chemotherapy.
The head and neck location comprises only 4% to 10% of newly diagnosed STS. 1 Anatomically, the scalp has minimal laxity and is in close proximity to critical structures, such as the skull. In order to obtain negative resection margins, scalp STS frequently requires en bloc resection of the skull periosteum. This precludes simple skin graft coverage of the wide resection defect. Consequently, reconstruction of scalp STS wide resection defects can be technically challenging.
For patients undergoing preoperative radiation or chemotherapy, there are often 2 to 3 months of treatment prior to surgical wide resection. In that situation, there is ample time for preoperative tissue expansion of uninvolved adjacent scalp. This can potentially allow for primary scalp local rotation flap closure of the wide resection defect, including hair-bearing scalp. The following patient cases illustrate this technique, utilizing the surgical placement of tissue expanders underlying adjacent, uninvolved scalp.
Patient 1
The patient is a 32-year-old African American male with neurofibromatosis who developed synchronous left upper arm and right occipital scalp high-grade malignant peripheral nerve sheath tumors (MPNST). The right scalp MPNST was 5.4 cm, after previous excisional biopsy with positive margins. Staging imaging was negative for metastatic disease. Following multidisciplinary discussion, the plan was for preoperative radiation therapy to both the left upper arm and the right occipital scalp STS sites. Prior to the initiation of the scalp radiation, a 7 × 12 cm rectangular tissue expander was placed 5 cm away from the radiation field, underlying the contralateral (left sided) scalp (Figure 1). The expander was initially inflated to 30 mL. Over the next 3 months, the expander was filled every 1 to 2 weeks to a total of 245 cc, the last expansion occurring 1 week prior to surgery. Staged wide resections were performed of the left arm MPNST followed by the right scalp MPNST 3 weeks later. The size of the right occipital scalp defect was 15 × 10 cm, including resection of the underlying skull periosteum (Figure 2). A fasciocutaneous scalp flap was raised laterally from over the tissue expander, including several releasing incisions of the capsular scar. There was enough laxity of the expanded scalp to provide primary coverage of the wide resection defect. The incision was closed primarily over a surgical drain. The rotated scalp flap included normal, hair-bearing tissue. The patient was discharged on post-operative day 1 with no post-operative wound healing issues.
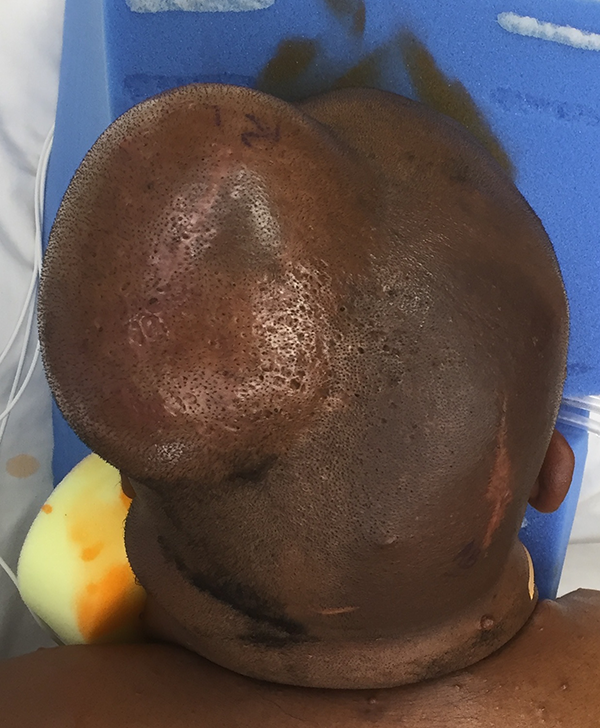
Preoperative scalp sarcoma with expander in place.
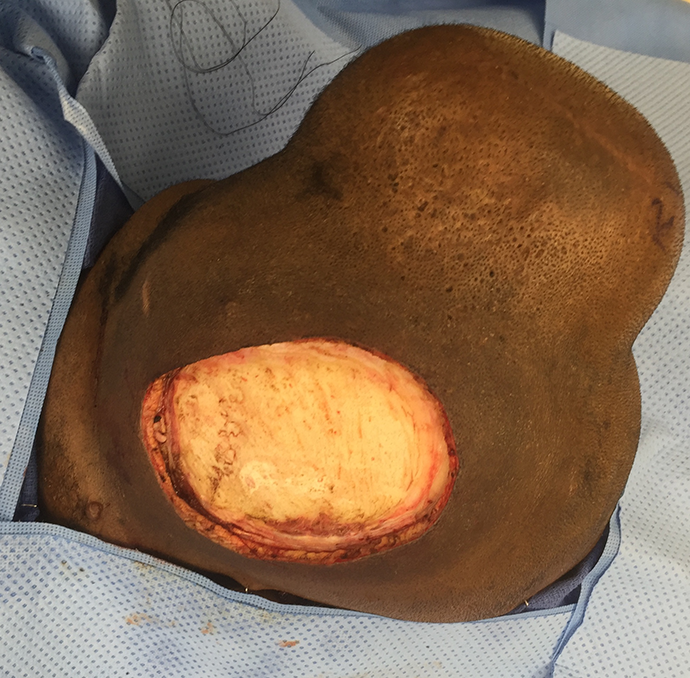
Post-resection of scalp sarcoma prior to advancement flap.
Patient 2
The patient is a 77-year-old white female with multifocal high-grade angiosarcoma of the right frontoparietal scalp. Staging imaging was negative for metastatic disease. After multidisciplinary discussion, the plan was for neoadjuvant paclitaxel chemotherapy secondary to a high risk for harboring occult distant metastases. An 11 × 9 cm tissue expander was surgically placed in the left parietal region through an incision adjacent to the left ear. Secondary to the patient’s distance from the treating facility, serial expansion to a total volume of 270 cc was performed closer to home by a local surgeon (Figure 3). Following 3 cycles of neoadjuvant chemotherapy, wide resection of the right scalp angiosarcoma was performed, including the underlying skull periosteum. The wide resection defect was 10 × 8 cm, covered with a rotational fasciocutaneous scalp flap from the expanded portion of the scalp (Figure 4). Primary closure over a surgical drain was performed, and the patient was discharged on post-operative day 1. There was slightly delayed healing of a small portion of the anterior aspect of the rotation scalp flap that healed by secondary intention.
Preoperative scalp sarcoma with expander in place.
Post-resection of scalp sarcoma prior to advancement flap.
Patient 3
The patient is an 81-year-old white female with a multifocal right frontotemporoparietal high-grade angiosarcoma. Staging imaging was negative for metastatic disease. After multidisciplinary discussion, the plan was for neoadjuvant paclitaxel chemotherapy secondary to a high risk for harboring occult distant metastases. Prior to initiating chemotherapy, a 10 × 8 cm crescent-shaped 250 cc volume tissue expander was placed. During chemotherapy, the expander was serially filled and 40- to 50-cc increments. Approximately 4 weeks into the expansion (to a total of 160 cc), there was progressive skin erosion overlying the expander, which required removal (Figure 5). Following completion of neoadjuvant chemotherapy, wide resection of the right frontotemporoparietal scalp angiosarcoma was performed, including the underlying skull periosteum (Figure 6). A fasciocutaneous scalp rotation flap was created from the previously expanded area. However, due to limited overall laxity, a complete primary closure was not possible. Therefore, a split thickness skin graft was used to cover a portion of the rotation flap donor site, which contained normal periosteum. There were no post-operative complications.
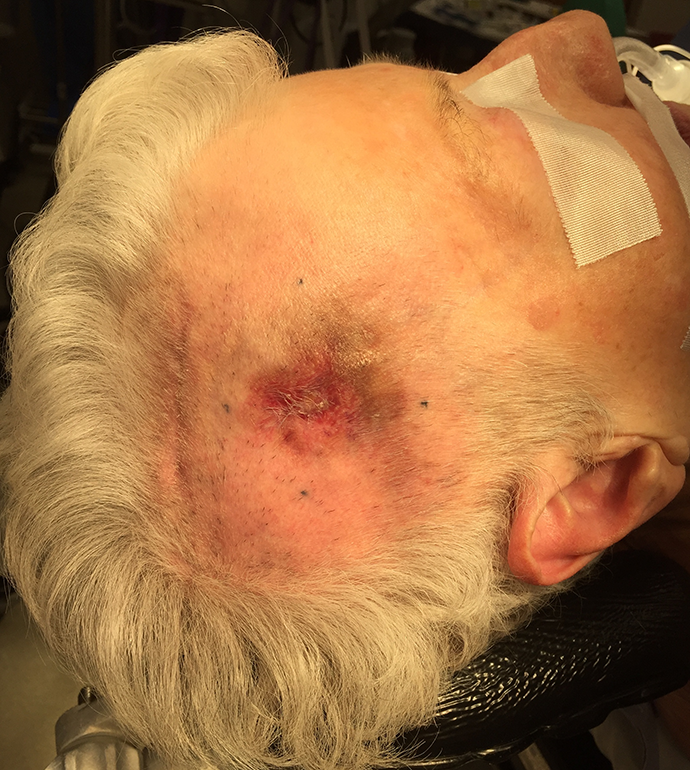
Preoperative scalp sarcoma (expander previously removed due to erosion).
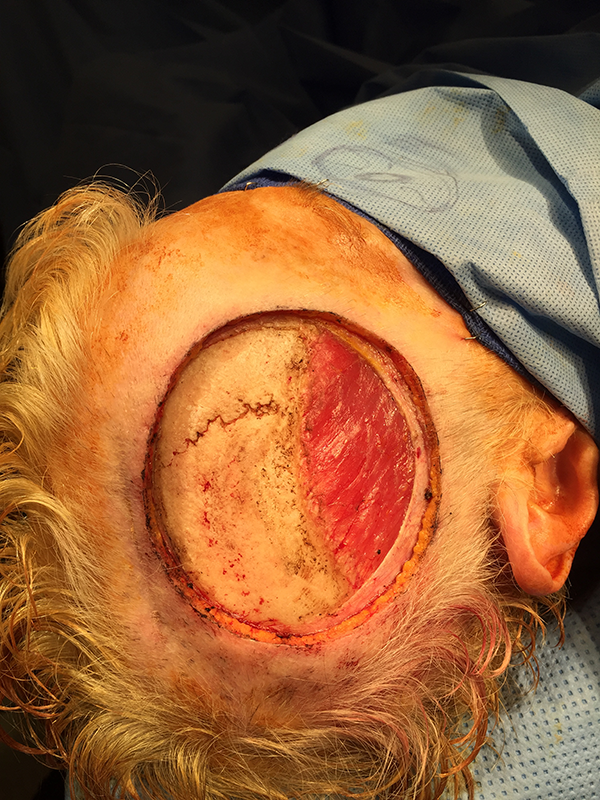
Post-resection of scalp sarcoma prior to advancement flap.
Patient 4
The patient is a 26-year-old white male without neurofibromatosis diagnosed with an 8 × 6 cm vertex scalp MPNST following positive margin excisional biopsy. Staging imaging was negative for metastatic disease. After multidisciplinary discussion, the plan was for preoperative radiation therapy. After consultation with plastic surgery regarding preoperative scalp expansion, it was felt that to separate tissue expanders would be placed. A 350-cc crescent-shaped expander was placed in the occipital region as well as a 100-cc rectangular expander in the right frontotemporoparietal region. Preoperative radiation therapy was administered at an outside facility, where the radiation field inadvertently included a portion of the scalp overlying the expanders. Total expansion during preoperative radiation was 190 cc for the occipital expander and 88 cc for the right frontotemporoparietal expander. Wide resection of the vertex scalp MPNST site was performed (including skull periosteum), resulting in a surgical defect of 8.5 × 7.5 cm (Figure 7). Primary closure over a drain was performed utilizing rotational fasciocutaneous scalp flaps from the regions of tissue expansion. Portions of the scalp rotation flaps had areas of alopecia due to inclusion in the radiation field. The patient was discharged on post-operative day 2 with no subsequent post-operative complications.
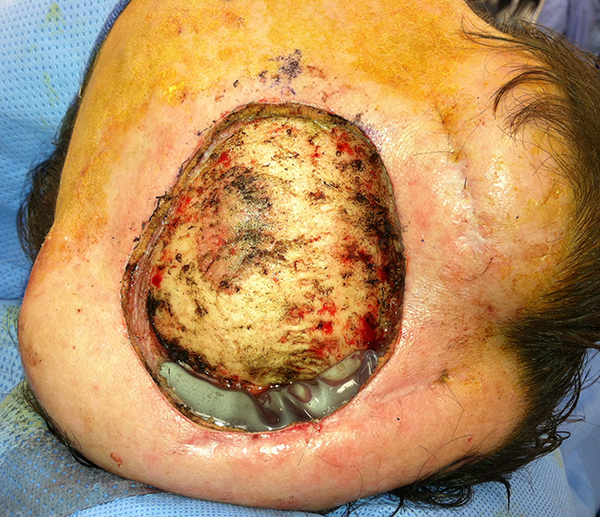
Post-resection of scalp sarcoma prior to advancement flap.
Patient 5
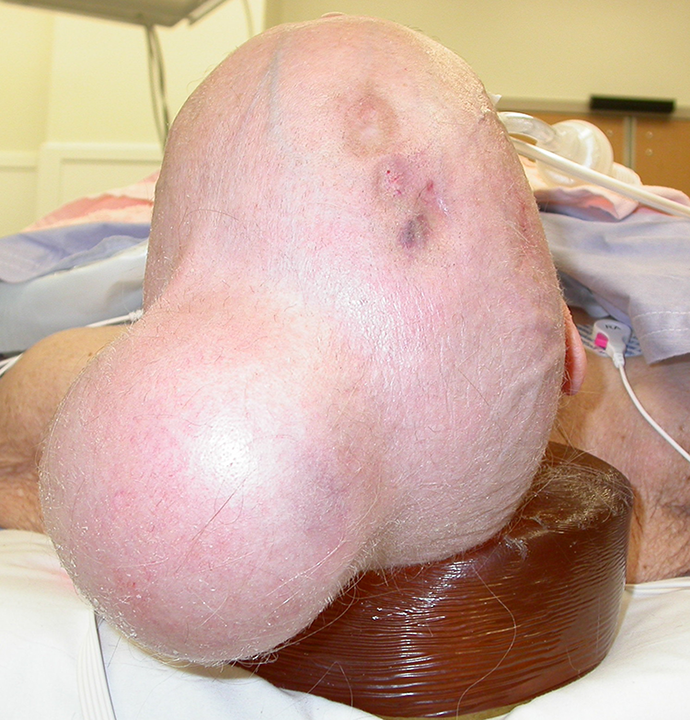
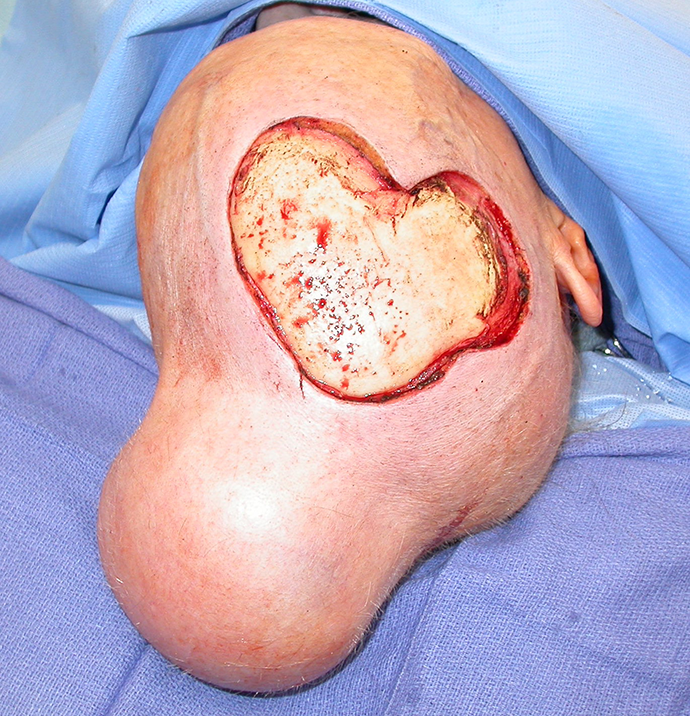
The patient is a 32-year-old white female without neurofibromatosis diagnosed with a 6.2-cm occipital scalp MPNST. Staging imaging was negative for metastatic disease. After multidisciplinary discussion, the plan was for preoperative radiation therapy. A 16-cm breast tissue expander was placed underlying the central frontoparietal scalp. Serial expansion was performed over a total of 6 weeks (Figure 8). Wide resection of the primary STS site was performed (including skull periosteum), resulting in an 11 × 9 cm defect (Figure 9). Primary closure of the wide resection defect was performed with a fasciocutaneous scalp advancement flap from overlying the tissue expander (Figure 10). This included abundant hair-bearing scalp. The patient was discharged on post-operative day 1. There was a small area of delayed wound healing at the edge of the flap that resolved with local wound care.
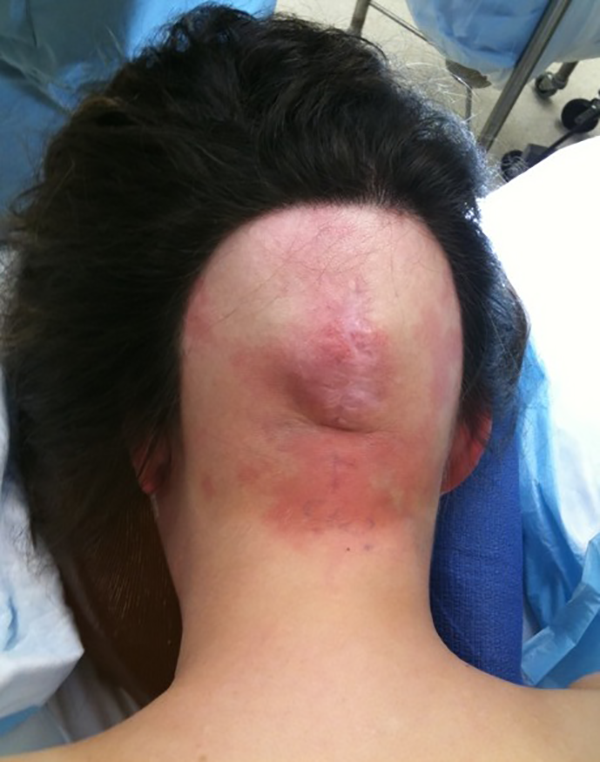
Preoperative scalp sarcoma with expander in place.
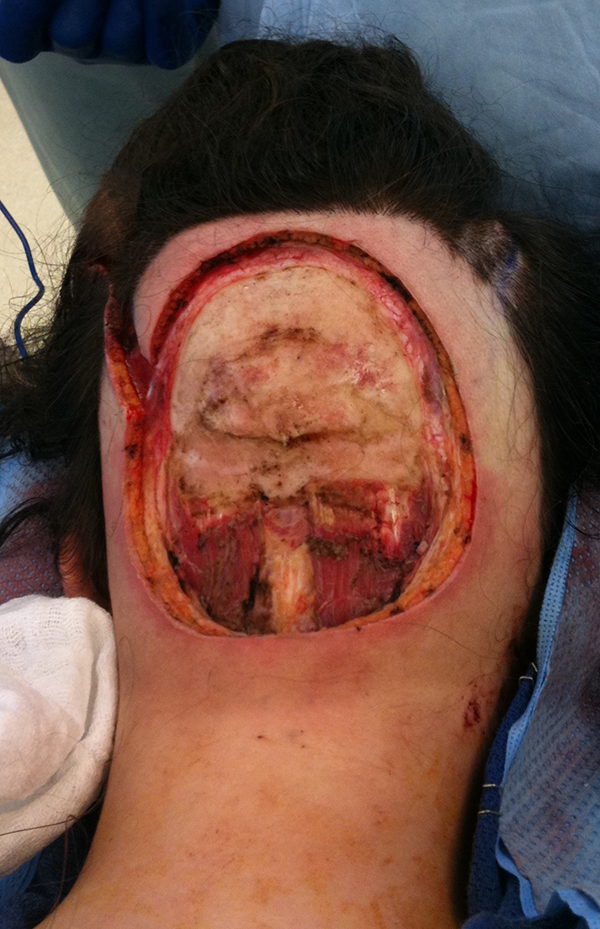
Intraoperative post-resection of scalp sarcoma.
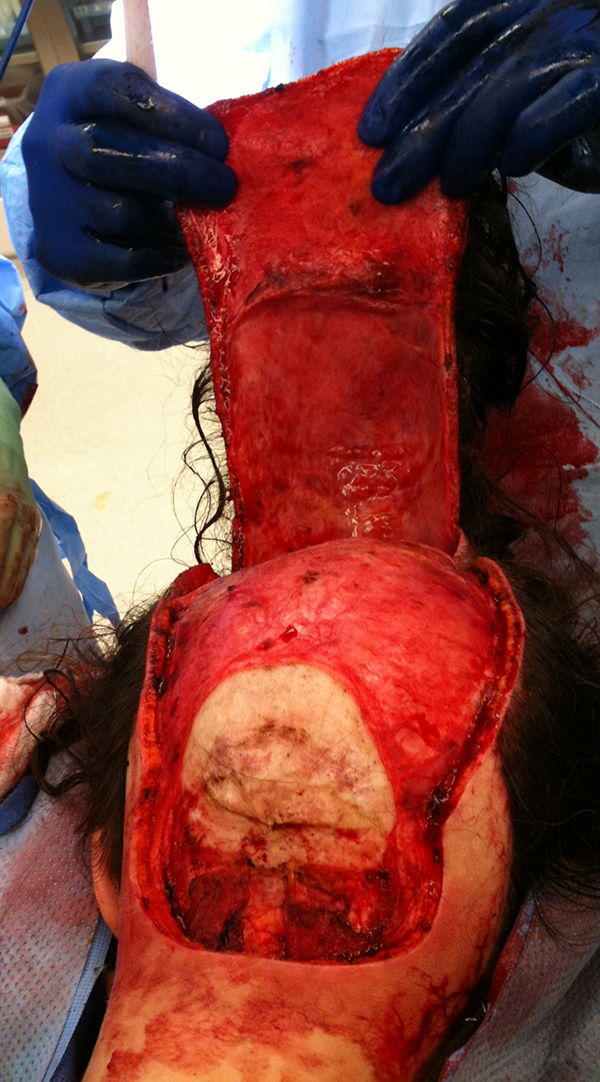
Advancement flap post-resection of scalp sarcoma.
Discussion
Tissue expansion is a natural phenomenon during pregnancy or following significant weight loss. The use of surgically placed tissue expanders was first described in 1956, where a balloon was placed in the temporal region in order to reconstruct a missing ear. 2,3 The concept of post-mastectomy breast reconstruction utilizing a temporary expander was subsequently described in 1982. 4,5 Although the physiologic basis of tissue expansion has remained the same, the technique and design of the expanders have significantly evolved over time. The scalp is inherently not very pliable. Consequently, primary closure of a scalp surgical resection defect is often not technically possible. For the surgical treatment of primary skin cancers of the scalp, the periosteum can be preserved. Therefore, a simple skin graft can be used to cover the surgical defect. However, a scalp STS wide resection requires en bloc removal of the underlying skull periosteum as an oncologically appropriate deep margin. This is also in a setting, where preoperative radiation may have been administered or post-operative radiation is being considered. These conditions prohibit the use of a skin graft for reconstruction. Although a scalp local rotation flap can be used to cover the STS wide resection defect, and the adjacent flap donor site (with intact periosteum) can be skin grafted, the skin grafted area can be very large, resulting in a significant area of permanent alopecia. In addition, scalp adjacent to the primary STS site may already have alopecia secondary to preoperative radiation. Large areas of scalp alopecia can be psychologically and emotionally stressful, especially for younger and female patients.
Some patients with a scalp STS will receive preoperative radiation or chemotherapy. For radiation, preoperative treatment is typically delivered over 5 weeks with an additional 4- to 8-week interval from the end of radiation until surgery. Most neoadjuvant chemotherapy regimens are 2 to 4 cycles, occurring over a comparable number of months. This period of preoperative therapy also provides a unique “window of opportunity” for preoperative tissue expansion. For a scalp STS, adequate expansion of the adjacent, uninvolved scalp can allow for primary closure of an otherwise complicated wide resection defect. Through the use of this technique, it is often possible to primarily close the wide resection defect using normal hair-bearing scalp, resulting in acceptable cosmetic outcomes without the need for more extensive techniques. 6,7
Tissue expansion of the scalp generally occurs through a biphasic progression. In the early stage of scalp expansion, there is a period of resistance, lasting several weeks, before the galeal barrier is gradually broken down. After the first process is complete, skin compliance markedly increases, allowing for the desired extent of tissue expansion. 7
Preoperative scalp tissue expansion requires close coordination between the members of the multispecialty care team, especially in the setting of preoperative radiation. Consequently, team communication is critical. The sarcoma surgical oncologist should identify the planned location and margins for the eventual wide resection, allowing the plastic surgeon to place the tissue expanders well away from the primary tumor and outside the surgical field. A reasonable estimation of the anticipated wide resection defect size also helps with planning the location, number, and final volume of the expanders. The radiation oncologist should create the preoperative radiation treatment plan prior to placement of the tissue expanders so that the plastic surgeon can place the expanders well outside the radiation field, if technically possible. The scalp expanders can then be surgically placed immediately prior to the start of radiation with a minimal risk for expander erosion through irradiated scalp. The placement of the expanders outside the radiation field will also minimize radiation associated alopecia involving the expanded scalp (which will be used for the reconstruction) and will also reduce the need for significant changes to the radiation treatment plan due to volume changes from the ongoing expansion. Finally, the expanders should be placed at location relative to the radiation treatment volume that will permit ongoing expansion without interfering with the immobilization or reproducibility of the daily radiation treatment setup. Following expander placement, the patient can proceed with simulation and the radiation treatment plan can be generated. It is often helpful to fuse a preliminary radiation planning scan prior to expander placement with the subsequent actual treatment planning scan as artifact from the tissue expander can obscure target definition and normal tissue anatomy.
Complications of tissue expansion include temporary hair loss, expander migration, seroma, infection, and hematoma. 6 Fortunately, the head and neck location has the lowest rate of complications, presumably due to the robust blood supply. 8 Skin necrosis overlying the expander is the complication that has the greatest negative impact on the treatment plan, as this requires immediate removal of the expander, frequently with subsequent loss of all of the previously gained expansion. This occurred in patient 3 in our series, which was the only patient who required adjacent scalp flap donor site skin grafting. Typically, the skin becomes necrotic secondary to pressure-induced ischemia from the underlying expander. This can be multifactorial: radiation-induced vascular changes, creation of thin skin flaps, pre-existing poor skin quality, and medical co-morbidities (diabetes, collagen vascular disorders, etc). 3 Therefore, the expander should be placed in a location that will allow the patient to sleep and perform activities of daily living without excessive pressure/stress on the area. Finally, there is a component of psychological distress with preoperative scalp expansion. Most of our patients had some concerns regarding the cosmetic appearance of the highly visible expanders, including undesired attention in public situations. It is important to refocus the patients on the ultimate long-term cosmetic benefit of primary closure of the wide resection defect with hair bearing scalp.
In conclusion, preoperative radiation or chemotherapy as part of the multimodality treatment of a scalp STS provides an adequate time frame for preoperative scalp tissue expansion. This technique allows for primary closure of the oncologically appropriate wide resection defect (ie, absent skull periosteum) with normal hair bearing scalp. In our experience, complications have been minimal and primary closure without the need for skin grafting was achieved in the majority of patients. The implementation of this preoperative scalp tissue expansion technique requires close communication and coordination between the plastic surgeon and the other members of the oncologic care team.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
The procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 and 2008.
Statement of Informed Consent
All participants included in the study provided informed consent for their photographs and information as seen in the manuscript.
