Abstract
Pyoderma gangrenosum (PG) is a rare dermatologic condition manifesting as sterile, rapidly progressing painful skin ulcerations. It is commonly associated with autoimmune disorders such as inflammatory bowel disease. This case report illustrates a severe case of post-surgical pyoderma gangrenosum (PSPG) following bilateral deep inferior epigastric perforator flap breast reconstruction in a 56-year-old Caucasian woman. Factors delaying diagnosis included negative personal/family history of inflammatory disorders and a clinical presentation more suggestive of breast cellulitis. Refractory antibiotic therapy, subsequent ulceration at multiple breast and abdominal incisions, and pathergic response to tissue biopsy supported an eventual clinical diagnosis of PSPG, 12 days postoperatively, despite equivocal histopathologic findings. Initiation of intravenous immunoglobulin, prednisone therapy, local wound care, and avoidance of debridement led to a full resolution by 5 months postoperatively. This study raises awareness for PSPG and highlights the importance of early recognition and urgent dermatology consultation to expedite optimal treatment and minimize subsequent fat necrosis.
Introduction
Pyoderma gangrenosum (PG) is a non-infectious neutrophilic dermatologic condition 1 that manifests as rapidly progressive, sterile pustules, evolving into deep, painful ulcerations with violaceous borders and undermined wound edges. 2,3 Pyoderma gangrenosum occurs in as few as 3 to 10 patients per million per year. 2 Pyoderma gangrenosum is notoriously considered a diagnosis of exclusion and is often misdiagnosed in its early stages, usually for infectious etiologies such as cellulitis or necrotizing fasciitis. 1 Although PG can be associated with autoimmune conditions such as rheumatoid arthritis and inflammatory bowel disease (IBD) or paraneoplastic presentations, 4 post-surgical pyoderma gangrenosum (PSPG) is also a distinct PG entity, especially following breast surgery. 5 Early recognition, initiation of immunosuppressive therapy, and avoidance of pathergic interventions such as wound debridement are considered the most effective treatment principles. 5,6
We present a rare case of PSPG following bilateral deep inferior epigastric perforator (DIEP) free flap breast reconstruction at an academic hospital. The patient had no past medical history of inflammatory disorders. This case report is presented in accordance with Delphi consensus-based Surgical CAse REport guidelines. 7
Case Presentation
A 56-year-old non-smoker Caucasian woman with locally advanced ductal carcinoma (Stage T3, N3) of the left breast was referred to our clinic for a breast reconstruction consultation 1 month after having left breast lumpectomy and axillary node dissection and subsequent left breast mastectomy. Her past medical history included gastroesophageal reflux disease and overactive bladder. She had no personal or family history of inflammatory or autoimmune disorders. The patient received neoadjuvant chemotherapy (AC-T and tamoxifen), adjuvant left breast radiotherapy, and a right breast prophylactic mastectomy as part of her oncologic treatment. The patient was consented for bilateral DIEP breast reconstruction, approximately 2 years after her initial oncologic surgical treatment.
The bilateral DIEP reconstruction was performed with no intraoperative complications. On the fourth postoperative day (POD 4), the patient was discharged afebrile with an unremarkable postoperative course and a slightly elevated white blood cell (WBC) count of 15.8 × 109 cells/L. She had intermittent fevers between POD 0 and POD3, with maximal temperature (T max) of 38.7°C on POD 3, although her fevers were not attributable to pneumonia, deep vein thrombosis, urinary tract infection, or surgical site infection. Return instructions were given to present to the emergency department if there were signs of local or systemic infection. Prophylactic oral clindamycin was prescribed, as the patient had a penicillin allergy, and was continued as long as the breast and abdominal drains were in situ.
On POD 6, the patient returned for her first follow-up since discharge. She reported a 2-day history of worsening erythema and tenderness of the left breast, intermittent fevers, and chills. The left DIEP flap had normal capillary refill and turgor. Serous drainage was noted from the left lateral breast incision site with no wound dehiscence. The patient was admitted to hospital on intravenous (IV) vancomycin with a presumed diagnosis of left breast cellulitis as she had fever (38.1°C), leukocytosis (WBC 24.3), and tachycardia with heart rate (HR) of 105. The infectious diseases (IDs) service was consulted on the date of admission and recommended changing empiric antibiotics to IV cefazolin and IV metronidazole prior to ordering left breast drain cultures and blood cultures. The breast drain output was serosanguinous throughout the admission.
On POD 7, the right breast developed a small area of erythema at the medial surgical incision with no ulceration, which was attributed to fat necrosis. Some epidermal sloughing was noted on the left breast at the initial site of cellulitis on admission. Preliminary blood cultures were negative. Interventional radiology was consulted to place a drain into a fluid collection underneath the left breast flap on POD 8, and drain cultures were subsequently negative. Leukocytosis fluctuated between POD 7 and POD 11 (WBC 21.6-34.8), and the patient remained intermittently febrile and tachycardic. Per os (PO) ciprofloxacin and IV vancomycin were initiated on POD 9.
By POD 11, erythema at the abdominal incision was noted and the bilateral breast incisions began to develop violaceous ulcerations. A punch biopsy was taken from the left abdominal wound at the border of ulceration and normal skin. The patient underwent a septic workup and was transferred to our surgical intensive care unit. At this point, the patient was febrile at 38.1°C with WBC 31.1, lactate 2.3 mM, and hemoglobin of 79 g/L. The biopsy results were equivocal, yielding a differential diagnosis of infection, ischemia, or PG. Of note, the biopsy itself initiated a pathergic response that was clinically observed. The biopsy did serve to rule out other possible entities, such as vasculitis, calciphylaxis, atypical mycobacterium, and deep fungal infections.
On POD 12, the ID service excluded infection as the primary diagnosis and discontinued antibiotic therapy, with the exception of IV cefazolin, which was continued until a definitive diagnosis was made. The dermatology service was consulted and confirmed a clinical diagnosis of PG. Intravenous immunoglobulin therapy was initiated on POD 12 for 3 days and the IV cefazolin was discontinued. The patient’s leukocytosis, tachycardia, and febrile episodes improved shortly after the immunosuppressant course was initiated. On POD 14, a PO prednisone taper was initiated at an initial daily dose of 75 mg. Pathergy related to frequent dressing changes was minimized by utilizing Acticoat silver-impregnated dressings, changed every 3 days, which also helped to prevent secondary infection of the ulcerated lesions. The maximal extent of wound ulceration was documented on POD 18 (Figure 1).
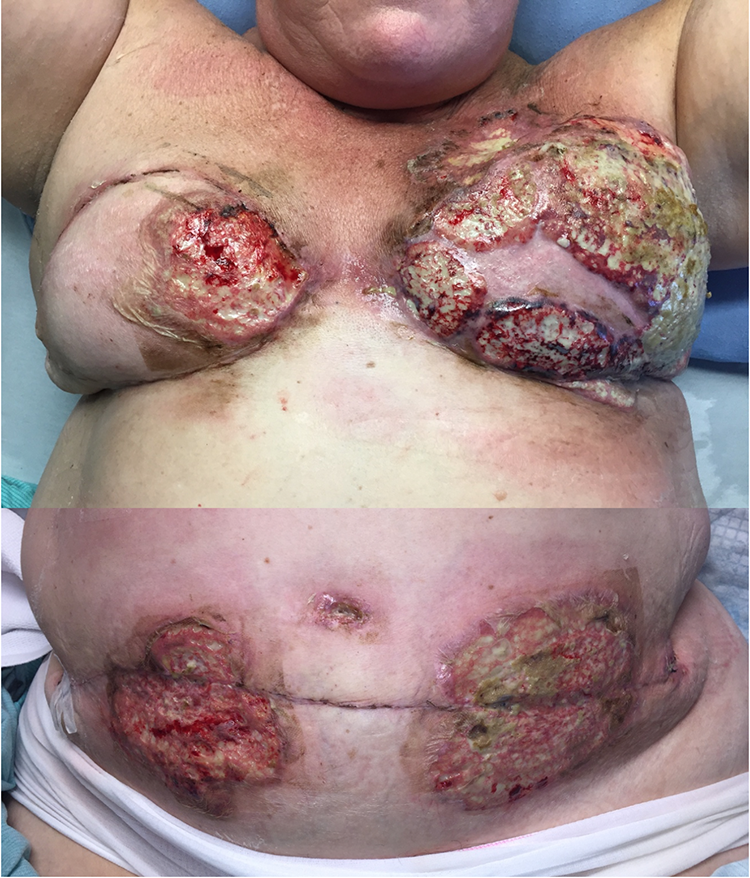
Appearance of PSPG 18 days following bilateral DIEP flap breast reconstruction. The involvement of bilateral breasts, umbilicus, and bilateral abdominal incisions represents the maximal extent of PG ulceration in this patient. DIEP indicates deep inferior epigastric perforator; PG, pyoderma gangrenosum; PSPG, post-surgical pyoderma gangrenosum.
By POD 19, the patient’s vitals (T 37.1°C, HR 71) and bloodwork (WBC 17.2, hemoglobin 90 g/L) stabilized, and she was ambulating and tolerating a full diet. She was discharged home on prednisone 60 mg PO daily with a tapered prescription that involved decreasing prednisone by 10 mg every 5 days to 40 mg, then slowly tapering over the ensuing 4 months. The tapering regime was determined by evidence of wound healing and pain levels.
Since discharge, the patient has had follow-up with her plastic surgeon and dermatologist (the senior authors of this study), initially weekly and then on a monthly basis. The left breast developed an eschar that was unroofed 5 weeks postoperatively and lifted by 9 weeks postoperatively with local wound care using intrasite gel. The right breast and abdominal ulcerations were first to re-epithelialize and were declared healed by 3 months postoperatively (Figure 2). Topical tacrolimus (calcineurin inhibitor) applied directly to the wound was noted to expedite wound healing. The patient was treated at one point for infection of the left breast wound with PO cefalexin and ciprofloxacin, and topical dilute acetic acid for a short period of time. She did not require readmission at any point and her PSPG wounds were declared healed at 5 months postoperatively (Figure 3), although there was significantly decreased left breast volume due to PSPG-related fat necrosis as compared to the right breast.
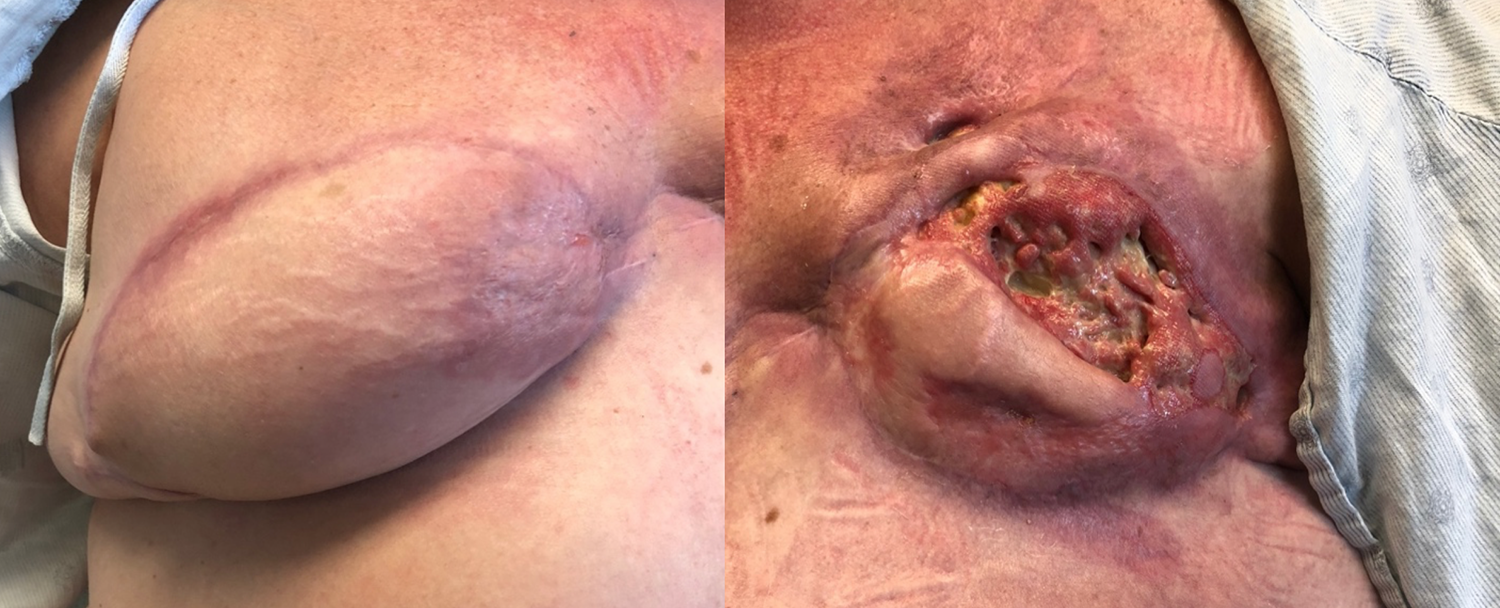
Appearance of PSPG 3 months following bilateral DIEP flap breast reconstruction. PSPG is entirely resolved for the right breast with ongoing prednisone therapy, while the left breast continues to receive local wound care and monitoring. There is significant fat necrosis of the left breast flap. DIEP indicates deep inferior epigastric perforator; PSPG, post-surgical pyoderma gangrenosum.
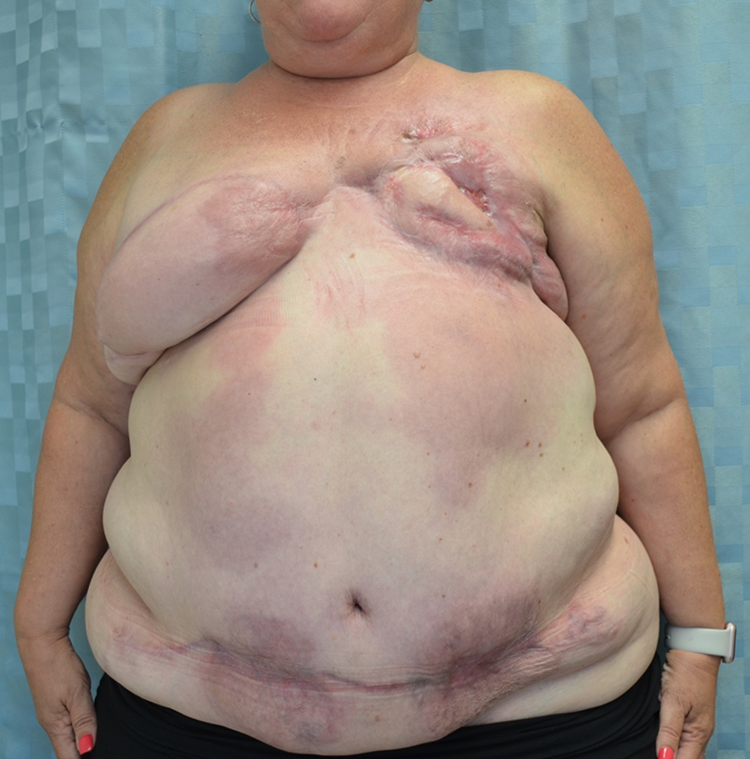
Appearance of PSPG 5 months following bilateral DIEP flap breast reconstruction. There is a complete resolution of ulceration with discontinuation of prednisone therapy and local wound care. Fat necrosis has resulted in significant volume loss of the reconstructed left breast. DIEP indicates deep inferior epigastric perforator; PSPG, post-surgical pyoderma gangrenosum.
Discussion
Pyoderma gangrenosum is a diagnosis of exclusion and may masquerade clinically as vasculitis, malignancy, drug reaction, exogenous tissue injury, or infection, as in our case report. 1,8 In a recent review of 2273 patients with PG, 77.4% of cases were initially misdiagnosed, with the most common misdiagnosis being cellulitis in 9.4% of cases. 2 In the study population, the mean age was 56 years, 66.4% of participants were female, and 71.1% were Caucasian. 2 These demographic findings are in keeping with our patient, a 56-year-old Caucasian woman.
Post-surgical pyoderma gangrenosum most commonly manifests between 4 days and 6 weeks postoperatively as ulcerated surgical wounds, with predominant involvement of the breast and abdomen, as compared to classic PG, which preferentially involves the extremities. 3 In a systematic review of 220 PSPG cases, 25% followed breast surgery, which represented the most common surgical etiology. 5 Twenty-five percent of the breast surgery cases were reconstructive in nature. It is unclear why breast surgery is the most common patient group for PSPG, but it may be due to a reporting bias in the literature. 5 In the same review, only 5.8% of PSPG patients had a medical history of IBD and 3.6% had rheumatoid arthritis, while 8.6% had a history of hematologic malignancy and 16.8% had a past history of PG. 5
In our case report, the patient presented with fever and leukocytosis. Tuffaha et al 6 performed a systematic review of 49 PSPG breast cases and reported that fever and leukocytosis occurred in 55% and 43% of cases, respectively. In 2 recent systematic reviews, 18 (86%) of 21 cases of PSPG following breast reconstruction, including DIEP and TRAM flaps, developed donor-site PG. 6,8 Abdominal donor-site involvement was present in our case and aided in recognition and diagnosis of PSPG.
Tissue biopsy was not diagnostic in our case of PSPG, although it did result in a mild, yet noticeable, pathergic response that aided diagnosis. Other research groups have suggested forgoing tissue biopsy and focusing instead on a clinical diagnosis of PG. 8 The consultant dermatologist involved in our patient’s inpatient care was confident in the PG diagnosis despite the equivocal pathology results. Indeed, previous PG diagnostic criteria proposed by Su et al 9 featured major criteria that did not require histopathologic confirmation. Instead, major criteria included the rapid progression (1-2 cm per day or 50% increase in 1 month) of a necrotic ulcer with pain out of proportion on clinical examination. According to Su et al, 9 other minor criteria suggestive of PG include the presence of ulceration secondary to pathergy, cribriform scarring, associated systemic diseases, histopathological clues (eg, sterile dermal neutrophilia with inflammation), and rapid decrease in ulceration size (at least 50%) within 1 month of starting systemic steroids. Thus, histopathology can be used to aid diagnosis, but it is not necessary or definitive.
In terms of treatment, a number of approaches have been attempted, with some showing a consistent success. Systemic steroids, cyclosporine, infliximab, and canakinumab have the strongest evidence as treatment regimens for PG 6,10 and are considered first-line therapy based on a recent literature review of 704 PG cases, 10 although a limited number of high-quality studies exist that address this topic. Application of topical tacrolimus, a T-lymphocyte activation inhibitor, has also been reported to expedite healing and reduce the duration of corticosteroid treatment. 11,12 Surgical treatment of PG is generally contraindicated and, if pursued, requires preoperative initiation of systemic immunosuppression. 13 Immunosuppressive tapering is to be cautiously employed once there is stabilization of PG wounds. 5 In general, debridement and reconstructive surgery (ie, skin grafting, flap coverage) should be delayed until PG resolution and optimization of perioperative steroid therapy are achieved. 5 In a systematic review of 16 cases of post-autologous breast reconstruction PG, 63% (10/16) underwent debridement, 25% (4/16) had no debridement, and 12% (2/16) were unknown. 8 Tuffaha et al 6 reported debridement in 67% (33/49) of cases involving breast PSPG. A 20-year retrospective chart review of 18 patients with PSPG by Tolkachjov et al 14 found that debridement was performed in 61% (11/18) of patients. In a large review of all types of PG, Sasor et al 2 found that only 5.3% of 2273 cases were treated with debridement. Any future reconstructive surgeries following PSPG should be approached with caution, as recurrence is most likely in patients with a past history of PG. 5 In the case of our patient, reconstruction to correct the volume loss of the left breast secondary to fat necrosis was contraindicated due to risk of PSPG recurrence.
Overall, the key elements in the successful treatment of PSPG in our patient case included urgent dermatology consultation once the diagnosis was suspected, initiation of immunosuppressive treatment, local wound care, and specific avoidance of debridement. The ulcerated wounds were able to heal secondarily and did not require skin grafting or subsequent reconstructive surgery. Earlier PSPG diagnosis and treatment is important to minimize volume loss from fat necrosis.
Footnotes
Statement of Human and Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5).
Statement of Informed Consent
Informed consent was obtained from all patients for being included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
