Abstract
Background
Adherence to a Mediterranean Diet (MeD) has been associated with lower disease severity in patients with psoriasis. However, the mechanism behind how this diet may lead to disease modification remain understudied. Recent studies have revealed dysbiosis of the gut microbiome in patients with psoriasis suggestive of inflammation and altered immune regulation. Diet affects the gut microbiome and this review aims to evaluate whether correcting this dysbiosis may be one theoretical mechanism by which the MeD may be associated with lower psoriasis severity.
Methods
A literature search of the PubMed database was conducted for the terms 1) ‘psoriasis’ and ‘microbiome’ or ‘microbiota,’ and 2) ‘Mediterranean diet’ and ‘microbiome’ or ‘microbiota’ with manual screening for relevant articles. In total, we identified 9 relevant primary research studies investigating the gut microbiome in patients with psoriasis and 16 relevant primary research studies investigating changes in the microbiota for those consuming a MeD.
Results
Though varying in exact levels of certain bacteria, studies analyzing the microbiome in psoriasis revealed dysbiosis. Those analyzing the effect of the Mediterranean diet on the microbiome revealed beneficial changes, including alleviating some of the same alterations seen in the microbiome of those with psoriasis.
Conclusion
Microbiota change is a possible mechanism why the MeD has previously been associated with lower psoriasis severity.
Keywords
Introduction
Psoriasis is a chronic inflammatory skin disorder, affecting 2%–4% of the Western population with incidence continuing to increase.1,2 Psoriasis affects more than just skin, and is associated with co-morbidities such as cardiovascular disease, metabolic syndrome, and psoriatic arthritis 1
While there have been exciting developments in medical treatments for psoriasis, the effects of lifestyle factors remains understudied. Lifestyle modification, especially dietary changes, is of interest to patients. 3 Studies show that most patients living with psoriasis have tried dietary changes in an attempt to control their disease. 4
One diet of interest is the Mediterranean diet (MeD) due to its anti-inflammatory effects. 5 The MeD focuses on increased consumption of nutrient and anti-oxidant rich foods such as vegetables, olive oil, and legumes while limiting meat and dairy. 6 The diet has been widely studied with benefits such as improved cardiometabolic health and slowed cognitive decline.5,6 In the few studies examining psoriasis and the MeD, results suggest the diet may be helpful in reducing psoriasis severity. 7 For instance, a cohort study of 3557 psoriasis patients found that dietary patterns more consistent with the MeD were associated with a lower psoriasis severity. 8 Given the MeD’s many benefits, efforts have been made to determine by what physiologic mechanism the MeD causes its positive impact. 6 Its effect on the gut microbiome is one mechanism that has been explored.
The gut microbiome is a population of microbes in the gastrointestinal tract, which impact metabolism and immune functioning. 9 While the gut microbiome’s existence has been known for many years, its association with disease and ability to be manipulated for better health outcomes is an exciting area of interest. The consumption of different foods alters the gut microbiome, with some microbiota associated with good health, and others associated with pro-inflammatory effects potentially leading to obesity and insulin resistance. 9
When discussing the microbiome, one should be familiar with the concepts of alpha- and beta-diversity, microbial diversity, microbial richness, and bacterial taxonomy. Microbial richness can be described as the number of taxa while microbial diversity is the different types of taxa. Alpha-diversity encompasses abundance, while beta-diversity is the variability in the identity of taxa within that community.
10
The taxonomic classifications of bacteria are important as changes can occur at any of these levels (Figure 1). An example of bacterial taxonomy in the gut.
11
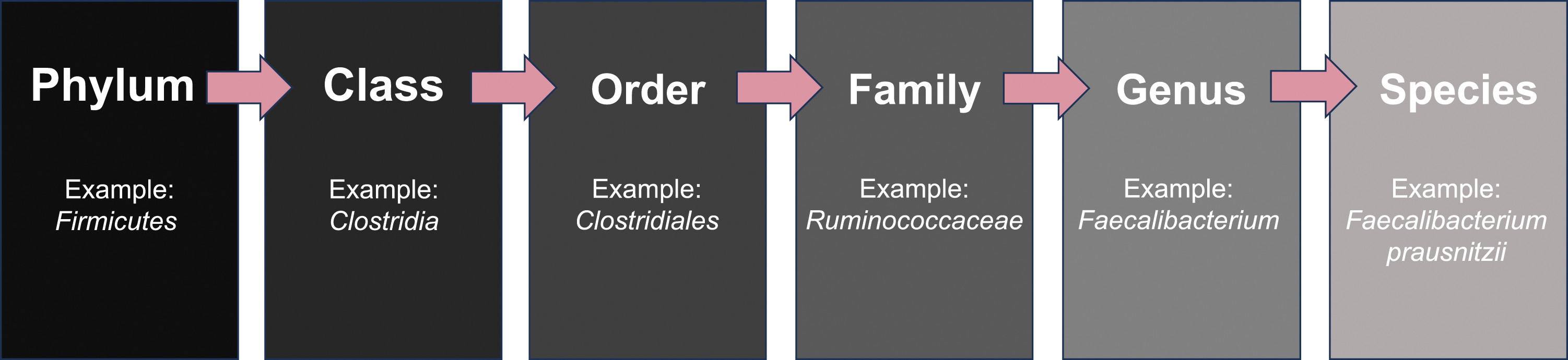
In this review, we evaluate what is known about the gut microbiome in patients with psoriasis and in those who follow a MeD. We also propose that changes in gut microbiota induced by a MeD as a theoretical mechanism for why the diet is associated with lower disease severity.
Methods
A literature search of the PubMed database was conducted for the terms 1) ‘psoriasis’ and ‘microbiome’ or ‘microbiota, ’ and 2) ‘Mediterranean diet’ and ‘microbiome’ or ‘microbiota’ Our search was limited to English-language articles and those published prior to August 11th, 2023. For review, the authors manually identified relevant articles discussing the gut microbiome and psoriasis or the MeD specifically. Duplicate articles were excluded. In total, we identified 9 relevant primary research studies investigating the gut microbiome in patients with psoriasis and 16 relevant primary research studies investigating changes in the microbiota for those consuming a MeD as the study group.
On September 8th, 2023, our literature search was updated to include an article published August 31st, 2023.
Results
The Gut Microbiome in Patients With Psoriasis
Complete Differences Across Studies Evaluating the Gut Microbiome in Patients With Psoriasis Separated by Taxonomic Groupings. A “-“ Indicates No Significant Findings in That Taxonomic Grouping for the Study, or the Study Did Not Perform Analysis Within That Grouping. F/B = Firmicutes/Bacteroidetes Ratio. Pso = Psoriasis. PsA = Psoriatic Arthritis. HC = Healthy Control.
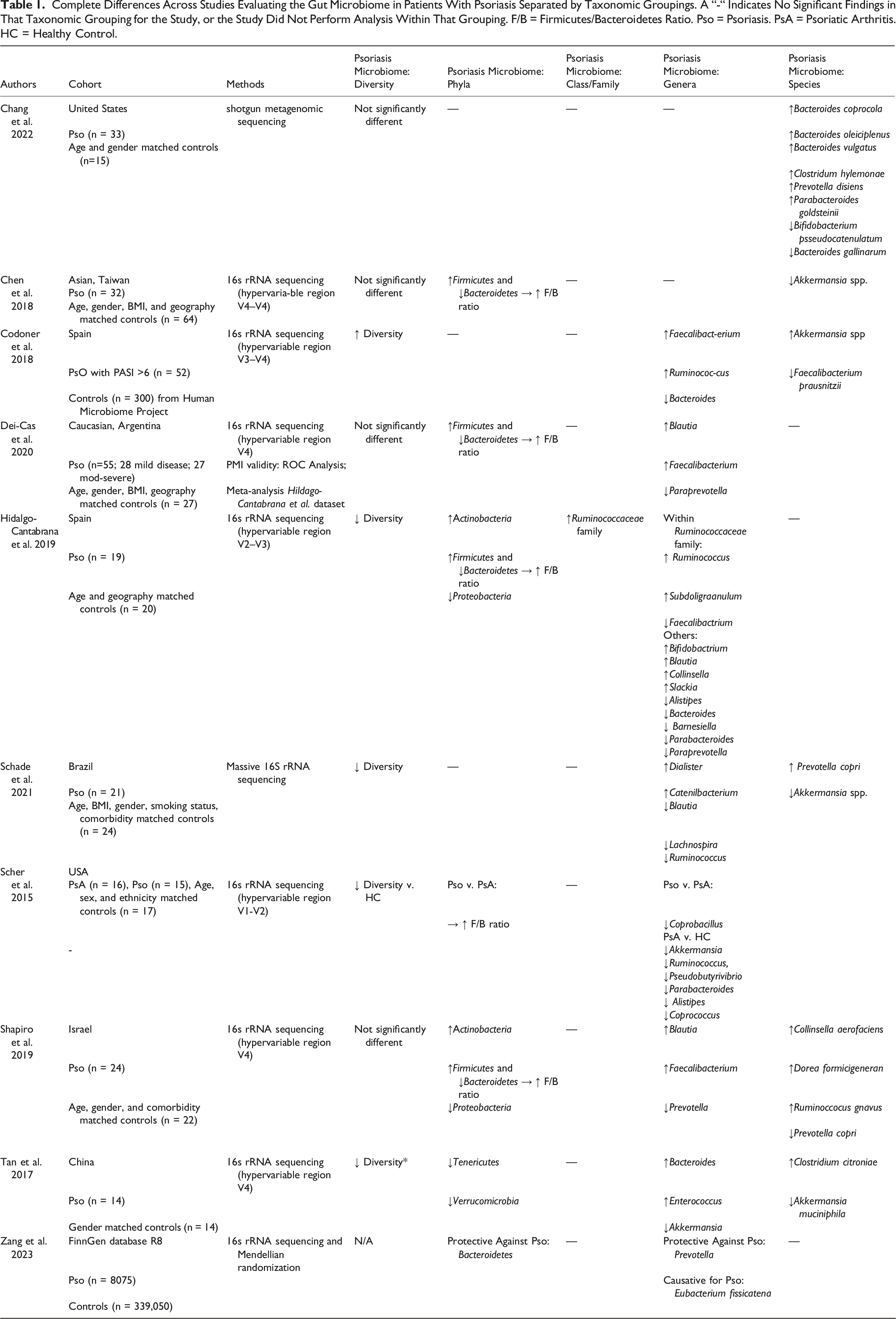
As seen in the cladogram in Figure 1, taxonomic categorization of bacteria begins at the phylum and is followed by class. 11 At the phyla level, the human gut microbiome is composed primarily of the Firmicutes and Bacteroidetes. High levels of Firmicutes compared to Bacteroidetes (increased F/B ratio) has been correlated with greater BMI and higher levels of inflammatory markers.21,22 In a 2018 Taiwanese study by Chen et al, researchers found an increased Firmicutes and decreased Bacteroidetes phyla in psoriasis patients when compared to age, gender, BMI, and geography matched controls, leading to an increased F/B ratio. Increased F/B ratios in psoriasis patients were found in three additional studies: a Caucasian cohort (n = 55) evaluated by Dei-Cas et al, Spanish psoriasis patients (n = 19) evaluated by Hidalgo-Cantabrana et al, and a cohort from Israel (n = 24) evaluated by Shapiro et al. 18 Short chain fatty acids (SCFAs) such as butyrate are produced by some bacteria and have been suggested to promote epithelial integrity and exert an anti-inflammatory effect. 23 A higher F/B ratio leads to altered SCFA production – including decreasing butyrate - potentially explaining the negative metabolic associations. 24 Interestingly, there was a reduction in the Bacteroidetes phylum found in the 2015 study by Scher et al 12 ’s study in psoriasis patients vs those with psoriatic arthritis, presumably raising the F/B ratio, though this was not commented on specifically and this finding did not reach significance. The most recent study on the gut microbiome in psoriasis performed mendelian randomization on published large-scale genome wide association studies comparing greater than 10000 patients with psoriasis to healthy controls. They found that Bacteroidetes have a protective role in psoriasis (OR .81 [95% CI .67-.98]), further supporting the findings of low Bacteroidetes in psoriasis patients from previous studies. 25
The next level of taxonomy is the genera level. In terms of significant findings for Scher at al., Parabacteroides genus was reduced compared to healthy controls which was replicated in one other study.12,17 Tan et al sequenced the microbiome in a Chinese cohort (n = 14) finding several significant changes, including an increase in the Bacteroides genus and decrease in the Akkermansia genus including the Akkermansia muciniphila species. Higher Bacteroides is typically seen in animal-based diets, and though some species in the genus have beneficial properties, an overgrowth can degrade important intestinal mucus leading to intestinal inflammation and is associated with colonic cancer. 26 Bacteroides can also be thought of in the context of the Prevotella/Bacteroides (P/B) ratio, where a high ratio (lower Bacteroides) is associated with improved metabolic status. 27 Decreases in Akkermansia muciniphila are associated with chronic inflammatory states including obesity. 19 This decrease in the Akkermansia genus was replicated in a cohort of psoriasis patients (n = 32) in Taiwan and another cohort (n = 21) from Brazil.13,15 However, other studies have challenged these findings with Codoner et al finding an increase in the Akkermansia genus and a decrease in the Bacteroides genus in their cohort of 52 psoriasis patients from Spain when compared to unmatched controls (n = 300) from the Human Microbiome Project. This decrease in Bacteroides was also seen in a separate cohort from Spain (n = 19) compared to age and geography matched controls. 17 These differences between studies may be due to the differences in severity of psoriasis, matching protocols used for controls, and geography of the study populations particularly considering both cohorts with a decrease in the Bacteroides genus were from Spain whereas 2/3 studies that found an Akkermansia genus decrease evaluated East Asian patients. Another consistency across both the studies out of Spain was increased Blautia genus in the psoriasis groups.14,17 However, despite analyzing the microbiome of patients from the same country of Spain, Codoner et al and Hidalgo-Cantabrana et al did have some conflicting findings, with Codoner et al finding increased Faecalibacterium genus which was supported by an additional study, while Hidalgo et al found a decrease in psoriasis patients.14,16,17 From the mendelian randomization study, the genus Prevotella was found to possibly have a protective impact (OR .87 [95% CI: .76-1.00] whereas Eubacterium fissicatena conferred an increase risk for psoriasis (OR 1.22 [95% CI: 1.10-1.35]). 25 The Prevotella genus was found to be decreased in a psoriasis cohort in a study that took place in Israel. 18 In the same family as Prevotella, the genus Paraprevotella was found to be decreased in two studies.13,17
Within genera, there are a few species of particular interest such as the discussed Akkermansia muciniphila species. Similarly, within the Faecalibacterium genus, one notable species is Faecalibacterium prausnitzii. Which is one of the main butyrate producers in the colon and its presence is reduced in inflammatory intestinal disorders. 28 In their study, Codoner et al found that although the Faecalibacterium genus was increased in their psoriasis cohort, F prausnitzii was actually reduced. 16 Hidalgo-Cantabrana et al did not report to the species level, though presumably F prausnitzii was reduced given the genus was reduced as a whole. 14 Prevotella copri is a member of the Prevotella genus, and two studies had conflicting significant results regarding levels in psoriasis patients.13,18 This species is associated with high fiber, low fat diets and often linked with desirable health, although conflicting reports exist. 29
The Gut Microbiome in Patients Consuming a Mediterranean Diet
Complete Differences Across Studies Evaluating the Gut Microbiome in Patients Following the Mediterranean Diet and Other Lifestyle Interventions. Changes in Microbiota Separated by Taxonomic Groupings. A “-“ Indicates No Significant Findings in That Taxonomic Grouping for the Study, or the Study Did Not Perform Analysis Within That Grouping. F/B = Firmicutes/Bacteroidetes Ratio. Pso = Psoriasis. WD = Western Diet. MD = Mediterranean Diet. FF = Fast Food. BMI = Body Mass Index. MetS-OB = Metabolic Syndrome and Obesity. NAFLD = non-alcoholic Fatty Liver Disease. VD = Vegetarian Diet. HIV = Human Immunodeficiency Virus.
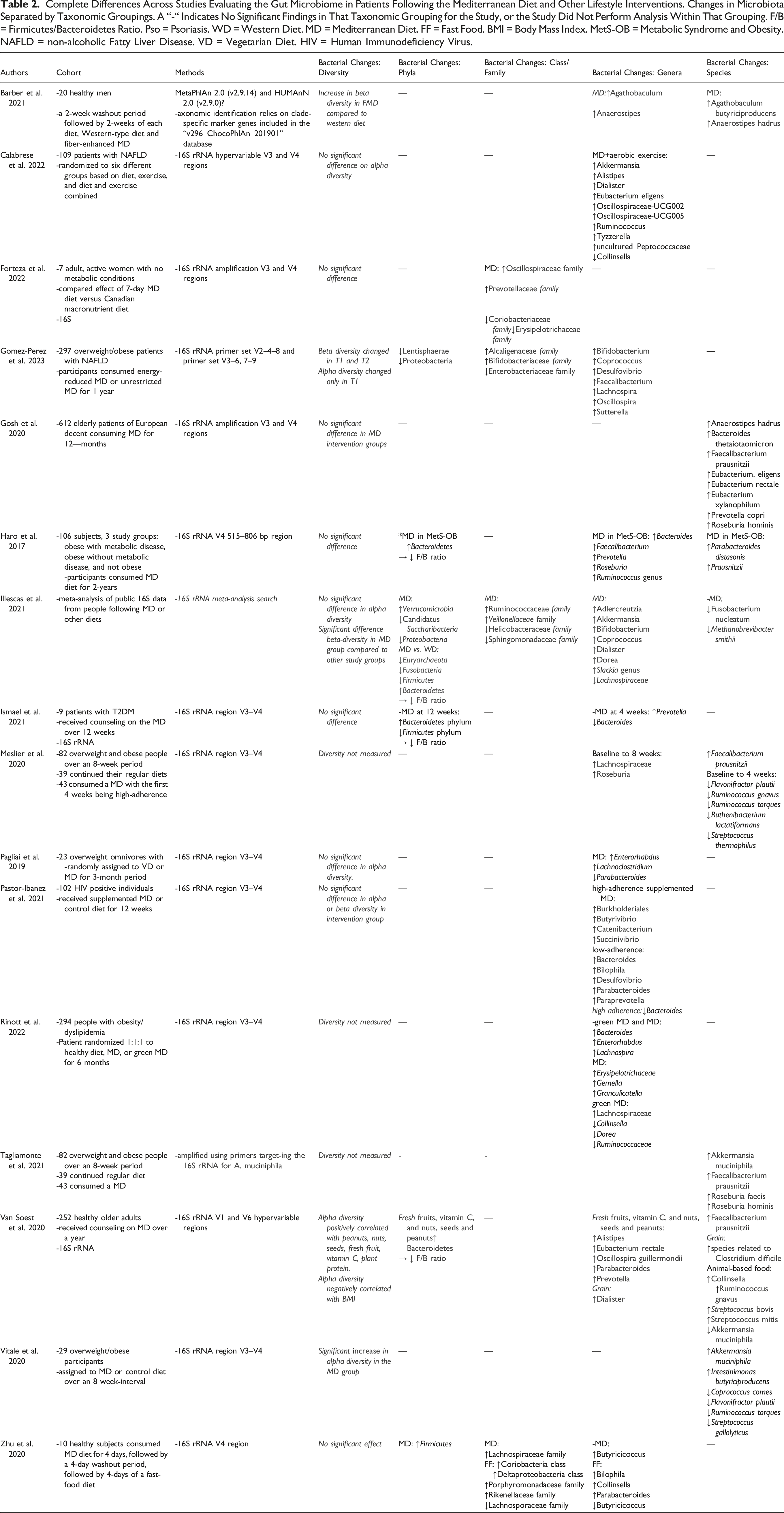
Starting at the phyla level, Haro et al 32 found that obese patients with metabolic syndrome were found to have decreased Bacteroidetes phylum at baseline which was increased following the MeD diet. Similarly, Van Soest et al, 21 Ismael et al, and Illesecas et al showed an increase in Bacteroidetes phylum following the MeD.21,30,34 Van Soest specifically showed a positive relationship between plant-based carbohydrates and the Bacteroidetes phylum and an inverse relationship between protein and fat. Zhu et al evaluated the MeD vs a fast food (FF) diet. They found that although the phylum Firmicutes was increased following the MeD, 40 there was no significant change found in F/B ratio, which was consistent with one additional study.9,19,36 Similar to previous studies associating a high F/B ratio with inflammation/obesity, Zhu et al found F/B ratio to be negatively correlated with HDL-C, emphasizing a possible negative metabolic impact.21,40
Genera was widely analyzed by the reviewed studies. Pagliali et al found a decrease in Parabacteroides following the MeD, while Zhu et al found an increase in this genus following a FF diet.38,40 Parabacteroides has previously been associated with hypertension. 43 Ismael et al 30 quantified their results after varying time points, and found that after 4 weeks of MeD counseling, a higher ratio of Prevotella to Bacteriodes was detected. This P/B ratio has been examined in other studies such as Meslier et al 42 who found that the MeD may improve insulin sensitivity in patients with high baseline levels of the generally inflammatory Bacteroides genus and lower levels of Prevotella genus. The anti-inflammatory Akkermansia genus was increased by the MeD in two studies.34,36
Finally, at the most individualistic level there are species. Van Soest et al and Meslier et al found that animal product-rich foods resulted in a significant elevation in species associated with inflammation such as R gnavus, while plant-based food led to an increase in anti-inflammatory species such as F prausnitzii, emphasizing the importance of a vegetable centric diet, such as the MeD for gut health.21,42 Tagliamonte also found a significant increase in F prausnitzii, in addition to Akkermansia muciniphila with the MeD which is decreased in T2DM, hypertension, obesity and IBD. 44 Vitale et al found similar results in their cohort of 29 overweight/obese individuals, with a significant increase in beneficial bacteria such as Akkermansia muciniphila, and a decrease in proinflammatory species. Specifically Akkermansia muciniphila is associated with increased production of butyrate, improving glucose metabolism and insulin sensitivity. 35 Results of Barber et al. study of 20 healthy similarly showed an increase in the butyrate-producing, colon protective species of Akkermensia genus. 41
Discussion
Based on the dysbiosis found in psoriasis patients and the possible changes induced by the MeD on the gut microbiome, this may be one mechanism that the MeD may exert benefit for these patients. One way is by decreasing the F/B ratio by increasing Bacteroidetes as found in 4 studies and thus helping restore SCFA balance and promoting butyrate production.21,30-33,45 This in turn could alleviate intestinal and systemic inflammation, having profound implications for psoriasis. Additional butyrate producers of the gut include species in the Akkermansia genus and Faecalibacterium prausnitzii, thus the MeD also promotes SCFA production through increasing these bacteria.21,31,32,34–37,42,44 This is of particular interest as the Akkermansia genus was found to be low in several studies on psoriasis and F prausnitzii or the Faacalibacterium genus as a whole was found to be reduced in two studies12,13,15,19 Overall, the MeD has been suggested to alter the microbiota in several ways to promote an immune-regulated and anti-inflammatory state that would presumably be beneficial to psoriasis.
Cardiovascular disease, metabolic syndrome, and obesity are co-morbidities associated with psoriasis and also can be improved by the MeD. 1 The study by Haro et al suggests the MeD-induced microbiota changes may be more pronounced in those with obesity and/or metabolic syndrome. The MeD diet reduces the risk of cardiovascular disease, type 2 diabetes, and neurodegenerative diseases.5,46 In Rinott et al, 45 the researchers employed the Green MED score to determine adherence to a plant-based diet. They discovered that higher adherence to the Green MED diet, which reduces animal-based food consumption even further than the traditional MeD, resulted in a more significant shift in the microbiome, as well as a reduction in markers linked to cardiometabolic disease. This is another argument for encouraging this dietary pattern in patients with psoriasis. By adopting a MeD, one has the potential to not only improve their skin disease but alter their microbiome in a way that improves overall health and wellness. This is especially pertinent to psoriasis patients, given their higher propensity to develop cardiovascular and metabolic co-morbidities. 47 Many studies proved the MeD diet’s positive effects as it pertain to metabolic syndrome by evaluating markers such as insulin sensitivity and hypertension.30,42,45
The precise foods that impart beneficial effects within the MeD has also been studied. 5 The MeD diet has a similar total fat intake to western diets, however more of that fat comes from omega-3s such as in fish and monounsaturated fatty acids such as olive oil vs saturated fats. 5 There is evidence that this pattern of fat intake reduces LDL and triglycerides while raising beneficial HDL cholesterol, providing cardiovascular benefit. 5 Furthermore, high omega-3 intake impacts prostaglandin metabolism by suppressing pro-inflammatory pathways, such has cycloxygenase-2 (COX-2), one of the targets of non-steroidal anti-inflammatory medications. 48 It is also important to note the lower intake of dairy products and land meats in the MeD than the western diet when considering its metabolic advantages. Furthermore, the high fiber in the MeD has a direct impact on the gut microbiome, positively influencing colonic production of butyrate. High fiber intake decreases risk of insulin resistance, another benefit. 5
Notably, Dei-cas et al developed a Psoriasis-Microbiota Index (PMI) that could discriminate between psoriasis patients and controls with high sensitivity and specificity. This study is the first to propose a PMI with the ability to discriminate between psoriasis patients and age-sex-and BMI matched controls and between samples from communities of different continents by performing a meta-analysis.
14
The successful development of a PMI suggests a signature dysbiosis of psoriasis patients Figure 2. The colon on the left represents the consequences of some of the proposed dysbiosis in psoriasis patients that the Mediterranean diet may alleviate. The colon on the right represents the relevant changes seen in the Mediterranean diet. The foods listed above the arrow are those eaten in large amounts in the Mediterranean diet, and those below the arrow are eaten sparingly.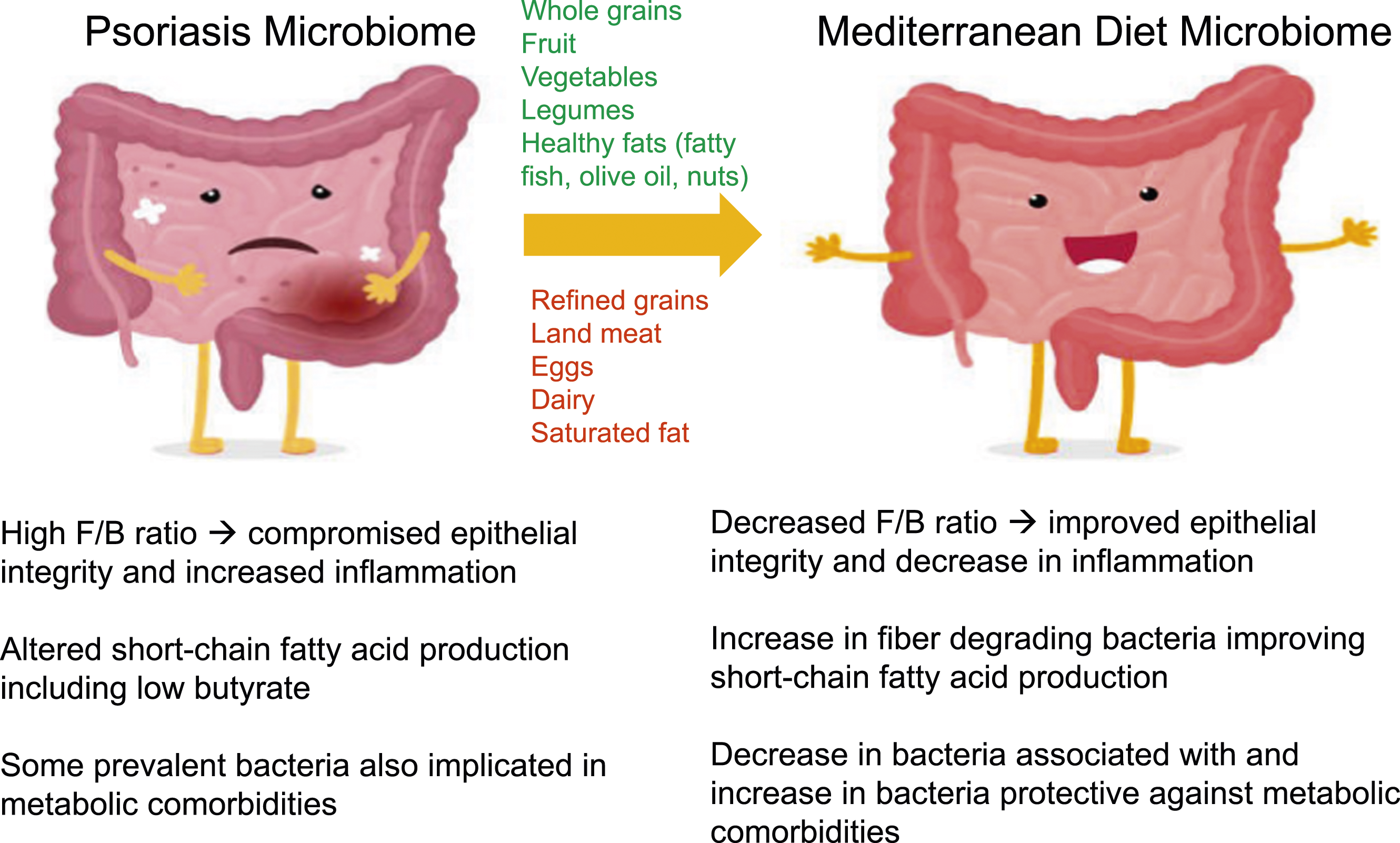
However, an important consideration is the ability to adhere to the MeD. Studies have indicated that significant changes in the microbiome only occur with a high level of adherence over extended periods of time. This suggests that to fully reap the benefits of microbial changes, one should follow a high-adherence pattern. In Meslier et al 42 the most significant changes in the microbiome were present during the high-adherence period, as defined by the MeD index. Pastor-Ibanez utilized a scoring system called MEDAS, with a score of 10 or higher being considered as high adherence. The study found significant results only in the high-adherence group, such as an increase in the presence of the fiber-degrading bacteria Burkholderiales and the anti-inflammatory bacteria Bifidobacterium and Lactobacillus. 39 Studies that showed no change in microbiota were shorter duration (4-7 days) with less time for the diet to be followed.33,40 To achieve a meaningful, anti-inflammatory change in one’s microbiome, the stricter one adheres to the MeD and for a longer timeframe the greater likelihood to reap benefits. A limitation of the studies evaluating the microbiome in those following a MeD was the study populations were not patients with psoriasis. Furthermore, it is feasible that an individual who adheres closely to a diet may also adhere more closely to their prescribed medications, which themselves will impact psoriasis severity and have even been suggested to affect the gut microbiome. 49
Conclusion
The MeD has numerous health benefits including an association with lower psoriasis severity. A relationship between the gut microbiome and the MeD has been established. The changes induced by the MeD in the gut microbiome may possibly be beneficial for patients with psoriasis based on the dysbiosis patterns found in several studies, but this cannot be said with certainty as none of the MeD-microbiome studies were performed using psoriasis patients. Regardless, given the potential benefit for psoriasis and the overall state of health promoted by the MeD, there is little harm in recommending this diet to psoriasis patients who are interested in exploring if diet can impact their disease. Patients who do pursue a MeD should not stop their prescribed topical or systemic medicines.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the T.B. is currently a principal investigator for studies being sponsored by Amgen, Castle, CorEvitas, Pfizer, and Regeneron. She has additional research funding from Novartis and Regeneron. She has served as an advisor for Abbvie, Arcutis, Boehringer-Ingelheim, Bristol Myers Squibb, Dermavant, Janssen, Leo, Lilly, Pfizer, Novartis, Sanofi, Sun, and UCB. W.L. has received research grant funding from Amgen, Janssen, Leo, Novartis, Pfizer, Regeneron, and TRex Bio.
CME Credit
To claim CME credit: https://www.eeds.com/em/8102
