Abstract
Background
Despite recent advances in biologics, there is a lack of significant evidence regarding the comparative efficacy of biologics in treating more resistant features of psoriasis, namely nail psoriasis. A systematic review synthesizing data from multiple studies is efficacious in assessing the comparative efficacy among biologics for the treatment of nail psoriasis.
Objective
To evaluate and compare the efficacy of biologics for the treatment of nail psoriasis.
Methods
Utilizing PRISMA guidelines, a systematic literature review was conducted using the Pubmed database on November 16, 2022. Studies selected were phase 3 or 4 randomized clinical trials, clinical studies, or other randomized trials with data on the treatment with biologics for adults with nail psoriasis.
Results
Sixteen studies meeting inclusion criteria were included for analysis. At 24 weeks, the highest mean NAPSI percent improvement achieved at week 24 was by brodalumab (76.9%) followed by etanercept (74%) and ixekizumab (70.5%) while the biologics achieving the greatest proportion of NAPSI 0 were adalimumab (44.6%) and ixekizumab (41%).
Conclusions
This study helps elucidate the comparative efficacy of biologics for the treatment of nail psoriasis. This review suggests that brodalumab and etanercept are associated with the highest percent improvement in nail psoriasis while adalimumab and ixekizumab are associated with the greatest probability of complete nail resolution.
Introduction
There have been great advances in the last few decades regarding biologics as treatment options for patients with psoriasis. Advances in biologic treatment of psoriasis include targeting specific immune pathways to inhibit certain cytokines including tumor necrosis factor (TNF), interleukin (IL)-17, and IL-23. 1 The US Food and Drug Administration and the European Medicines Agency have approved the use of the following drugs for moderate to severe psoriasis/psoriatic arthritis (PsA): secukinumab, ixekizumab, and brodalumab (IL-17 inhibitors), ustekinumab (IL-12/23 inhibitor), tildrakizumab-asmn, guselkumab, and risankizumab-rzaa (IL-23 inhibitors), adalimumab, etanercept, infliximab, golimumab, and certolizumab pegol (tumor necrosis factor inhibitors), and abatacept (costimulation modulator). 1
Even with these new therapeutics, there is a lack of significant evidence on the comparative efficacy of biologics in treating more resistant features of psoriasis, such as nail involvement. Although there have been some recent head-to-head studies comparing efficacy of various biologics, there continues to be a paucity of evidence in comparing the wide variety of biologics currently available. Additionally, determining which biologic to use for patients with certain features can be challenging without significant data on direct comparison as all biologics differ slightly in safety, dosing, and efficacy. 1 Therefore, a systematic review synthesizing data from multiple studies is efficacious in assessing the comparative efficacy among biologics for the treatment of nail psoriasis.
Methods
Search strategy
Utilizing PRISMA guidelines, a systematic literature review was conducted using the Pubmed database on November 16, 2022 and updated on December 4, 2022. The query terms used included: “biologics and psoriasis and nails.” Additional studies which may have been missed during the query were found by referencing comprehensive review articles. The selection process is illustrated below (Figure 1). Selection process.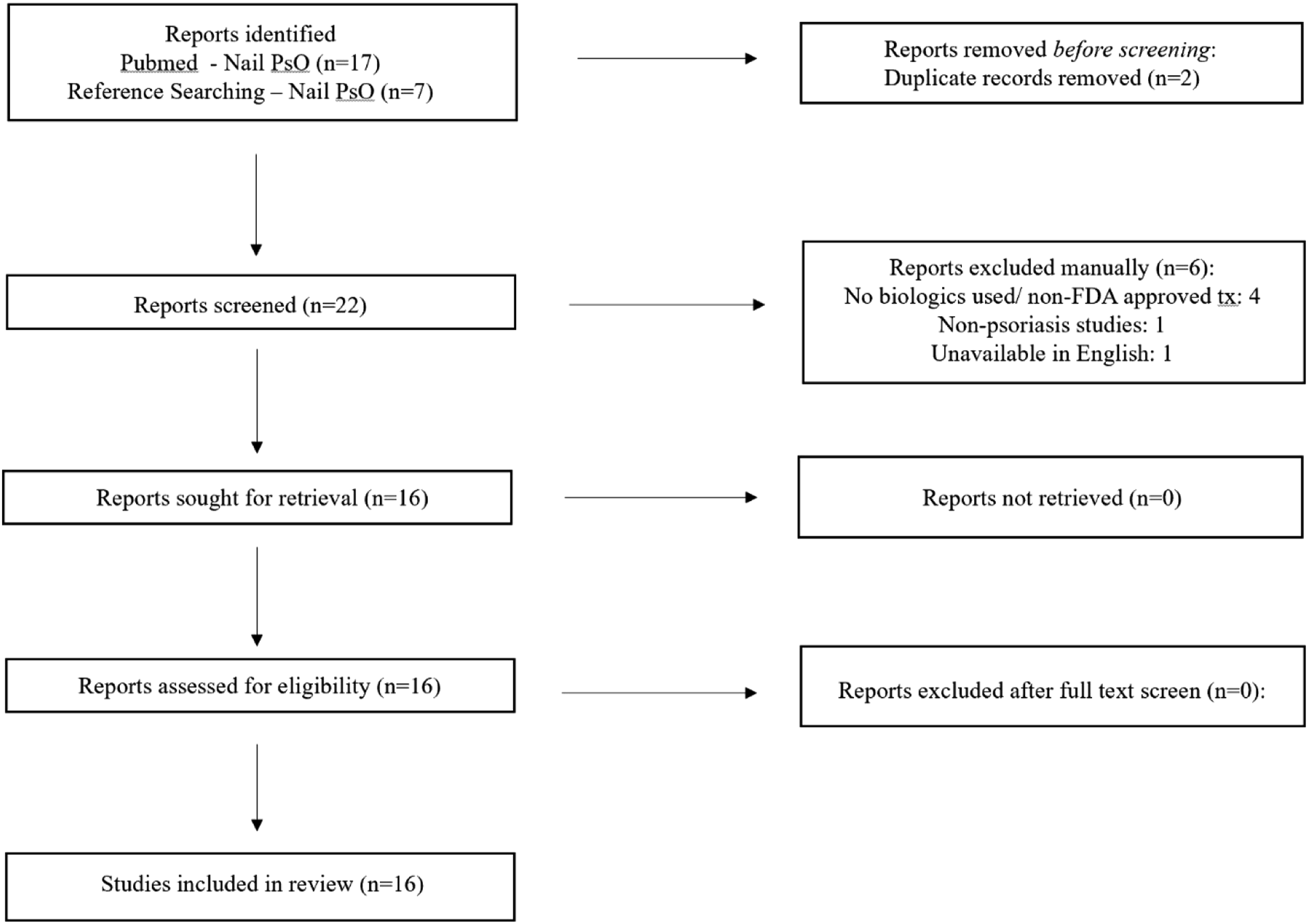
Inclusion and exclusion criteria
Inclusion criteria consisted of phase 3 or 4 randomized clinical trials, clinical studies, or other randomized trials (i.e. head to head comparisons with blinded assessors) of treatment with biologics for adults with psoriasis involving nails with data on the nail psoriasis severity index (NAPSI) or modified nail psoriasis severity index (mNAPSI). Exclusion criteria consisted of studies with nonhuman models, books, meta-analysis, reviews, systematic reviews, abstracts, and lack of availability in English.
Two independent reviewers (MK and CW) performed the initial search as well as independently screened articles. Both independently performed data extraction. The following data were extracted from all articles: study design, study duration, treatments and dose utilized, total number of patients, patient characteristics, and results of the treatments (NAPSI/mNAPSI score, mean percent improvement from baseline, and the proportion with a NAPSI/mNAPSI score of 0. A third reviewer (FA) settled any discrepancies.
Comparators
The biologics compared in this systematic review consisted of anti-TNF agents (etanercept, 50 mg twice weekly or 50 mg once weekly; adalimumab, 40 mg every 2 weeks; certolizumab pegol, 400 mg every 4 weeks or 200 mg every 2 weeks; infliximab, 5 mg/kg at weeks 0, 2, and 6, then every 8 weeks thereafter; golimumab, 50 mg every 4 weeks, or 100 mg every 4 weeks), anti-IL-17 agents (secukinumab, 300 mg or 150 mg at baseline, weeks 1, 2, 3, and 4, then every 4 weeks; ixekizumab, 160 mg, at week 0, then 80 mg every 4 weeks or 80 mg every 2 weeks; brodalumab, 210 mg at weeks 0 and 1 then every 2 weeks), and anti-IL-23 agents (guselkumab, 100 mg, at weeks 0, 4, then every 8 weeks; risankizumab-rzaa, 150 mg, at weeks 0, 4, then every 12 weeks; ustekinumab, 45 mg or 90 mg at weeks 0, 4, then every 12 weeks). The varied doses of individual biologics were pooled into a single treatment arm and the average of the outcome data is represented in the Tables. The data for those studies which provided stratified results based on dose differences are discussed in the results section below.
Outcomes
The outcomes of interest in this review were the mean NAPSI or mNAPSI percent improvement, and the proportion of patients achieving a NAPSI score of 0. A NAPSI or mNAPSI score is an assessment tool utilized to provide a standardized way of scoring nail psoriasis severity. A NAPSI score is determined by discerning 8 clinical features of nail psoriasis (leukonychia, red spots in lunula, nail plate crumbling, pitting, oil drop dyschromia, nail bed hyperkeratosis, splinter hemorrhages, and onycholysis) in each of 4 quadrants of the greatest affected nail. 2 An mNAPSI score is calculated without dividing the nail into quadrants and also takes into account the severity of the most common clinical nail psoriasis features (nail pitting, nail crumbling, and onycholysis) on a scale of 0-3 while the rest of the features are given a score of 0 if absent and 1 if present. 2 Thus, the mNAPSI scores range from 0-14 for an individual nail, or 0-140 for all 10 fingernails. The NAPSI scores reported in the studies were measured either as a target fingernail NAPSI score for an individual nail (0-8) or as an overall/total NAPSI which is a sum of all target fingernail NAPSI scores (0-80).
Results
Study characteristics
Patient Characteristics.

alack of data provided in some studies for nail psoriasis patients specifically -excluded from total.
Outcomes
Comparison of Overall Biologic Efficacy at 24 Weeks.
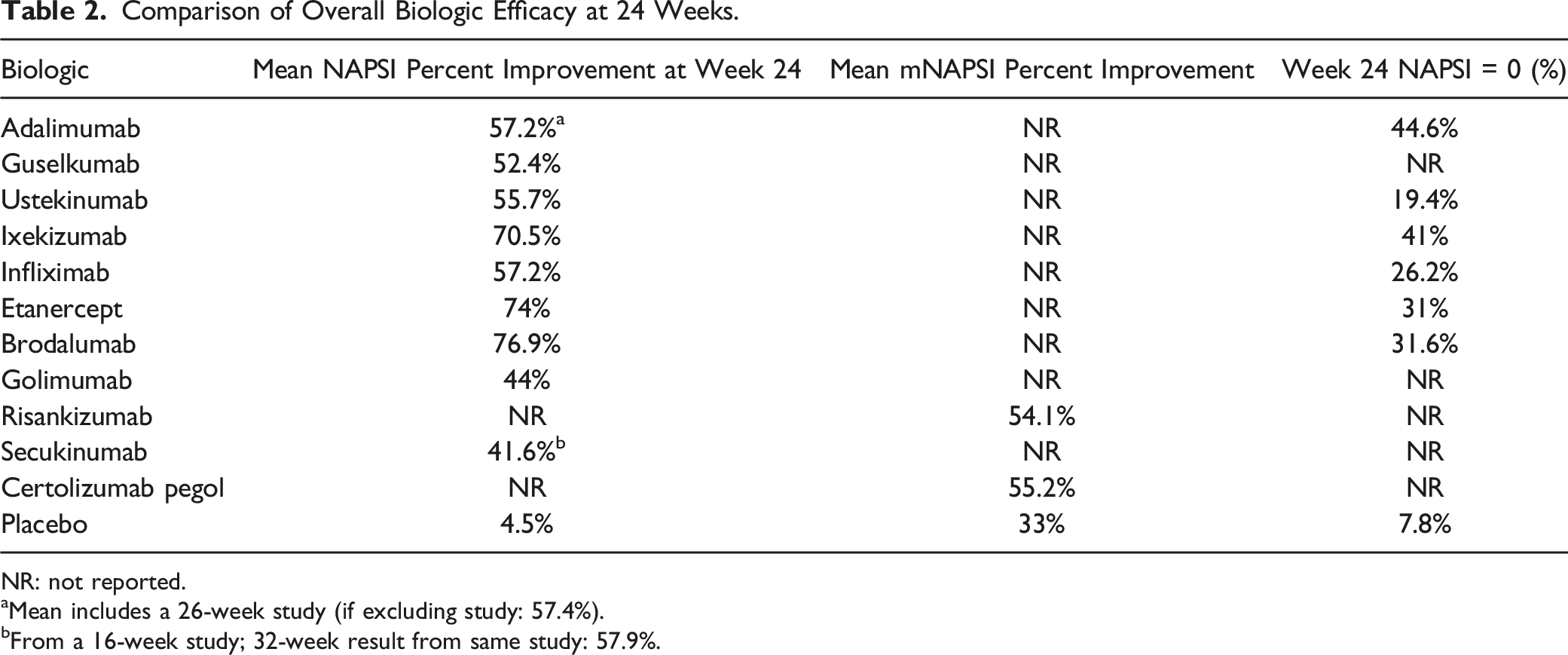
NR: not reported.
aMean includes a 26-week study (if excluding study: 57.4%).
bFrom a 16-week study; 32-week result from same study: 57.9%.
Comparison of Overall Biologic Efficacy at 52 Weeks.
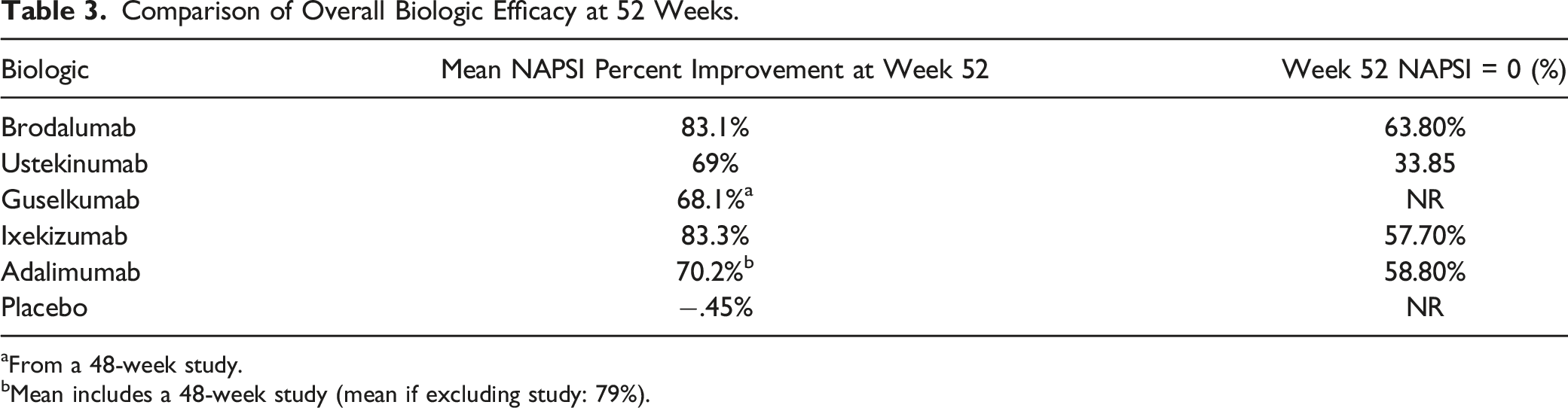
aFrom a 48-week study.
bMean includes a 48-week study (mean if excluding study: 79%).
Study specifics
Adalimumab
Six studies reviewed the effect of Adalimumab (ADA) on nail psoriasis resolution.
Study 1
Elewski et al. examined the efficacy of ADA vs placebo with the primary endpoint of at least 75% improvement in total-fingernail modified NAPSI (mNAPSI75). Patients received a starting dose of 80 mg then 40 mg ADA every other week or placebo. The baseline mean total-fingernail NAPSI was 47.9 in the ADA group and 46.8 in the placebo group. The baseline mean total-fingernail mNAPSI was 57.6 for the ADA group and 58.1 for placebo. The mNAPSI75 response rate at 26 weeks was 46.6% (P < .001) for ADA and 3.4% for placebo. The mNAPSI of 0 was 6.6% (P < .01) for ADA and 0% for placebo. The mean NAPSI percent improvement was 56.2% (P < .001) for ADA and 11.5% for placebo. The 5 additional studies which included ADA are described in the guselkumab and ixekizumab sections below.
Brodalumab
Elewski et al. evaluated the efficacy of Brodalumab (BRO) (210 mg every 2 weeks) vs ustekinumab (45-mg/90-mg weight-based dosing every 12 weeks) in the treatment of scalp and nail psoriasis. The baseline mean total NAPSI in the BRO group was 9.6 and the baseline in the ustekinumab (UST) group was 9.9. The mean NAPSI percent improvement at 52 weeks was 83.1% for BRO and 75% for UST; at 24 weeks, the mean percent improvement was 76.9% (P < .05) for BRO and 58.9% for UST. NAPSI of 0 at week 52 was achieved by 63.8% (P < .05) in the BRO group and 39.1% in the UST group.
Certolizumab
Mease et al. studied Certolizumab (CZP) at 2 doses (200 mg every 2 weeks or 400 mg every 4 weeks) vs placebo in patients with PsA in which modified NAPSI was determined as a secondary endpoint. The baseline mNAPSI scores were 3.4 for the placebo group, 3.4 for the 400 mg group, and 3.1 for the 200 mg group. The mNAPSI mean percent improvement from baseline was 51.6% (P = .003), 58.8% (P < .001), and 32.4% in the 200 mg, 400 mg, and placebo groups, respectively.
Etanercept
Ortonne et al. investigated Etanercept (ETN) at 2 different doses (50 mg twice weekly for 12 weeks followed by once weekly for 12 weeks or 50 mg weekly for 24 weeks) for the treatment of nail psoriasis in patients with moderate-to-severe psoriasis. The baseline fingernail NAPSI in the twice weekly group was 6 vs 5.8 in the weekly group. The twice weekly group resulted in a mean NAPSI percent improvement of 71.7% vs 75.9% for the weekly group (P < .0001). Interestingly, NAPSI 0 results were 14.3% (P < .05) and 31% (P < .001) in the twice weekly and weekly groups, respectively.
Golimumab
Kavanaugh et al. studied 2 doses of Golimumab (GOL) (50 mg or 100 mg every 4 weeks through week 20) vs placebo for the treatment of psoriatic arthritis with effects on nail psoriasis as a secondary endpoint. The baseline fingernail NAPSI score for the 50 mg, 100 mg, and placebo groups were 4.7, 4.6, and 4.4, respectively. The median percent improvement in NAPSI at 24 weeks was determined to be 54% in the 100 mg group, 33% in the 50 mg group, and 0% in the placebo cohort (P < .001).
Guselkumab
There were 2 studies included which evaluated Guselkumab (GUS) in the treatment of psoriasis with nail disease resolution as a secondary endpoint.
Study 1
Reich et al. investigated GUS (100 mg at weeks 0 and 4, then every 8 weeks) compared to placebo with crossover to GUS (weeks 0, 4, and 12 then guselkumab at weeks 16 and 20, then every 8 weeks) and ADA (80 mg week 0, 40 mg week 1, then 40 mg every 2 weeks through week 23). The baseline mean fingernail NAPSI was 5, 4.8, and 4.5 for placebo, GUS, and ADA, respectively. At 24 weeks, the mean NAPSI percent improvement for GUS and ADA was 55% and 53.7%, respectively while the week 16 NAPSI percent improvement for placebo was 1.8%.
Study 2
Blauvelt et al. also evaluated GUS (100 mg at weeks 0 and 4, then every 8 weeks) compared to placebo with crossover to GUS (weeks 0, 4, and 12 then guselkumab at weeks 16 and 20, then every 8 weeks) and ADA (80 mg week 0, 40 mg week 1, then 40 mg every 2 weeks through week 47). The baseline mean fingernail NAPSI was 4.7, 4.9, and 4.6 in the placebo, GUS, and ADA groups, respectively. At 48 weeks, the mean NAPSI percent improvement for GUS and ADA was 68.1% and 61.4%, respectively while the week 16 NAPSI percent improvement for placebo was −.9%.
Infliximab
Rich et al. studied the efficacy of Infliximab (INF) in patients with moderate-to-severe psoriasis with baseline nail disease. Patients were given either INF (5 mg/kg) or placebo at weeks 0, 2, 6, and every 8 weeks through week 46, with those in the placebo group crossing over to INF at week 24. The baseline mean fingernail NAPSI was 4.3 for the placebo group and 4.6 for the INF group. The mean NAPSI percent improvement was 57.2% (P < .001) for INF and −4.1% for placebo while NAPSI 0 was achieved by 26.2% (P < .001) going up to 44.7% by week 50 in the INF group compared to 5.1% at week 24 in the placebo group.
Ixekizumab
Six studies reviewed the efficacy of Ixekizumab (IXE) in nail psoriasis.
Study 1
Mease et al. studied the efficacy of IXE vs ADA and placebo for the treatment of PsA in biologic naïve patients. Patients were assigned to receive either IXE 80 mg every 2 weeks (IXEQ2W), IXE 80 mg every 4 weeks (IXEQ4W), ADA 40 mg every 2 weeks, or placebo. The baseline total mean NAPSI scores were 19.8, 21.3, 25, and 20.9 in the placebo, IXEQ4, IXEQ2, and ADA cohorts, respectively. The mean NAPSI percent improvement from baseline at week 24 were 12.1%, 65.7% (P < .001), 62%, (P < .001) and 51.2% (P < .001) in the placebo, IXEQ4, IXEQ2, and ADA cohorts, respectively. NAPSI of 0 was achieved by 18.9%, 25.7%, 36.5% (P < .05), and 39.4% (P < .01) of the cohort for the placebo, IXEQ4, IXEQ2, and ADA groups, respectively.
Study 2
Nash et al. conducted a study evaluating IXE vs placebo in patients with active psoriatic arthritis and prior insufficient response to tumour necrosis factor inhibitors. Patients received either IXE 80 mg every 4 weeks (IXEQ4), IXE 80 mg every 2 weeks (IXEQ2), or placebo with patients in the IXE group receiving an initial 160 mg dose. The baseline total mean NAPSI scores were 18.7, 20.5, or 21 in the placebo, IXEQ4, and IXEQ2 groups, respectively. The mean NAPSI percent improvement from baseline at week 24 for all 3 groups were −5.3%, 51.2% (P < .0001), and 59.5% (P < .0001) for the placebo, IXEQ4, and IXEQ2 groups respectively. NAPSI of 0 was achieved by 7%, 20% (P = .02), and 30% (P = .0005) of the cohort for the placebo, IXEQ4, and IXEQ2 groups respectively.
Study 3
Van der Heijde et al. conducted a 52-week analysis comparing 2 doses of IXE (80 mg every 2 weeks or 80 mg every 4 weeks with both receiving an initial 160 mg dose) in biologic naïve patients with PsA and concomitant nail psoriasis. Ixekizumab was also compared to cohorts of patients receiving placebo or adalimumab 40 mg every 2 weeks. At week 24, patients receiving placebo or ADA were rerandomized to either IXE every 2 weeks (IXEQ2) or IXE every 4 weeks (IXEQ4) with a 160 mg initial dose at week 24 for placebo patients. Patients previously assigned to ADA entered an 8-week washout period and started IXE at week 32 without the initial 160 mg dose. The total mean NAPSI (the average of the sum of all involved fingernail scores for each patient) at baseline were reported as 19.1, 26.7, 21.2, 19.9, 15.6, and 26.1 for the placebo/IXEQ4, ADA/IXEQ4, IXEQ4, placebo/IXEQ2, ADA/IXEQ2, and IXEQ2 groups, respectively. At week 52, the mean NAPSI percent improvements were 55%, 62.9%, 78.2%, 64.3%, 66.7%, and 82.8% for the placebo/IXEQ4, ADA/IXEQ4, IXEQ4, placebo/IXEQ2, ADA/IXEQ2, and IXEQ2 groups, respectively. NAPSI of 0 was 32.3%, 39.4%, 46.9%, 34.3%, 45.7%, and 40.6% for the placebo/IXEQ4, ADA/IXEQ4, IXEQ4, placebo/IXEQ2, ADA/IXEQ2, and IXEQ2 groups, respectively.
Study 4
Mease et al. examined IXE vs ADA in the treatment of PsA in biologic maive patients. Patients received an initial IXE 160 mg dose at week 0, followed by 80 mg every 4 weeks starting at week 4 onwards (if patients met criteria for moderate-to-severe psoriasis, in they received 80 mg IXE every 2 weeks from week 2 to week 12, followed by IXE every 4 weeks. Those randomized to receive ADA were given a 40 mg initial dose followed by 40 mg every 2 weeks starting at week 2 (if they met criteria for moderate-to-severe psoriasis, patients received an 80 mg starting dose of ADA, followed by 40 mg ADA every 2 weeks starting at week 1). The results were not stratified by disease severity. The total mean NAPSI at baseline was 19.7 for the IXE group and 19.1 for the ADA group. The mean NAPSI percent improvement at 24 weeks was 80.7% (P < .001) for IXE and 65.6% for ADA while NAPSI 0 was 58.1% (P < .082) for IXE vs 49.7% for ADA.
Study 5
Wasel et al. conducted a post hoc analysis comparing the efficacy of IXE vs UST for the treatment of nail psoriasis in patients with moderate-to-severe plaque psoriasis. Patients were randomized to either the IXE group (160-mg initial dose, then 80 mg every 2 weeks for 12 weeks followed by 80 mg every 4 weeks) or UST (45-mg/90-mg weight-based dosing at weeks 0, 4 and every 12 weeks thereafter). The baseline total mean NAPSI scores were 28.3 in the IXE group and 24.8 in the UST group. The mean NAPSI percent improvement at week 52 was 79% (P < .001) for IXE compared to 62.9% for UST and NAPSI 0 at week 52 was 61.9% (P < .001) for IXE compared to 28.6% for UST. For those patients deemed to have significant baseline nail psoriasis (NAPSI>16), the NAPSI 0 at week 52 was 57.4% (P < .001) and 17.5% for IXE and UST, respectively.
Study 6
Smolen et al. looked at the efficacy of IXE vs ADA for the treatment of PsA. Patients receiving treatment with IXE received a 160 mg starting dose followed by either IXE 80 mg every 4 weeks from week 4 or 80 mg IXE every 2 weeks from week 2 to week 12, followed by IXE every 4 weeks for those with moderate-to-severe disease. Those in the ADA group received ADA 40 mg every 2 weeks starting at week 2 or 40 mg ADA every 2 weeks starting at week 1 after an 80 mg starting dose at week 0. The results were not stratified by disease severity. The baseline total mean NAPSI scores were 19.7 in the IXE group and 19.1 in the ADA group. The mean NAPSI percent improvement at 52 weeks was 90.3% (P = .005) and 79% for IXE and ADA, respectively whereas NAPSI 0 was 67.5% (P = .06) for IXE compared to 58.8% for ADA.
Risankizumab
Kristensen et al. evaluated the efficacy of Risankizumab (RIS) (150 mg at weeks 0, 4, and 16) compared to placebo in the treatment of PsA with nail features. The total mean mNAPSI at baseline was 18.1 for the RIS group and 16.6 for placebo. The mean percent improvement in mNAPSI at week 24 was 54.1% (P < .001) and 33.7% in the RIS and placebo groups, respectively.
Secukinumab
Reich et al. conducted a study looking at 2 doses of secukinumab (SEC) (150 mg or 300 mg weekly for 5 weeks, then every 4 weeks thereafter until 16 weeks) vs placebo for the treatment of moderate-to-severe nail psoriasis. The total mean NAPSI at baseline was reported as 45.5, 39.1, and 43.2 in the 300 mg, 150 mg, and placebo groups, respectively. The mean percent improvement in NAPSI at week 16 was 45.3%, 37.9%, and 10.8% for the 300 mg, 150 mg, and placebo groups, respectively (P < .001). At 32 weeks, the mean percent improvement in NAPSI was 63.2% and 52.6% for the 300 mg and 150 mg groups, respectively. The patients were further stratified into those with and without concomitant psoriatic arthritis (PsA). For the 300 mg group without PsA, the mean NAPSI percent improvement at 32 weeks was 73.3% while the 300 mg group with PsA was 65.1%. The 150 mg group without PsA had a mean NAPSI percent improvement of 50.6% while in those with PsA it was 72.3%.
Discussion
Due to the variety of biologics available, it is difficult for clinicians to select the most effective biologic for nail psoriasis due to insufficient comparative data. Our study’s objective was to provide a comparative evaluation of various biologics and elucidate their efficacy in treating PsA and nail psoriasis. Results from our study suggest that brodalumab, etanercept, and ixekizumab are associated with the highest improvement in mean NAPSI percentage for nail psoriasis. These results partially align with a 2018 meta-analysis studying therapeutic efficacy in plaque psoriasis in which brodalumab and ixekizumab were among the highest performing biologics for both short and long term PASI 75, 90, and 100 rates, although effects on nail psoriasis were not discussed.1,19 Another network meta-analysis conducted in 2017 concluded that brodalumab was associated with a significantly higher PASI 100 score compared to secukinumab, ustekinumab, and etanercept but this study did not discuss the effects on nail psoriasis. 20
One network meta-analysis by Reich et al. in 2022 examined complete resolution of nail psoriasis at 24-26 weeks (based on NAPSI, mNAPSI, or Physician's Global Assessment of Fingernails) amongst 7 randomized controlled trials. 21 They found that ixekizumab (46.5%) had the greatest probability of achieving complete resolution of nail psoriasis. The 5 other biologics included in this analysis included brodalumab (37.0%), adalimumab (28.3%), guselkumab (27.7%), ustekinumab (20.8%), and infliximab (.8%). Huang et al. also conducted a 2021 network meta-analysis in which they also found that ixekizumab illustrated the highest efficacy for complete nail psoriasis resolution and improvement in NAPSI score when compared to other biologics as well as small molecule inhibitors. 22 Finally, a 2021 meta-analysis by Zhang et al. further illustrated that ixekizumab achieved the most significant improvement in nail psoriasis. 23 Our results were similar to the above findings as ixekizumab and adalimumab were the top 2 biologics resulting in complete nail psoriasis resolution. Brodalumab, etanercept, infliximab had similar rates of complete nail psoriasis resolution, while ustekinumab had the lowest probability of achieving complete nail psoriasis resolution.
Limitations
This study was able to combine data from multiple independent trials of various biologics to increase the sample size and power for determining NAPSI or mNAPSI responses. Despite this, there remains a paucity of statistically significant evidence, making it difficult to make definitive claims regarding the best biologic for nail psoriasis. Additionally, only 1 database was utilized for our review, limiting our results to the studies found within that platform.
The evidence pooled from these studies does provide more insight into which biologics may be more efficacious for nail psoriasis; however, our results were not calculated as a weighted average, possibly skewing efficacy of a particular biologic over another. Although all studies were randomized controlled trials, some of the evidence also stemmed from open-label studies and non-placebo compared trials. Additionally, there were studies that did not specify if the results were statistically significant. We also only utilized NAPSI/mNAPSI criteria for nail psoriasis as these were the most widely studied. Information on Physician’s Global Assessment of Fingernails (PGA-F) was not compared as there were few studies that reported these scores. Furthermore, differences in the conducting of multiple trials and lack of available stratified patient characteristics for nail psoriasis may alter the therapeutic efficacy results and create bias. Another element contributing to potential bias is prior biologic use therefore we could not stratify our results due to several studies not disclosing prior biologic administration.
The sample size of patients evaluated for biologic efficacy on nail psoriasis is substantial (n = 5400) but there remains a dearth of studies when compared to the vast number of studies on PsA or plaque psoriasis. Much of the data obtained for nail psoriasis was from studies on PsA or plaque psoriasis, making it challenging to obtain specifics on patient characteristics and previous biologic use for nail psoriasis. It is imperative to obtain more research, ideally in the form of randomized head-to-head and placebo-controlled trials, specifically studying biologic efficacy for nail psoriasis. Because of these limitations, there is a need for more research evaluating multiple biologics in the treatment of nail psoriasis.
Conclusions
There is a lack of sufficient head-to-head clinical trials comparing the efficacy of multiple biologics in the treatment of nail psoriasis. Nevertheless, this study provides a comparative overview on the efficacy of multiple biologics in the treatment of nail psoriasis based on a variety of clinical trials. At 24 weeks, we found that brodalumab, etanercept, and ixekizumab were associated with the highest improvement in mean NAPSI percentage for nail psoriasis. Complete resolution of nail psoriasis was highest for adalimumab and ixekizumab. Further studies are required to better understand the statistically significant efficacy of biologics for nail psoriasis treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics,IRB,and Informed Consent Statement:
This study did not require IRB approval given its nature as a systematic review. Informed consent was not required given data was only based on prior, publicly available published datasets.
