Abstract
Background
Electronic health records (EHRs) offer the possibility of using data entry templates to simultaneously document routine clinical care and capture disease-specific measures as discrete data elements that can be used for health services research (HSR). The objective of this study was to determine factors associated with meaningful treatment escalation (MTE) of psoriasis as a pilot study for future real-world HSR studies.
Methods
We conducted a retrospective, observational cohort study of psoriasis patients by using data collected during routine clinical care from an EHR using EpiCare® SmartForms. The psoriasis SmartForm records psoriasis disease severity measures and descriptive findings to generate visit notes. These data were extracted and analyzed to identify factors associated with MTE, defined as changing or adding, phototherapy, systemic, or biologic therapy.
Results
473 psoriasis patients met study criteria; 239 underwent MTE between their first and third observed visits. Patients who experienced MTE had more severe disease at Visit 1—assessed by BSA, pPGA, oPGA, and a patient-reported disease severity measure--than patients who did not experience MTE. Other factors associated with MTE included use of topicals only or no active treatment at Visit 1, palmoplantar disease, and involvement of other difficult-to-treat body areas. Patients who underwent MTE experienced larger improvements in disease severity than those who did not.
Conclusions
This study highlights how data collected during routine clinical practice can be readily used for real-world retrospective HSR when disease measures are captured as discrete elements. This approach could provide a cost-effective platform to conduct real-world HSR.
Keywords
Background
Psoriasis is a complex, chronic immune-mediated skin disease with varying phenotypic presentations, associated comorbid conditions, and severity. Psoriasis usually requires long-term management with immunomodulatory treatment such as corticosteroid and non-corticosteroid topical agents, ultraviolet light (phototherapy), oral systemic drugs, and biologics. 1 Since 2004, more than 15 new oral and injectable biologic systemic therapies have come to market for plaque psoriasis. Although robust clinical trials have brought them to regulatory approval, long-term real-world safety and efficacy data are lacking. The United States Food and Drug Administration has required pharmaceutical sponsors to provide a risk evaluation and mitigation strategy for new systemic agents, 2 prompting the development of robust registries, the largest of which are the PSOLAR and CorEvitas psoriasis registries.3,4 These registries have enrolled thousands of patients to rigorously collect prospective safety and efficacy data at 6-month intervals alongside routine clinical care.
Registries are robust resources for comparative effectiveness and other types of health services research (HSR) but are expensive, time consuming to conduct, do not collect data in real time, and are duplicative in nature as much of the same data are collected and documented in patients’ electronic health records (EHRs). EHRs offer the prospect of utilizing clinical data for real-world health services research (HSR). Compared with alternative data sources and study approaches, such as clinical trials and cohort studies, data sourced from EHRs are already captured, thus reducing administrative efforts, costs, and sample selection bias. 5 The EpiCare® EHR, in use by many academic centers, provides robust functionality to syngerize data extraction and clinical workflows. EpiCare® provides a tool known as the SmartForm (SF), which is a clinical documentation tool that can be customized, with minimum burden, to mix free form, structured, and coded data. 6 SFs are ideal for documenting and assisting the management of chronic conditions where complex clinical data can be input during routine care, organized for optimal data review, and incorporated into the visit note. 6 Structured SF data can then be used for analysis without the need for natural language processing, manual data extraction, or manual data entry. The data can be efficiently extracted and exported directly to a database for data management and analysis.
To that end, in 2015, the staff from the Information Technology (IT) services department at the University of Utah Health (UUH) and clinicians in the UUH Department of Dermatology partnered to design and implement custom SFs for several chronic skin conditions including psoriasis. The UUH psoriasis SF allows providers to document past and present therapies, symptoms, body location(s) affected, extent, and morphology of the lesions. SFs also promote documentation of physician-reported severity measures such as body surface area (BSA) and Physician Global Assessment (PGA) (healthcare providers received extensive training on the BSA and PGA as they have all conducted numerous psoriasis clinical trials). These measures are standard endpoints for registries and trials and are also required for insurance prior authorization of therapeutic agents and for institutional and federal quality assessment programs.
The prescribing patterns for psoriasis are not well understood. Individuals with psoriasis often must switch therapies for a variety of reasons (tolerance, safety, efficacy, cost, formulary design changes), but a greater understanding of the factors associated with these changes is needed. Registries and other databases are commonly queried to assess treatment patterns and predictors of therapy switches. We, therefore, conducted this study for two purposes: first, to establish the feasibility of using psoriasis SFs for future retrospective or prospective real-world analyses and second, to compare outcomes and clinical features of psoriasis patients who had meaningful treatment escalation (MTE) with those who did not. We defined MTE as initiation of, switching to, or adding a new non-topical therapy (e.g., phototherapy, oral systemic, or biologic).
Methods
Data Source
This retrospective, observational cohort study was approved by the University of Utah’s Institutional Review Board (IRB). In collaboration with the UUH IT Services and the Department of Dermatology, customized psoriasis SFs were developed and implemented in 2015. The psoriasis SFs collect psoriasis-specific subjective and objective measures in a structured fashion. These measures include: the therapy selected at the visit, disease duration, symptoms, disease triggers, phenotype (e.g., plaque, palmoplantar, pustular, guttate, inverse, nail, erythrodermic), body locations, and body surface area (BSA; 0%-100%). In addition to these clinical measures, the SFs also collect data from a Patient-reported disease severity numeric rating scale (PtNRS; 0-10), a six-point plaque Physician Global Assessment (pPGA) instrument in which erythema, scaling, and induration scores were averaged and rounded to the nearest whole number (0-5), and a six-point overall static Physician Global Assessment (oPGA) instrument for which the pPGA and BSA were factored together to create an overall score (0 = clear, 1 = almost clear, 2 = mild, 3 = moderate, 4 = severe, and 5 = very severe). The psoriasis SFs also capture, as discrete data elements, past and current treatments that are unique to the visit. This is preferred to using a medication list which may or may not be historically accurate or updated. Therapy categories recorded include topical medications (vitamin D analogs, topical retinoids, topical corticosteroids, calcineurin inhibitors, combination corticosteroids plus vitamin D), phototherapy (narrowband ultraviolet (UV)-B, broadband UV-B, psoralen plus UV-A), individual oral systemic therapies (e.g., methotrexate, acitretin, cyclosporine), and individual biologic therapies.
In addition to all psoriasis SFs data from Visits 1 and 3, we also extracted demographic data (age, sex) and medical problems/history based on selections from predefined lists as well as International Classification of Diseases ninth and 10th editions codes listed at Visit 1 from the EHR. Data were prepared and exported by the University’s IT Data Sciences personnel. In compliance with IRB guidance and the Health Insurance Portability and Accountability Act (HIPAA) of 1996 Federal Privacy Rule, direct patient identifiers were removed before transferring the limited dataset to a third-party for analysis.
Study Sample and Treatment Groups
The study sample consisted of individuals who were ≥18 years of age, had three consecutive visits at the UUH Department of Dermatology with a primary diagnosis of psoriasis between October 2015 and October 2018, and were not missing all psoriasis SF data. To maximize the number of eligible patients, we did not impose any minimum or maximum times between visits (Visit 1 did not have to be their first clinic visit). We classified patients into one of two treatment groups based on treatments received between Visits 1 and 3: those who received MTE and those who did not. MTE was defined as initiation of, switching to, or adding a new non-topical therapy (e.g., phototherapy, oral systemic, or biologic) by Visit 3. Patients were classified as not receiving MTE if they: (1) did not change their treatment plan between Visits 1 and 3 or (2) the only change experienced was addition of, or switching to, topical therapies between Visits 1 and 3.
Data Analysis
To avoid bias caused by deleting cases missing data (46 for PtNRS, 81 for BSA, 98 for pPGA, 101 for oPGA), all missing values for those variables were imputed using the sequential chained equations predictive mean matching multiple imputation approach as implemented by the mi command in Stata 17.0, 7 which created ten imputed datasets; results of analyses involving the severity measures based on the multiple datasets were pooled using standard methods. 8 The original (unimputed) data are summarized in Appendix A in the Supplement Material.
Clinical and demographic characteristics at Visit 1 were summarized using means and standard deviations for continuous variables and counts and proportions for categorical variables. Two-tailed P-values comparing patient characteristics between the two groups were computed using the Student’s t-test and the chi-square test for continuous and categorical variables, respectively. Treatment changes between Visits 1 and 3 were summarized using transition matrices, in which each cell,
We assessed the simultaneous associations of Visit 1 patient characteristics with the probability of experiencing MTE via odds ratios (OR) estimated from a multiple logistic regression model. Propensity scores, i.e., the probability of experiencing MTE, were computed from the coefficients of the fitted logistic regression model and the values of the patients’ Visit 1 characteristics. Changes in the psoriasis severity measures between Visits 1 and 3 (i.e.,
Results
Sample Selection and Characteristics
We initially identified 1115 adult patients who had at least one visit with a primary diagnosis of psoriasis during the study period. Forty-two percent (n = 473) of those patients had at least three visits with a primary diagnosis of psoriasis and who were not missing all SF data for the first three observed visits. (Figure 1) Of the 473 patients, 239 (51%) met the definition for MTE between their first and third observed visits (Visits 1 and 3). Patient selection and attrition flowchart.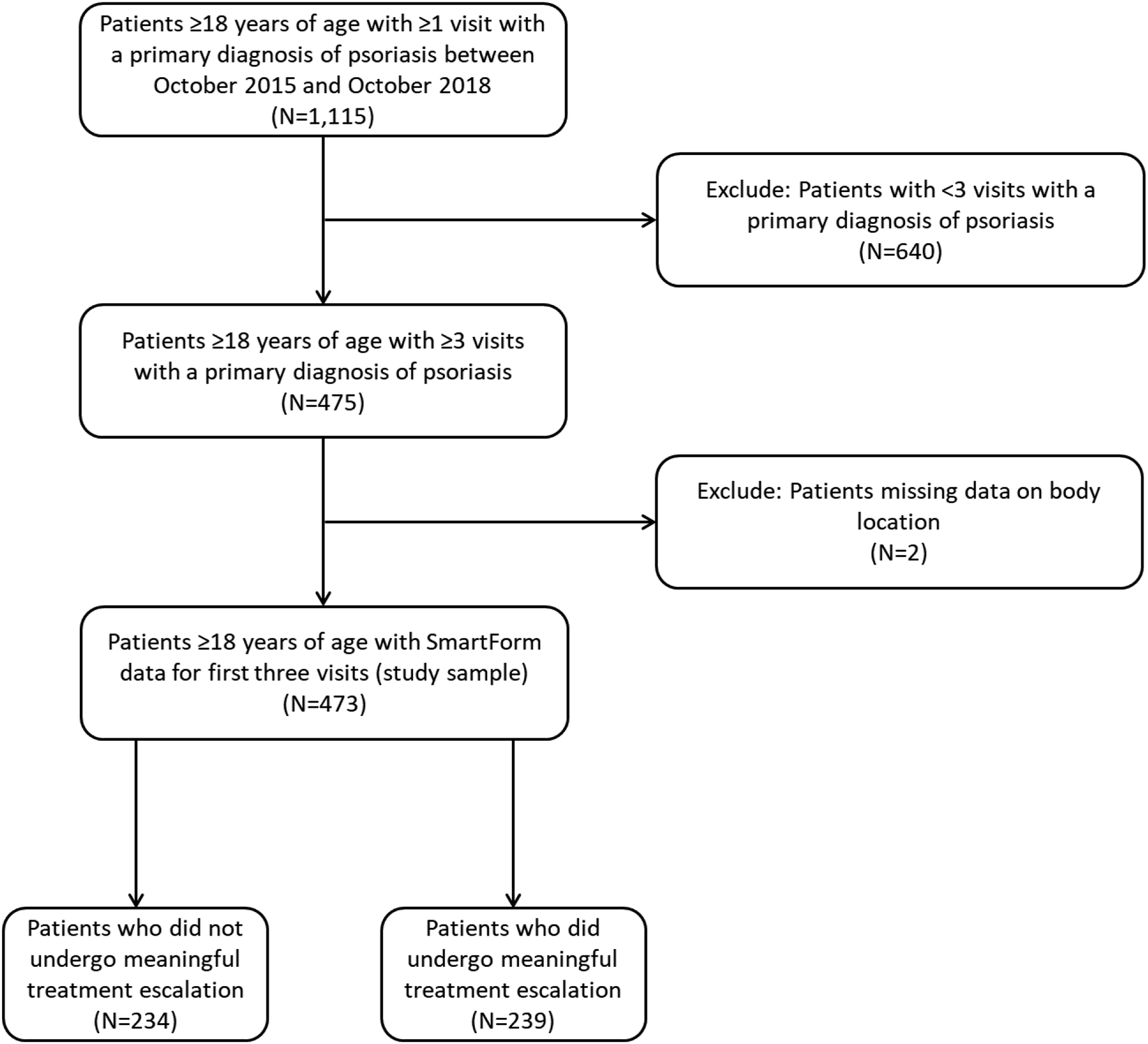
Patient Characteristics at Visit 1, by Treatment Escalation Group.
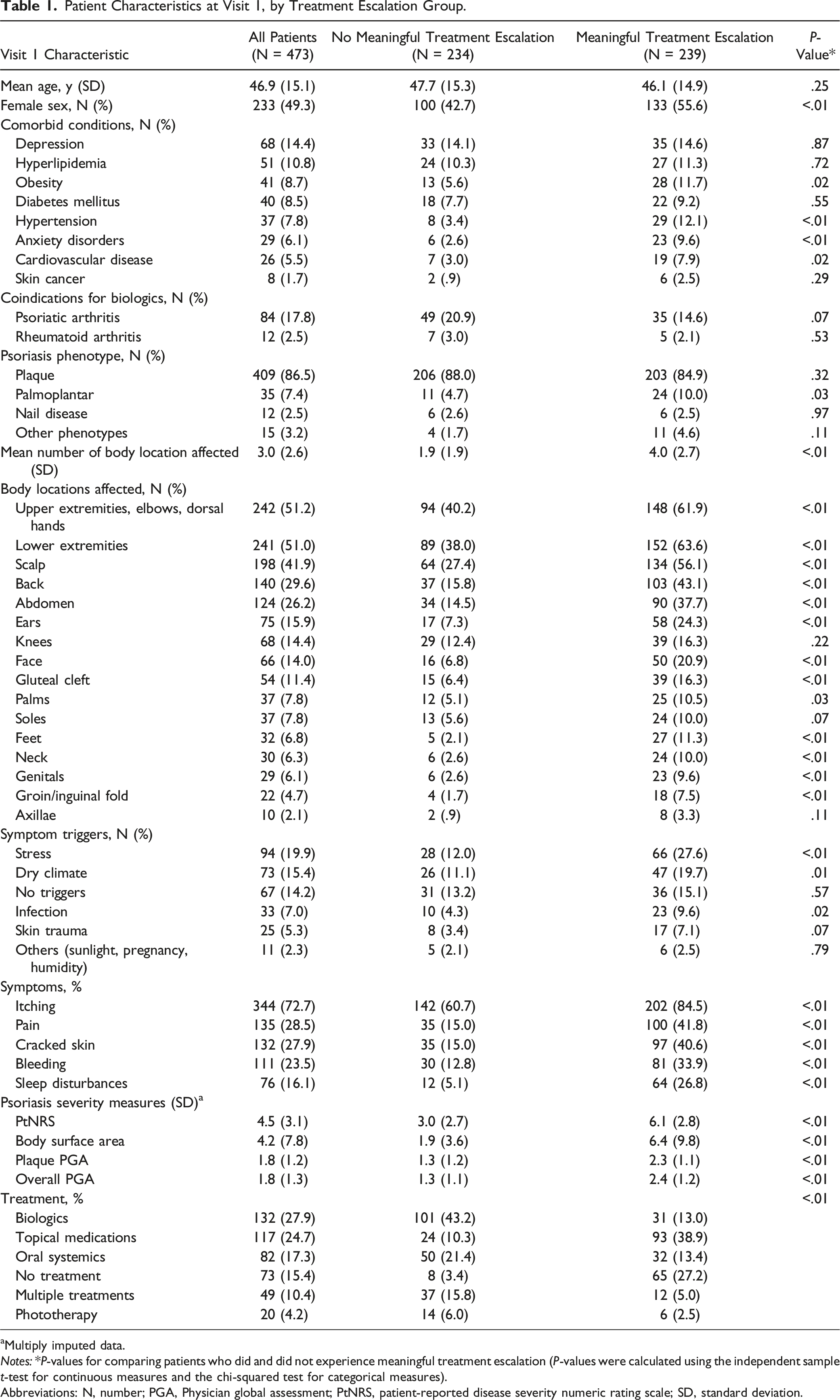
aMultiply imputed data.
Notes: *P-values for comparing patients who did and did not experience meaningful treatment escalation (P-values were calculated using the independent sample t-test for continuous measures and the chi-squared test for categorical measures).
Abbreviations: N, number; PGA, Physician global assessment; PtNRS, patient-reported disease severity numeric rating scale; SD, standard deviation.
Transition Matrices for Treatment Changes Between Visits 1 and 3.
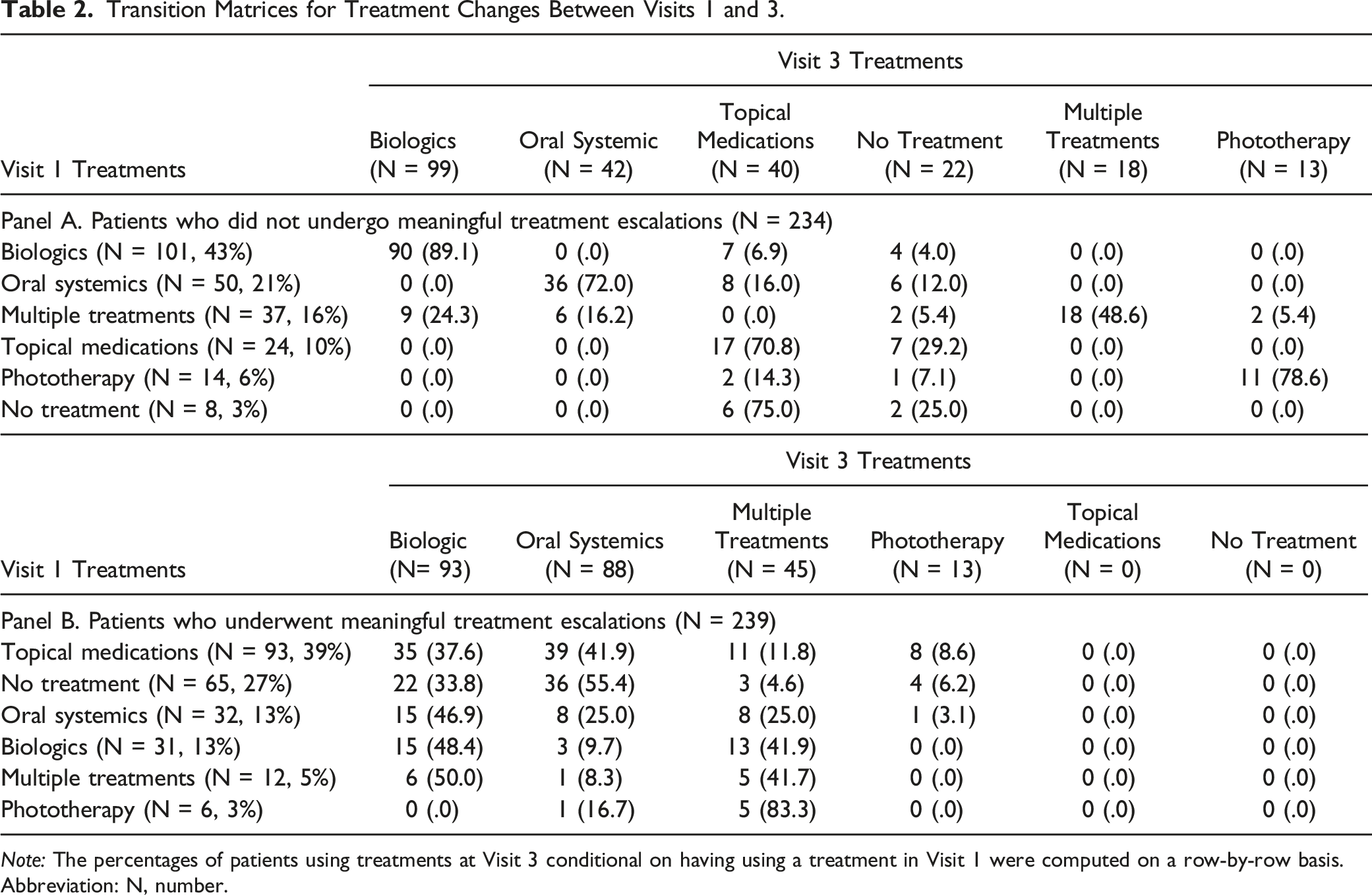
Note: The percentages of patients using treatments at Visit 3 conditional on having using a treatment in Visit 1 were computed on a row-by-row basis.
Abbreviation: N, number.
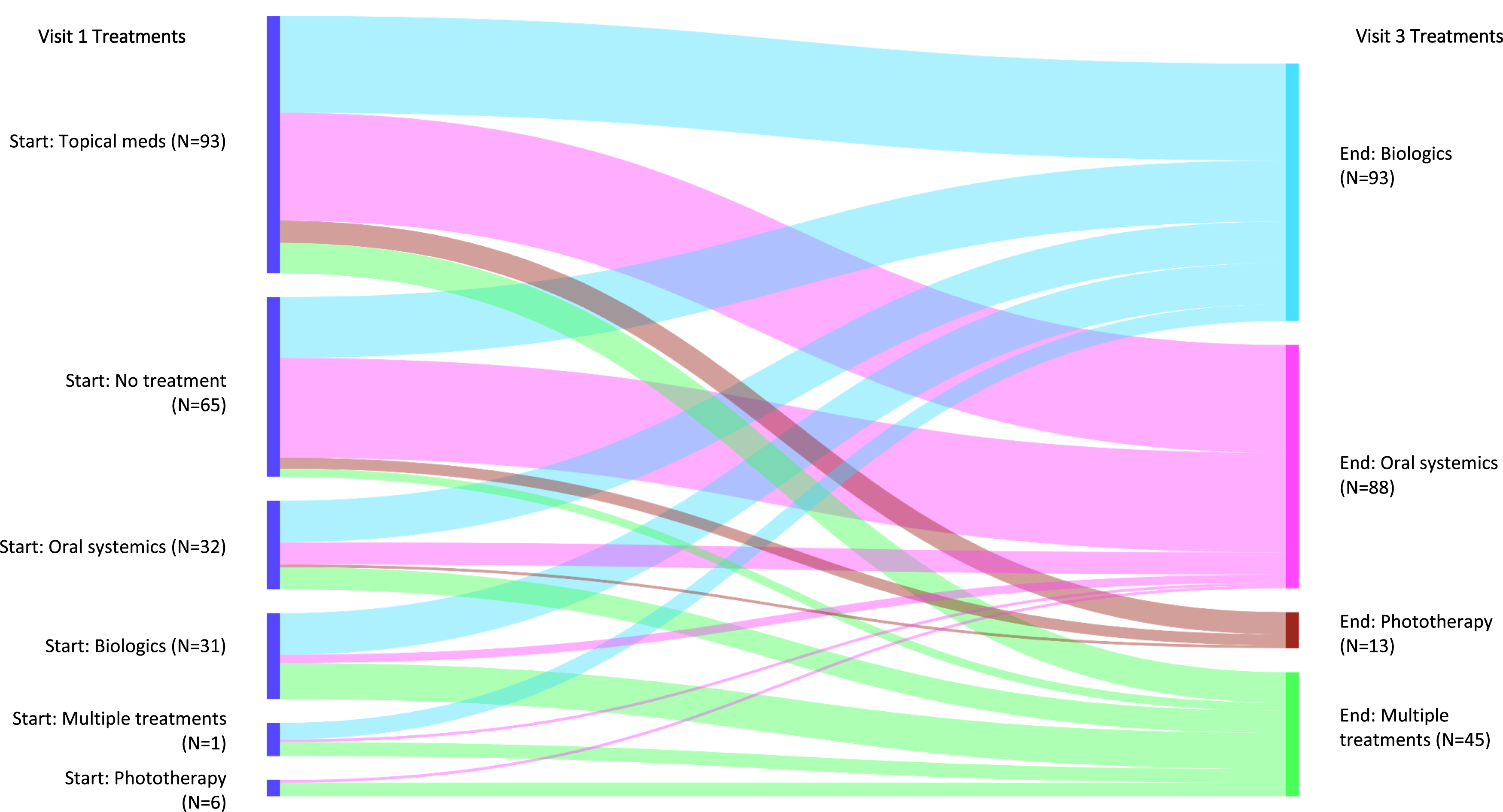
Treatment transitions between visits 1 and 3 for patients who experienced meaningful treatment escalation. Note: The widths of the bands represent the proportions of patients who used a treatment at visit 1 and who used another (possibly the same) treatment at visit 3. Abbreviation: N, number.
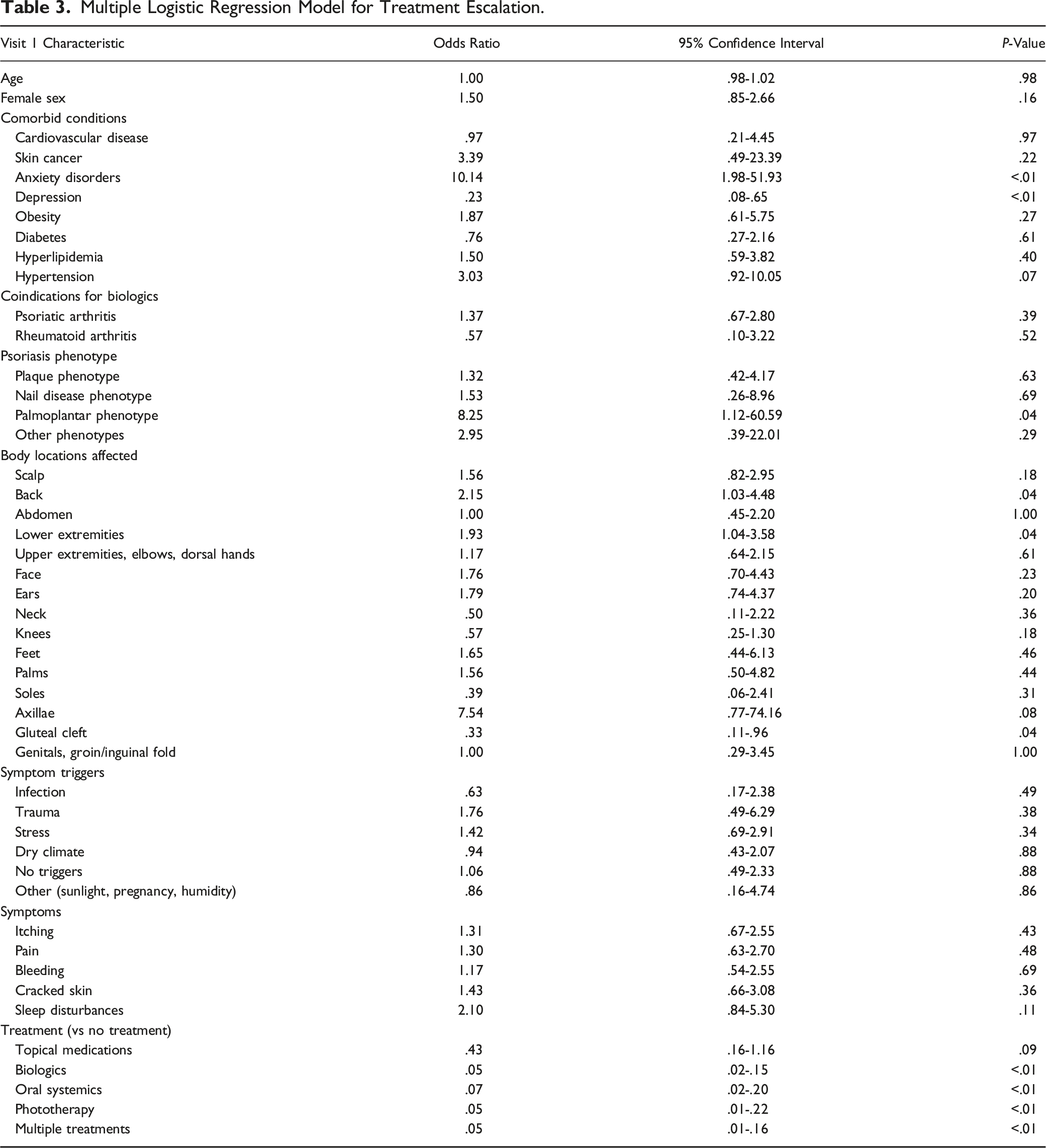
Multiple Logistic Regression Model for Treatment Escalation.
Changes in Psoriasis Severity Measures
Changes in Psoriasis Severity Measures, by Treatment Escalation Group (Multiply Imputed Data).
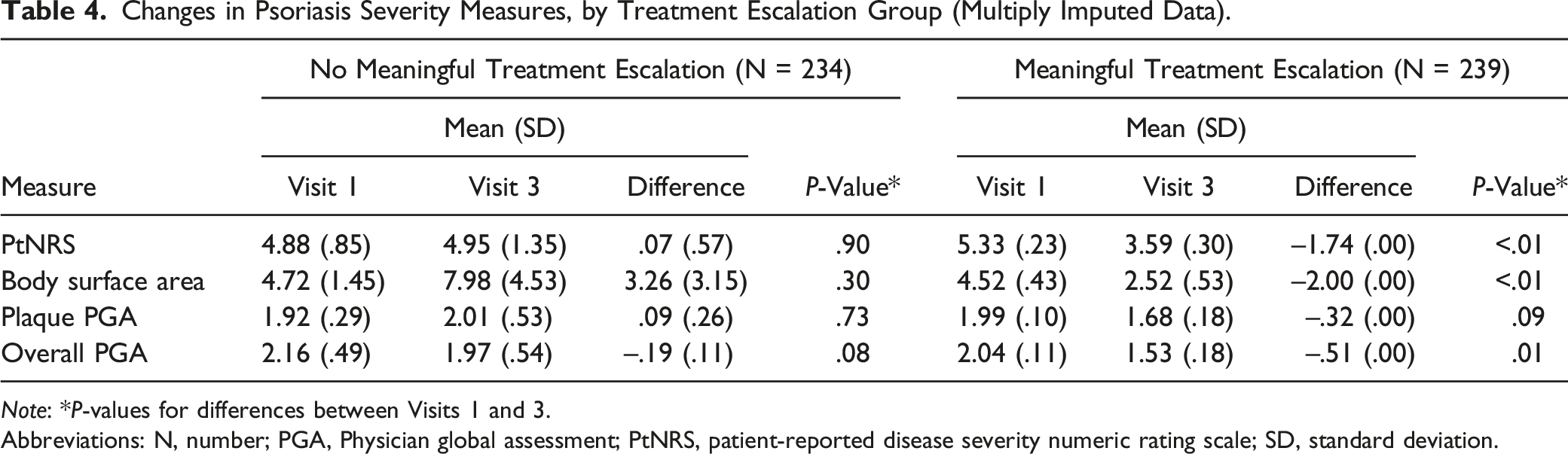
Note: *P-values for differences between Visits 1 and 3.
Abbreviations: N, number; PGA, Physician global assessment; PtNRS, patient-reported disease severity numeric rating scale; SD, standard deviation.
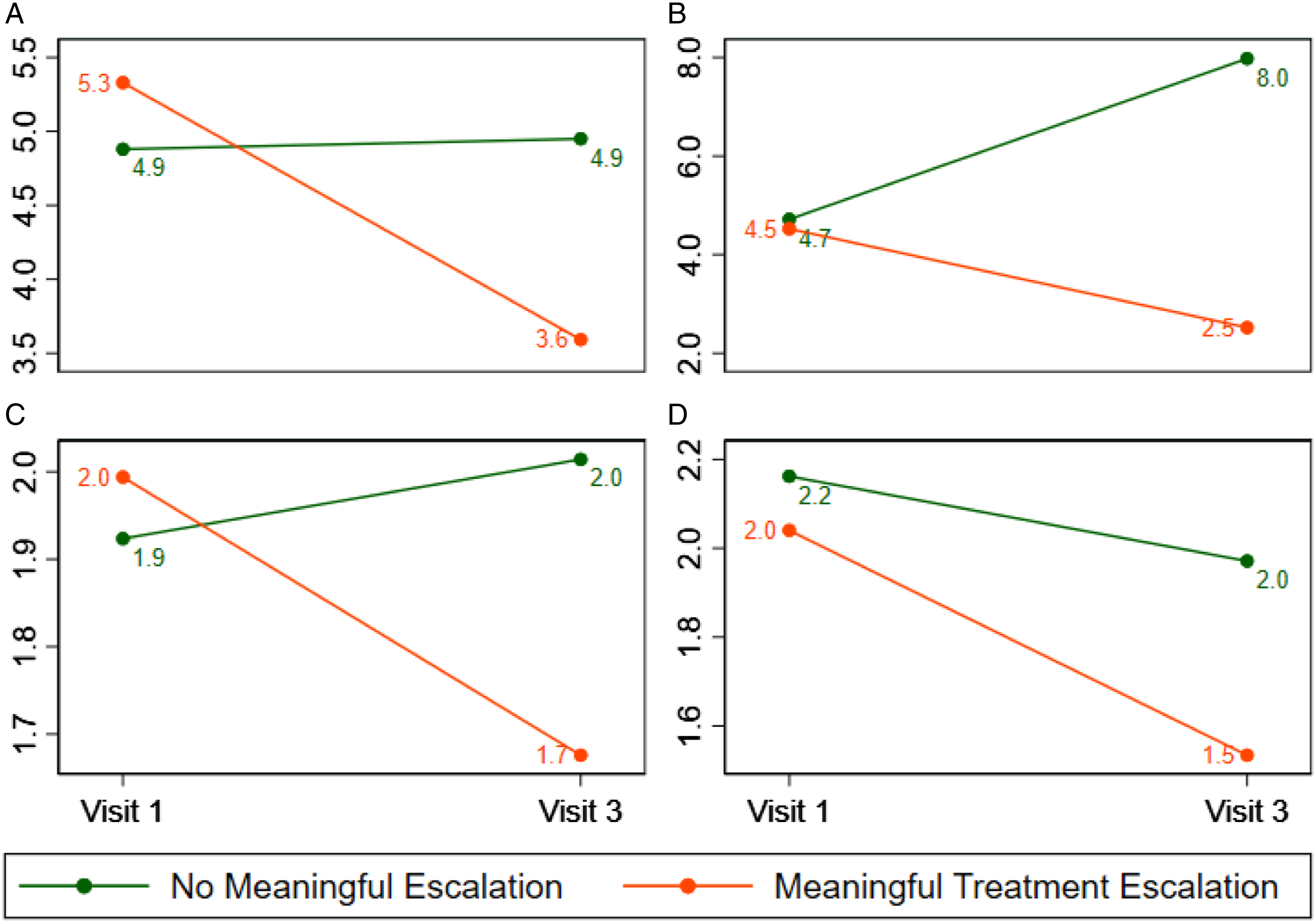
Adjusted changes in psoriasis severity measures, by treatment escalation group (multiply imputed data). (A) Patient reported seventy (B) body surface area (%) (C) Physician global assessment, plaque (D) Physician global assessment, overall.
Discussion
This study was designed to establish the feasibility of using customized visit note templates (SFs) as data collection tools for real-world outcomes research in chronic skin disease. This study demonstrates that a HIPAA-compliant dataset containing psoriasis-specific variables without patient identifiers could be efficiently generated within a matter of weeks, allowing researchers to query data that typically requires formal prospective registries to collect. Disease-specific study outcomes and other measures of interest can be easily added to SFs that already capture information important for clinical care (e.g., a score from a validated instrument that assesses itch). While the University of Utah’s IT services collaborated with clinicians and researchers to generate customized SFs locally, significant efficiencies could be gained if such SFs were developed centrally and shared across institutions. The primary barrier to this approach is a lack of consensus around those measures considered pertinent to clinical care and the research community. More recently, interoperability applications (Substitutable Medical Applications and Reusable Technologies on Fast Healthcare Interoperability Resources) provide opportunities to collect standard of care measures across different EHR platforms, which, for example, has been successfully implemented for assessing bilirubin. 9
The second aim of our study was to examine factors associated with MTE in patients with psoriasis. We observed that anxiety, palmoplantar disease phenotype, and certain affected body locations, such as involvement of the lower extremities, were associated with MTE. Anxiety is well known to be associated with psoriasis10-12 and patients commonly express anxiety about their disease worsening, which could explain why anxiety is associated with therapy escalation. Palmoplantar psoriasis is a very challenging phenotype to treat and having the disease on the hands may precipitate the need to escalate therapy. Lower extremities, which are commonly felt by clinicians to be more recalcitrant, are another area that may require treatment escalation to clear. These observations, generated from structured data, deserve further consideration in trials designed to rigorously address causality.
As expected, we found that severity scores improved more among patients who experienced MTE than those who did not. However, despite adjusting for differences between patients who did and did not experience MTE, severity scores such as BSA and oPGA at Visit 3 for patients who experienced MTE were not as low as those who did not experience MTE suggesting potential unmet treatment needs or that it may take more than three visits to achieve desired improvement. This type of analysis can be used to assess quality (e.g., determining if patients are at target as defined, for example, by the merit-based incentive payment system program)13-15 and increase our ability to initiate therapies in a timely manner by supplying clinical data required for prior authorizations. Likewise, analyzing treatment transitions between visits could be used to gain insights at a local, system, or national level into treatment patterns. With data from only 473 evaluable patients across three visits, we were able to conduct unadjusted and adjusted analyses to identify factors associated with treatment escalation. With a larger dataset generated from multiple centers, this analysis could have been used to identify factors that predict switching from one class of therapy (e.g., anti-tumor necrosis factor-alpha therapies to anti-interleukin-23 therapies) or, even, from one drug to another, establishing patterns that could inform guidelines for which biologics should be used as first- or second-line therapies.
Our findings should be interpreted considering the several limitations inherent to using EHR data and due to our study design.16,17 Given that this is a single-center study with a design limited to drawing observations about association, our conclusions may lack internal validity and may not be generalizable. Adopting SFs across other institutions and applying our model across multiple centers could overcome the limit of generalizability. An important limitation to note is that of incomplete data. The degree to which data are incomplete, or missing, is highly dependent on clinician-level factors such as workflow, patient volume, burnout, and familiarity with the disease-specific measures to be recorded; furthermore, data can be missing if patients received relevant psoriasis care from healthcare providers outside of the UUH or who did not use the EpiCare® system. Scribes, which are commonly employed in specialty clinics, may not always collect the patient-reported disease severity. Although missing BSA, or other severity measures, can be confidently interpreted as missing when not recorded, that is not necessarily the case when other disease measures are missing. For example, we cannot be certain if a body location was not affected or simply not recorded if the corresponding field in the psoriasis SFs was not completed. Similarly, although we assume that the lack of a diagnosis code for a comorbidity indicates the lack of that condition, we cannot be certain that this assumption is true. Additionally, the visits we analyzed were arbitrarily selected and our analyses were not able to account for time on treatment. Another limitation is the lack of access to patient-reported outcome data, which is one of the advantages to analyses done with modern-day psoriasis registries. For example, having access to patient-reported treatment satisfaction data would have allowed us to determine the extent to which improvements seen by patients who underwent MTE (and the lack of relative improvement seen by patients who did not undergo MTE) were due to levels of treatment satisfaction. Although patient questionnaire data were not available to us due to our study’s timeframe, the addition of validated questionnaires that are dermatology- or psoriasis-specific are now active and feasible for future studies. An embedded prospective design and slight revisions in how structured data are entered, with more proactive collection of patient-reported outcome data, could overcome these limitations.
Conclusions
This study highlights an efficient and potentially cost-effective approach to design specialty-relevant data entry templates that generate routine clinical documentation and granular disease-specific data to improve patient care and conduct HSR in the longitudinal management of chronic diseases like psoriasis. Future efforts are needed to gain consensus on physician and patient-reported outcomes and quality measures that are feasible to collect at all visits, which could be driven by development of core outcome sets. This will be critical to establishing the larger datasets and research networks needed for rigorous retrospective or prospective real-world data analyses.
Supplemental Material
Supplemental Material - Factors Associated With Treatment Escalation for Psoriasis: An Analysis of Electronic Health Records Data
Supplemental Material for Factors Associated With Treatment Escalation for Psoriasis: An Analysis of Electronic Health Records Data by Jamie L. W. Rhoads, William N. Malatestinic, Russel Burge, Michael L. Ganz, and Kristina C. Duffin in Journal of Psoriasis and Psoriatic Arthritis®.
Footnotes
Acknowledgments
The authors would like to thank Donald O’Hara and Shanmugapriya Saravanan for their help managing and analyzing the data for this study and Douglas S. McAllister and the UHealth Data Science Services team for their support in data extraction.
Author Contributions
JLWR, WM, RB, and KCD conceived and designed the study, interpreted the data, provided critical review of the manuscript, and revised the manuscript. MLG analyzed and interpreted the data and drafted and revised the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: WM and RB are employees and minor shareholders of Eli Lily and Company. JLWR has received consulting fees from Eli Lilly and Company, Boehringer Ingelheim, Genentech, and Argenx. KCD has received grants and personal fees from Eli Lilly and Company, Amgen, Janssen, Pfizer, UCB, Novartis, Celgene, AbbVie and Sienna; and has received personal fees from Ortho Dermatologica. MLG is an employee of Evidera Inc., an independent research company that received payment from Eli Lilly and Company for the conduct of this study and the development of this manuscript. MLG is a minor shareholder of Thermo Fisher Scientific.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research and editorial support were funded by Eli Lilly and Company, which was involved in the design of the study; interpretation of the data; preparation, review, and approval of the manuscript; and decision to submit the manuscript for publication.
Ethical Statement
Data Availability Statement
The data that support the findings of this study are not openly available to protect patient confidentiality. Please contact the corresponding author with questions about access to the data.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
