Abstract
Background
Palmoplantar pustulosis (PPP) is a rare skin disease characterized by episodes of neutrophilic pustules on the palms of the hands and soles of the feet. Current treatments for PPP have limited efficacy, and there is little real-world evidence characterizing the disease burden of PPP in patients.
Objective
To describe and compare the clinical characteristics and patient-reported outcomes (PROs) of patients with PPP with those of patients with plaque psoriasis.
Methods
This real-world study used data from patients with PPP or plaque psoriasis enrolled in the CorEvitas Psoriasis Registry. Disease characteristics, historical medical data, PROs, and quality-of-life measures were compared between patients with PPP and those with plaque psoriasis. These measures were also compared following patient stratification by body surface area involvement or Psoriasis Area and Severity Index. This study is purely descriptive; no hypothesis testing was performed.
Results
In this data set, patients with PPP (n = 64) reported higher mean overall pain, using the visual analog scale, relative to those with plaque psoriasis (n = 4894). Patients with PPP also reported a greater proportion of work hours missed or affected; greater impairment of work; and greater impairment of daily activity than patients with plaque psoriasis. Similar effects were observed irrespective of patient stratification.
Conclusion
Generally, PPP was associated with worse PROs and a greater impact on quality of life compared with plaque psoriasis. These findings highlight a need to develop effective therapies for PPP to improve disease management.
Introduction
Palmoplantar pustulosis (PPP) is a painful, heterogeneous, neutrophilic skin disease characterized by episodes of eruption of sterile, macroscopic pustules on the palms of the hands and soles of the feet.1-3 Robust data on PPP prevalence are lacking, although some studies have reported the prevalence as ranging from .009% to .120%.4-6 Typically, the disease affects women aged 50-70 years and is often associated with tobacco use, thyroid-gland abnormalities, and nickel sensitivity.1,2,7,8 The disease course is usually chronic and relapsing, and PPP is generally refractory to treatment.1,2 Furthermore, PPP is associated with a significant clinical burden, with patients often experiencing pain, fatigue, and comorbidities such as dyslipidemia and hypertension, all of which can greatly impact quality of life (QoL).1,9-12
Currently available treatment options for PPP typically include those used to treat plaque psoriasis, such as potent topical corticosteroids, and systemic therapies such as biologics and apremilast, as well as cyclosporine, excimer laser, methotrexate, psoralen–ultraviolet A, and retinoids.1,2,8 Unlike in plaque psoriasis, these options generally either have limited efficacy in the treatment of PPP or are associated with toxic short- and long-term side effects.1,2 Indeed, there is a lack of definitive, high-quality evidence to support efficacy for the most widely used treatments in chronic PPP.8,9 To date no biologic therapies are centrally approved in the USA for the treatment of PPP; the interleukin (IL)-23 inhibitor guselkumab has been approved in Japan based on the results of a Phase III trial. 13
Various clinical, genetic, and histopathologic features of PPP are distinct from plaque psoriasis.1,2 Further elucidation of these distinguishing features and the pathophysiologic mechanisms of PPP may augment research into molecularly targeted therapies for PPP; 2 improve individual disease-management programs; and, ultimately, enhance overall outcomes for patients with PPP.
Here we use data from the CorEvitas (formerly Corrona) Psoriasis Registry 14 to describe and compare the clinical characteristics and patient-reported outcomes that differentiate PPP from plaque psoriasis in US-based patients using current treatments for psoriasis.
Methods
This real-world, cross-sectional study included patients with PPP or plaque psoriasis enrolled in the CorEvitas Psoriasis Registry. 14 In collaboration with the US National Psoriasis Foundation, CorEvitas developed the Psoriasis Registry with the aim of enhancing the extent of real-world data on patients with psoriasis. The prospective, multicenter, observational, disease-based registry collects clinical information on patients with a diagnosis of psoriasis, including those with PPP, who are under the care of a dermatologist. As of January 10, 2020, a total of 10 026 patients with psoriasis were enrolled in the CorEvitas Psoriasis Registry. To join the Psoriasis Registry, patients must have a dermatologist-confirmed diagnosis of psoriasis, be aged ≥18 years, provide written informed consent, and have started or switched to a US Food and Drug Administration–approved systemic psoriasis treatment within the prior 12 months or at the time of enrollment. Thus, the registry records a diverse range of outcomes, including clinical, safety, and patient-reported outcomes (PROs), many of which are validated endpoints included in randomized controlled trials.
All participating investigators were required to obtain full board approval for conducting research involving human subjects. Sponsor approval and continuing review was obtained through a central IRB (IntegReview, Protocol number is Corrona-PSO-500). For academic investigative sites that did not receive a waiver to use the central IRB, approval was obtained from the respective governing IRBs and documentation of approval was submitted to the Sponsor prior to initiating any study procedures. All registry subjects were required to provide written informed consent prior to participating.
Clinical characteristics reported in this study include data on the duration of disease, presence and duration of psoriatic arthritis, body surface area (BSA) involvement, and Psoriasis Area Severity Index (PASI). Historical medical data are also reported, detailing prior treatment experience and existing comorbidities. PROs and QoL measures analyzed in this study are the itch visual analog scale (VAS); fatigue VAS; pain VAS; Patient Global Assessment (PtGA); employment status; work hours missed; work impairment; and work hours affected.
For this study, patients were classified as having PPP if they had a dermatologist-confirmed diagnosis of localized palmoplantar pustules (.6% of the registry) or as having plaque psoriasis if they had a dermatologist-confirmed diagnosis of plaque psoriasis without pustules and no other form of psoriasis (48.8% of the registry). Of note, patients were classified as having PPP if they had any history of a PPP diagnosis, with or without concomitant plaque psoriasis. Consequently, patients with PPP may not have had active PPP disease at the time of enrollment in the registry, and their clinical assessment may therefore have been based on symptoms of plaque psoriasis rather than those specific to PPP. Furthermore, the definition of PPP in the registry does not distinguish PPP from palmoplantar pustular psoriasis; therefore patients with a history of palmoplantar pustular psoriasis could have been included in the PPP patient sample.
At enrollment into the registry, the patient data collected included clinical assessments, lifestyle factors, medical history, medication history, and patient demographics. Patient comorbidities, disease characteristics, medications, PROs, and sociodemographics were assessed as variables in this study, and all comparisons were conducted using data collected when patients were enrolled into the registry. A full list of all variables assessed at the study enrollment visit is available online in the Supplemental Information (Table S1).
This study had three main objectives in comparing patients with PPP vs those with plaque psoriasis. These were, first, to describe their clinical characteristics; second, to compare their PROs; and third, to compare characteristics and PROs when stratifying patients by BSA involvement or PASI.
Owing to the small PPP sample size and the exploratory nature of this study, no hypothesis testing was performed; all reported results are descriptive and apply solely to the CorEvitas Psoriasis Registry sample (no inferences can be made about the population from which the registry sample was taken). Categorical variables from the registry were summarized using frequencies and percentages, and continuous variables were described using the mean with standard deviation and the median with interquartile range.
For the third study objective, patients were stratified based on BSA involvement or PASI. Low BSA was defined as <3%, and patients with ≥3% BSA were classified as medium-to-high BSA. Similarly, a PASI of ≤5 was used as the threshold for low PASI, and patients with a score of >5 were classified as medium-to-high PASI. Comparisons were conducted for stratified samples between patients with PPP and those with plaque psoriasis. All analyses were conducted using Stata® release 15 (StataCorp, LLC, College Station, TX, USA).
Results
Patient Sociodemographics
Overall, 64 patients with PPP and 4894 patients with plaque psoriasis were identified and included in the analyses; 41/64 of patients with PPP (64.1%) also had concurrent plaque psoriasis whereas 18/64 of patients with PPP (28.1%) had no other disease morphology. Enrolled patients with PPP and plaque psoriasis were similar in mean age (52.5 for PPP; 50.6 for plaque psoriasis). The PPP group had a higher proportion of female patients compared to the plaque psoriasis group (71.9% vs 44.4%). The 2 patient groups were similar in education level, work status, and body mass index (Table S2). A higher proportion of patients with PPP were current smokers at the time of assessment compared with patients with plaque psoriasis (43.5% vs 17.0%) (Table S2).
Disease Characteristics, Comorbidities, and Prior Treatments
Disease Characteristics, Comorbidities, and Prior Treatment Regimens of Patients with Palmoplantar Pustulosis or Plaque Psoriasis.
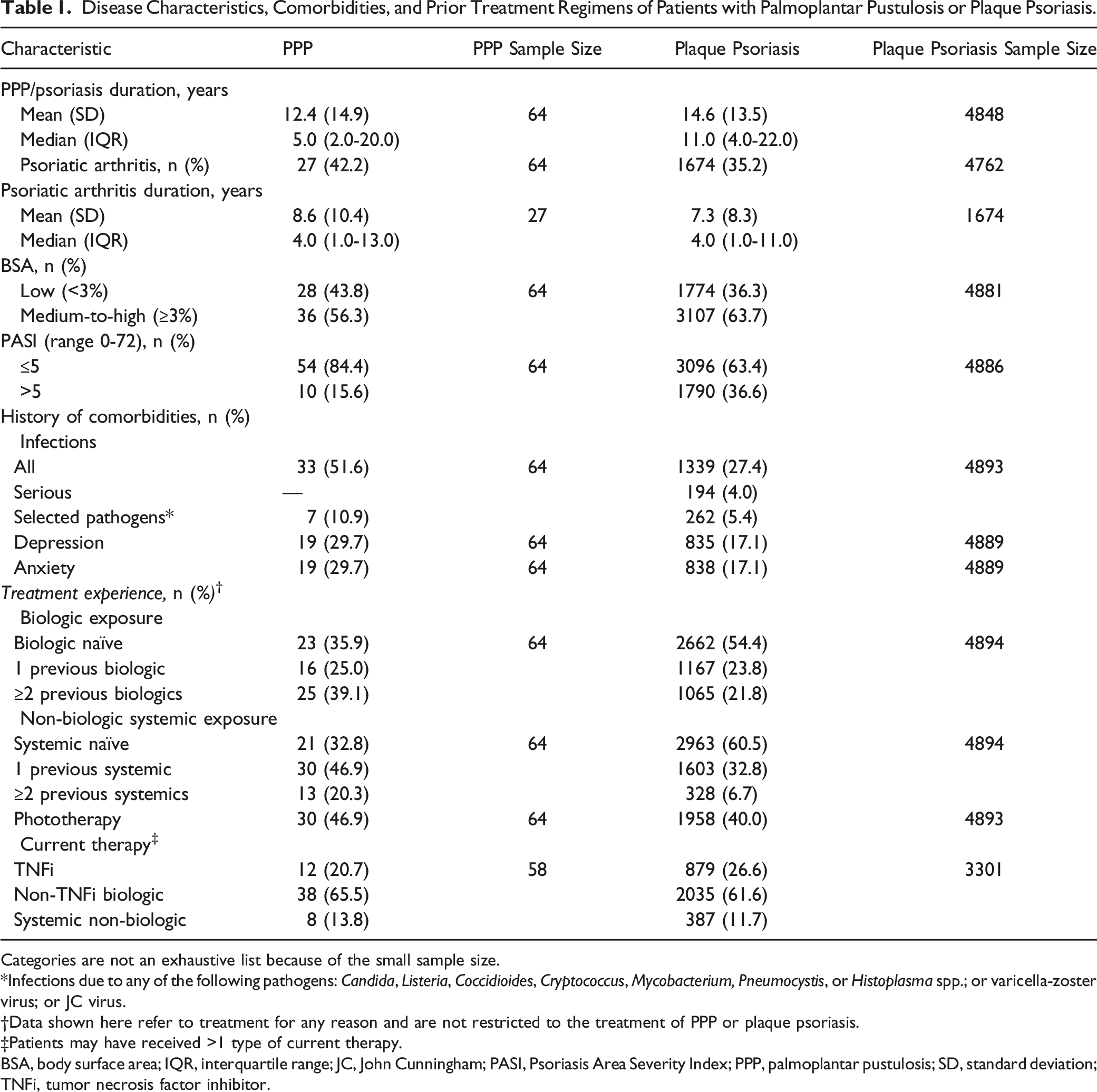
Categories are not an exhaustive list because of the small sample size.
Infections due to any of the following pathogens: Candida, Listeria, Coccidioides, Cryptococcus, Mycobacterium, Pneumocystis, or Histoplasma spp.; or varicella-zoster virus; or JC virus.
Data shown here refer to treatment for any reason and are not restricted to the treatment of PPP or plaque psoriasis.
Patients may have received >1 type of current therapy.
BSA, body surface area; IQR, interquartile range; JC, John Cunningham; PASI, Psoriasis Area Severity Index; PPP, palmoplantar pustulosis; SD, standard deviation; TNFi, tumor necrosis factor inhibitor.
PROs and quality of life
Patient-Reported Symptom and Quality-of-Life Measures.
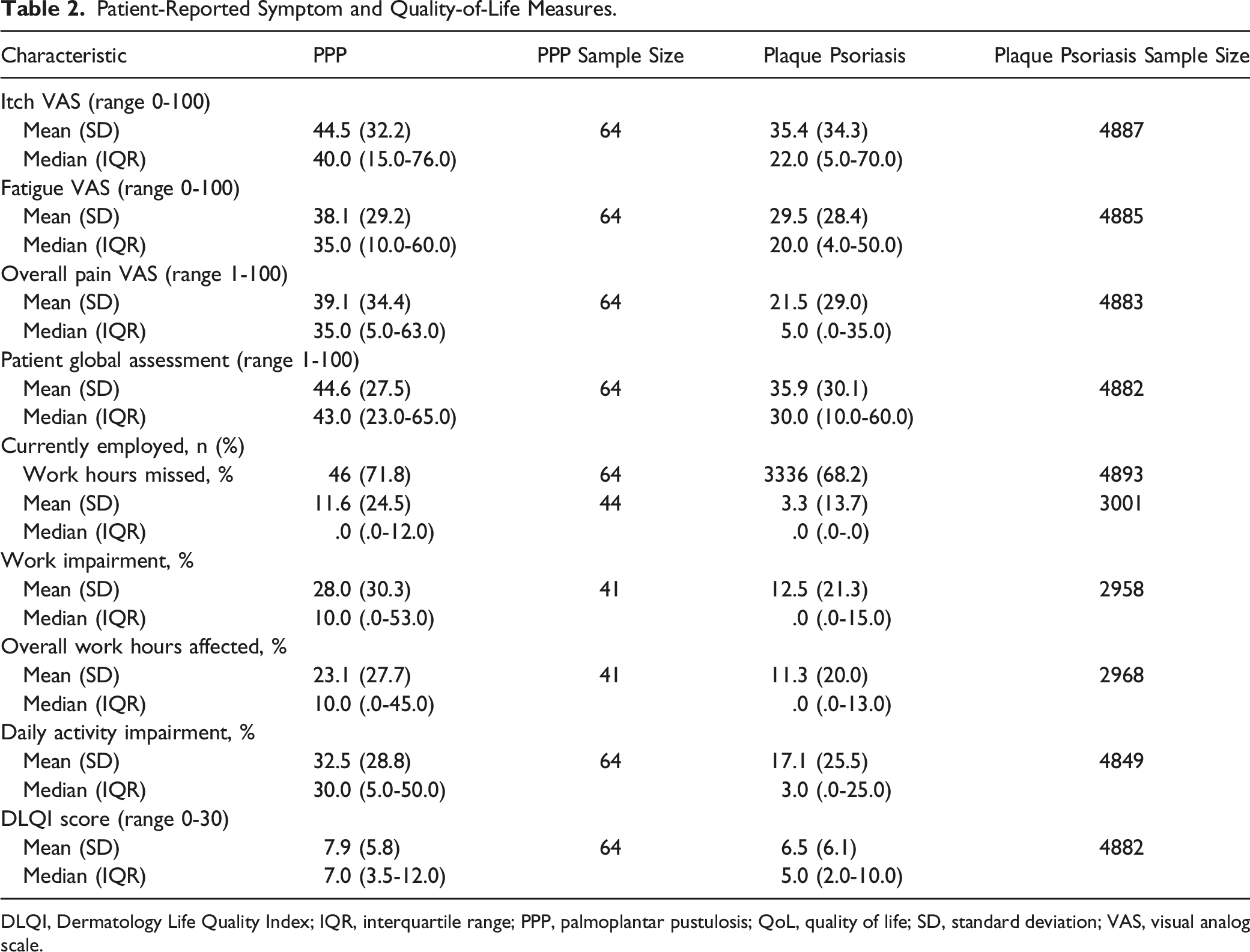
DLQI, Dermatology Life Quality Index; IQR, interquartile range; PPP, palmoplantar pustulosis; QoL, quality of life; SD, standard deviation; VAS, visual analog scale.
Patient stratification by BSA or PASI involvement
Clinical characteristics and PROs of patients with PPP or plaque psoriasis were analyzed following stratification by BSA involvement and PASI. Low BSA and low PASI were classified as <3% and ≤5, respectively, with values above this threshold classified as a medium-to-high score.
PPP and Plaque Psoriasis Characteristics Stratified by Percentage BSA Involvement.
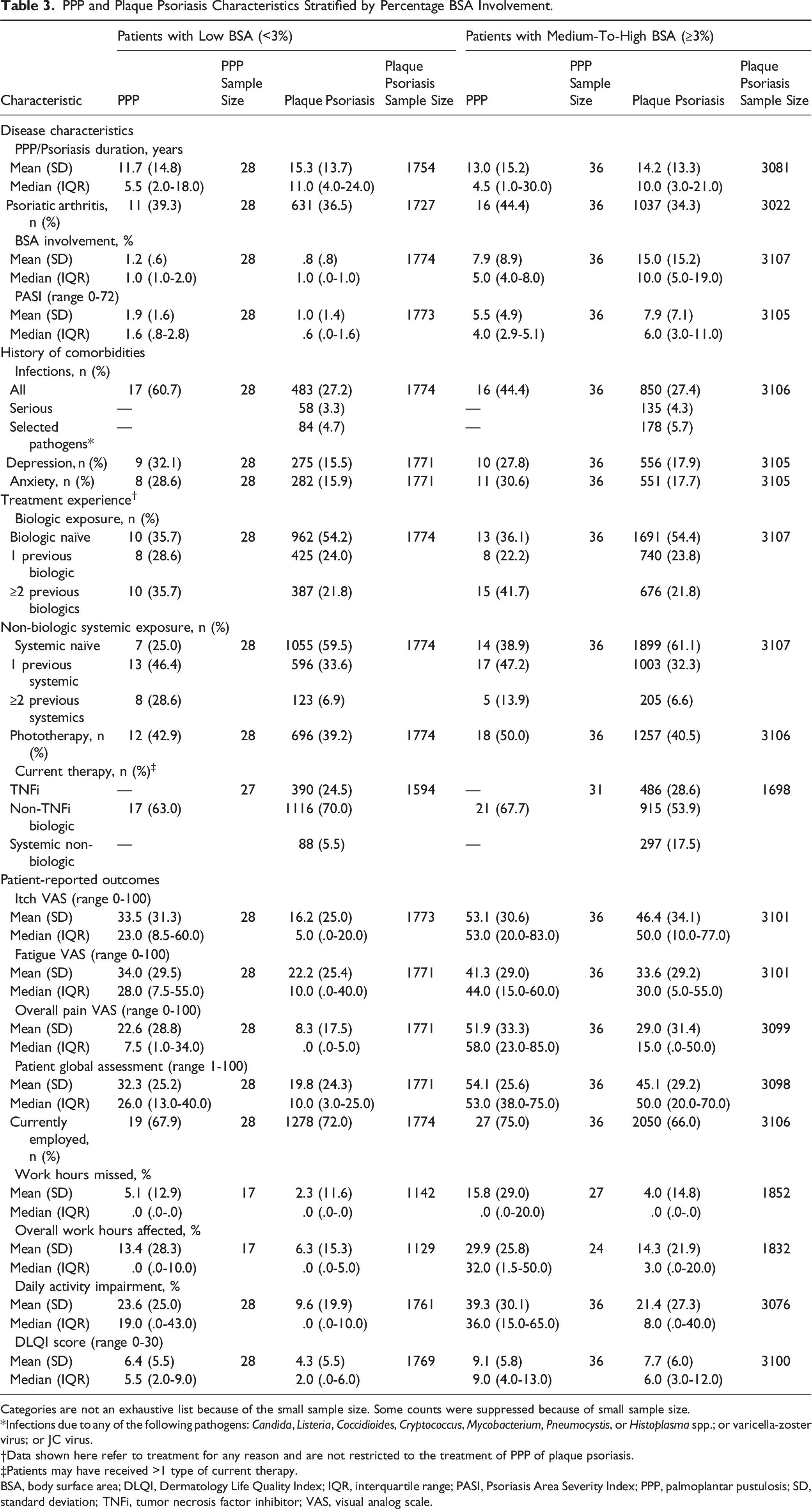
Categories are not an exhaustive list because of the small sample size. Some counts were suppressed because of small sample size.
Infections due to any of the following pathogens: Candida, Listeria, Coccidioides, Cryptococcus, Mycobacterium, Pneumocystis, or Histoplasma spp.; or varicella-zoster virus; or JC virus.
Data shown here refer to treatment for any reason and are not restricted to the treatment of PPP of plaque psoriasis.
Patients may have received >1 type of current therapy.
BSA, body surface area; DLQI, Dermatology Life Quality Index; IQR, interquartile range; PASI, Psoriasis Area Severity Index; PPP, palmoplantar pustulosis; SD, standard deviation; TNFi, tumor necrosis factor inhibitor; VAS, visual analog scale.
PPP and Plaque Psoriasis Characteristics Stratified by PASI.
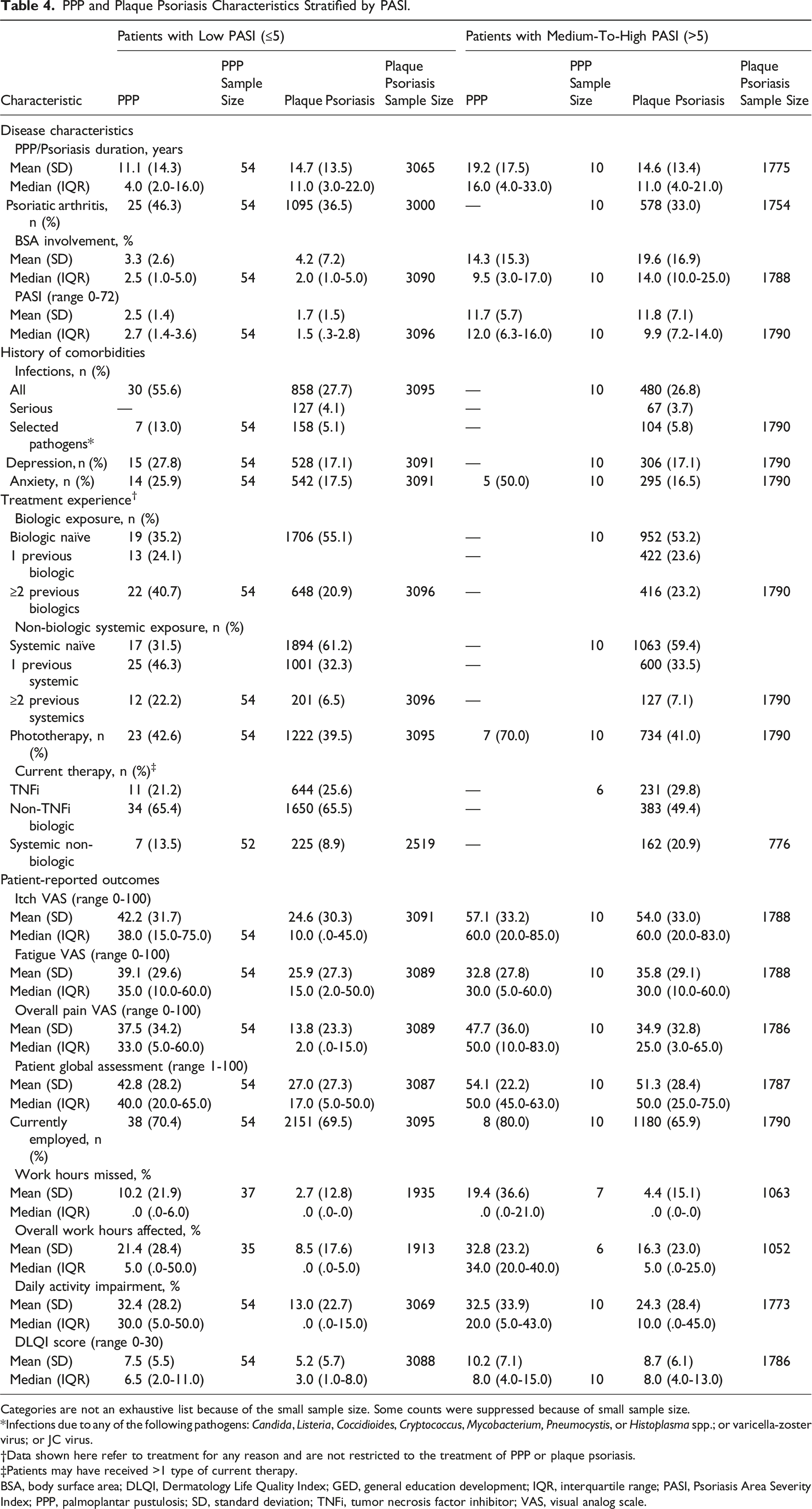
Categories are not an exhaustive list because of the small sample size. Some counts were suppressed because of small sample size.
Infections due to any of the following pathogens: Candida, Listeria, Coccidioides, Cryptococcus, Mycobacterium, Pneumocystis, or Histoplasma spp.; or varicella-zoster virus; or JC virus.
Data shown here refer to treatment for any reason and are not restricted to the treatment of PPP or plaque psoriasis.
Patients may have received >1 type of current therapy.
BSA, body surface area; DLQI, Dermatology Life Quality Index; GED, general education development; IQR, interquartile range; PASI, Psoriasis Area Severity Index; PPP, palmoplantar pustulosis; SD, standard deviation; TNFi, tumor necrosis factor inhibitor; VAS, visual analog scale.
Discussion
In this sample, PPP was associated with worse PROs (eg, pain, itch, and/or fatigue, and impairment of work and daily activities) than plaque psoriasis, particularly when these outcomes were analyzed after stratification for BSA affected or PASI. Of note, in this sample, patients with PPP reported more hours of work missed or affected due to their disease relative to patients with plaque psoriasis, irrespective of their stratification by either disease measure. This is concordant with previous studies, underscoring the significant clinical impact of PPP and the impaired QoL of patients with the disease.11,15,16 Importantly, the results of this study highlight the unsuitability of BSA involvement as a measure of disease severity for PPP, which affects a low percentage value of BSA by definition.
These findings parallel those seen in a study of patients with generalized pustular psoriasis (GPP) in the CorEvitas Psoriasis Registry; patients with GPP had a greater severity of symptoms of pain (mean, 33.1 vs 21.5), fatigue (mean, 42.6 vs 29.5), and itch (mean, 47.7 vs 35.4) and experienced a greater impact on QoL than patients with plaque psoriasis. 17 However, it should be noted that no formal comparison between patients with PPP or GPP has been made in the CorEvitas registry.
There are limitations to this study. First, the sample size of the PPP patient group was small compared with the plaque psoriasis sample (n = 64 vs n = 4894). Owing to the small sample size, which reflects the rarity of PPP, it was decided not to perform hypothesis testing. Furthermore, as this was an observational study, there is the potential for findings to be biased by confounding factors. For instance, the data reported on the number of infections in each patient sample could have been confounded by prior treatment experience; indeed, a greater proportion of patients with PPP had prior treatment with biologics than did patients with plaque psoriasis. It is also possible that prior treatment experience may have had a confounding effect on QoL measures through adverse events or treatment burden. It is important to note that the treatment experience data presented in this study do not specify the amount of time spent on treatment and therefore may not fully capture a patient’s treatment exposure.
In addition, an overlap in disease morphologies may have confounded the results. As PPP is rare and can coexist with plaque psoriasis (or other disease morphologies), 64.1% of patients in the PPP sample had concurrent plaque psoriasis, whereas patients in the plaque psoriasis sample could not have other morphologies. However, it is possible that characteristics reported here for patients with PPP were based on symptoms of active plaque psoriasis. Indeed, it may be the case in the stratified PPP patient cohort with medium-to-high BSA involvement. Similarly, it is possible that the treatments captured in this registry for patients with PPP may have been prescribed to treat non-PPP disease. Moreover, as the PPP cohort was classified as patients with a history of PPP with or without plaque psoriasis, and PPP was not distinguished from palmoplantar pustular psoriasis, it is unclear whether patients presented with active PPP at the time of enrollment and assessment. It is also important to note that the severity measures used in this study, such as the PASI and BSA involvement, are specifically used for the assessment of plaque psoriasis. Therefore, these may not fully reflect the extent of PPP symptoms, highlighting a requirement for PPP-specific disease measures.
Overall, this study provides key, real-world insights into the clinical characteristics and disease burden faced by patients with PPP, which serves as an important prelude to further clinical research into potential targeted treatment options. The findings of this study highlight a clear need to develop detailed and effective approaches for the management of PPP so that the overall burden of this rare disease can be reduced and, ultimately, PROs and patient QoL can be improved.
Supplemental Material
Supplemental Material - Palmoplantar Pustulosis has a Greater Disease Burden than Plaque Psoriasis: Real-World Evidence From the CorEvitas Psoriasis Registry
Supplemental Material for Palmoplantar Pustulosis has a Greater Disease Burden than Plaque Psoriasis: Real-World Evidence From the CorEvitas Psoriasis Registry by Mark G. Lebwohl, MD, Rose Anne Medeiros, PhD, Rachel H. Mackey, PhD, Wendell C. Valdecantos, MD, Kimberly G. Brodovicz, DrPH, Apinya Lertratanakul, MD, and Bruce Strober, MD, PhD in Journal of Psoriasis and Psoriatic Arthritis
Footnotes
Acknowledgments
All authors meet the criteria for authorship as recommended by the International Committee of Medical Journal Editors and made the decision to submit the manuscript for publication. The authors did not receive payment related to the development of the manuscript. Agreements between Boehringer Ingelheim and the authors included the confidentiality of the study data. In the preparation of this manuscript, David Murdoch, BSc (Hons), and Isabella Goldsbrough, PhD, of OPEN Health Medical Communications (London, UK) provided medical writing, editorial support, and/or formatting support, which was contracted and funded by BI. BI was given the opportunity to review the manuscript for medical and scientific accuracy as well as intellectual property considerations. We thank all the patients and study investigators who participated in the clinical study described here.
Declaration of Conflicts of Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MGL is an employee of Mount Sinai and receives research funds from AbbVie, Amgen, Arcutis, Avotres, Boehringer Ingelheim, Cara Therapeutics, Dermavant Sciences, Eli Lilly, Incyte, Janssen Research and Development, LLC, Ortho Dermatologics, Regeneron, and UCB, Inc., and is a consultant for Aditum Bio, Almirall, AltruBio, Inc., AnaptysBio, Arcutis, Inc., Arena Pharmaceuticals, Aristea Therapeutics, Arrive Technologies, Avotres Therapeutics, BiomX, Brickell Biotech, Boehringer Ingelheim, Bristol Myers Squibb, Cara Therapeutics, Castle Biosciences, CorEvitas, Dermavant Sciences, Dr Reddy’s Laboratories, Evelo Biosciences, Evommune, Inc., Facilitation of International Dermatology Education, Forte Biosciences, Foundation for Research and Education in Dermatology, Helsinki Therapeutics, Hexima Ltd, LEO Pharma, Meiji Seika Pharma, Mindera, Pfizer, Seanergy, and Verrica. RAM was an employee of CorEvitas, LLC, at the time of the study being conducted but is currently employed by R.A. Medeiros Statistical Consulting. RHM was an employee of CorEvitas, LLC, at the time of the study being conducted but is currently employed by Premier, Inc. WCV, KGB, and AL are full-time employees of Boehringer Ingelheim. BS declares receiving consultant honoraria from AbbVie, Almirall, Amgen, Arcutis, Arena, Aristea, Asana, Boehringer Ingelheim, Immunic Therapeutics, Bristol Myers Squibb, Connect Biopharma, Dermavant, Eli Lilly, Equillium, GlaxoSmithKline, Janssen, LEO Pharma, Maruho, Meiji Seika Pharma, Mindera, Novartis, Ortho Dermatologics, Pfizer, Regeneron, Sanofi-Genzyme, Sun Pharma, UCB, and Ventyx Biosciences; has been a speaker for AbbVie, Eli Lilly, Janssen, and Sanofi-Genzyme; is a co-scientific director (receiving a consulting fee) for the CorEvitas Psoriasis Registry; is an investigator for AbbVie, Cara Therapeutics, the CorEvitas Psoriasis Registry, Dermavant, Dermira, and Novartis; and is the editor-in-chief (receiving an honorarium) of the Journal of Psoriasis and Psoriatic Arthritis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by CorEvitas, LLC, and the analysis was funded by Boehringer Ingelheim. Access to study data was limited to CorEvitas and CorEvitas statisticians completed all the analysis; all authors contributed to the interpretation of the results. CorEvitas has been supported through contracted subscriptions in the past 2 years by AbbVie, Amgen, Inc., Arena, Boehringer Ingelheim, Bristol Myers Squibb, Celgene, Chugai, Eli Lilly and Company, Genentech, Gilead Sciences, Inc., GlaxoSmithKline, Janssen Pharmaceuticals, Inc., LEO Pharma, Novartis, Ortho Dermatologics, Pfizer, Inc., Regeneron Pharmaceuticals, Inc., Sanofi, Sun Pharmaceutical Industries Ltd, and UCB S.A. The CorEvitas Psoriasis Registry was developed in collaboration with the National Psoriasis Foundation.
Authors’ Note
Data are available from CorEvitas, LLC, through a commercial subscription agreement and are not publicly available. No additional data are available from the authors.
IRB Approval
All participating investigators were required to obtain full board approval for conducting research involving human subjects. Sponsor approval and continuing review was obtained through a central IRB (IntegReview, Protocol number is Corrona-PSO-500). For academic investigative sites that did not receive a waiver to use the central IRB, approval was obtained from the respective governing IRBs and documentation of approval was submitted to the Sponsor prior to initiating any study procedures. All registry subjects were required to provide written informed consent prior to participating.
Supplementary Material
Supplementary material for this article is available online.
CME Credit
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
