Abstract
Introduction
Retrolental membranes in pediatric patients present a diagnostic challenge owing to their broad differential diagnosis, which includes persistent fetal vasculature (PFV), retinoblastoma, Coats disease, retinopathy of prematurity (ROP), familial exudative vitreoretinopathy, and inflammatory or infectious etiologies. Accurate differentiation is critical, as management strategies and prognoses vary significantly among these conditions.
Ocular toxocariasis, a parasitic infection caused by
We report a case of ocular toxocariasis initially suspected to represent PFV, highlighting the importance of multimodal imaging, intraoperative assessment, and targeted laboratory evaluation in establishing the correct diagnosis.
Case Report
A 4-year-old Guatemalan boy, born at term with no significant medical history or prior ocular trauma, presented with a 3-week history of right eye irritation. Visual acuity (VA) was limited to counting fingers in the right eye and 20/40 in the left eye. Intraocular pressure was soft to palpation bilaterally. Pupils were round and reactive without a relative afferent pupillary defect, and extraocular movements were full. Slitlamp examination of the right eye revealed a retrolental membrane with a poor view of the fundus. The left eye examination was unremarkable. B-scan ultrasonography of the right eye showed a funnel-shaped hyperechoic membrane extending from the optic nerve to the lens.
Due to limited cooperation at the bedside, an examination under anesthesia (EUA) of the patient was performed, revealing intraocular pressure of 14 mm Hg OD and 18 mm Hg OS. Axial lengths were 22.7 mm OD and 23.2 mm OS. Anterior segment findings were consistent with the prior slitlamp examination, demonstrating a retrolental membrane (Figure 1A). B-scan ultrasonography of the right eye showed a hyperechoic membrane extending from the optic nerve to the lens (Figure 1B). No solid mass or calcifications were identified, and there was no evidence of tractional retinal detachment. Optical coherence tomography demonstrated a flat macula in both eyes with no signs of epiretinal membrane. Indirect ophthalmoscopy of the right eye revealed a funnel-shaped avascular vitreous stalk. No exudation or vascular telangiectasia was noted, and the optic nerve appeared normal. Fluorescein angiography (FA) revealed peripheral avascular retina with areas of hyperfluorescence and leakage in the superior peripheral retina (Figure 2). The left eye examination was unremarkable.
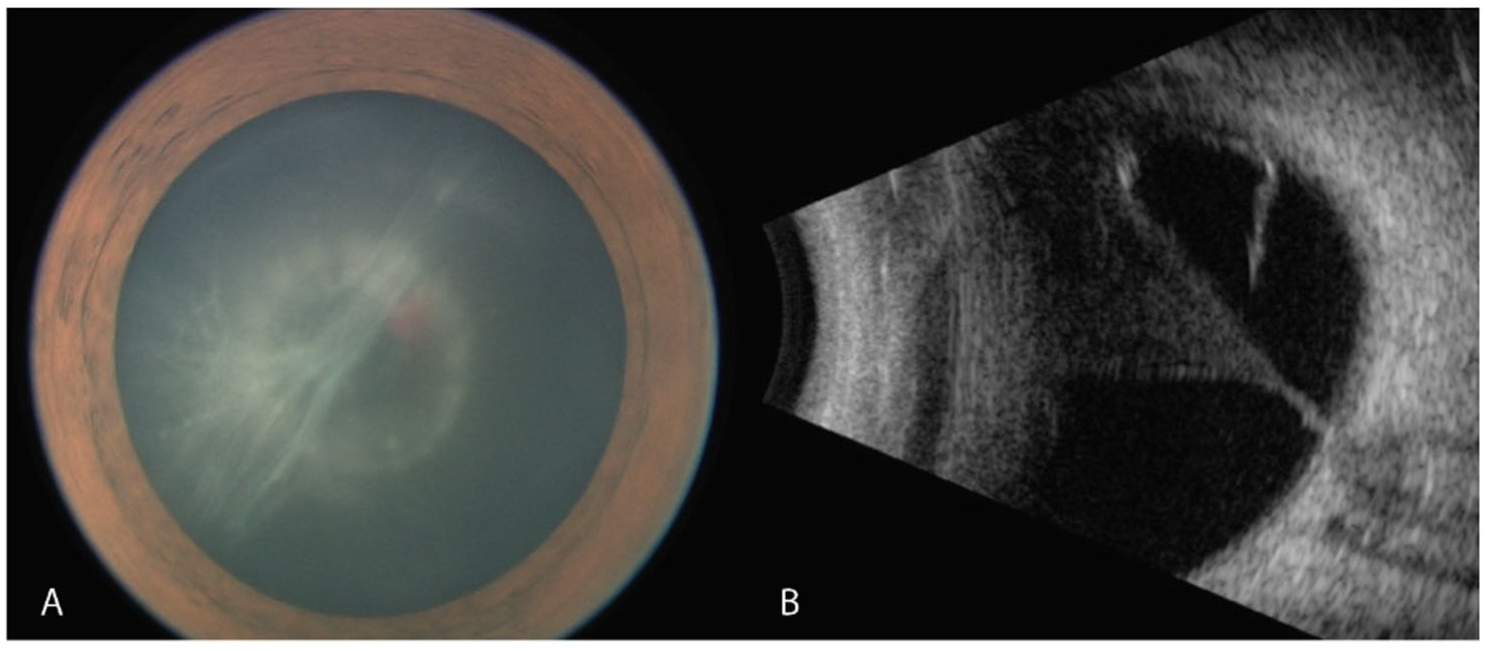
Anterior segment photograph and B-scan ultrasonography of the right eye obtained during examination under anesthesia. (A) Anterior segment photograph shows white membranous vitreous strands behind the lens in the inferotemporal region. (B) B-scan ultrasonography demonstrates a funnel-shaped hyperechoic membrane extending from the optic disc to the posterior lens capsule.
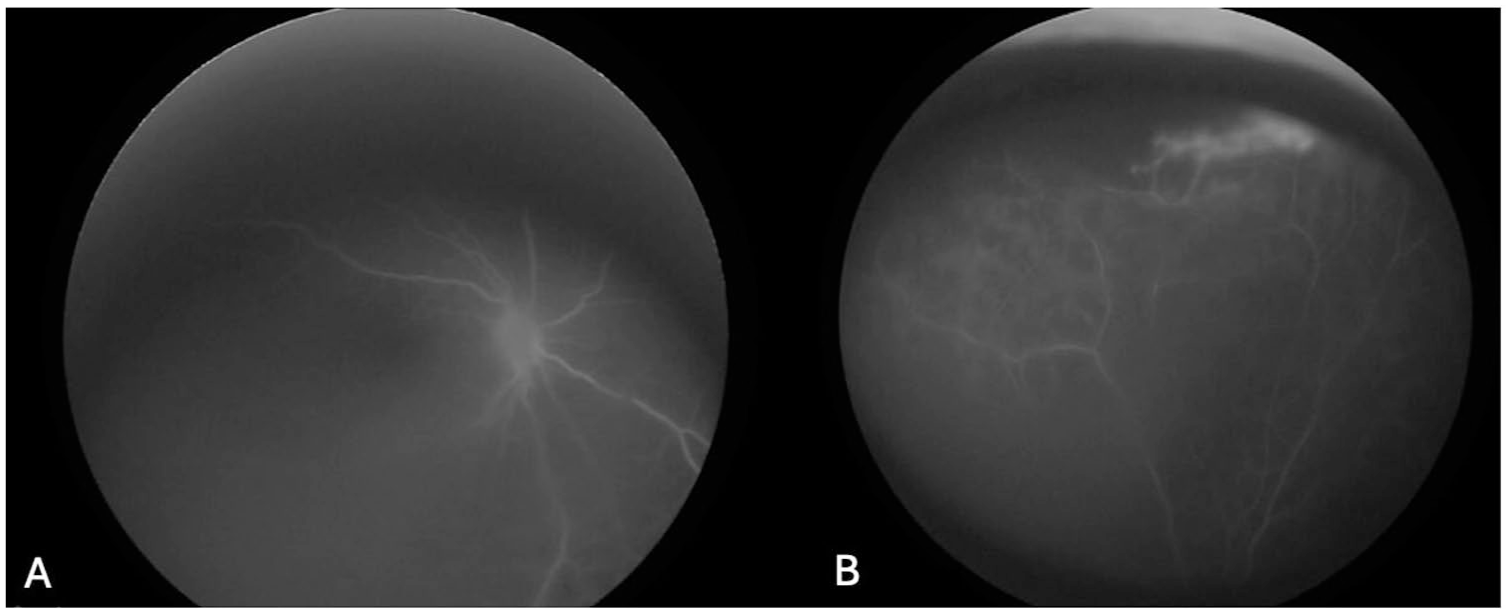
Early-phase fluorescein angiography of the right eye shows (A) avascularity along the vitreous stalk and (B) avascular areas of the superior peripheral retina with associated capillary leakage.
After EUA, pars plana vitrectomy and lensectomy with peripheral iridectomy were performed due to the visually significant retrolental membrane. The lens was removed first to improve the visualization of the posterior segment. The vitreous stalk was wispy and avascular and was carefully trimmed down to the optic nerve. Intraoperatively, areas of ischemic temporal retina and 2 inferonasal chorioretinal granulomatous scars at the 5 and 6 o’clock positions were noted (Figure 3). The scars appeared fibrotic and inactive; however, associated vitritis and diffuse peripheral sclerotic vessels suggested chronic inflammation. The peripheral vitreous was meticulously shaved down to the vitreous base using scleral depression, being careful not to unroof the granulomas. No tractional retinal detachment was observed. Inferior endolaser photocoagulation was applied to the inferior retina surrounding each granulomatous scar and to adjacent avascular areas. An inferior peripheral iridectomy was created, and the patient was left aphakic at the conclusion of the procedure.
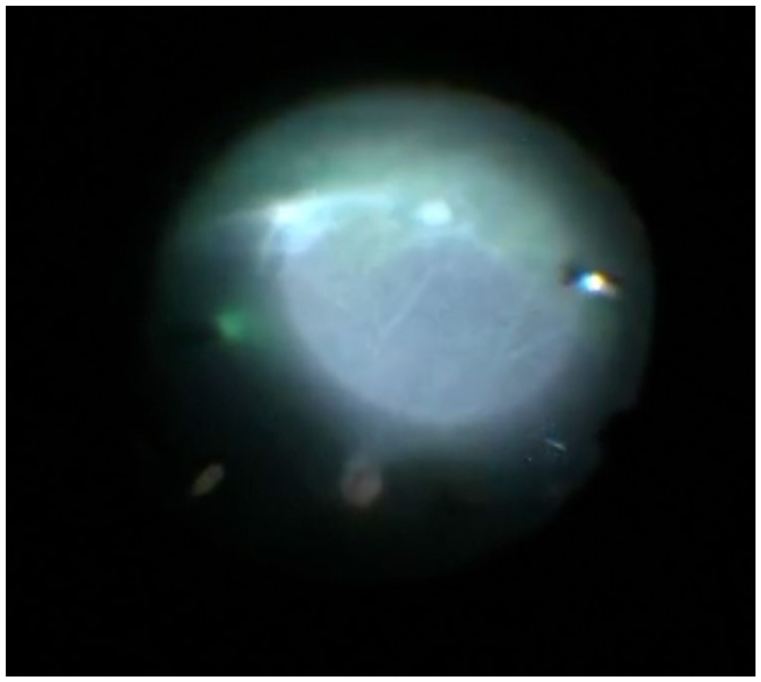
Intraoperative surgical video freeze frame (surgeon’s view, right eye) demonstrates 2 chorioretinal granulomas at the 5 and 6 o’clock positions. The granulomas were not visualized on fluorescein angiography due to limited posterior segment view through the retrolental membrane.
Intraoperative findings of a retrolental membrane arising from an inferior granuloma, peripheral ischemic retina, and vascular leakage raised a high index of suspicion for an infectious etiology. To establish a diagnosis, polymerase chain reaction of vitreous fluid was performed for both
Postoperatively, topical and systemic corticosteroids reduced intraocular inflammation, which resolved after 4 months (Figure 4). Aphakic spectacles were prescribed, along with patching of the left eye for amblyopia therapy. VA improved to a range of 20/80 to 20/150. However, at 6 months, VA declined to counting fingers, accompanied by recurrent anterior chamber inflammation. Hourly topical corticosteroids led to partial improvement, but inflammation persisted, and VA remained limited to counting fingers. After consultation with pediatric infectious disease specialists, oral corticosteroids and albendazole were initiated to address persistent inflammation in the setting of a confirmed infectious etiology. Subsequently, VA improved to 20/80 with aphakic correction and continued amblyopia patching therapy.
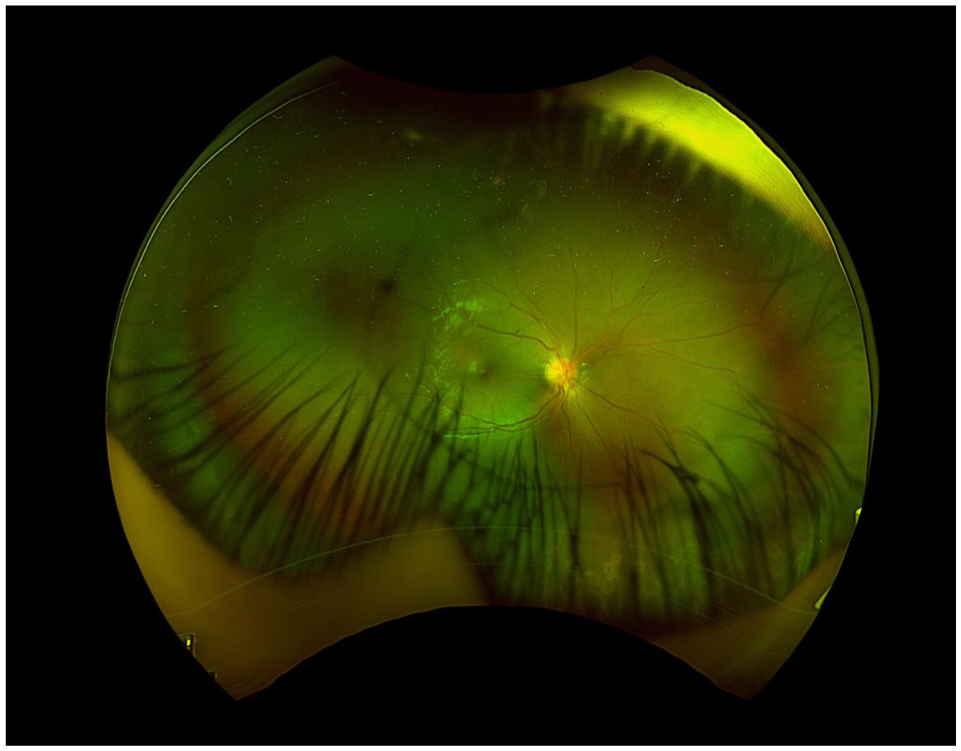
Ultra-widefield pseudocolor fundus image of the right eye obtained 2 weeks postoperatively shows a clearer posterior segment view, attached retina, and inferior laser photocoagulation scars.
Conclusions
We describe a case of a 4-year-old child presenting with a retrolental membrane. In pediatric patients, the differential diagnosis of a retrolental membrane includes PFV, retinoblastoma, Coats disease, ROP, familial exudative vitreoretinopathy, infectious causes, and noninfectious inflammatory etiologies.1–4 The finding of a retrolental membrane and a funnel-shaped hyperechoic membrane extending from the optic nerve to the lens on ultrasonography made PFV the leading diagnosis in our differential. PFV occurs when the embryonic hyaloid vasculature, which normally runs from the optic disc to the lens, fails to regress. 5 Although the vitreous membrane in this case was initially suggestive of PFV, detailed examination using multimodal imaging revealed a peripheral granuloma and vascular leakage, which indicate inflammatory causes rather than a developmental malformation.
Inflammatory causes may be infectious or noninfectious; however, infectious etiologies should always be ruled out first. Microorganisms that may present as posterior pole granulomas include bacteria (eg,
Ocular toxocariasis classically presents as a posterior granuloma, peripheral granuloma, or chronic endophthalmitis. Granulomas can cause retinal traction and membrane formation, leading to retinal folds, tractional retinal detachments, and retinal tears. These complications can manifest as leukocoria or retrolental membranes.7,8 Our patient exhibited a PFV-like presentation, with a vitreous stalk extending from the optic nerve head to the posterior lens capsule, beyond the peripheral retina. The classic
The current standard treatment for ocular toxocariasis involves corticosteroids to manage active intraocular inflammation. While immunomodulatory drugs may be beneficial for long-term inflammation control, they are unsuitable for immediate management as their therapeutic effect requires several weeks. 9 In this case, the patient was initially treated with oral and topical corticosteroids to address intraocular inflammation. Although oral albendazole is the drug of choice for systemic toxocariasis, its role in ocular toxocariasis remains controversial due to a lack of controlled clinical trials and limited data on the ocular pharmacokinetics and pharmacodynamics of anthelmintic agents. Nevertheless, studies suggest that combined systemic corticosteroids and anthelmintic therapy may yield favorable outcomes and have been shown to significantly reduce recurrence rates compared with corticosteroid monotherapy. 8
We present the case of a 4-year-old boy whose initial ocular examination was highly suggestive of PFV. However, detailed evaluation in the operating room, combined with multimodal imaging and intraoperative findings, shifted our suspicion toward infectious inflammatory causes. Subsequent serologic testing confirmed the diagnosis of ocular toxocariasis. Postoperatively, the patient’s VA improved to 20/150 with amblyopia therapy, aphakic correction, a course of oral and topical corticosteroids, oral albendazole, and placement of a secondary sulcus intraocular lens.
This case emphasizes the importance of a thorough history, comprehensive ophthalmic examination, multimodal imaging, and laboratory workup to establish an accurate diagnosis, rule out potentially life-threatening diseases, and provide appropriate treatment.
Footnotes
Ethical Approval
This case report was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information was performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
In accordance with the institutional review board guidelines, patient consent was not required for this case report owing to the fully de-identified nature of all information presented herein.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed and submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Dr. Hua has served on advisory boards for AbbVie/Allergan, Alcon, Alimera/ANI, EyePoint, and Genentech. None of the other authors declared potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH Center Core Grant (P30EY14801) and an unrestricted grant from Research to Prevent Blindness (GR004596-1).
