Abstract
Keywords
Introduction
Approximately 15 million infants are born prematurely each year, including nearly 50 000 infants born in the United States. 1 Infants born at less than 30 weeks of gestation or at very low birth weights are at risk for retinopathy of prematurity (ROP), 2 in which abnormally developed retinal vasculature may result in retinal detachment and permanent visual impairment, including blindness. 3 Thus, appropriate treatment for ROP is critical to prevent adverse visual outcomes and to maximize the quality of life of those patients meeting the criteria. The traditional treatment for sight-threatening ROP has been panretinal photocoagulation (PRP), although intravitreal (IVT) bevacizumab is now widely used as a primary treatment.4,5
There are significant variations in the choice of treatment globally, likely relating to resources and costs. 6 Treatment of ROP with PRP is more definitive and may require fewer follow-up visits compared with IVT bevacizumab monotherapy. However, PRP has been associated with increased risks of high myopia in addition to permanent scarring of the peripheral retina and peripheral visual field deficits. 7 Additionally, a recent survey of ophthalmologists who treat ROP found that systemic challenges accompany the use of PRP and present barriers to treatment, including lack of access to anesthesia or an operating room as well as the length of time needed for the procedure. These barriers were much less problematic with IVT bevacizumab monotherapy. 8
IVT bevacizumab is now increasingly used because it avoids permanent destruction of the peripheral retina. 9 However, both early recurrence of stage 3 ROP and late recurrences of stage 4 and 5 ROP have been reported infrequently.10–12 In their approved guidelines, the American Academy of Pediatrics recommends weekly eye examinations after treatment with IVT bevacizumab until 65 weeks postmenstrual age to ensure there are no recurrences requiring treatment. 13 Due to the risk of late recurrences of treatment-requiring ROP and the presence of chronic peripheral vascular arrest, some providers prefer supplementation with rescue laser photocoagulation in infants treated with IVT bevacizumab before discharge from the neonatal intensive care unit, or approximately 55 to 60 weeks post-conceptual age (PCA).14,15 Although IVT bevacizumab monotherapy prevents some of the complications of PRP, more frequent follow-up examinations and possibly further treatment are required, thus potentially increasing the cost of treating ROP.
ROP has a significant economic impact, with Rothschild et al 16 estimating that the cost per infant treated in the US in 2014 was $9821 using treatment modalities including antivascular endothelial growth factor (anti-VEGF) therapy or PRP. The purpose of this study was to evaluate the economic impact of monotherapy with IVT bevacizumab and compare it with combined IVT bevacizumab and PRP for the treatment of ROP at a single site in the US.
Methods
Patient Population
The study was conducted according to the tenets of the Declaration of Helsinki and was approved by the institutional review board. Premature infants undergoing treatment for ROP at the University of Florida between 2014 and 2019 were included in the study. Informed consent was obtained from all parents or guardians of the infants participating in the study. Screening for ROP was conducted for all infants born at ≤30 weeks gestational age, weighing ≤1500 g, or infants with an unstable clinical course. ROP was classified according to the International Classification for Retinopathy of Prematurity.17,18 Our cohort included infants who met the Early Treatment of Retinopathy of Prematurity threshold criteria for treatment 19 and infants with aggressive ROP. Infants presenting with stage 4 or 5 ROP, as well as those who did not meet the Early Treatment of Retinopathy of Prematurity treatment threshold, were excluded from the study.
Infants meeting the Early Treatment of Retinopathy of Prematurity threshold guidelines 19 initiated their treatment with either IVT bevacizumab monotherapy (Group 1: 42 infants/64 eyes) or IVT bevacizumab with PRP (Group 2: 29 infants/58 eyes). In all cases in Group 2, PRP was performed concurrently with administration of IVT bevacizumab during the same session, mitigating the effects of inflammation after laser and the incidence of cystoid macular edema (CME) and exudation secondary to laser, irrespective of recurrence status.20–22
The choice of treatment modality was determined by the physician in conjunction with the infants’ legal guardians. Infants were not randomized to either treatment. The infants in Group 1 were seen weekly/biweekly until 55 weeks postmenstrual age and then had full eye examinations with cycloplegic refraction at 60 weeks postmenstrual age. All Group 1 infants had an examination under anesthesia (EUA) with Retcam and intravenous fluorescein angiography (FA) every 6 to 8 months starting at 65 weeks postmenstrual age. The frequency of 6-month follow-up visits is per institutional practice, based on previous literature. 10
Cost Analysis
The medical records of all infants treated for ROP were reviewed. Data collected included the number of inpatient or outpatient examinations, PRP sessions, IVT bevacizumab injections, EUA, and Retcam fundus photographs. The cost (physician fee and facility fee) for each service was determined based on Current Procedural Terminology (CPT) codes and 2024 national payment amounts, as specified by the US Centers for Medicare & Medicaid Services reimbursement allowables (https://www.cms.gov/medicare/physician-fee-schedule/search). Relative value units were also exported from the Centers for Medicare & Medicaid Services and were used to calculate the physician fee (relative value unit multiplied by 32.74) for each service. For both groups, the direct costs based on actual treatment received were calculated. We compared the cost billed for Group 1 with Group 2 with regards to different forms of bevacizumab (Avastin) available on the market (vial: $74.07, bottle: $849). The cost of bevacizumab injections (vial and bottle) was determined based on the Medicare Part B average sales price for fiscal year 2024 (Quarter 3) using Healthcare Common Procedure Coding System code J9035 ($74.07), sourced from https://www.cms.gov/medicare/payment/part-b-drugs/asp-pricing-files, and on pricing information from https://www.drugs.com/price-guide/avastin, respectively.
The costs of care in the neonatal intensive care unit, anesthesia, and indirect cost to parents from time away from work and transportation costs were not included in this study.
Statistical Analysis
For descriptive analyses, categorical variables were presented as count (%), and continuous variables were summarized using mean (95% CIs). All statistical analyses were performed using commercially available software (version 17, Stata, StataCorp LP). The alpha level (type I error) was set at 0.05.
Results
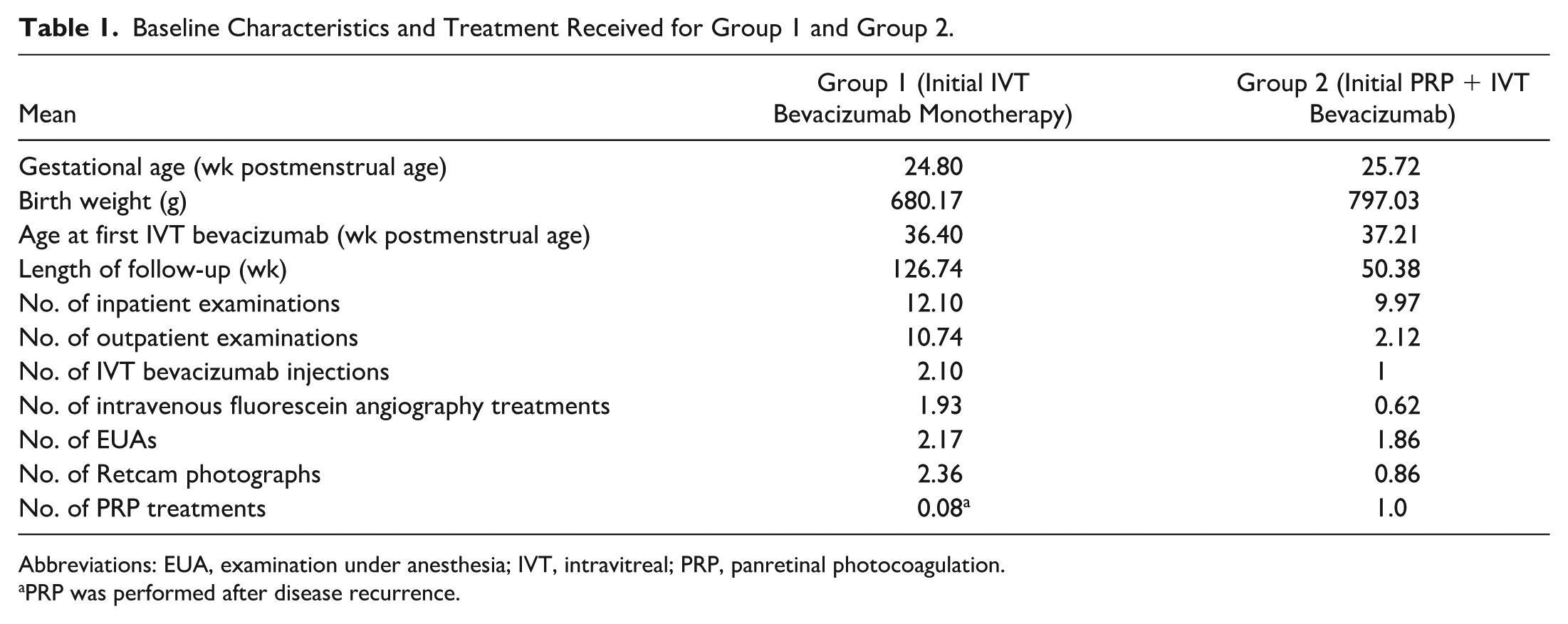
Group 1 included 42 infants (64 eyes) initially treated with IVT bevacizumab monotherapy, while Group 2 included 29 infants (58 eyes) initially treated with IVT bevacizumab combined with PRP. The demographics and average treatments received for both groups are described in Table 1.
Baseline Characteristics and Treatment Received for Group 1 and Group 2.
Abbreviations: EUA, examination under anesthesia; IVT, intravitreal; PRP, panretinal photocoagulation.
PRP was performed after disease recurrence.
The mean gestational age at birth and birth weight were similar between both groups. Infants in Groups 1 were initially treated with IVT bevacizumab at a mean postmenstrual age of 36.40 weeks (range, 31-46) and infants in Group 2 at 37.21 weeks (range, 32-46). The mean postmenstrual age of follow-up in Group 1 was 126.73 weeks (range, 51-246) and was 50.37 weeks (range, 41-69) in Group 2. Twelve eyes (18.75%) in Group 1 developed a recurrence of ROP at a mean postmenstrual age of 48 weeks. A total of 8 eyes (12.50%) in Group 1 were treated with repeat injections after recurrence. Three infants in Group 1 with a recurrence (6.25%; 4 eyes) received PRP after initial IVT bevacizumab. For these 4 eyes, the parents or legal guardians elected for PRP at the time of recurrence instead of repeat IVT bevacizumab monotherapy. Notably, 1 eye (1.56%) in an infant in Group 1 had an initial recurrence at 63 weeks postmenstrual age and was retreated with an injection; subsequently, a late recurrence developed at 91 weeks postmenstrual age that was successfully managed with barrier laser for localized peripheral fibrovascular proliferation, without further complications. 12 This infant was retained in Group 1 for analysis. None of the eyes in Group 2 experienced a recurrence. In all cases in Group 2, PRP was performed concurrently with administration of IVT bevacizumab. There were no repeat PRP procedures performed for any of the infants in either group. In Group 1, 52 eyes (81.25%) received a single IVT bevacizumab injection, while the remaining 12 eyes (18.75%) required 2 or more injections.
The mean number of inpatient and outpatient examinations for both groups, as well as the average number of IVT bevacizumab treatments and EUAs, are presented in Table 1. All infants in both groups were deemed to have good results at their last examination, with no infants requiring surgical treatment for stage 4 or 5 ROP.
The details of the cost (physician fee, facility fee, and total bill) for each bill component by Medicare are described in Table 2. When we first calculated the cost of treatment in Groups 1 and 2 using an Avastin vial, the total costs and proportion of costs per eye were $3231.03 (64.04%) for the facility fee and $1658.76 (32.88%) for the physician fee in Group 1 and in Group 2 were $2811.73 (64.64%) for the facility fee and $1464.12 (33.66%) for the physician fee. The total cost and proportion of cost per eye for the Avastin vial were $155.55 (3.08%) in Group 1 and $74.07 (1.70%) in Group 2. In addition, the total costs and proportion of costs per eye were $132.49 (2.63%) in Group 1 and $1656.13 (38.07%) for PRP in Group 2. When using an Avastin vial, the total cost per eye in Group 1, including the physician fee, facility fee, and injections, for the extrapolated follow-up was $5045.34, compared with $4349.92 in Group 2 when using Avastin vials (Table 3).
Costs of Each Treatment.
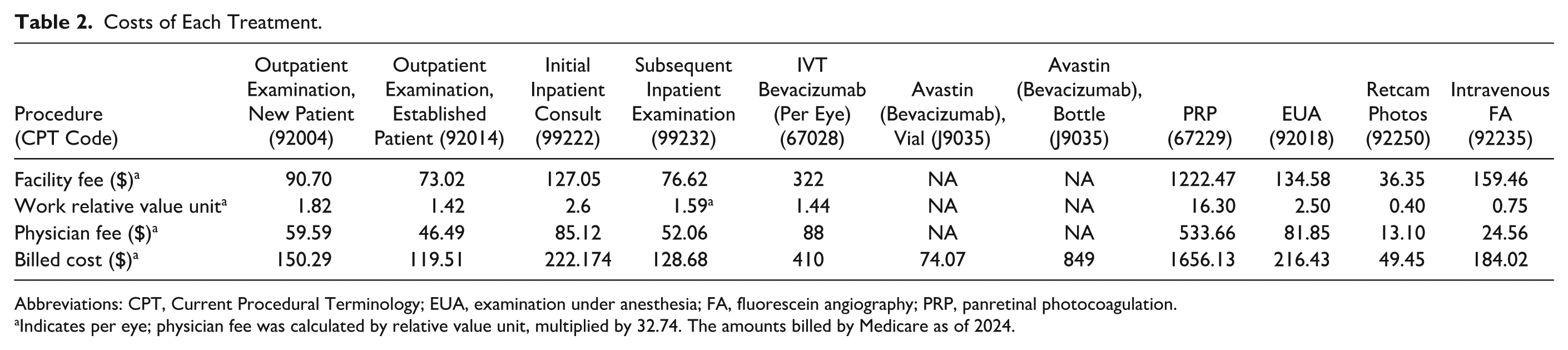
Abbreviations: CPT, Current Procedural Terminology; EUA, examination under anesthesia; FA, fluorescein angiography; PRP, panretinal photocoagulation.
aIndicates per eye; physician fee was calculated by relative value unit, multiplied by 32.74. The amounts billed by Medicare as of 2024.
Total Costs of Injection, PRP, Physician Fees, and Facility Fees for Group 1 and Group 2 With Regards to Different Types of Avastin.
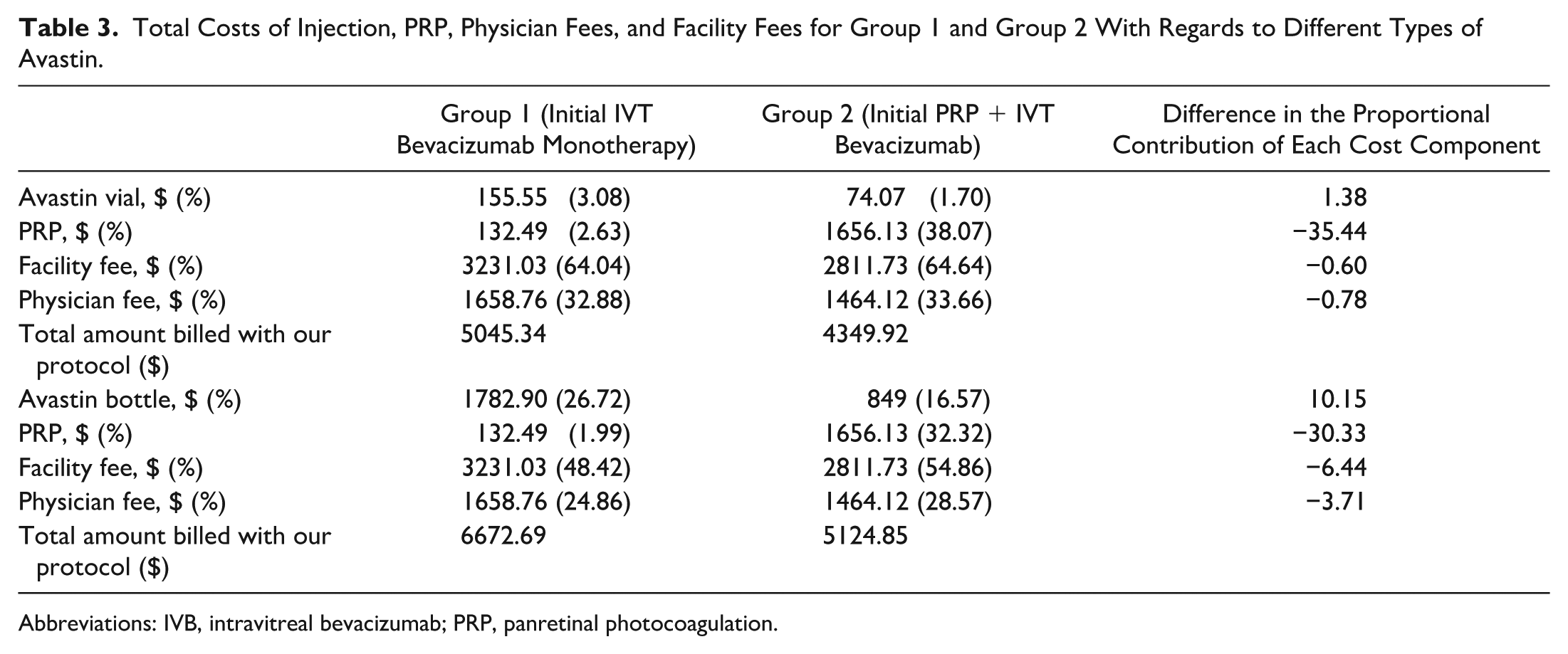
Abbreviations: IVB, intravitreal bevacizumab; PRP, panretinal photocoagulation.
We also calculated the cost of treatment for each group when using the Avastin bottle. In Group 1, the total costs and proportion of costs per eye were $3231.03 (48.42%) for the facility fee and $1658.76 (24.86%) for the physician fee, compared with $2811.73 (54.86%) for the facility fee and $1464.12 (28.57%) for the physician fee in Group 2. The total cost and proportion of cost per eye for the Avastin bottle were $1782.90 (26.72%) in Group 1 and $849 (16.57%) in Group 2. In addition, the total costs and proportion of costs per eye were $132.49 (1.99%) in Group 1 and $1656.13 (32.32%) for PRP in Group 2. When using the Avastin bottle, the total cost per eye in Group 1 for the extrapolated follow-up, including the physician fee, facility fee, and injections, was $6672.69, compared with $5124.85 in Group 2 (Table 3).
Supplementary Tables 1 and 2 report the cost of each service for facility fees, physician fees, and total costs in the model calculation when using the Avastin vial and bottle, respectively. When using the Avastin vial, the largest share of total treatment costs in Group 1 was from subsequent inpatient visits, amounting to $1428.31 (28.31%). In contrast, the largest share of costs in Group 2 was for PRP, which totaled $1656.13 (38.07%).
The largest share of physician fees in Group 1 was also for subsequent inpatient visits, at $577.83 (11.45%). In Group 2, the largest share of physician fees was split between subsequent inpatient visits at $466.95 (10.73%) and PRP at $533.66 (12.27%). For facility fees, the largest share in Group 1 was for subsequent inpatient visits, totaling $850.48 (16.86%). In Group 2, the largest share of facility fees was for PRP, amounting to $1122.47 (25.80%).
The largest share of total treatment costs in Group 1 was from use of the Avastin bottle, totaling $1782.90 (26.72%). In contrast, the largest share of costs in Group 2 was for PRP, which amounted to $1656.13 (32.32%). The largest share of physician fees in Group 1 was also for subsequent inpatient visits, at $577.83 (8.66%). In Group 2, the largest share of physician fees was split between subsequent inpatient visits at $466.95 (9.11%) and PRP at $533.66 (10.41%). For facility fees, the largest share in Group 1 was for subsequent inpatient visits, totaling $850.48 (12.75%). In Group 2, the largest share of facility fees was for PRP, amounting to $1122.47 (21.90%).
The difference in the proportional contribution of each cost component to the total cost per eye between the 2 treatment groups is shown in Table 3. The current cost-utility analysis found that when using Avastin vials, the costs for the treatment of Group 1 were 1.38% higher for the injection fee, 0.60% lower in the facility setting, 0.78% lower for the physician fee, and 35.44% lower for the laser fee compared with Group 2. The injection fee in Group 1 was the principal cost driver, whereas the 1-time laser fee was the principal cost driver in Group 2, leading to a lower injection fee and ultimately a lower total cost. Similarly, when using Avastin bottles, the costs for the treatment of Group 1 were 10.15% higher for the injection fee, 6.44% lower in the facility setting, 3.71% lower for the physician fee, and 30.33% lower for the laser fee compared with Group 2. In the current study, the injection fee in Group 1 remained the principal cost driver, while the 1-time laser fee in Group 2 led to a lower injection fee and ultimately a lower total cost.
Conclusions
With the emergence of bevacizumab monotherapy as a common treatment for ROP, it is important to directly assess the cost comparison between IVT bevacizumab monotherapy and combined IVT bevacizumab and PRP. In our study, based on a clinical cohort, there was an increased amount billed in Group 1 when compared with Group 2, despite the higher up-front cost of PRP over IVT bevacizumab.
The reason for the higher cost in Group 1 was largely due to the requirement for longer and more frequent follow-up. Group 1 had 8.9 more outpatient examination visits, and the total number of visits was almost double that of Group 2. It is reasonable to assume that there would be an associated indirect economic burden for parents who missed employment. Of note, there are alternative, higher cost anti-VEGF agents approved for the treatment of ROP.9,23,24 The follow-up period was significantly longer for Group 1, with patients followed for years longer than Group 2 because of late complications being reported until 3 years of age. EUA were required because of the difficulty of assessing the peripheral retina before 3 years of age.
One of the challenges in calculating the cost-effectiveness in the treatment of ROP stems from the fact that reimbursement rates vary greatly between insurers, as well as geographically. Additionally, there is significant variability in follow-up protocols after treatment. To our knowledge, there is only 1 other study directly comparing the costs of PRP with IVT bevacizumab monotherapy. The study took place in South Africa and found that, in their center, the cost of PRP was almost double that of IVT bevacizumab. However, the study took place in a private hospital, with follow-up consisting only of clinic visits. 25 Given the significant difference in these variables, it is not surprising that these 2 treatment modalities were much more similar in cost in the current study.
In terms of cost-effectiveness, both anti-VEGF and laser are acceptable. The economic savings of appropriate treatment of ROP are staggering. Rothschild et al 16 estimated that the direct cost of raising a blind child from birth to 18 years in the US in 2016 was $239 307. Loss of caregivers’ productivity was $2 329 653, and the loss of productivity for blind individuals was $249 913. The cost per neonate treated in the Rothschild study was $9821, which varied greatly from our study; however, both studies would deem treatment with either group’s approach to be cost-effective. Costs of treatment were obtained from hospital costs, not the billed amounts or amounts actually reimbursed by insurance.
A previous survey highlighted the difference in practice patterns among physicians who treat ROP, indicating the length of follow-up in infants treated with anti-VEGF monotherapy varied between 45 weeks to beyond 80 weeks postmenstrual age. However, 80% of survey respondents continued examinations past 55 weeks postmenstrual age, although the frequency of examinations varied even among these respondents. 26 In the absence of further data determining the optimal duration and frequency of follow-up in infants treated with anti-VEGF monotherapy, we advocate for prolonged follow-up beyond 1 year of age to ensure sequelae from undetected late ROP reactivation are not missed.10–12
An important aspect to consider is the stark difference in relative value unit assignments between IVT bevacizumab and PRP. The higher relative value unit for laser treatment, combined with fewer follow-ups, often incentivizes providers to opt for PRP rather than manage long-term care after IVT bevacizumab. Additionally, administering IVT bevacizumab in a premature infant in the neonatal intensive care unit is significantly more time-consuming and complex compared with administration to an adult in a clinic under topical anesthesia. It requires sedation, local anesthesia, and continuous monitoring, as the procedure can be stressful for the infant.
It is important to acknowledge that practice patterns have evolved since our study period (2014–2019). In many centers worldwide, PRP has become less common as a primary management strategy, with IVT bevacizumab often used first and, depending on the vascular response, PRP considered subsequently. Nevertheless, combined IVT bevacizumab and PRP continues to be used in selected cases to minimize the risk of late recurrences, and practice patterns remain heterogeneous globally. Thus, while our data reflect institutional practices during the study period, the cost analysis remains relevant for understanding the financial impact of different treatment approaches, particularly in centers where adjunctive PRP is still performed.
When comparing Avastin vials and bottles, we observed that the difference in total billed costs between the 2 treatment groups was markedly greater with bottles. Using vials, combined treatment was modestly less costly than IVT bevacizumab alone (≈$695.42 difference), whereas with bottles, the cost savings of combined treatment were much more pronounced (≈$1547.84 difference). This highlights that procurement method (vial vs bottle) can significantly influence the relative cost-effectiveness of treatment strategies for ROP, particularly when PRP is incorporated.
The selection of treatment modality was primarily determined by the treating physician in consultation with the infant’s legal guardians. Parental social factors played a secondary role in decisions regarding PRP. Neither birth weight nor gestational age alone dictated treatment; instead, choices were guided by disease severity and zone per Early Treatment of Retinopathy of Prematurity criteria, along with clinical judgment. Although a single physician administered all treatments, decisions were made collaboratively with parents, who generally followed the physician’s recommendation. In cases of incomplete vascular maturation after IVT bevacizumab, repeat injections were considered to minimize the risk of reactivation or complications such as CME or exudation.
There are limitations to this study, including the absence of an analysis of the costs of care in the neonatal intensive care unit, anesthesia costs, and the indirect costs to parents for transportation and missed employment. Additionally, the protocol of EUA with Retcam photography and intravenous FA every 6 to 8 months for infants in Group 1 may be more conservative than at other institutions. There were good outcomes with this approach, with no cases of stage 4 or 5 ROP and 100% sensitivity for reactivation. One may consider not including a PRP monotherapy group a limitation; however, in recent times, primary PRP is no longer the standard practice. Furthermore, reimbursements vary based on insurer and location, and thus our results may not be applicable to different geographic locations.
In conclusion, bevacizumab monotherapy was associated with a significantly higher cost and required more frequent visits and sedated examinations compared with the combination of bevacizumab and PRP to the avascular retina. Both modalities resulted in successful management of all infants in the study, with good outcomes. Further studies are needed to evaluate the indirect costs of treatment, as well as the difference in quality of life for patients treated with PRP and IVT bevacizumab monotherapy. These findings underscore the importance of optimizing treatment strategies for ROP to balance the financial implications and patient care demands. Future research and policy decisions could benefit from these cost analyses to refine ROP management guidelines, potentially reducing the burden on healthcare systems while ensuring effective outcomes for patients.
Supplemental Material
sj-docx-1-vrd-10.1177_24741264261433974 – Supplemental material for Cost Analysis of Anti-VEGF With Laser Photocoagulation in the Treatment of Retinopathy of Prematurity at an Academic Referral Center
Supplemental material, sj-docx-1-vrd-10.1177_24741264261433974 for Cost Analysis of Anti-VEGF With Laser Photocoagulation in the Treatment of Retinopathy of Prematurity at an Academic Referral Center by Nimesh A. Patel, Golnoush Mahmoudinezhad, Sandra Hoyek, Celine Chaaya, Yasmin Islam and Swati Agarwal-Sinha in Journal of VitreoRetinal Diseases
Supplemental Material
sj-docx-2-vrd-10.1177_24741264261433974 – Supplemental material for Cost Analysis of Anti-VEGF With Laser Photocoagulation in the Treatment of Retinopathy of Prematurity at an Academic Referral Center
Supplemental material, sj-docx-2-vrd-10.1177_24741264261433974 for Cost Analysis of Anti-VEGF With Laser Photocoagulation in the Treatment of Retinopathy of Prematurity at an Academic Referral Center by Nimesh A. Patel, Golnoush Mahmoudinezhad, Sandra Hoyek, Celine Chaaya, Yasmin Islam and Swati Agarwal-Sinha in Journal of VitreoRetinal Diseases
Footnotes
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of patient health information was performed in a Health Insurance Portability and Accountability Act-compliant manner.
Statement of Informed Consent
Not applicable.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Patel is a consultant for Alcon, Alimera, Allergan, Apellis, Atheneum, Biogen, DORC, Eye Point, Genentech, Gerson Lehrman Group, Inc, Guidepoint, Kyoto Drug Company, Lifesciences, Regeneron, and Regenx Bio. The other authors have no relevant financial disclosures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
