Abstract
Introduction
Familial exudative vitreoretinopathy (FEVR) is a rare inherited disorder of retinal vascular development, classically diagnosed in childhood.1–3 It is often associated with fundus findings such as incomplete peripheral vascularization, neovascularization, and retinal detachment (RD).1–3 Initial presentation in adulthood is uncommon and may reflect a missed diagnosis in childhood. 4 In adults with pathologic myopia, FEVR-related peripheral vascular abnormalities may be misattributed to myopic degeneration. It is important to distinguish between these conditions, as their management and prognosis differ significantly. Genetic testing can play a pivotal role in establishing the correct diagnosis. We report a case of autosomal dominant FEVR in an adult with high myopia, highlighting the diagnostic challenges posed by overlapping retinal pathologies.
Case Report
A man in his early 60s presented to our emergency department with acute vision loss in the left eye. His ocular history was significant for a prior rhegmatogenous retinal detachment (RRD) in the same eye, repaired with pars plana vitrectomy (PPV) in 2003, bilateral pseudophakia following cataract extraction, high myopia, and medically managed primary open-angle glaucoma. He also reported a family history of vision loss, although details were limited. Preoperative refractive data were unavailable for review.
On examination, best-corrected visual acuity was 20/150 in the right eye and hand motion in the left eye. A grade 3 relative afferent pupillary defect was present in the left eye. Intraocular pressure was 7 mm Hg in both eyes. Anterior segment examination of the left eye revealed endothelial and stromal corneal scarring nasally at the 3 and 7 o’clock positions, along with superior keratoneovascularization. The anterior vitreous was Shafer positive, raising concern for a retinal tear or detachment.
Dilated fundus examination of the left eye demonstrated a staphylomatous configuration with significant foveomacular atrophy, vessel straightening, and peripheral temporal atrophy (Figure 1). The nasal macula showed a large RRD (Figure 2). Examination of the right eye revealed macular atrophy, vessel straightening, peripheral lattice degeneration with associated pigmentary changes, and a sharply demarcated posterior staphyloma (Figure 3).
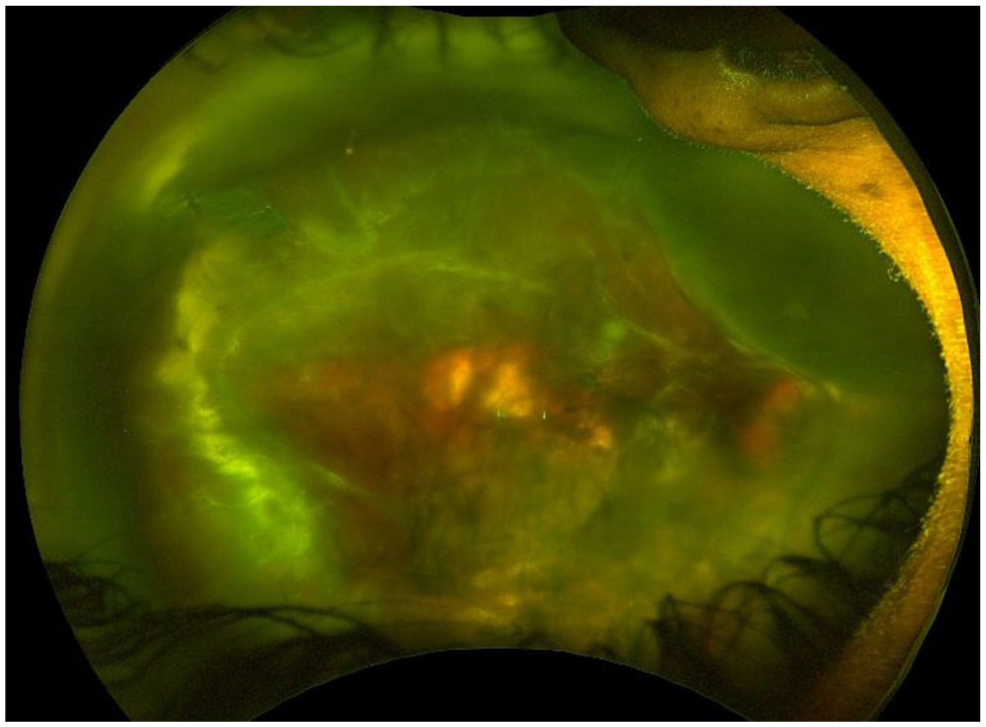
Color fundus photograph of the left eye demonstrates temporal chorioretinal atrophy, nasal intraretinal vs subretinal hemorrhage, and a tractional retinal detachment with overlying fibrotic membranes and proliferative vitreoretinopathy.

Optical coherence tomography image of the left eye demonstrates retinal detachment with prominent nasal atrophy.
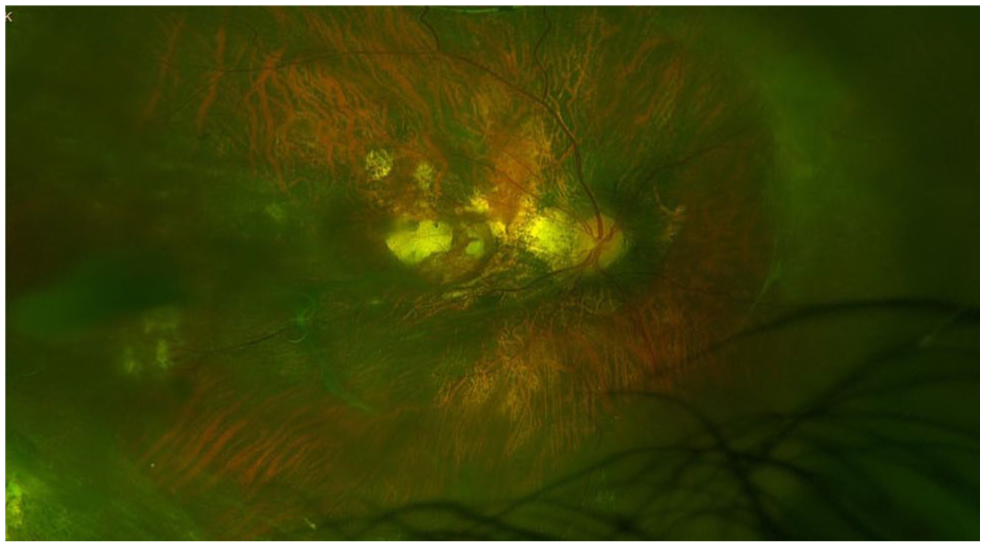
Color fundus photograph of the right eye demonstrates macular atrophy, posterior staphyloma, vessel straightening, and peripheral lattice degeneration with associated pigmentary changes.
The patient subsequently underwent scleral buckling combined with 25-gauge PPV, perfluorocarbon liquid instillation, extensive membrane dissection, endolaser retinopexy, placement of a 240 encircling scleral buckle, and silicone oil tamponade. Intraoperative adjunctive pharmacologic therapy included intravitreal bevacizumab and methotrexate.
Given the patient’s high myopia, vessel straightening, family history of vision loss, and abnormally posterior vitreous insertion, genetic testing was performed using the Invitae vitreoretinal dystrophy panel. A pathogenic heterozygous variant in FZD4 (c.1482G>A; p.Trp494*) was identified, consistent with autosomal dominant FEVR. At the most recent follow-up, vision in the patient’s affected eye was limited to counting fingers, with persistent posterior RD. He continued to receive intravitreal antivascular endothelial growth factor injections.
Conclusions
Inherited retinal disease may remain undiagnosed well into adulthood. While FEVR is classically diagnosed in childhood, this case illustrates how its features can be mistaken for more common conditions such as pathologic myopia.5–7 When missed in childhood, identifying FEVR in adults can be difficult, particularly when coexisting pathologies alter the fundus appearance. In our patient, however, fluorescein angiography (FA; Figure 4) revealed peripheral nonperfusion, supporting the diagnosis.
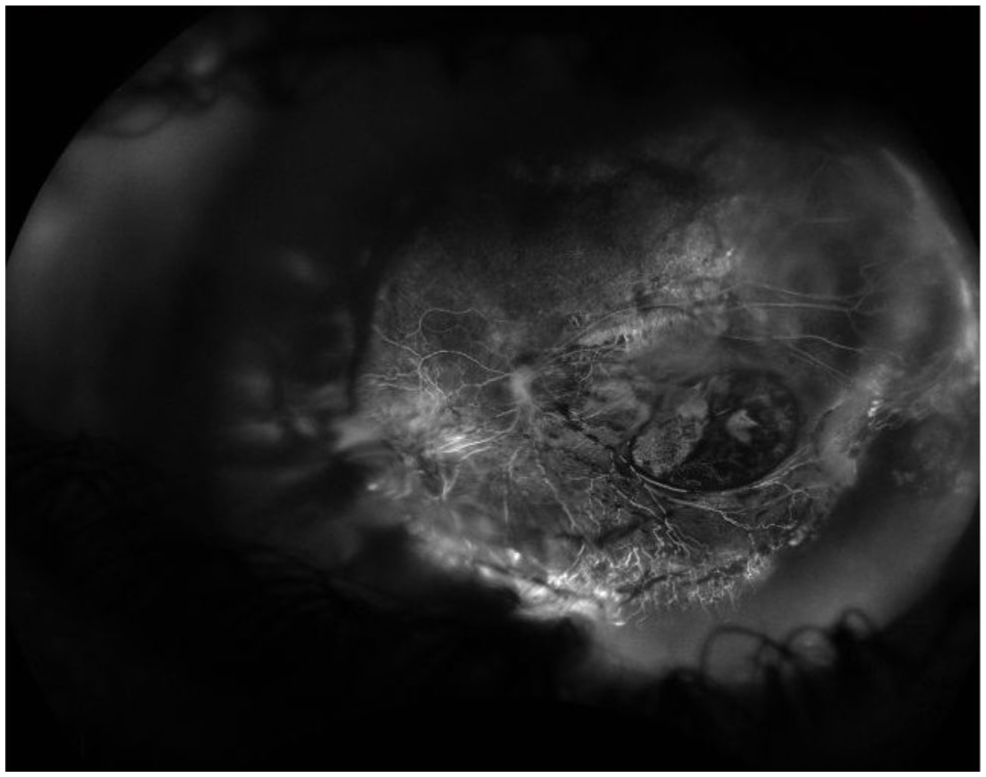
Widefield fluorescein angiography of the left eye demonstrates peripheral retinal nonperfusion, abrupt vascular termination, and abnormal vascular remodeling.
Late diagnosis of FEVR has been reported but remains uncommon, largely owing to the wide phenotypic variability linked to FZD4 mutations.4,8,9 Most published series focus on early-onset or familial pediatric cases, which often demonstrate bilateral, symmetric involvement.5,10 When one eye maintains good vision and peripheral vascular changes are subtle, the diagnosis may be missed until adulthood. This is particularly likely in patients with high myopia, in whom complications such as RD may be the first manifestation.
In this patient, the combination of high myopia, staphylomas, vessel straightening, and RD prompted further investigation for FEVR. While these findings are individually nonspecific, their combination has been found in adult presentations of FEVR and other retinal vasculopathies. This raises the possibility that such cases may be misclassified as myopia-associated RD.11,12
Genetic testing played a critical role in confirming the diagnosis. The identified mutation in FZD4 (p.Trp494*) has been previously reported in familial FEVR, although infrequently in adult cases.13,14 This diagnostic gap suggests that similar cases may fall outside the FEVR spectrum without genotyping. Given the relatively high carrier frequency of FZD4 variants and the known incomplete penetrance, it is plausible that adult patients with similar fundus findings remain undiagnosed or are misclassified as having myopic or idiopathic retinal pathology.15–17 Greater access to molecular genetics, along with an appropriate index of suspicion, may help clinicians interpret unusual retinal and retinal vascular findings in adults.
This case also raises important considerations regarding surgical management in similar patients. Obtaining an FA before surgery may help delineate the extent of pathology and identify areas of abnormal vitreoretinal traction. Areas of avascular retina may be particularly susceptible to iatrogenic breaks due to underlying atrophy. Despite limited success in this case, a growing body of evidence suggests that intravitreal methotrexate may modulate fibroproliferative responses in high-risk RD.18,19 Its use is better established in adult RRD.20–23 Ongoing trials, such as the GUARD study, may help clarify its utility. 24 Although methotrexate has been explored in pediatric or syndromic tractional diseases (eg, FEVR, retinopathy of prematurity), guidance regarding its use in adult FEVR remains limited.20,21 In this case, methotrexate was administered on a trial basis, and its benefit remains uncertain.
Ultimately, this case underscores the importance of maintaining a high index of suspicion for inherited retinal vascular diseases such as FEVR in unconventional RDs with abnormal vasculature. Genetic analysis is increasingly important for diagnosis, surgical decision-making, and prognostication in FEVR. As access to genetic testing expands, it will play an important role in refining diagnosis and expanding our understanding of the clinical spectrum of FEVR.
Footnotes
Acknowledgements
The authors thank Giselle De Oliveira, Senior Ophthalmic Photographer, for her contributions to clinical imaging.
Ethical Approval
The University of Miami Institutional Review Board waived ethical approval for this case report because the patient data were de-identified. This report adhered to the tenets of the Declaration of Helsinki.
Statement of Informed Consent
Informed consent was not obtained because no patient identifiers were included.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Berrocal is a consultant for Alcon, Allergan, Dutch Ophthalmic Research Center, Novartis, Oculus, ProQR, and Zeiss. None of the other authors declared potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Bascom Palmer Eye Institute is supported by National Institutes of Health (core grant no.: P30EY014801) and Research to Prevent Blindness (GR004596 unrestricted award).
Data Availability
No datasets were generated or analyzed during the current study.
