Abstract
Introduction
Uveitis is one of the smallest specialties in medicine. A recent geodemographic study identified only 223 specialists nationwide, with 15 states lacking any uveitis specialist and 8 states having only 1 provider. Most specialists are concentrated in urban tertiary care centers, leaving 36.7% of the United States population more than 1 hour from the nearest provider. 1 On average, uveitis patients require longer office visits than comprehensive ophthalmology patients, 2 and they demonstrate improved functional outcomes when treated by subspecialists. 3
Despite the clear need for uveitis specialists, 60.4% report limited support staff, and 56.3% report inadequate reimbursement for the time and effort involved in care as barriers to practice. 4 The combination of complex patients, a limited workforce, declining ophthalmology reimbursement, 5 and potential workforce shortage 6 may disincentivize uveitis care, hinder recruitment, and worsen existing barriers to access. Therefore, we sought to measure the total cost of uveitis care and assess how well reimbursement reflects the demands of complex care provision.
Time-driven activity-based costing is a well-described, bottom-up micro-costing approach, 7 with emerging applications in ophthalmology.8–11 Time-driven activity-based costing assesses the capacity of individual resources, allocates costs using time as the common denominator, and helps identify relevant cost drivers. It is well suited to capture the full cost of care. Given the time-intensive nature of uveitis visits—which often require medical record review before the visit, complex lab workup, and interdisciplinary medication management afterward—we hypothesized that the true total cost of new patient uveitis care is largely nonremunerated.
Methods
Time-driven activity-based costing analysis was performed for consecutive new uveitis patient visits meeting inclusion criteria, seen over a 3-week period by 2 uveitis specialists (SG, AST) in a university-based practice (Vanderbilt Eye Institute) and a private practice (Tennessee Retina). Process flow maps were created for new uveitis patients at both sites. De-identified electronic health records (EHRs), internal financial statements, and manual duration recordings were used to build the time-driven activity-based costing model as previously described.7,9 This study was approved by the Vanderbilt Institutional Review Board and complied with the tenets of the Declaration of Helsinki.
Process Flow Map
A process flow map was created from direct observation of consecutive new uveitis patient visits with Common Procedural Terminology (CPT) codes 99204 and 99205 (Figure 1). To capture the full cost of care, processes were categorized as pre-visit, in-visit, and post-visit. Imaging-related costs, waiting time, and expenses were excluded because they are billed and reimbursed separately. Time-stamped data were extracted from the EHR (Epic Systems) after staff and providers were instructed to log each step in the process using Epic Systems event stamps. All processes were also measured in a cohort of 44 new uveitis patients using manual time recording. Stages were defined by use of clinical space and personnel (Figure 1).
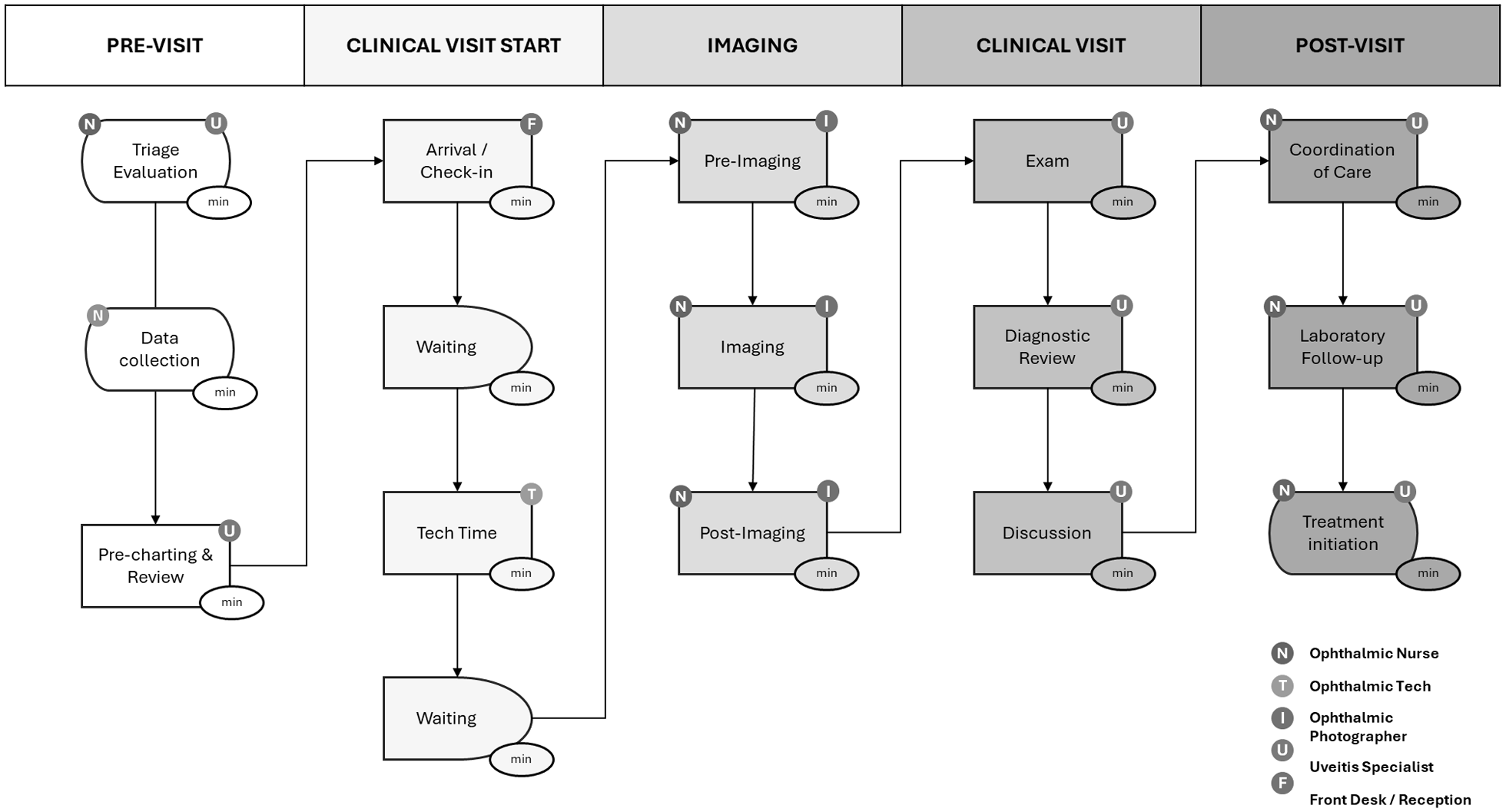
Process flow diagram of new uveitis patient visits.
Duration data for steps that could not be adequately measured were excluded, and physician data for patients seen by trainees were removed to avoid bias from teaching-related time. Pre-visit was defined as up to 24 hours before the clinic visit and included physician or nursing time spent collecting and reviewing outside records and lab work, as well as discussions with the patient or referring physician when appropriate. In-visit time included patient arrival and check-in, waiting, technician workup, specialist evaluation, and check-out. Imaging was excluded. Post-visit included physician time required to coordinate or follow up on diagnostic studies, initiate treatment, and communicate with the patient and referring physician.
Financial Data
Financial data were obtained from the Vanderbilt Eye Institute and Tennessee Retina offices, including supply costs, office space expenses (new construction per square footage and maintenance), and de-identified staff salaries. To standardize physician costs across practice types, the 2021 Medical Group Management Association median salary for ophthalmology was used to calculate the capacity cost rate. Costs were then compared with Centers for Medicare and Medicaid Services reimbursement for CPT codes 99204 and 99205, based on the 2024B physician fee schedule (conversion factor $33.2875).
CPT codes 99205 and 99204 represent moderate- and high-complexity new patient visits, respectively. The appropriate code is determined by the number and complexity of medical diagnoses addressed, the amount and complexity of data reviewed, and the risk of complications or morbidity/mortality associated with management options. Complex medical diagnoses typically require longer visits, more extensive decision-making, and higher-risk treatment plans. 12 Margins were calculated using the non-facility limiting charge, which represents the maximum amount a nonparticipating provider can bill. Private third-party insurer rates were not included in this analysis. Imaging procedures—and their associated costs and wait times—were excluded, as they are separately reimbursed.
Labor, Space, Equipment, and Material Costs
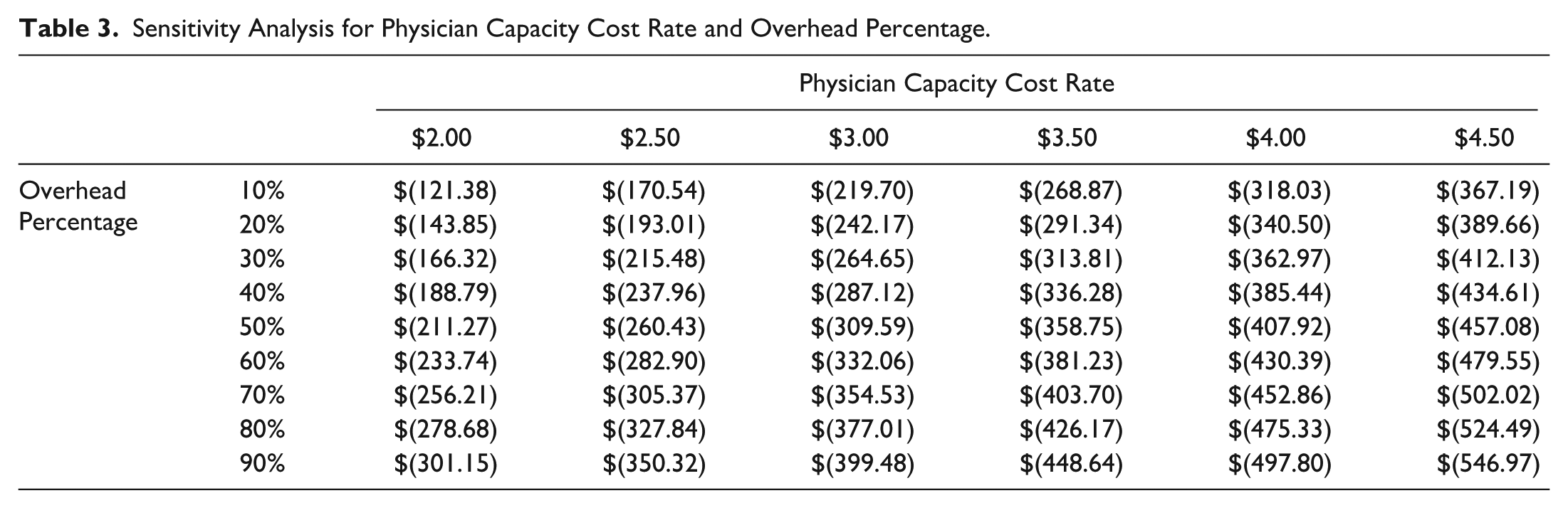
Costs were calculated as previously described, 8 including a conservative overhead rate of 25.27% for base-case assumptions. Capacity for space, equipment, and personnel was defined from standard clinical schedules using departmental data. Base-case physician compensation was sourced from the 2021 Medical Group Management Association national average salary for an ophthalmologist in the South, 13 to account for regional salary variation; this assumption was tested in sensitivity analysis. Using these inputs, the capacity cost rate (cost per minute) was calculated from the total costs and available capacity in minutes for relevant personnel, equipment, and clinical space. Sensitivity analyses were performed for both reimbursement rates and physician time. Statistical testing was conducted using Microsoft Excel and RStudio.
Results
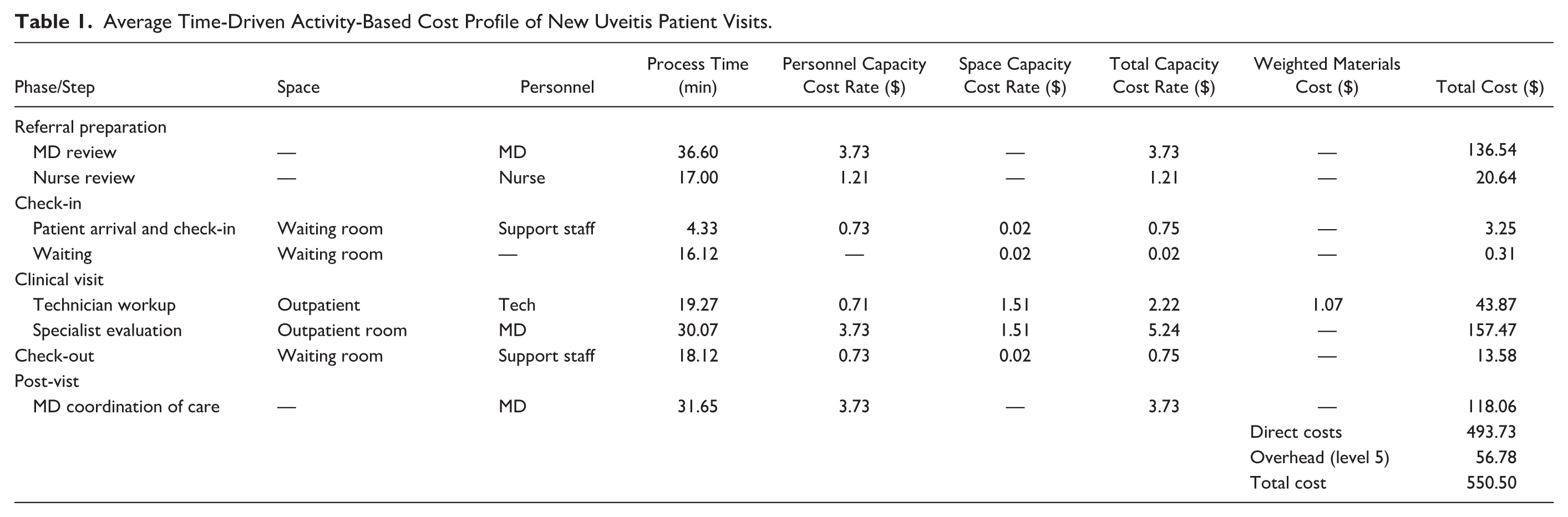
Across both institutions, time-driven activity-based costing analysis was performed for 44 new uveitis patient encounters (24% anterior, 24% intermediate, and 52% posterior or panuveitis). On average, visits required 36.60 ± 38.65 minutes (95% CI, 24.7 to 48.50) of physician time pre-visit, 30.07 ± 13.33 minutes (95% CI, 25.34 to 34.79) during the visit, and 31.70 ± 16.50 minutes (95% CI, 25.87 to 40.13) post-visit. The time-driven activity-based costing profile averaged $157.18 (28.6%) pre-visit, $218.49 (39.7%) during-visit, $118.06 (21.4%) post-visit, with an additional $56.78 (10.0%) attributed to overhead, resulting in a total average cost of $550.50 (95% CI, $493.74 to $607.27) (Table 1). In comparison, the maximum Medicare reimbursement (non-facility limiting charge) was $224.72 for CPT code 99205 and $170.34 for CPT code 99204, resulting in negative margins of $325.75 and $380.16, respectively.
Average Time-Driven Activity-Based Cost Profile of New Uveitis Patient Visits.
The average total cost profile varied by uveitis subtype: anterior ($371.11; 95% CI, $240.68 to $501.53), intermediate ($571.00; 95% CI, $403.36 to $738.65), and posterior or panuveitis ($351.29 to $450.98). There was a trend toward increased time and cost for intermediate and posterior/panuveitis encounters relative to anterior uveitis, primarily driven by physician time (Figure 2). However, the Kruskal-Wallis test comparing total physician time across uveitis subtypes (anterior, intermediate, panuveitis/posterior uveitis) was not significant (P = .12). Dunn’s test indicated longer physician time for intermediate and posterior/panuveitis relative to anterior uveitis, but these comparisons did not reach significance after Bonferroni correction.
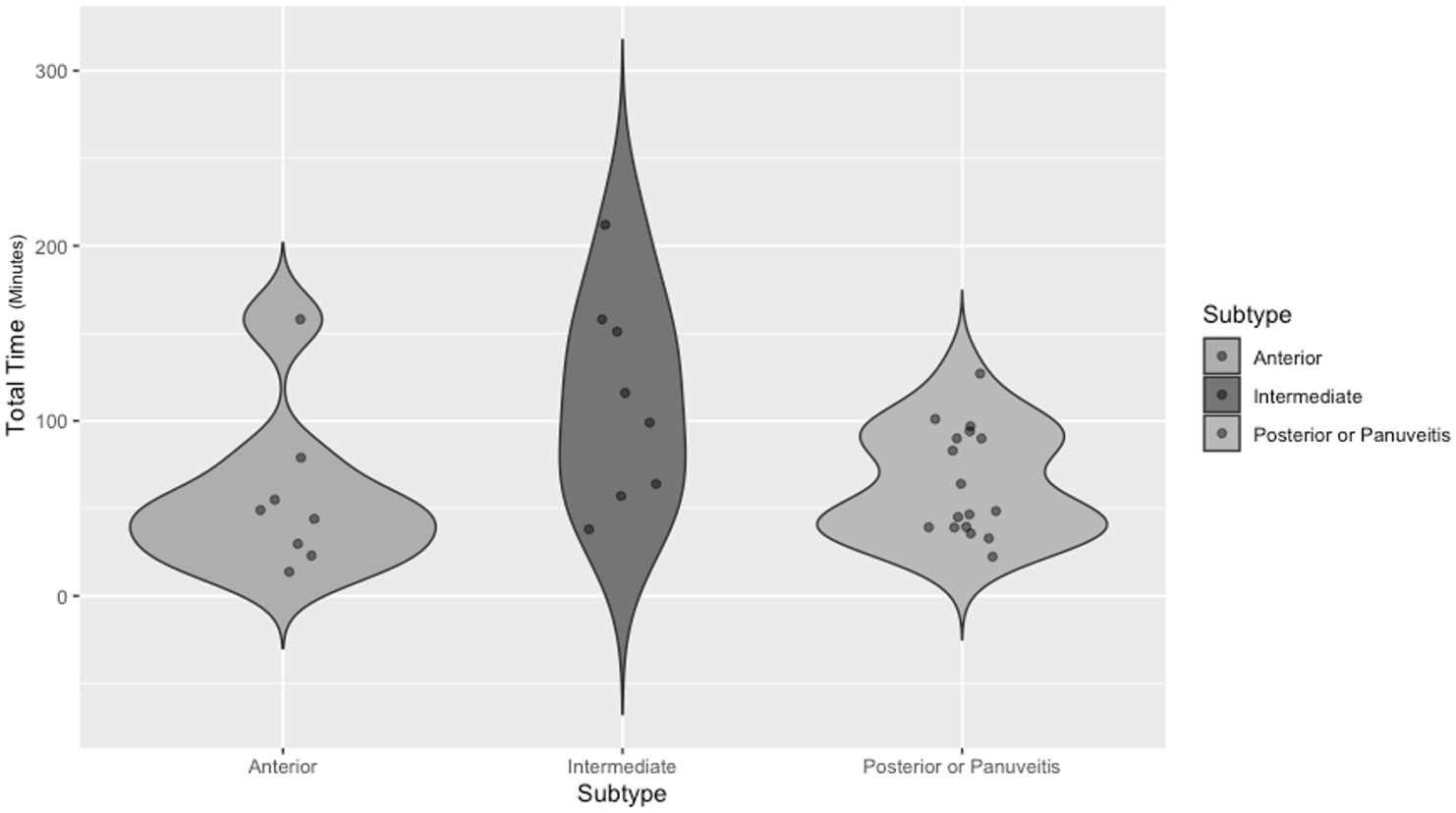
Total visit time profile in minutes by uveitis subtype.
When excluding pre- and post-visit time, cost, and overhead, the average during-visit cost was $218.49. Compared with the maximum Medicare reimbursement (non-facility limiting charge) of $224.72 for CPT code 99205 and $170.34 for CPT code 99204, this resulted in a positive contribution margin of $6.23 for CPT code 99205 and a negative margin of $48.15 for CPT code 99204. Total episode-of-care margins varied widely across encounters; however, all but one resulted in negative margins (Figure 3).
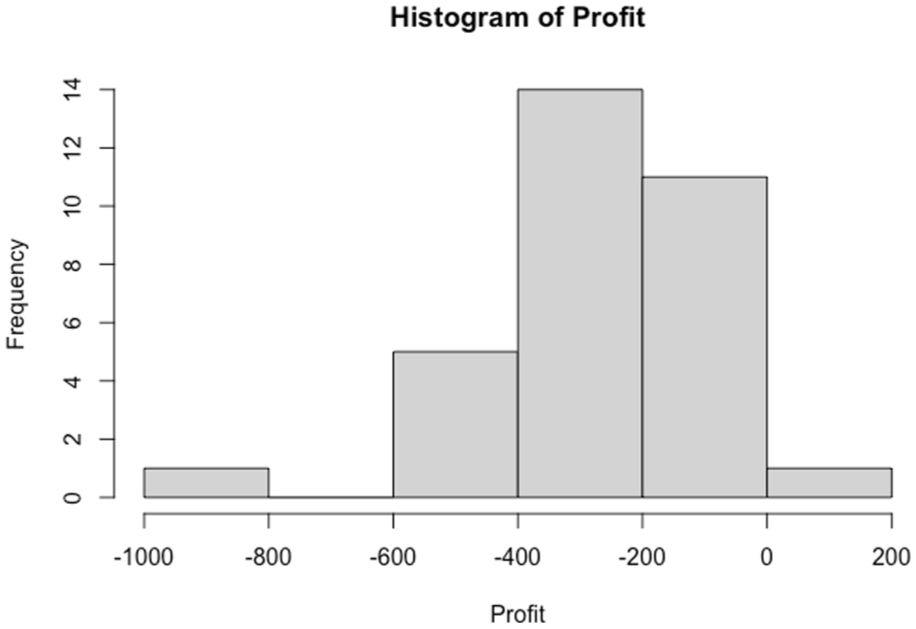
Histogram of new uveitis patient visit margin in United States dollars.
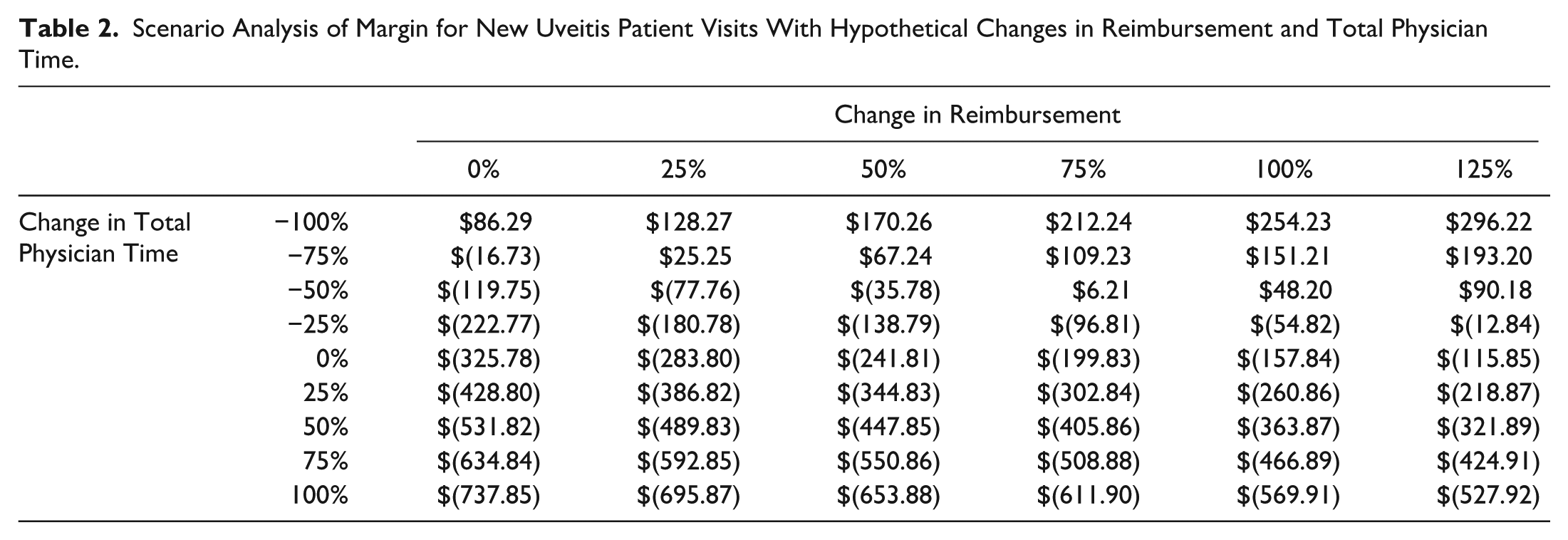
The G2211 add-on CPT code, which describes longitudinal care for a complex condition, has a maximum Medicare reimbursement (non-facility limiting charge) of $16.96. This amount compensates for only 3.23 minutes of in-visit physician time and had a minimal impact on the average time-driven activity-based costing margin. Scenario analysis demonstrated that costs would not be fully remunerated unless physician times were reduced by 79% or reimbursement increased by 194% (Tables 2 and 3).
Scenario Analysis of Margin for New Uveitis Patient Visits With Hypothetical Changes in Reimbursement and Total Physician Time.
Sensitivity Analysis for Physician Capacity Cost Rate and Overhead Percentage.
Conclusions
Time-driven activity-based costing analysis of new uveitis patients demonstrated average losses of $325.78 for complex level 5 (CPT code 99205) visits and $380.16 for level 4 (CPT code 99204) visits relative to maximum Medicare reimbursement. Although not statistically significant, there was a clear trend toward increased time and cost among intermediate and posterior/panuveitis patients, indicating that these complex new visits are financially disincentivized. These findings align with known trends in Medicare reimbursement and highlight the economic pressures facing uveitis subspecialists, with potential implications for access to care and long-term care provision.
The present analysis demonstrates that the resources, physician time, and subsequent costs of new uveitis patients exceed current Medicare reimbursement for complex new patient visits. Uveitis specialists typically manage complex, chronic cases that have failed prior treatments, often in association with infectious or systemic autoimmune diseases with high morbidity risks. Uveitis specialists rely on extensive diagnostic evaluation, including a review of prior records and systemic treatment options. Although a small positive margin exists when considering only same-day, direct, during-visit physician costs, the time-driven activity-based costing profile shows that most of the time required for a new patient visit is split between pre-visit activities (communication, triage, and diagnostic and outside record review) and post-visit care coordination (diagnostic follow-up, treatment initiation, and collaboration with other providers).
Patients with uveitis frequently experience ocular medical and surgical sequelae of inflammation, including cataracts, glaucoma, 14 and systemic disease, requiring coordination with both ophthalmologic subspecialties and general medical providers. Although these activities are not explicitly included in current reimbursement mechanisms, the pre- and post-visit time and resources invested are what make the during-visit time more efficient. Notably, the average positive contribution margin of $6.23 for during-visit time would turn negative if the uveitis specialist spent more than 1.67 additional minutes with the patient. These narrow margins disincentivize comprehensive patient care for complex cases. Scenario analyses further highlight the reimbursement disparity, indicating that reimbursement would need to increase by 194% to achieve a non-negative margin. The G2211 add-on CPT code provides only $16.96—equivalent to 3.23 minutes of during-visit physician time—suggesting its insufficiency to account for the additional time and resources needed for complex cases.
We believe these findings highlight the need for policy changes, including a new CPT code that properly accounts for the time and complexity of new uveitis patients. Current declines in reimbursement—without inflationary adjustment to the physician fee schedule—further disincentivize clinicians from treating the most complex cases requiring significant time and resources. Racial and gender disparities in healthcare utilization among Medicare beneficiaries with inflammatory and infectious eye diseases 15 may be exacerbated by financial disincentives. Uveitis patients managed by subspecialists experience improved functional outcomes. 3
Provision of patient care at a financial loss is systemically problematic. Low reimbursement can hinder recruitment into the field and limit the feasibility of uveitis-only practice to tertiary academic centers, where losses may be offset through recoupment or cross-subsidization. Comprehensive ophthalmologists and other subspecialists likely manage more routine cases of ocular inflammation, selectively referring the most complex cases for subspecialist evaluation and management. 16 This referral pattern ensures complex disease is managed by providers with advanced training and expertise in efficiently managing uveitis.
Future study comparing clinic visit time for uveitis-trained specialists vs non-uveitis-trained specialists could demonstrate that uveitis specialists save patients, other ophthalmologists, and the broader medical system significant time and opportunity cost. However, if the most complex patients are seen at a larger financial loss, uveitis specialists may be incentivized to shift toward more routine cases to sustain their practices. These economic pressures may contribute to the clustering of uveitis specialists in metropolitan geographies 1 and the lack of specialists in underserved areas.
Established patient visits may be more efficient after the initial visit and workup, warranting further study. Although imaging procedures are separately reimbursed, resource-intensive imaging such as fluorescein angiography can also be financially disincentivized. 9 Uveitis specialists frequently pursue multimodal imaging during the initial workup, 17 yet reimbursement is often partial owing to payor bundling requirements. The impact of bundling on uveitis and other ophthalmology practices should be further examined.
Strengths of this study include data from 2 practice settings within the same geographic region with a similar payor mix. Although diverse settings were included, cost profiles may vary nationally by geography and practice structure and should be calculated individually. Limitations include the potential for disproportionately high-complexity patients during the study window despite consecutive recruitment, and the possibility that the workflows of 2 uveitis specialists are not representative of national averages.
Manual recording of pre- and post-visit time limited the sample size and the ability to find statistically significant differences in subgroup analyses. Overhead, inputs, and referral patterns may also vary substantially across practices. Use of national estimates and a unified process flow for visit costs minimized some differences between academic and private practice settings, but cost comparison across practice types remain limited. Despite these limitations, the economic pressures demonstrated here emphasize the need to critically evaluate Medicare reimbursement and support the necessity of advocacy efforts to ensure appropriate access to care for disadvantaged, complex patient populations.
The present analysis demonstrates that the total cost profile for new uveitis patients exceeds maximum Medicare reimbursement in both academic and private practice settings. Costs are driven by the time and expertise needed to evaluate patients before and during clinic visits. Intermediate and posterior/panuveitis cases—those carrying the highest risk of permanent visual impairment and blindness—are also the most time-consuming and costly encounters. These findings may inform policy discussions on reimbursement and applicable modifiers that better capture the time and expertise required for complex uveitis evaluation and management.
Footnotes
Ethical Approval
This study was approved by the Vanderbilt Institutional Review Board and complied with the tenets of the Declaration of Helsinki.
Statement of Informed Consent
Informed consent was not required as no patient-specific data were collected.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Finn has served on advisory boards for AbbVie, Alimera, Apellis, and Iveric Bio, and as a consultant for Genentech. Dr. Thomas has served as a consultant for Alimera/ANI, Allergan, Avesis, Bausch + Lomb, Eyepoint, Genentech, and Zeiss.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by an unrestricted grant from Research to Prevent Blindness to the Vanderbilt Eye Institute. The sponsor or funding organization had no role in the design or conduct of this research.
