Abstract
Introduction
Epiretinal membranes (ERMs) are characterized as fibrocellular proliferation at the vitreoretinal interface above the internal limiting membrane (ILM). The prevalence is reported in the literature as 7% to 11.8%, with increasing age being the most important risk factor.1–3
ERMs can be classified as idiopathic or secondary based on the initiating event.4,5 Idiopathic ERMs develop as the vitreous ages and retinal adhesions weaken, predisposing to posterior vitreous detachment (PVD) and subsequent fibroglial proliferation. 4
Secondary ERMs have been linked to various ocular pathologies, such as uveitis, central or branch retinal vein occlusion, and retinal detachment (RD). 5 They are also associated with PVD, cataract surgery, ocular trauma, and retinal surgery.1,6–9 Preoperative risk factors for ERMs include proliferative vitreoretinopathy, choroidal detachment, and laser photocoagulation. 10
An understanding of concomitant ocular conditions is paramount to the initial diagnosis and the prognostication of long-term vision preservation after treatment of choroidal melanoma. Although tumor growth, the effect on vision, and the potential for metastasis have been well documented, the association of choroidal melanoma with secondary ERM has not. Using a PubMed search with the terms “melanoma”, “epiretinal membrane”, “choroid”, “vision”, and “traction”, we could find no publications focused on choroidal melanoma–associated ERMs. However, we found predisposing factors that may link choroidal melanoma to ERMs, including pretreatment and posttreatment cystoid macular edema, ocular inflammation (eg, necrotic melanoma, toxic tumor syndrome), acute glaucoma, and vitreous hemorrhage.6,11–13
In this study, we investigated the association between ERMs and choroidal melanoma. Furthermore, we examined the effects of the radiation dose, intravitreal (IVT) injections of antivascular endothelial growth factor (anti-VEGF), radiation retinopathy, and RD as risk factors for ERM development.
Methods
A retrospective review of charts and imaging data for 427 patients treated with palladium-103 plaque brachytherapy for choroidal melanoma was performed.14,15 The choroidal melanomas were staged according to the 8th edition of the American Joint Committee on Cancer (AJCC) criteria. 16 All patients provided written informed consent before the study.
No case selection was performed. However, participants were excluded from analysis if their scans were low in quality as a result of poor layer discrimination, severe motion artifacts, or ocular conditions that prevent adequate visualization of the posterior segment, such as dense cataracts, hemorrhage, and severe corneal opacification (n = 43). In addition, patients who had pars plana vitrectomy for ERM removal were excluded from analysis to ensure the integrity of the data for the untreated progression of ERM and the visual acuity (VA) outcomes (n = 11). The VA was recorded every 12 months over 60 months.
Clinical Ophthalmoscopic Characteristics of ERM
ERMs were identified during ophthalmoscopy (indirect, contact lens, photographic), fundus photography, or both. Clinically, an ERM was characterized as an opaque membrane with superficial retinal folds or traction lines with or without dysmorphic macular retinal vessels and a foveal reflex.
Optical Coherence Tomography Criteria of ERM
The ERM diagnosis was confirmed and further characterized using optical coherence tomography (OCT). OCT imaging was performed using a spectral-domain OCT device (Spectralis, Heidelberg Engineering). The ERM appeared as a highly reflective, typically white membrane overlying the macular retina. Key OCT parameters included the central foveal thickness and inner segment ellipsoid band integrity. The inner segment ellipsoid band appeared clear and consistent, while a disrupted band appeared blurred, interrupted, or absent. Associated OCT findings included evidence of vitreous traction, macular thickening, or cystoid intraretinal edema.
In addition to the initial comprehensive examination, examinations for ERM occurred 3 months, 6 months, 12 months, 24 months, 36 months, 48 months, and 60 months after ophthalmic plaque brachytherapy. New cases of ERM were defined by the absence of ERM at baseline and its presence at a follow-up visit.
Statistical Analysis
SPSS software (version 28, IBM) was used to analyze the data. Continuous variables were expressed with the mean ± SD. Categorical variables were expressed as percentages and compared using χ2 tests.
Logistic regression models were used for multivariate analysis of potential risk factors, such as radiation retinopathy, anti-VEGF treatment, and RD at the initial consultation. Each factor was adjusted for age and sex. The logistic regression model calculated odds ratios (ORs) with 95% CIs to identify significant associations between risk factors and the development of ERMs.
The Cox proportional hazards model was used to evaluate the time-to-event data for the development of ERMs over a follow-up of up to 60 months. The dependent variable was the time to ERM development, and the independent variables included the radiation dose to the fovea, presence of radiation retinopathy, use of anti-VEGF injections, and presence of RD at the initial diagnosis. Anti-VEGF was used to mitigate complications associated with radiation retinopathy, including macular edema and neovascularization, thereby preserving vision. 17 Age and sex were also adjusted as potential confounders. The model estimated hazard ratios (HRs) with 95% CIs to quantify the relative risk for ERM development associated with each factor. This analysis allowed a determination of the risk for ERM development in patients treated for choroidal melanoma compared with the control group.
Changes in VA were evaluated with paired-sample t tests to assess the statistical significance of differences between baseline measurements and follow-up measurements. Statistical significance was set at P < .05.
Results
This study included 373 eligible patients and 746 eyes; the contralateral eye was used as the control. The mean patient age was 67 ± 11 years (range, 21-87); 205 patients (55%) were biologically female, and 168 (45%) were biologically male. The mean follow-up was 52 ± 6 months (range,12-60). Staged according to the 8th edition AJCC criteria, 197 choroidal melanomas (53%) were cT1, 146 (39%) were cT2, 26 (7%) were cT3, and 4 (1%) were cT4. Most tumors were posterior and temporally located (Table 1).
Tumor Location, Radiation Dose, and Plaque Distance to Fovea.

As is common for choroidal melanomas, most tumors were posterior and temporally located (Nagendran ST, Finger PT, Campolattaro BN. Extraocular muscle repositioning and diplopia: associated with ophthalmic plaque radiation therapy for choroidal melanoma. Ophthalmology. 2014;121(11):2268-74. doi:10.1016/j.ophtha.2014.05.037).
Choroidal Melanoma and ERM at Presentation
At presentation, ERMs were significantly more likely to present in eyes with choroidal melanoma than in control eyes (127 eyes [34%] vs 45 eyes [12.1%]) (P = .001, χ2 test ). The ERM was bilateral in 23 patients (6%).
Eighty-nine (45%) of 197 eyes with AJCC cT1 tumors had an ERM at baseline. Compared with contralateral control eyes, the OR of the baseline ERM with AJCC cT1 tumors was 1.17 (P = .093; 95% CI, 0.82-1.70). AJCC cT2 tumors with an ERM at baseline were present in 88 (58%) of 146 eyes. Compared with control eyes, the OR was 2.38 (P = .048; 95% CI, 1.01-5.64). Nineteen (73%) of 26 eyes with AJCC cT3 tumors had an ERM at baseline. Compared with control eyes, the OR of the ERM was 2.9 (P = .016; 95% CI, 1.22-6.82). Three (75%) of 4 eyes with AJCC cT4 tumors had an ERM at baseline. The OR was 4.8 (P = 0.154; 95% CI 0.56-40.98). Thus, increasing tumor size was significantly associated with the presence of ERM at baseline (P < .014 for trend).
Development of ERM After Plaque Radiation
The mean onset of the appearance of new ERMs in eyes with choroidal melanoma was 16 months after plaque radiation. In contralateral control eyes, the mean time to the appearance of new ERMs was 41 months. During the 5-year follow-up, 99 additional cases of ERM were identified in eyes with choroidal melanoma and in 44 control eyes. Cox proportional hazards model analysis showed a higher risk for ERM development in eyes with choroidal melanoma than in control eyes during the 60-month study period (HR, 2.669; 95% CI, 1.863-3.909; P < .001). Therefore, eyes with choroidal melanoma were significantly associated with the development of ERM and an earlier onset of ERM progression. (P < .001).
Radiation Dose and ERM Development
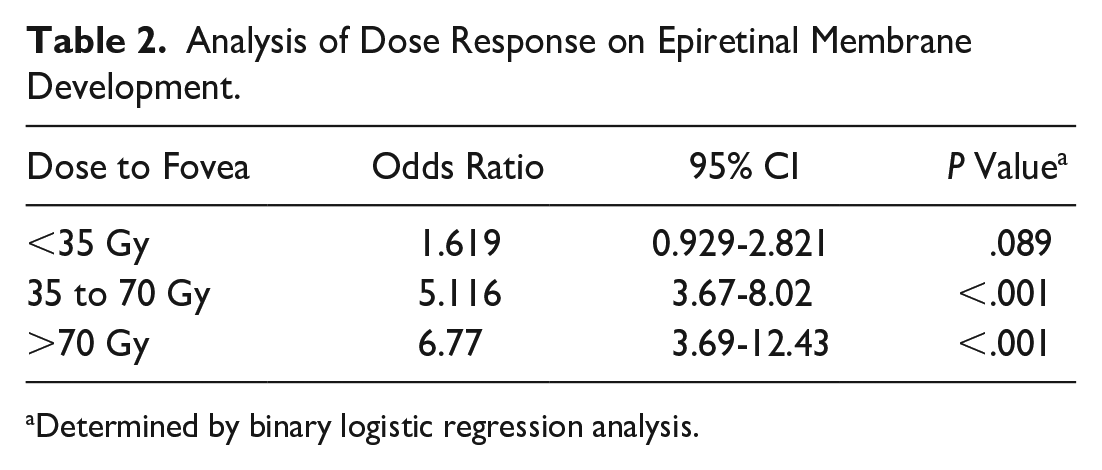
Table 1 shows the radiation dose to the fovea as well as the plaque distance to the fovea. The mean radiation dose to the fovea was 46 Gy, and the mean plaque distance to the fovea was 6.5 mm. A significant dose-response relationship was found between the dose to the fovea and the development of an ERM.
Table 2 shows an analysis of the effect of the dose response on ERM development. During the follow-up, 114 patients (31%) were treated with radiation doses from 0 to 35 Gy. Of these patients, 92 (81%) did not develop an ERM, although 22 (19%) later developed an ERM. The difference between study eyes and control eyes was not statistically significant (P = .089).
Analysis of Dose Response on Epiretinal Membrane Development.
Determined by binary logistic regression analysis.
Of the patients, 204 (55%) were treated with radiation doses from 35 to 70 Gy. Of these, 101 (49.5%) did not develop an ERM, although 103 (50.5%) later developed an ERM. The difference between study eyes and control eyes was statistically significant (P < .001).
Fifty-five patients (15%) were treated with radiation doses of 70 Gy or more. Of these, 21 (37%) did not develop an ERM, although 34 (63%) later developed an ERM. The difference between study eyes and control eyes was statistically significant (P < .001).
Association Between Radiation Retinopathy and ERM Development
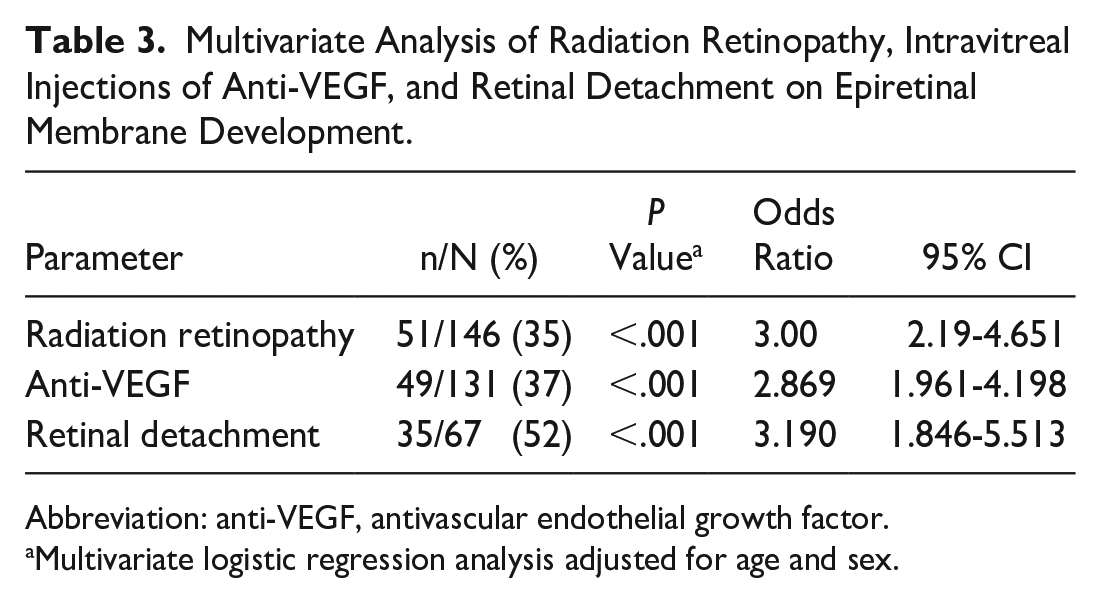
Table 3 shows the multivariate analysis of the association between radiation retinopathy and ERM development. During the 5-year follow-up, 146 patients (39%) developed radiation maculopathy. Of these, 95 (65%) did not have an ERM, although 51 (35%) later developed an ERM. As the OR value in Table 3 shows, radiation retinopathy was significantly associated with ERM development (P < .001).
Multivariate Analysis of Radiation Retinopathy, Intravitreal Injections of Anti-VEGF, and Retinal Detachment on Epiretinal Membrane Development.
Abbreviation: anti-VEGF, antivascular endothelial growth factor.
Multivariate logistic regression analysis adjusted for age and sex.
Association Between Intravitreal Anti-VEGF Injections and ERM Development
Table 3 also shows the association between IVT anti-VEGF injections and ERM development. During the 5-year follow-up, 131 eyes (35%) received IVT anti-VEGF injections. Of these, 82 (63%) did not develop an ERM, although 49 (37%) subsequently developed an ERM. As the OR value in Table 3 shows, IVT injections were significantly associated with a higher prevalence of ERM in eyes with choroidal melanoma (P < .001).
Association Between Preexisting Retinal Detachment and ERM Development
In addition, Table 3 shows the association between a preexisting RD and ERM development. No eye had an RRD or traction RD. However, 67 eyes (18%) had an exudative RD at baseline. Of those, 32 (48%) did not develop an ERM, although 35 (52%) subsequently developed an ERM. The mean time to ERM development was 18 months (median, 12; range, 6-48). As the OR value in Table 3 shows, RD was the strongest predictor of ERM development compared with the other risk factors.
Visual Acuity
Contralateral Control Eyes
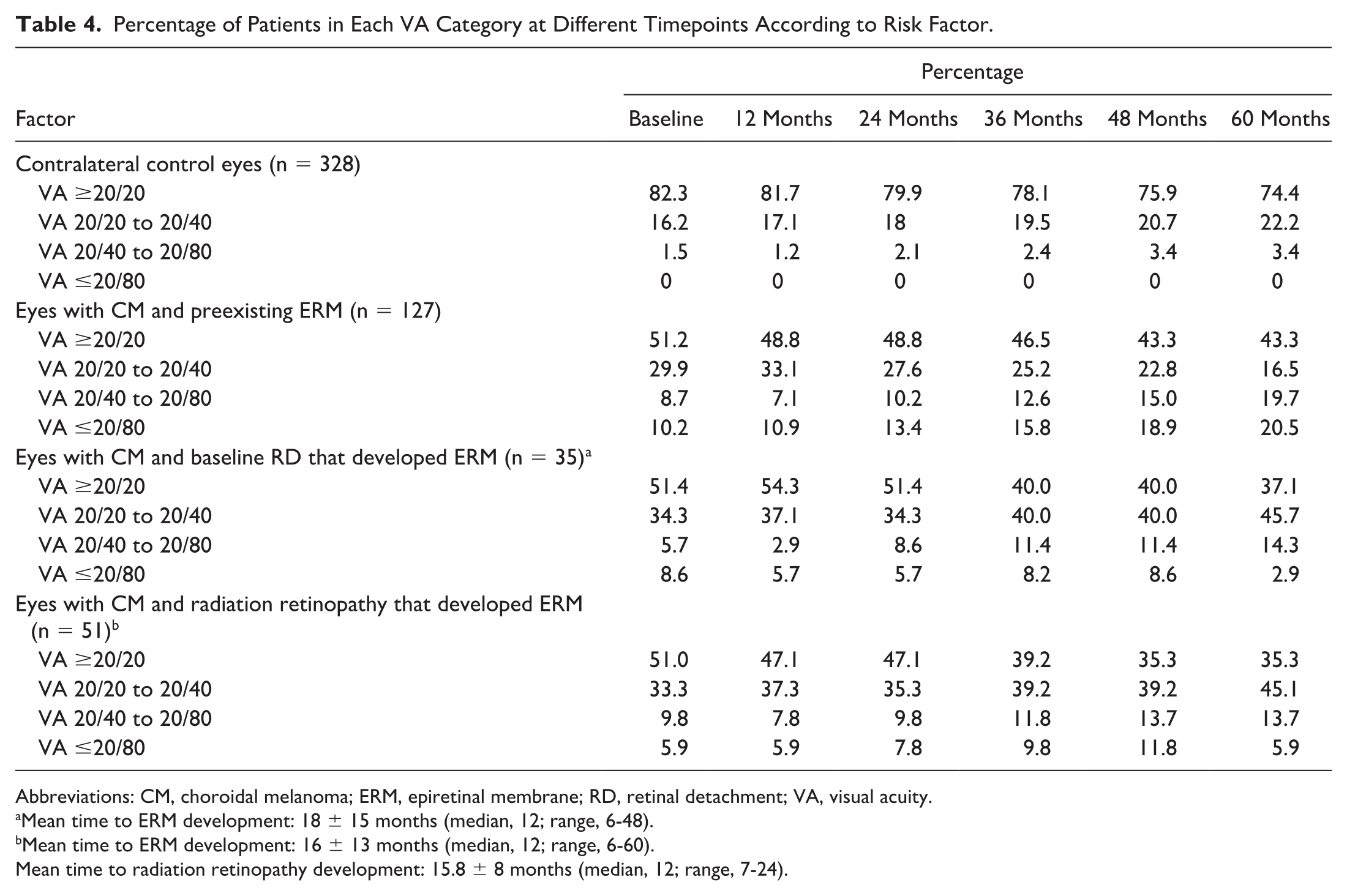
Of the contralateral control eyes, 268 (82%) had a baseline VA of 20/20 or better. At the end of the 60-month follow-up, 242 (74.4%) had a VA of 20/20 or better. (Table 4 and Figure 1).
Percentage of Patients in Each VA Category at Different Timepoints According to Risk Factor.
Abbreviations: CM, choroidal melanoma; ERM, epiretinal membrane; RD, retinal detachment; VA, visual acuity.
Mean time to ERM development: 18 ± 15 months (median, 12; range, 6-48).
Mean time to ERM development: 16 ± 13 months (median, 12; range, 6-60).
Mean time to radiation retinopathy development: 15.8 ± 8 months (median, 12; range, 7-24).
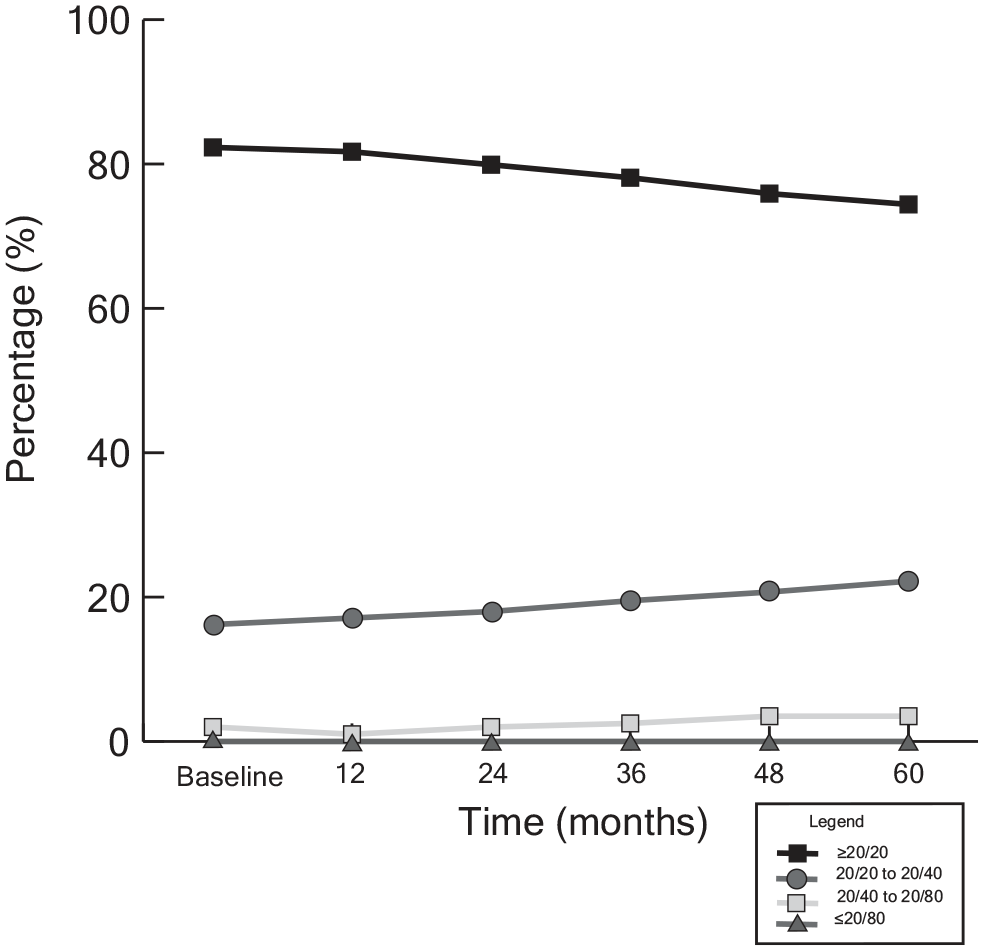
Percentage of contralateral control eyes in each visual acuity (VA) category (N = 328). Most contralateral control eyes showed a minimal change in VA over time, as shown by the stable trend in the percentage in each VA category across follow-up visits.
The mean change in logMAR VA from baseline was 0.003, 0.009, and 0.02 at 12 months, 36 months, and 60 months, respectively (Table 5). As expected for untreated eyes, no statistically significant differences in VA were observed between follow-up visits and the baseline visit.
Change in LogMAR VA Over Time.

Abbreviations: anti-VEGF, antivascular endothelial growth factor; CM, choroidal melanoma; ERM, epiretinal membrane; RD, retinal detachment; VA, visual acuity.
Eyes With Choroidal Melanoma Presenting With an ERM
Of the 127 patients with choroidal melanoma and a preexisting ERM, 64 (51%) had a baseline VA of 20/20 or better, 38 (30%) had a VA of 20/20 to 20/40, 11 (9%) had a VA between 20/40 and 20/80, and 12 (10%) had a VA of 20/80 or worse (Table 4 and Figure 2). At 60 months, fewer patients (54 [43%]) had a VA of 20/20 or better, and fewer patients (21 [17%]) had a VA of 20/20 to 20/40, corresponding to an increase in patients with a VA of 20/40 to 20/80 (25 [20%]). There was an increase in VAs worse than 20/80 (26 [21%]).
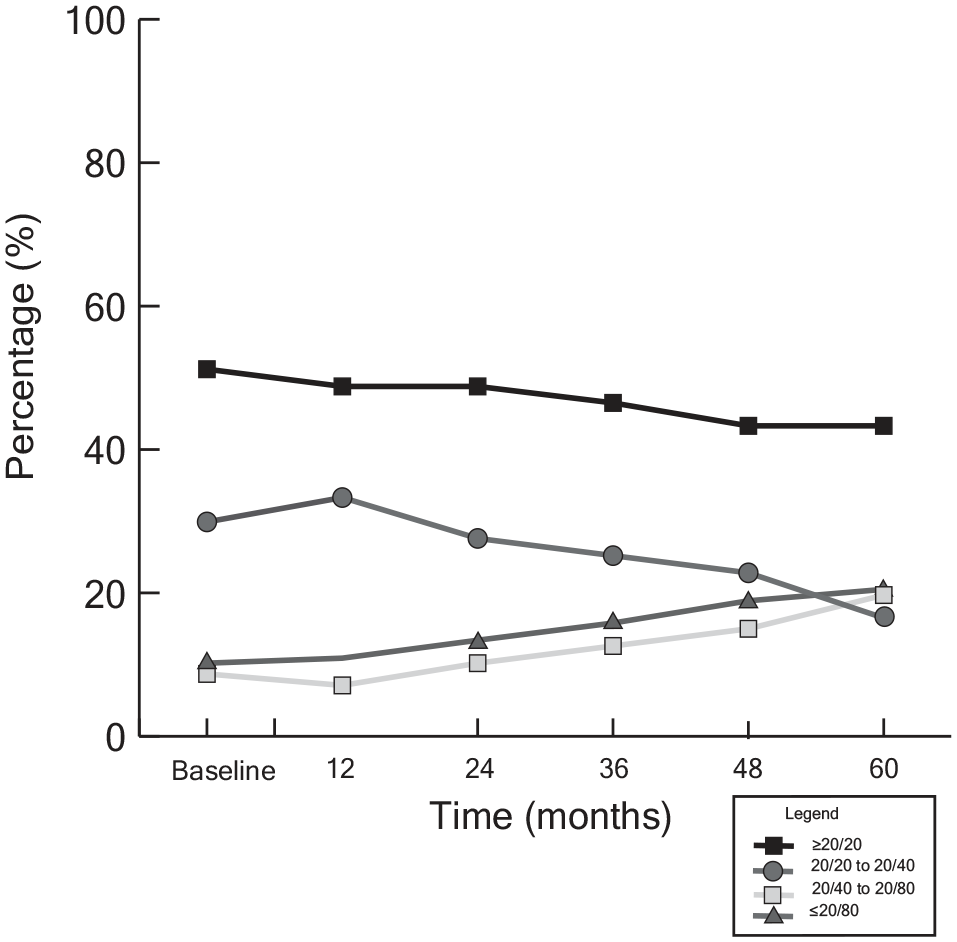
Percentage of eyes with preexisting epiretinal membranes (ERMs) in each visual acuity (VA) category (n = 127). There was a decline in VA over time. This is shown by the reduction in the distribution of patients with a VA of 20/40 or better and an increase over time in patients with a VA worse than 20/40.
There was a significant reduction in logMAR VA in eyes with choroidal melanoma diagnosed with an ERM before irradiation, with a mean VA change from baseline of 0.002, 0.02, 0.04, 0.05, and 0.06 at the 12-month, 24-month, 36-month, 48-month, and 60-month visits (Table 5). Therefore, a preexisting ERM corresponded to higher proportions of VAs of 20/40 or worse over time, especially when compared with control eyes.
Eyes With Choroidal Melanoma That Presented With an RD and Developed an ERM
Of the 35 patients with choroidal melanoma who presented with an RD and developed an ERM, 17 (51%) had a baseline VA of 20/20 or better, 12 (34%) had a VA of 20/20 to 20/40, 2 (6%) had VA between 20/40 and 20/80, and 3 (9%) had VA of 20/80 or worse (Table 4 and Figure 3). At 60 months, fewer patients (13 [37%]) had a VA of 20/20 or better, more patients (16 [46%]) had a VA of 20/20 to 20/40, and more patients (5 [14%]) had a VA of 20/40 to 20/80. At 60 months, 1 patient (3%) had a VA worse than 20/80. No differences were observed in the VA at the 12-month, 24-month, 36-month, and 48-month follow-up visits (Table 5).
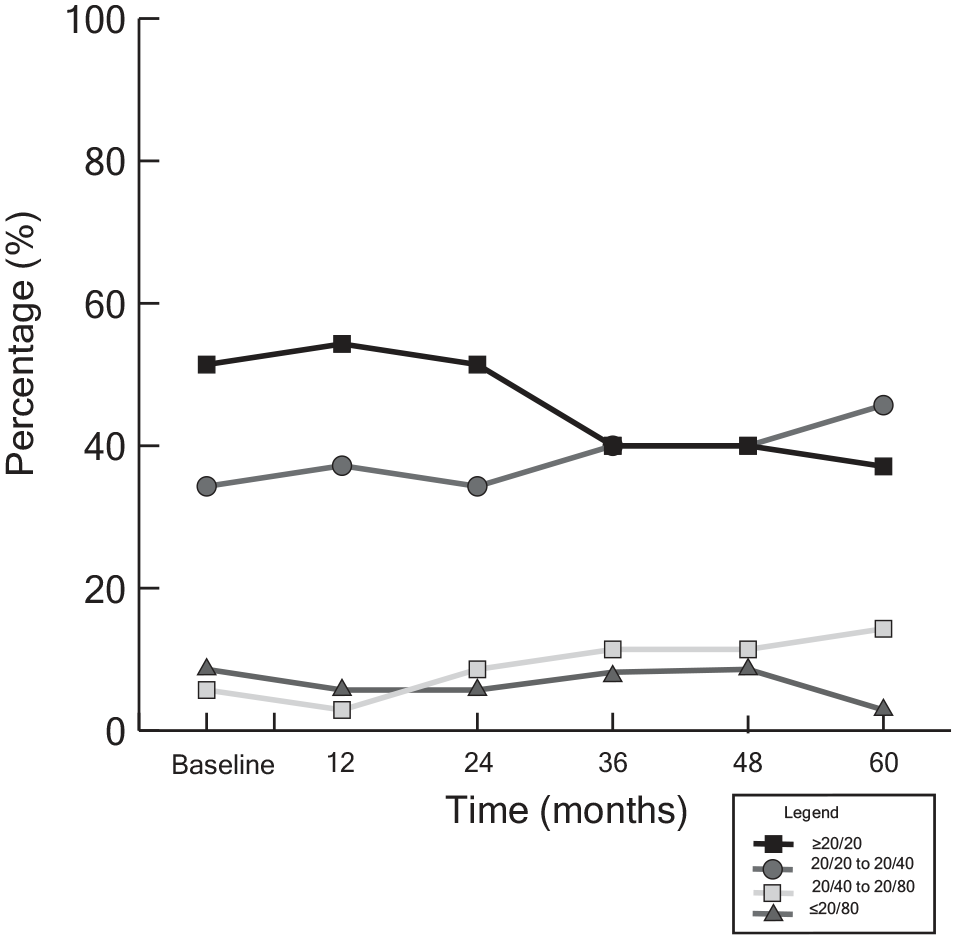
Percentage of eyes with choroidal melanoma and a baseline retinal detachment that developed an epiretinal membrane in each visual acuity (VA) category (n = 35). These patients had an initial improvement in VA, as shown by the increase in the percentage with a VA of 20/40 or better in the first 12 months. Between 24 months and 60 months, the percentage with a VA from 20/20 to 20/80 increased. The percentage with a VA of 20/20 or better declined. In addition, the percentage with a VA of 20/80 or worse declined.
A significant reduction in logMAR VA of 0.04 was observed at the 60-month visit (P = .016). Thus, RD was significantly associated with worse visual outcomes over time.
Eyes With Choroidal Melanoma That Developed Radiation Retinopathy
Of the 51 eyes with choroidal melanoma that developed radiation retinopathy and ERM, 26 (51%) had a baseline VA of 20/20 or better, 16 (33%) had a VA of 20/20 to 20/40, 5 (10%) had a VA between 20/40 and 20/80, and 3 (6%) had VA of 20/80 or worse (Table 4 and Figure 4). At 60 months, fewer patients (18 [35%]) had a VA of 20/20 or better, more patients (23 [45%]) had a VA of 20/20 to 20/40, more patients (7 [14%]) had a VA of 20/40 to 20/80; 3 patients (6%) had a VA worse than 20/80. No differences were observed in VA at the 12-month, 24-month, and 36-month follow-up visits.
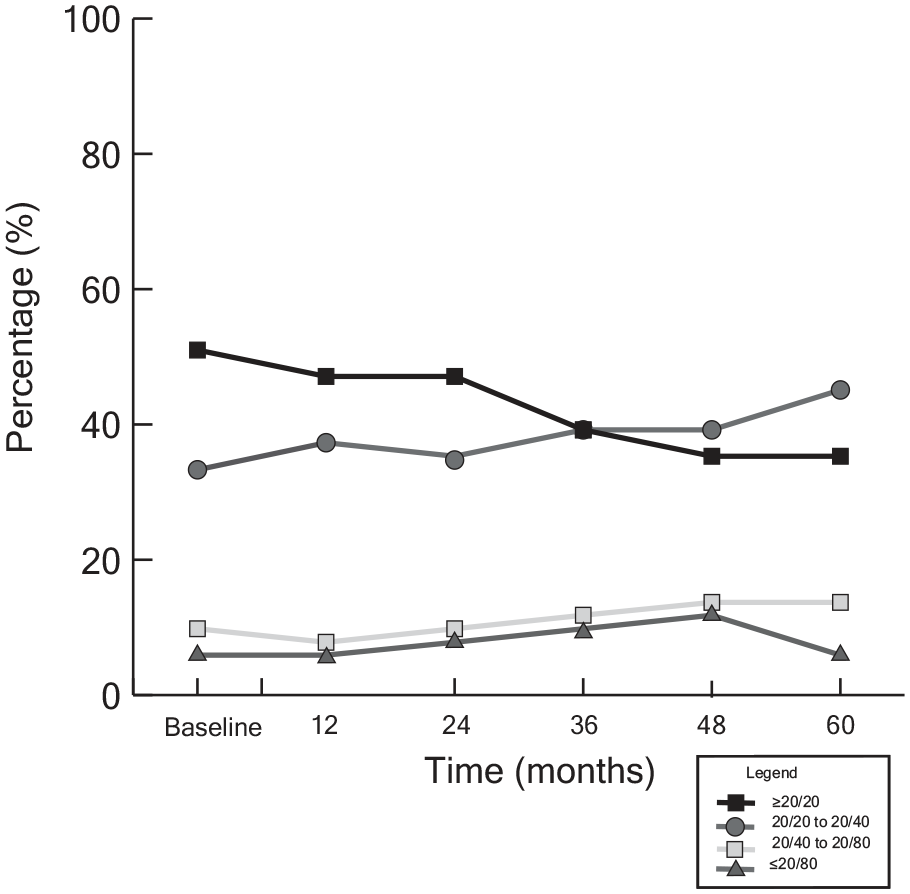
Percentage of eyes with choroidal melanoma in each visual acuity (VA) category that developed radiation retinopathy (n = 51). The percentage with a VA of 20/20 or better showed a general decline over time, while there was an increase in the distribution with a VA from 20/40 to 20/80.
There was a significant reduction in logMAR VA of 0.02 at the 48-month visit and 0.03 at the 60-month visit (P = .042 and P = .010, respectively) (Table 5). Therefore, radiation retinopathy–associated ERMs were associated with worsening VA over time.
Eyes With Choroidal Melanoma Treated With Anti-VEGF for Radiation Maculopathy
Of the 127 eyes with choroidal melanoma treated with anti-VEGF for radiation maculopathy and subsequently developed an ERM, 64 (51%) had a baseline VA of 20/20 or better, 127 (30%) had a VA of 20/20 to 20/40, 11 (9%) had a VA between 20/40 to 20/80, and 12 (10%) had a VA of 20/80 or worse (Table 4 and Figure 5). At 60 months, fewer patients (54 [43%]) had a VA of 20/20 or better and fewer patients (21 [17%]) had a VA of 20/20 to 20/40. There was a corresponding increase in patients with a VA from 20/40 to 20/80 (25 [20%]).
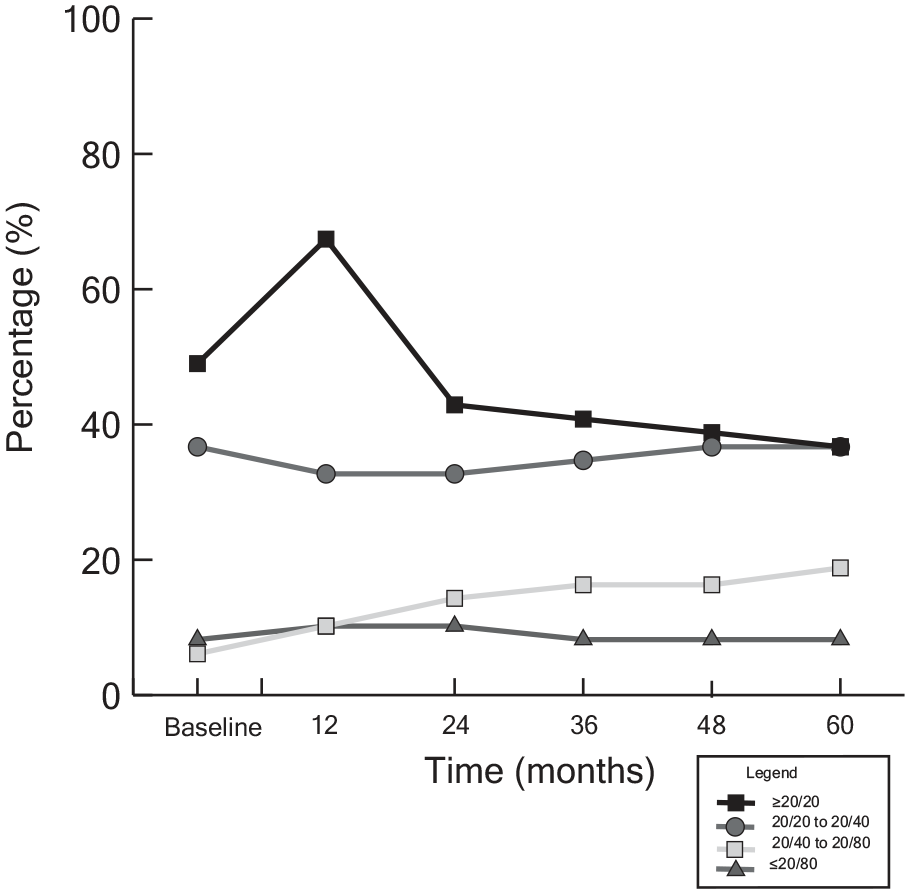
Percentage of eyes with choroidal melanoma treated with antivascular endothelial growth factor in each visual acuity (VA) category (n = 49). There was an initial increase in the percentage with a VA of 20/20 or better at the 12-month follow-up. After 12 months, the percentage with 20/20 or better declined. The percentage with a VA of 20/20 to 20/40 or of 20/80 or worse showed minimal change in the distribution. The percentage in the 20/40 to 20/80 category increased over the follow-up period, from 6.1% to 18.4%.
There was a significant reduction in logMAR VA from baseline of 0.02 at the 48-month visit and of 0.03 at the 60-month visit (P = .039 and P = .026, respectively) (Table 5). Although it is difficult to attribute the decline in VA over time solely to an ERM in eyes treated with anti-VEGF, an increased proportion of patients with declining VA was noted despite this vision-saving intervention.
Conclusions
This study identified a significantly higher prevalence of preexisting ERM in eyes with choroidal melanoma than in control eyes, suggesting that an ERM can be considered a diagnostic finding. This is reasonable because the primary pathophysiological mechanism involves the upregulation of inflammatory cytokines such as tumor necrosis factor-α, interleukin (IL)-6, IL-8, fibroblast growth factor, platelet-derived growth factor, and IL-1β, which are produced in response to neoplastic cells in the choroid.18–21 These cytokines can disrupt normal retinal pigment epithelium (RPE) cell function and promote the proliferation and migration of glial and fibrous cells onto the retinal surface, leading to ERM formation. 22
In addition, radiation retinopathy, anti-VEGF treatment, and radiation dose are interrelated factors contributing to the development of ERMs. Radiation can cause direct damage to retinal blood vessels, leading to ischemia and subsequent upregulation of inflammatory cytokines, which promote ERM formation. Although they reduce neovascularization and edema, anti-VEGF treatments can also influence the inflammatory milieu within the eye, highlighting the multifactorial nature of ERM development in this context. We acknowledge that this interplay is complex and multifactorial.
After ophthalmic plaque brachytherapy, 99 additional ERM cases occurred compared to 44 cases in contralateral eyes. This was significant because the Cox hazard regression analysis showed a higher risk for developing an ERM in irradiated eyes than in control eyes (HR, 2.669; P < .001; 95% CI, 1.863-3.909). This finding suggests the presence of choroidal melanoma and the use of radiation treatment are associated with a further risk for an ERM. Moreover, this study found a relationship between an increased radiation dose to the fovea and the development of ERM (Table 2). Given that more radiation is required to treat larger choroidal melanomas, it is no wonder that ERM formation was associated with an increasing AJCC cT category. In addition, the advent of radiation maculopathy and the use of anti-VEGF therapy were associated with the development of ERMs.
Radiation maculopathy was associated with an ERM in 51 patients (35%) (OR, 3.00; 95% CI, 2.190-4.561; P < .001). Radiation maculopathy has been characterized by macular edema, microaneurysms, retinal hemorrhaging, and capillary nonperfusion. This suggests our finding of an increased ERM prevalence with radiation maculopathy was likely related to secondary tissue ischemia, secondary neovascularization, and fibrovascularization.9,18,23,24 This study also suggests that the stimulus of chronic progressive radiation maculopathy outweighed or outlasted any possible antifibrotic properties of radiation therapy.25,26
Independent of the presence of radiation retinopathy, IVT injection of anti-VEGF medications was found to be associated with increased odds of developing an ERM in eyes with choroidal melanoma (OR, 2.869; 95% CI, 1.961-4.198; P < .001). This was not surprising because it is known that IVT injections can induce a PVD.27,28 After PVD alone, glial cells and cortical vitreous remnants are known to proliferate in the vitreomacular interface. 4 However, although it is known that IVT injections can induce PVD, we could find no literature proving that IVT anti-VEGF injections alone cause ERMs.4,7,8,28
An ERM occurred in 52% of cases presenting with a melanoma-associated exudative RD. In the multivariate analysis comparing radiation treatment, the advent of radiation maculopathy, the use of IVT anti-VEGF therapy, and the existence of RD at presentation, RD at presentation was found to be the strongest predictor of ERM development (OR, 3.190; 95% CI, 1.846-5.513; P < .001).
The literature suggests that with RRD, RPE cells migrate through the retinal break into the vitreous and then settle on the retinal surface.4,5,29,30 Subsequently, those RPE cells undergo fibrous metaplasia, forming plaques within a matrix of collagenous tissue on the ILM. 31 Although localized RPE hyperplasia has been associated with exudative choroidal tumors, choroidal melanomas are rarely associated with or induce retinal holes. 32 Exceptions include those associated with transpupillary thermotherapy or during transretinal biopsy.33–36
VA retention is complex and dependent on melanoma size, tumor location, and tumor-associated side effects (exudative RD, hemorrhage, glaucoma). 37 After treatment, additional risks include radiation-associated complications (maculopathy, optic neuropathy, and cataract). 38 Therefore, attributing differences in VA solely to the development of an ERM is not possible, even in this large series. However, the present study offers insight into the presence of an ERM, associated risk factors, and overall VA outcomes. For example, a preexisting ERM was associated with worse VA at all follow-up visits. This cohort had the most patients (20.5%) with a VA of 20/80 or worse at the 60-month follow-up (Table 4).
Eyes with choroidal melanoma that presented with an exudative RD typically had improvements in VA. During the first 12 months after treatment, slightly more than 50% of patients showed improvement to 20/20 or better, while 9% improved to better than 20/80. Despite these improvements, neither was statistically significant.
We acknowledge the limitations of using fellow eyes as controls given the inherent systemic predispositions that can potentially influence ERM development. However, these eyes were suitable controls because of age parity. To mitigate bias, we compared our prevalence rates with epidemiology data (Table 6).1,39–41 The Blue Mountains Eye Study reported a prevalence of 7.2% to 11.6% in age-matched adults, and the Beaver Dam Eye Study reported a prevalence of 11.8%.1,2 The prevalence in these large epidemiology studies closely match the prevalence in our fellow control eyes (12.1%). This approach remains valuable for isolating the influence of choroidal melanoma on ERM formation. Other potential weaknesses of this study include that the natural history of secondary ERMs has not been well characterized. The ERMs were diagnosed along with several comorbidities, including choroidal melanoma with and without RD, radiation treatment, radiation maculopathy, and anti-VEGF therapy. Therefore, this study does not indicate a specific etiology.
Summary of ERM Prevalence and Reported Risk Factors in Population-Based Epidemiology Studies.
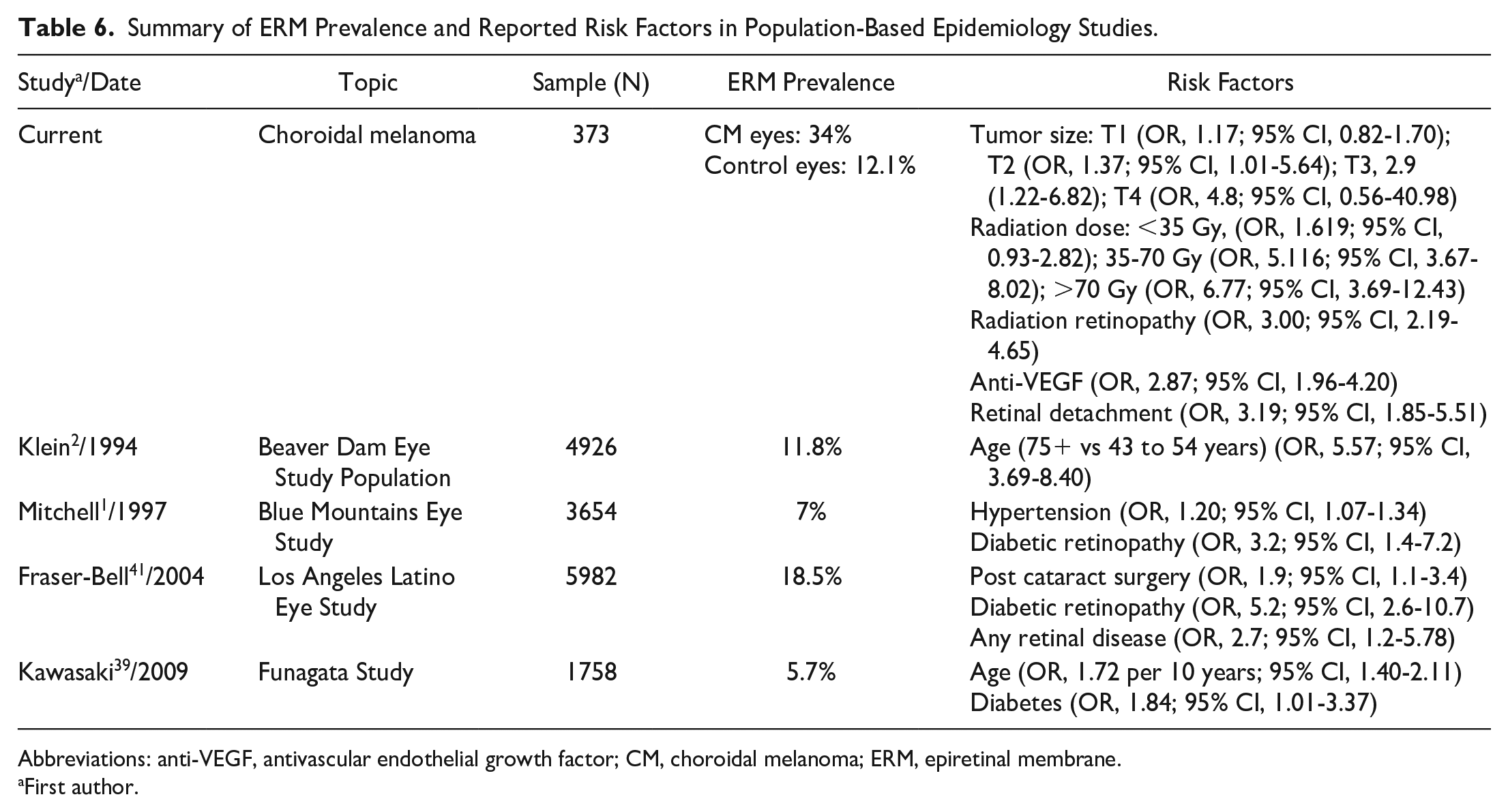
Abbreviations: anti-VEGF, antivascular endothelial growth factor; CM, choroidal melanoma; ERM, epiretinal membrane.
First author.
In addition, we found the most ERMs in eyes with AJCC cT1 and cT2 choroidal melanomas. Patients with larger T3-category and T4-category tumors present less frequently to our center and are associated with characteristics that preclude OCT (tumor size, larger RDs, more anterior locations).16,42
Strengths of this study include that it was performed in an established ophthalmic oncology center. In addition, a significant number of patients were included, evaluated, and staged for ERMs over a 5-year duration.
This study found that eyes with choroidal melanoma were more likely to present with and later develop ERMs. Although the radiation dose to the fovea, radiation maculopathy, and IVT anti-VEGF injection therapy are risk factors for subsequent ERM development, the strongest predictor was the presence of an RD at the initial diagnosis. These results can be used to explain the presence of an ERM in patients presenting with choroidal melanoma, in particular when they have a synchronous exudative RD. Thus, it can be used to explain the occurrence of ERM after vision-sparing treatment of radiation maculopathy.
Footnotes
Ethical Approval
This retrospective clinical case analysis research was performed in compliance with the tenets of the Declaration of Helsinki and directives of the US Health Insurance Portability and Accountability Act of 1996. Approval from the Institutional Review Board of The New York Eye Cancer Center was obtained to perform a retrospective review of charts and imaging data for 427 patients treated with palladium-103 plaque brachytherapy for choroidal melanoma between 2013 and 2022.
Statement of Informed Consent
All patients signed a written consent form to use their data for scientific research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of the article.
Funding
The Eye Cancer Foundation supplied an unrestricted grant in support of this work. This research was also supported in part by the Louisiana Board of Regents Endowed Chairs for Eminent Scholars program.
