Abstract
Introduction
Retinal and choroidal diseases are a major cause of vision loss globally, especially among older patients. 1 Treatments for neovascular age-related macular degeneration (nAMD), diabetic retinopathy (DR), diabetic macular edema (DME), and uveitis often require frequent, long-term intravitreal (IVT) injections. The high burden of treatment can be unsustainable for patients and caregivers, and low patient adherence and undertreatment can lead to suboptimal outcomes.2,3 In addition, anxiety about injections can lead to missed or rescheduled appointments with delays in treatment.2,4
The anatomy of the eye serves as a natural barrier to drugs and proteins in the systemic circulation, which makes ocular drug delivery challenging. 5 To address these barriers, IVT delivery platforms have been developed that release drugs in a controlled and sustained manner, thus improving bioavailability and maintaining a therapeutic concentration for extended periods. New therapies for these diseases would ideally have a favorable safety profile, have the ability to stabilize pathologic vasculature locally, and provide sustained delivery to support patient adherence to improve long-term outcomes.2,6
This review discusses multiple therapies, all of which are biocompatible (nontoxic to living tissue, avoiding immunological reactions). Some are biodegradable (undergo hydrolysis due to the presence of hydrolytically and/or enzymatically susceptible functional groups and then are absorbed into surrounding tissues), and some are bioerodible (erode through chemical and/or mechanical interactions with biological fluids that solubilize the material and enable its absorption into the surrounding tissues). 7 While the word implant may be part of the generic names of injectable IVT inserts, in this review the term implant refers to a surgically implanted platform and insert refers to an injectable platform.
Methods
The literature search for this review was performed using PubMed. The primary search included articles published between 2017 and 2023, and other articles were added because of their relevance. The following search terms were included: “ocular biomaterials”, “drug release kinetics”, “biodegradability”, “intravitreal implant”, “ocular implant”, “hydrogel”, “sustained drug delivery”, and “retinal pharmacodelivery”. Original articles and literature reviews were included.
Results
Key Biomaterials for Intraocular Delivery
Many biocompatible materials are used as part of ocular drug-delivery platforms for extended drug release. Optimal biomaterials for a sustained drug supply in the vitreous should ideally meet the following parameters6,8:
preserve the bioactivity of the drug by protecting its structure from unintended enzymatic degradation, changes in pH, and denaturation;
have the ability to deliver the drug to the relevant ocular tissues in a minimally invasive manner;
have high drug encapsulation efficiency (large amount of drug in a minimal volume);
avoid increasing intraocular pressure (IOP) during administration;
maintain the drug’s release at a predictable rate for an extended period;
avoid interference with vision by being biocompatible and small or optically transparent in the vitreous humor;
have an acceptable safety profile;
be readily manufactured, stored, and delivered.
This section describes the key biomaterials used in approved and investigational platforms for various indications involving the posterior segment.
Poly(lactic-co-glycolic acid)
Poly(lactic-co-glycolic acid) (PLGA) is a repeating carbon chain with oxygen moieties. More specifically, it is a synthetic, biodegradable polymer composed of repeating units of lactic acid and glycolic acid (Figure 1A). 9 PLGA is biocompatible and used widely for drug delivery.5,10 Under physiologic conditions, it is predominantly biodegraded through nonenzymatic hydrolysis of ester linkages,10,11 and it can be customized to degrade at specific rates, allowing for the controlled release of a drug over an extended period.10,12 Selecting PLGA with a specific molecular weight and level of polyglycolide allows the rate of degradation to be customized. Therefore, drug-delivery platforms can be tailored to the needs of different ocular diseases and therapeutic strategies.10,12 Once degraded, the monomeric components of the polymer are removed via biologic pathways. 10
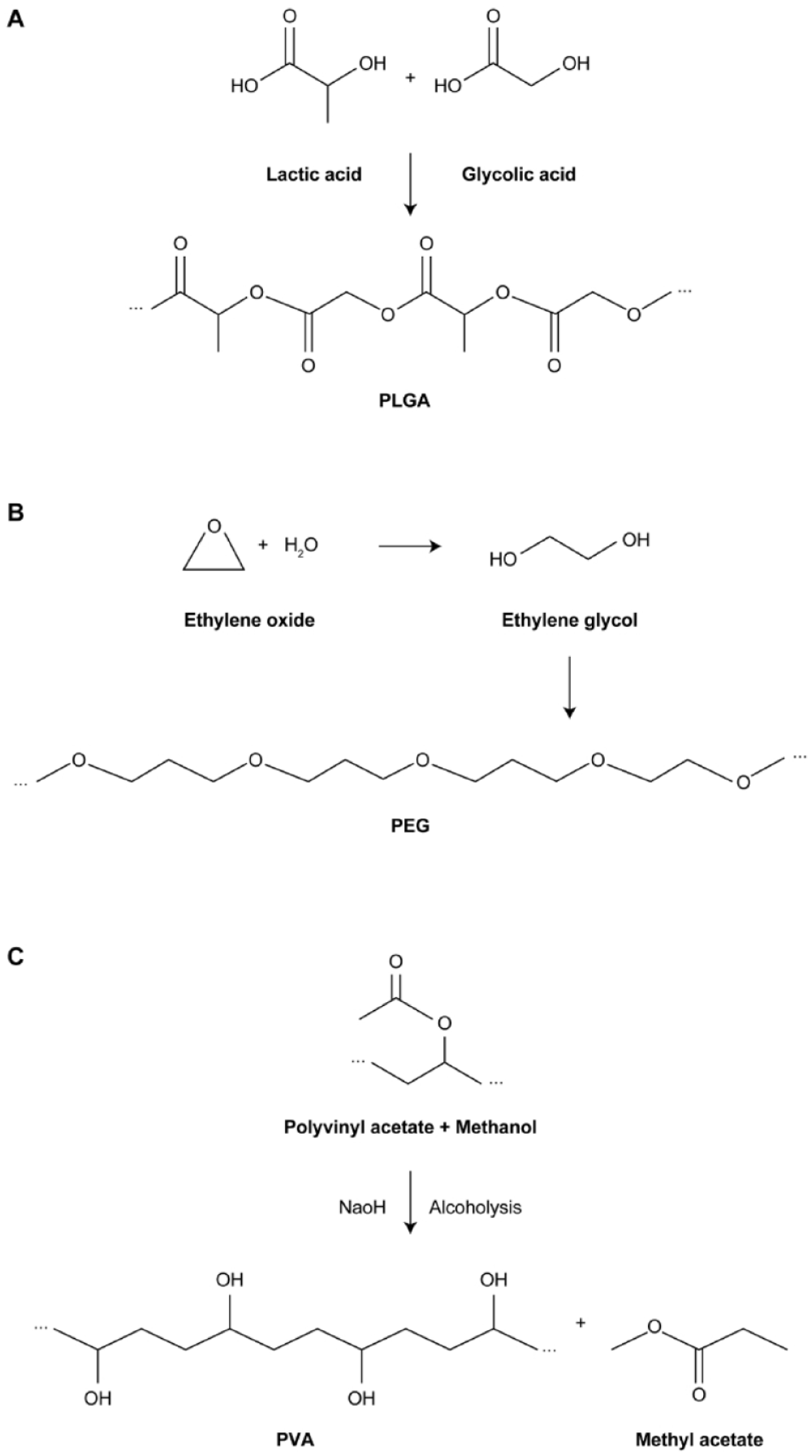
Structures of key polymers used in ocular drug delivery. (A) PLGA. (B) PEG. (C) PVA.
PLGA can encapsulate a wide range of drugs, including small molecules and proteins. 13 PLGA can be used in various forms, such as microparticles, often prepared as microspheres, which are microscopic objects (1 to 1000 μm in diameter) used to encapsulate drugs for specific applications. 14 Once inside the eye, PLGA particles degrade gradually, releasing the encapsulated drug over time. 10 Being biodegradable and biocompatible, PLGA is generally well tolerated in the eye. 13 However, in aqueous environments, such as the vitreous and aqueous humor, PLGA breaks down by hydrolysis into lactic and glycolic acids, creating an acidic microenvironment with a pH of 3 or lower, which has the potential to cause local inflammation and tissue damage in the eye, although this is rarely observed.15,16
Polyethylene Glycol and Polyethylene Glycol–Based Hydrogels
Polyethylene glycol is a hydrophilic polymer composed of repeating units of ethylene glycol derived from ethylene oxide (Figure 1B). 17 It is widely used in various industries, including pharmaceuticals, and is characterized by biocompatibility, solubility in aqueous media and the ability to augment drug stability and optimize efficacy.18,19 These properties have been useful in ocular drug delivery, including the application of polyethylene glycol–based hydrogels (3-dimensional networks that retain large amounts of water) designed to encapsulate drugs and release them in a controlled manner. 20 Polyethylene glycol can also be used to form micelles for drug encapsulation. 18 Such structures can improve the solubility of a drug and facilitate its absorption across ocular barriers.18,21
Hydrogels can be injected into the eye for controlled release of a drug, potentially overcoming challenges such as poor bioavailability, a short retention time, and rapid clearance.22–24 A hydrogel can hold a variety of substances within its crosslinked polymer matrix, including hydrophobic or hydrophilic agents, small molecules, or macromolecules. 8 Polyethylene glycol hydrogels are biocompatible and biodegradable and have a high water content, similar to the eye. 22 They typically have a burst release of drug at the beginning and end of treatment. In the initial burst release phase, as much as one half of the total drug may diffuse into the vitreous. 6 IVT hydrogels are injected as a solid insert or as a suspension that solidifies on contact with the vitreous fluid. 14 Challenges with these approaches include ensuring rapid solidification of the suspension to prevent leakage of drug particles into the vitreous and maintaining hydrogel integrity throughout release of the drug.25,26
Polyvinyl Alcohol
Polyvinyl alcohol is a synthetic, bioerodible polymer derived from vinyl acetate that has been polymerized and then hydrolyzed to remove acetate groups (Figure 1C). Polyvinyl alcohol is water soluble and biocompatible, and it is used in various industries including pharmaceuticals. 27 It is well tolerated in ocular tissues. 28
Polyvinyl alcohol has been used in ocular drug delivery in the form of hydrogels, eyedrops, and ophthalmic inserts. Polyvinyl alcohol–based inserts are formed as films or rods for sustained drug release in the eye, with the goal of prolonging therapeutic efficacy. 28 As with the polymers discussed above, polyvinyl alcohol–based platforms are used to enhance drug bioavailability by prolonging the presence of a drug at the target site, thereby reducing administration frequency and potential adverse effects. 29
Drug-Release Kinetics: Zero-Order vs First-Order
To appreciate the pharmacokinetics associated with the platforms currently available and in development, an understanding of drug-release kinetics is important. Ocular sustained-release platforms typically release drugs through zero-order or first-order kinetics.
The essential difference between zero-order and first-order kinetics is the elimination rate from the device compared with the total drug concentration within the device (Figure 2). Zero-order kinetics occurs when a constant amount of the drug is eluted from the device per unit of time, such as 1 µg every day; this rate of drug release is constant and independent of the total drug content in the device. Time is the rate-limiting factor in zero-order kinetics. 30 Thus, the target tissue receives a consistent level of drug over time. Polyvinyl alcohol–based IVT implants and inserts used to deliver ganciclovir or fluocinolone acetonide, respectively, are examples of sustained-release platforms that use zero-order kinetics. 14
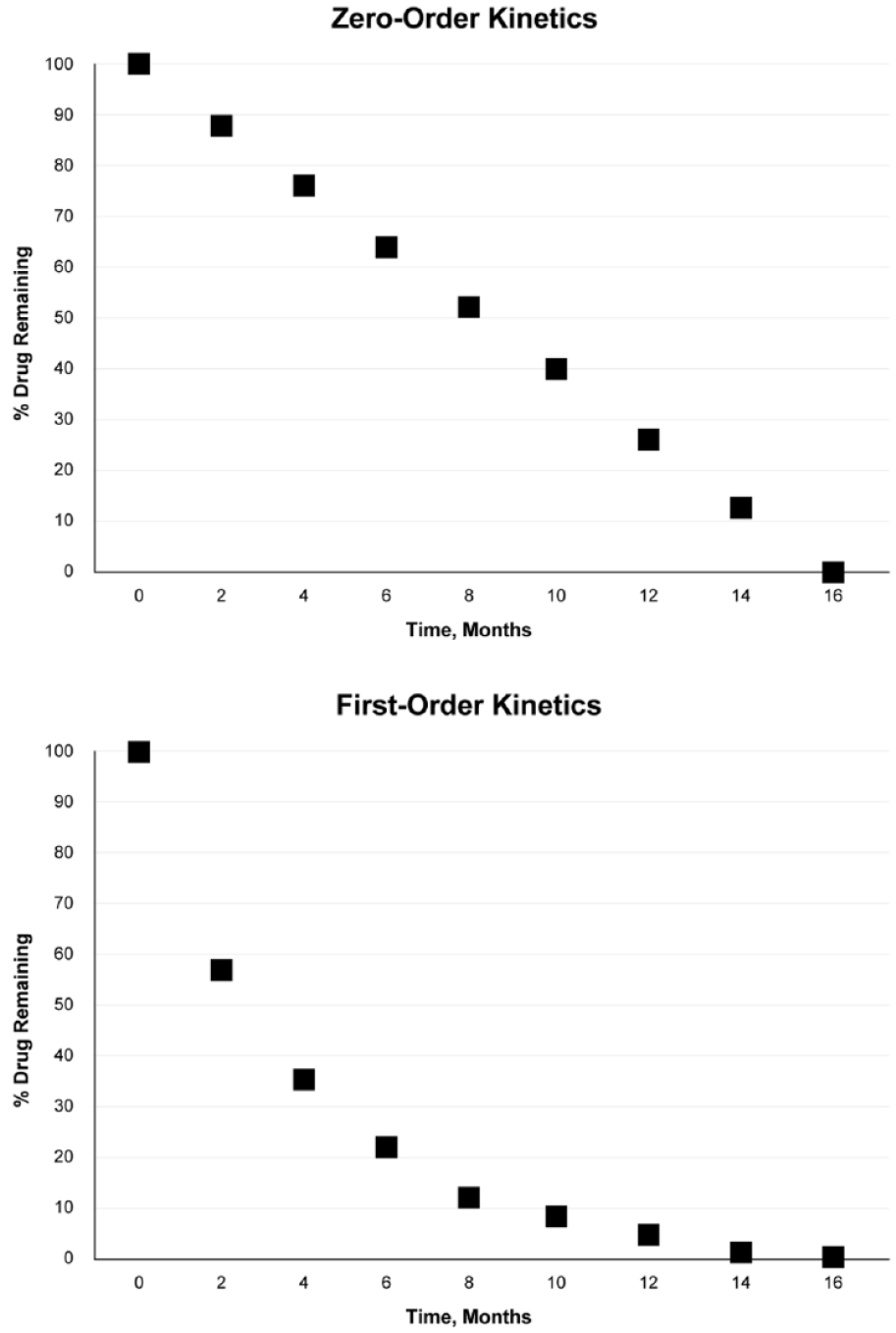
Graphs showing the difference between zero-order and first-order drug-delivery kinetics.
First-order kinetics occurs when a constant proportion, as opposed to an amount, of the drug is eliminated per unit of time. The rate of elimination is therefore proportional to the amount of drug in the device; overall, the more drug in the device (and the higher the concentration), the greater the amount of drug eliminated per unit of time. For example, a drug concentration of 50 ng/mL within an insert may be reduced to 25 ng/mL in the first month, 12.5 ng/mL in the second month, and 6.25 ng/mL in the third month (Figure 2). 30 The amount of drug eluted from the device is reduced over time, which may have therapeutic implications. The PLGA-based dexamethasone IVT insert is an example of a sustained-release platform with near first-order kinetics, 31 and some polyethylene glycol–based hydrogels have shown first-order kinetics.20,32
Biodegradability and Bioerodibility
Intraocular sustained-release platforms can be nonbiodegradable (insoluble) or composed of soluble, biodegradable materials that break down into nontoxic compounds that the body metabolizes and eliminates, bypassing the need for removal of the implant. 5 Biodegradable substances can undergo hydrolysis under physiologic conditions because of the presence of hydrolytically and/or enzymatically susceptible functional groups (eg, anhydride, ester, or amide bonds). Biodegradation can result in division of the polymer backbone or cleavage of side chains that are soluble in water. The cleavage products can then be metabolized and removed by the body. 7
Some materials used for these platforms are bioerodible, which means that they are biodegradable via chemical and/or mechanical interactions with biological fluids, such as the vitreous, that solubilize the polymer and enable absorption into the surrounding tissue.7,33 Polyethylene glycol–based hydrogels are often described as bioresorbable, which implies the polymer is absorbed into the surrounding tissue. 7
An advantage of bioerodibility is the potential to alter the erosion rate by modifying the chemical structure and adding surfactants that can be anionic or cationic. When the platform comes in contact with vitreous fluid, the matrix within the insert begins to bioerode, resulting in a controlled and sustained release of the drug contained in the matrix. Drug release from a bioerodible insert involves chemical and/or mechanical interactions with a biological fluid, which results in the dissolution of the polymer into the fluid. The polymer matrix undergoes bulk or surface hydrolysis, and the resulting release kinetics in either case are expected to be zero-order. 33
With biodegradable platforms, a final burst stage can occur when hydrolysis of the polymer reaches a critical point during release of the remaining drug. 28
IVT Injection vs Surgical Implantation
At present, IVT injection and surgical implantation are the 2 main routes of administration of ocular drug-delivery platforms for sustained release to manage retinal and choroidal diseases. In general, IVT inserts are smaller than implants because they are injected into the vitreous cavity and must be able to fit in a needle. A specialized applicator is typically needed for the successful placement of these inserts. The drug-release rate for this type of insert can be modified by adjusting the chemical composition of the matrix, and theoretically these inserts can be designed to release the drug over months or years depending on the objectives.28,34
Intraocular sustained-release implants that are surgically placed are historically larger than injectable inserts. 34 To date they have all been biocompatible and nonbiodegradable, and they may have to be removed or exchanged when all the drug has been released. These implants typically include a drug-releasing component, such as a polymer film or reservoir.28,34 They are typically placed in the posterior segment and are designed to release the drug over an extended period, typically months to years. 28
Another type of implant is the Port Delivery System (PDS), which is nonbiodegradable. The PDS differs from other ocular implants because it is intended to be permanent and refillable. It is placed in the sclera and contains a drug reservoir with a release control element that allows drug release into the vitreous over many months. 34 The PDS was voluntarily recalled by its manufacturer (Genentech) in October 2022 because of cases of septum dislodgement. 35 The PDS is to be reintroduced to the market in 2024.
US Food and Drug Administration–Approved Platforms
This section will describe some of the key US Food and Drug Administration (FDA)–approved platforms used for sustained drug release in the vitreous.
Biodegradable PLGA-Based IVT Insert
The dexamethasone IVT implant (Ozurdex, Allergan) is an injectable insert that delivers the corticosteroid to the back of the eye for between 3 months and 6 months. 36 It has been FDA approved for ME associated with retinal venous occlusive disease as well as for posterior noninfectious uveitis and DME and has demonstrated improved anatomic and visual outcomes for each indication. 37 This insert is approximately 0.46 mm × 6 mm and incorporates microparticles of dexamethasone in a PLGA-based polymer matrix that releases 0.7 mg of dexamethasone as it degrades over time (Table 1 and Figure 3).14,28,36,38 In preclinical studies in monkeys, it exhibited biphasic release with an initial phase of high dexamethasone concentration followed by a second phase of low drug concentration, exhibiting near first-order kinetics.31,37,39
FDA-Approved Platforms and Platforms Recently or Currently Studied in Clinical Trials.
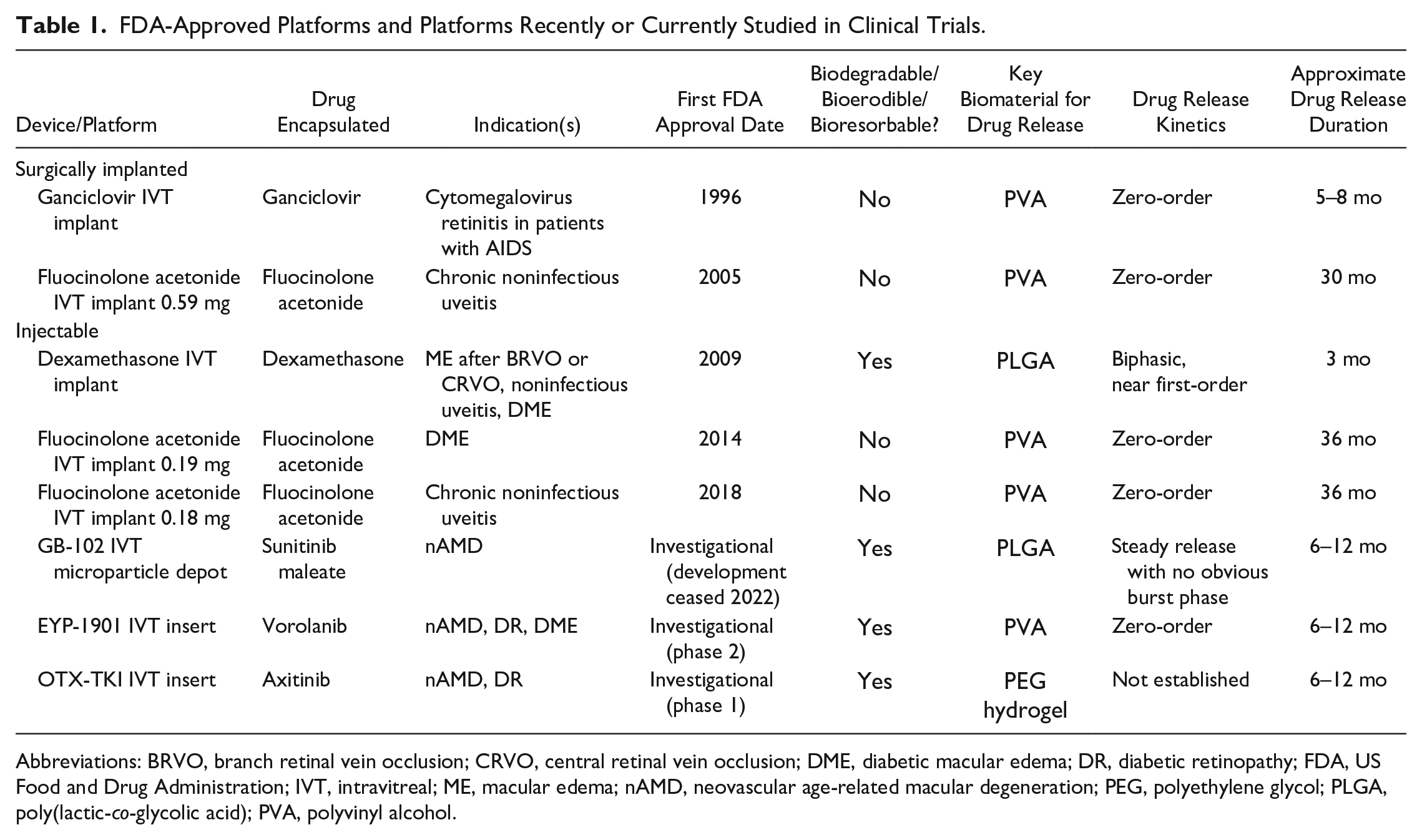
Abbreviations: BRVO, branch retinal vein occlusion; CRVO, central retinal vein occlusion; DME, diabetic macular edema; DR, diabetic retinopathy; FDA, US Food and Drug Administration; IVT, intravitreal; ME, macular edema; nAMD, neovascular age-related macular degeneration; PEG, polyethylene glycol; PLGA, poly(lactic-co-glycolic acid); PVA, polyvinyl alcohol.
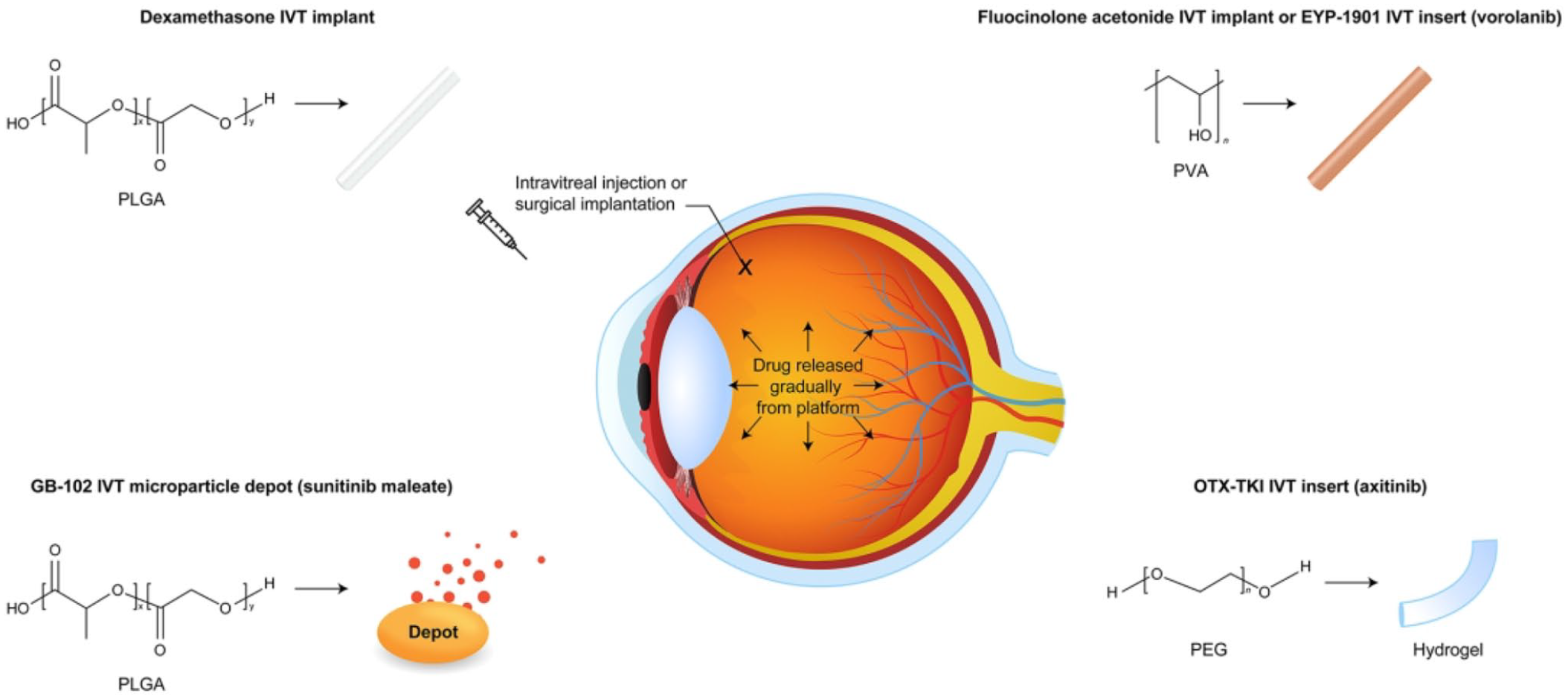
Various types of drugs have been embedded in sustained-release matrices or depots based on materials including PLGA polyvinyl alcohol, or polyethylene glycol hydrogel. The drug is released gradually in the vitreous over months or years, with kinetics dependent on the formulation.
PLGA is degraded into nontoxic lactic acid and glycolic acid under physiologic conditions, which reduces the local pH, and this may affect local tissues.40,41 Migration of this implant into the anterior chamber has been documented in case series, and corneal edema and endothelial damage can result from mechanical and chemical toxicity after such migration. 42 IOP elevation and cataracts are also potential adverse events with this insert. 36
Nonbiodegradable Polyvinyl Alcohol–Based IVT Implants and Inserts
There are multiple FDA-approved, nonbioerodible polyvinyl alcohol–based implants and injectable inserts for use in the vitreous. This type of platform enables customized release durations, zero-order release kinetics, and high drug loading. 14
The ganciclovir IVT implant (Vitrasert; discontinued in the US) was developed for the treatment of cytomegalovirus retinitis (Table 1) and was approved by the FDA in 1996. 28 It measures 10.0 mm × 4.0 mm × 3.5 mm and contains 4.5 mg of ganciclovir in a device composed of a restricted membrane made from the polymer ethylene vinyl acetate, which limits drug release, and a frame of the permeable polyvinyl alcohol polymer that regulates the diffusion of ganciclovir into the vitreous.11,28,43,44 The device is implanted in the vitreous cavity and sutured to the sclera. 45 Each implant is designed to release the drug over 5 to 8 months and can be removed and replaced after drug depletion, as evidenced by clinical findings. 44 The release rate of ganciclovir from the implant is 1 μg per hour. The implant allows diffusion of vitreous fluid into its interior, which dissolves the drug, allowing diffusion of the drug into the vitreous at a constant rate, with zero-order kinetics.29,45 The only abnormality observed in histopathologic studies of enucleated rabbit eyes containing the implant was granulomatous inflammation surrounding the scleral silk anchoring sutures. 43 The ganciclovir implant was the first nonbiodegradable/nonbioerodible implant for ocular use and has shown superiority over systemic ganciclovir therapy for control of retinitis. 28
Three fluocinolone acetonide IVT implants/inserts are approved by the FDA. All are nonbiodegradable, and they differ by indication, administration route, and drug dose, among other features.
The first device, a fluocinolone acetonide IVT implant 0.59 mg (Retisert, Bausch + Lomb) was approved in 2005 and is implanted surgically (Table 1). It is a 3.0 mm × 2.0 mm × 5.0 mm drug reservoir enclosed in a silicone elastomer cup lined with a polyvinyl alcohol membrane that controls drug release.28,46 It is surgically anchored to the sclera by a suture. 28 This implant is indicated for chronic noninfectious uveitis affecting the posterior segment. The implant contains a 0.59 mg tablet of fluocinolone, and it releases 0.6 μg of drug per day in the first month and 0.3 to 0.4 μg per day over the following 30 months. It can be replaced after drug depletion as determined by clinical efficacy. 46 In rabbits, this implant showed constant drug concentrations in the posterior segment and the vitreous, suggesting near zero-order release kinetics for at least 1 year. 31 Clinical trials showed a significant reduction in uveitis recurrence rates between 34 weeks preimplantation and 34 weeks postimplantation. 46 Potential problems associated with this implant include dissociation of its components over time, which could lead to visual issues, complications of removal, and hemorrhage.28,46 Drawbacks of this implant also include a high rate of elevated IOP and the need for filtering procedures to control IOP in many eyes. 46
The second and third fluocinolone IVT sustained-release devices are the fluocinolone acetonide IVT implant 0.19 mg (Iluvien, Alimera Sciences), approved in 2014 for DME, and the fluocinolone acetonide IVT implant 0.18 mg (Yutiq, Alimera Sciences), approved in 2018 for chronic noninfectious posterior uveitis (Table 1).28,31,47 These nonbioerodible, injectable, cylindrical inserts measure 3.50 mm × 0.37 mm. They contain fluocinolone combined with polyvinyl alcohol enclosed in an impermeable polyimide tube with a permeable polyvinyl alcohol membrane at 1 end of the tube, and they are designed to release the drug at an initial rate of 0.25 µg/day and to last for 36 months.28,48,49 In rabbit studies, drug levels peaked in ocular tissues in the first week, reached steady-state levels by month 3, gradually decreased through month 24, and were still detectable at 2 years. 50 Constant vitreous levels in rabbit studies have suggested near zero-order release for at least 2 years. 31 These inserts do not degrade and remain in the vitreous cavity after drug release is complete. 14 In clinical studies, treatment of DME with the 0.19 mg insert resulted in improved best-corrected visual acuity (BCVA) vs sham 45 and treatment of uveitis with the 0.18 mg insert resulted in a significantly lower percentage of patients who had uveitis recurrence within 6 months. 47 Multiple devices may have to be given over time, and migration of the inserts into the anterior chamber has been documented.28,47
Investigational Drug-Delivery Platforms
Biodegradable PLGA-based IVT Microparticle Depot
GB-102, previously in development by Graybug Vision, is an injectable microparticle depot loaded with the tyrosine kinase inhibitor (TKI) sunitinib maleate (Table 1).14,51 Sunitinib and some other TKIs, such as vorolanib and axitinib, inhibit multiple receptor tyrosine kinases simultaneously, including all receptors of vascular endothelial growth factor (VEGF). 51 The depot that makes up the structure of GB-102 was synthesized from PLGA and was designed to sustain the release of sunitinib in the posterior segment for more than 4 months, allowing the reduction of anti-VEGF injection frequency for patients with nAMD.14,31 After injection, microparticles with an average diameter of 12 μm consolidate and aggregate in the posterior vitreous, forming a depot to prolong drug release.31,51 The microparticles ideally remain close to the injection site, outside the visual axis. 2
In vitro, the depot released drug steadily for up to 28 days without an obvious burst release phase. In rat eyes, it prolonged ocular drug retention and suppressed neovascularization without ocular inflammation or foreign-body responses. 51 Although this platform showed evidence of efficacy in nAMD in early clinical studies, there was evidence of particle dispersion and of anterior chamber migration of the microparticles, and mild ocular hypertension was reported. 2
Bioerodible Polyvinyl Alcohol–Based IVT Insert
EYP-1901, in development by EyePoint Pharmaceuticals, is an investigational, injectable, bioerodible insert designed for the sustained release of the TKI vorolanib for patients with nAMD, DR, or DME (Table 1 and Figure 3). 2 The technology is similar to the fluocinolone injectable inserts discussed above, with the meaningful distinction being that EYP-1901 is bioerodible and completely dissolves over time. Based on preclinical studies, this insert allows for consistent vorolanib treatment over at least 6 months with zero-order kinetics. 52
In rabbit studies, the EYP-1901 insert showed a burst in drug release that rapidly resulted in steady-state levels in ocular tissues with an average zero-order drug-release rate of approximately 8% of the total dose per month through 8 months; this was followed by a rapid decrease through 10 months. 53 In a phase 1 clinical trial, adverse events after a single EYP-1901 insert injection were mostly mild and related to the injection, the BCVA and central subfield thickness (CST) of the retina were stable over 12 months, and the mean anti-VEGF injection frequency was reduced compared with the 12 months before eyes received the EYP-1901 insert. 54 The insert is being studied in phase 2 clinical trials in nAMD, DR, and DME. 52 Preparation for the global phase 3 LUGANO and LUCIA trials in nAMD have been initiated.
Biodegradable Polyethylene Glycol–Based Hydrogel IVT Insert
OTX-TKI, in development by Ocular Therapeutix, is an investigational, injectable, biodegradable, rod-shaped hydrogel insert that contains the TKI axitinib in a polyethylene glycol–based, crosslinked hydrogel matrix (Table 1 and Figure 3). The OTX-TKI insert is designed to offer sustained release of axitinib for 9 to 12 months for the treatment of VEGF-mediated retinal diseases.14,31,55 The hydrogel creates a meshwork that encapsulates axitinib and gradually dissolves in the vitreous, allowing the drug to be released gradually. 2
The ocular distribution of axitinib from OTX-TKI after a single IVT injection was characterized in nonhuman primates. Axitinib concentrations were above the half-maximum inhibitory concentration of VEGF receptor 2 in the retina, choroid, and retinal pigment epithelium consistently through 9 months. Hydrogel degradation was observed at approximately 5 to 6 months, with released axitinib particles visible in the vitreous at month 9. 25 In two phase 1 trials in patients with nAMD, the OTX-TKI insert was well tolerated and demonstrated biological activity.56,57 In one of those trials, the OTX-TKI insert reduced the frequency of anti-VEGF injections and maintained BCVA and CST, which were comparable to those in the aflibercept control arm that received injections every 8 weeks. 56 The OTX-TKI insert is also being evaluated for the treatment of DR in the phase 1 HELIOS trial. 58 The US-based phase 3 SOL trial has been initiated.
Conclusions
Therapies for diseases of the posterior segment are evolving and include multiple sustained-release intraocular platforms in commercial use and under development. Current FDA-approved platforms include biodegradable/bioerodible and nonbiodegradable/nonbioerodible devices that are implanted surgically or injected; all use biomaterials, such as polymers and hydrogels, to encapsulate the drug and deliver it gradually over months or years. These are in use for patients with noninfectious uveitis, infectious retinitis, retinal venous occlusive disease, and DME.
Footnotes
Acknowledgements
Professional medical writing support was provided by Scott Salsman of Two Labs Pharma Services and funded by EyePoint Pharmaceuticals.
Ethical Approval
Ethical approval was not applicable.
Statement of Informed Consent
Informed consent was not applicable.
Declaration of Conflicting Interests
Dr. Wykoff: Consulting—4DMT, AbbVie, Adverum, Aerie, AGTC, Alcon, Annexon, Apellis, Arrowhead, Ascidian, Bayer, Bausch+ Lomb, Boehringer Ingelheim, Cholgene, Clearside, Curacle, Eyebiotech, EyePoint, Foresite, Frontera, Genentech, Gyroscope, Iveric Bio, Janssen, Kato, Kiora, Kodiak, Kriya, Merck, Nanoscope, Neurotech, NGM, Notal Vision, Novartis, Ocular Therapeutix, Ocuphire, OcuTerra, ONL, Opthea, Oxular, Palatin, PerceiveBio, Perfuse, Ray, RecensMedical, Regeneron, RegenxBio, Resonance, Roche, Sanofi, SciNeuro, Stealth, Surrozen, Suzhou Raymon, THEA, TissueGen, Valo. Grants—4DMT, Adverum, AffaMed, Alexion, Alimera, Alkahest, Allgenesis, Amgen, Annexin, Annexon, Apellis, Asclepix, Bayer, Boehringer Ingelheim, Chengdu Kanghong, Clearside, Curacle, EyePoint, Gemini, Genentech, GlaxoSmithKline, Gyroscope, Ionis, iRenix, Iveric Bio, Janssen, Kodiak, LMRI, McMaster University, Nanoscope, Neurotech, NGM, Novartis, Ocular Therapeutix, Ocuphire, OcuTerra, OliX, Ophthotech, Opthea, Oxurion, Oxular, Oyster Point, PerceiveBio, Regeneron, RegenxBio, Rezolute, Roche, SamChunDang Pharm, Sandoz, Shanghai Henlius, UNITY, Verily, Xbrane. Stock options (not owner)—ONL, PolyPhotonix, RecensMedical, TissueGen, Visgenx, Vitranu.
Dr. Kuppermann: Consulting—Allegro Ophthalmics, Allergan/AbbVie, Aviceda Therapeutics, Bausch+Lomb, Boehringer Ingelheim, Clearside, EyeBio, Eyedaptic, EyePoint, Genentech Inc, Glaukos Corporation, InflammX Therapeutics, Iveric Bio, jCyte, Molecular Partners, Neurotech, Novartis Pharmaceuticals, Ocular Therapeutix, Regeneron Pharmaceuticals Inc, ReVana Therapeutics, Ripple Therapeutics, Roche Pharmaceuticals, Stealth, Theravance Biopharma. Research support—Allergan, Apellis, Clearside, Genentech Inc, Ionis, Iveric Bio, Novartis Pharmaceuticals, Regeneron Pharmaceuticals Inc, RegenxBio. Speaker fees—Allergan, Genentech, Roche. Dr. Kuppermann acknowledges an unrestricted grant from Research to Prevent Blindness to the Gavin Herbert Eye Institute.
Dr. Regillo: Consulting—4DMT, Adverum, Allergan, Annexon, Apellis, Aviceda, Bausch and Lomb, Clearside, Cognition, Eyepoint, Genentech, Iveric, Janssen, Kodiak, Lineage, Merck, NGM, Novartis, Ocugen, Ocuterra, Occuphire, Opthea, Ray, RegenxBio, Stealth, Thea, Zeiss. Research grant support—Adverum, Allergan, Annexon, Apellis, Astellas, EyePoint, Genentech, Gyroscope, Iveric, Janssen, Kodiak, Lineage, NGM, Notal, Novartis, Ocugen, Ocuterra, Opthea, Regeneron, RegenxBio.
Dr. Chang: Consulting—Genentech, RegenxBio, Iveric Bio. Research support—Regeneron, Allergan, Novartis, NGM, Opthea, Eyebio, OcuTerra, Alexion, Mylan.
Dr. Hariprasad: Consulting/speaker fees—Alimera Sciences, Allergan, EyePoint, Regeneron, Biogen, Apellis, Iveric Bio.
Drs. Duker, Altaf, and Saïm are employees of EyePoint Pharmaceuticals.
Funding
This work was supported by EyePoint Pharmaceuticals.
