Abstract
Introduction
Relapsing-remitting multiple sclerosis (RRMS) management has recently benefited from the approval of ponesimod (Ponvory, Janssen), a second-generation modulator of the sphingosine 1-phosphate1 receptor (S1P1R). 1 Ponesimod iterates on previous S1PR modulators such as fingolimod and offers increased selectivity for the S1P1R. The medication overwhelms the naturally present S1P concentration gradient, a crucial component of lymphocyte trafficking.2 –4 As a result, ponesimod was shown to lower the relapse rate, decrease magnetic resonance imaging endpoints, and provide symptomatic relief when compared with teriflunomide in the OPTIMUM (Oral Ponesimod Versus Teriflunomide In Relapsing Multiple Sclerosis) phase III trial. 5 However, the trial also identified a ponesimod-related adverse effect in that 5 of 565 treated individuals developed cystoid macular edema (CME).
Older S1P structural analogues, namely fingolimod, have similarly been implicated in ophthalmological pathologies, termed fingolimod-associated macular edema (FAME).6 –8 Fortunately, FAME occurs in only approximately 0.5% of patients and typically resolves after medication cessation. We report a case of macular edema in a patient with RRMS being treated with ponesimod. Cessation of ponesimod and initiation of topical 1% prednisolone acetate along with topical 0.07% bromfenac resulted in complete remission of the patient’s macular edema.
Case Report
A 75-year-old woman with a history of RRMS was referred for retinal consultation regarding macular edema in the left eye. The patient was started on ponesimod approximately 1 year prior for treatment of her RRMS. A baseline eye examination performed by the referring provider before ponesimod therapy was started showed no evidence of macular edema. On follow-up with the referring provider 9 months later, the patient was noted to have developed CME in the left eye. She had no visual or ocular complaints at that time. The patient was started on topical therapy with 1% prednisolone acetate twice daily and bromfenac once daily and was referred to our practice for further treatment.
On presentation to our clinic, the visual acuity (VA) was 20/40−1 OD and 20/30−1 OS. The intraocular pressures were normal, and the anterior segment examination findings were unremarkable except for a mild cataract in each eye. A dilated fundus examination and optical coherence tomography (OCT) results were unremarkable in the right eye. However, in the left eye, an examination and testing revealed macular thickening of 354 µm and trace cysts of intraretinal fluid consistent with CME (Figure 1). When compared with prior OCT scans from the referring doctor, a slight improvement in the macular edema was noted. The patient was visually asymptomatic and reluctant to discontinue ponesimod given its good systemic results. Therefore, the decision was made to taper the current topical therapy to once-a-day doses and to closely monitor the patient.
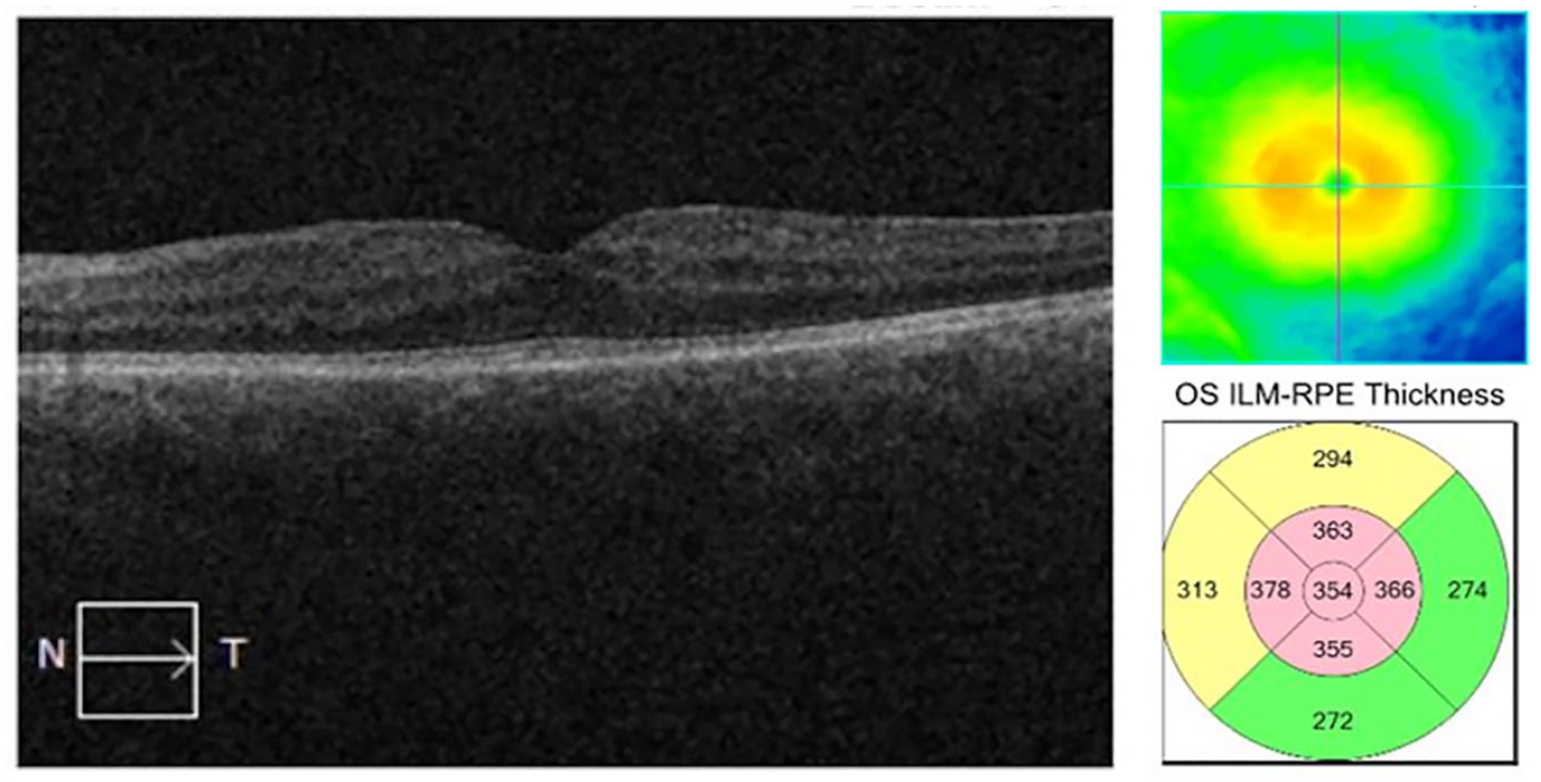
Optical coherence tomography of the left eye at initial presentation to our clinic shows intraretinal cysts consistent with cystoid macular edema.
On the patient’s return 1 month later, the VAs remained stable; however, OCT of the left eye revealed an interval increase in intraretinal fluid (Figure 2). The central macular thickness (CMT) had increased to 402 µm. The right eye remained unaffected. The patient’s neurologist was notified of these changes, and the recommendation was made to discontinue ponesimod and transition to an alternative therapy. At the direction of her neurologist, ponesimod was stopped and the patient was switched to teriflunomide—a pyrimidine synthesis inhibitor known to have lower ophthalmological risk. 7 Concurrently, her topical 1% prednisolone acetate dose was increased to twice daily while her topical bromfenac was maintained at a daily dose.
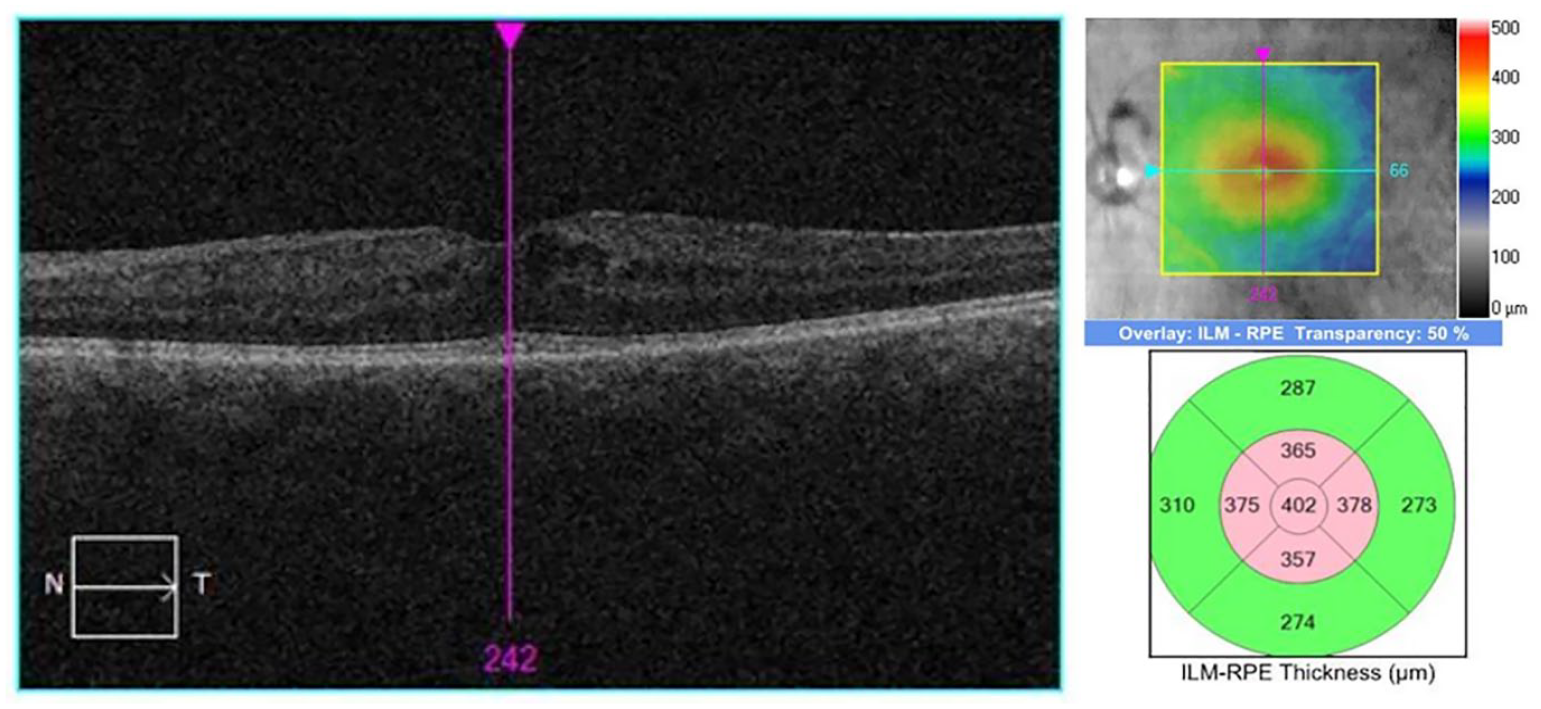
Optical coherence tomography of the left eye shows worsening intraretinal fluid at the patient’s 1-month follow-up.
The patient was reevaluated 1 month after stopping ponesimod and increasing her topical regimen. At this visit, OCT showed resolution of the intraretinal cysts (Figure 3), with the CMT returning to 326 µm. The topical prednisolone acetate 1% and bromfenac were discontinued. At her most recent follow-up, there was no recurrence of CME since the cessation of ponesimod (Figure 4) and the CMT had decreased to 284 µm. The VA remained stable and measured 20/30 OD and 20/25 OS. The patient had no visual or ocular complaints, and her RRMS was well controlled on teriflunomide.
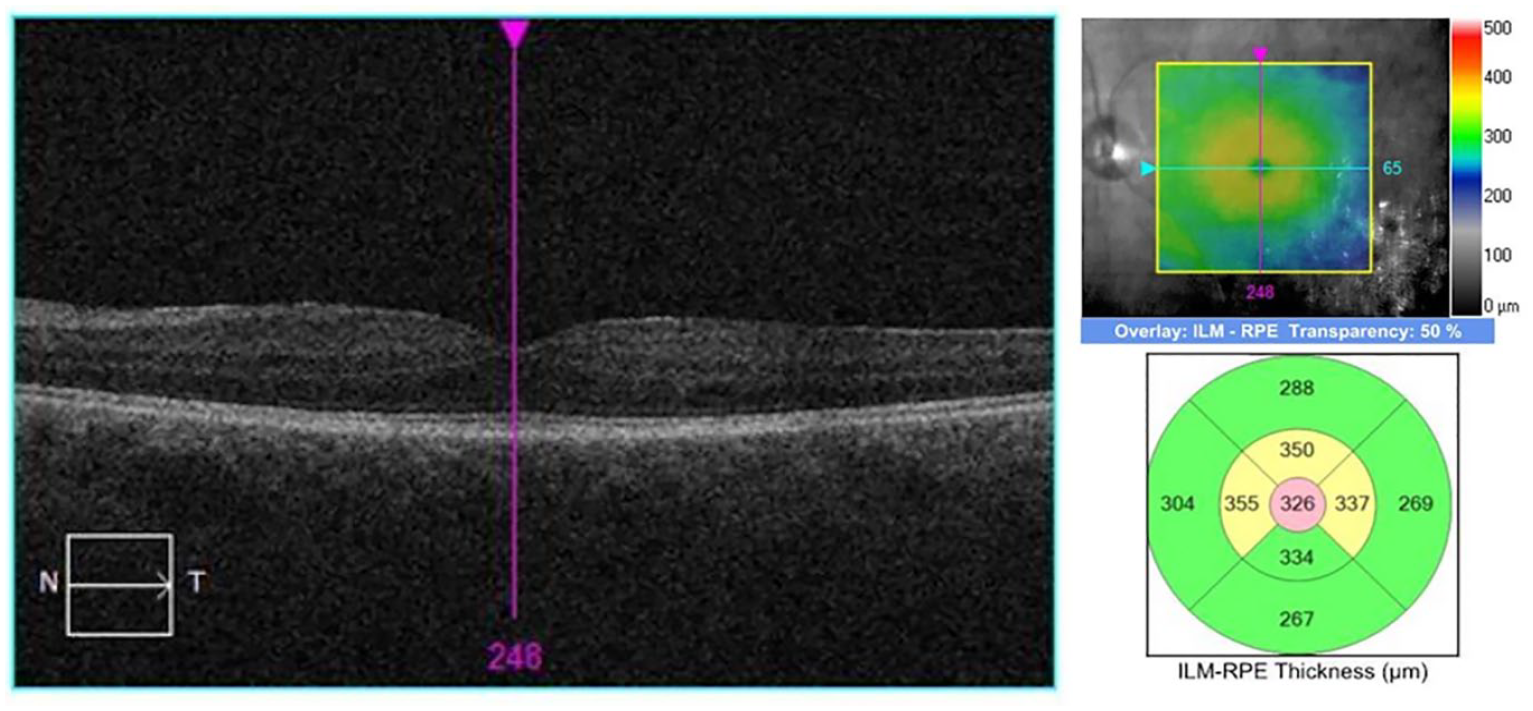
Optical coherence tomography of the left eye 1 month after cessation of ponesimod shows resolution of macular edema.
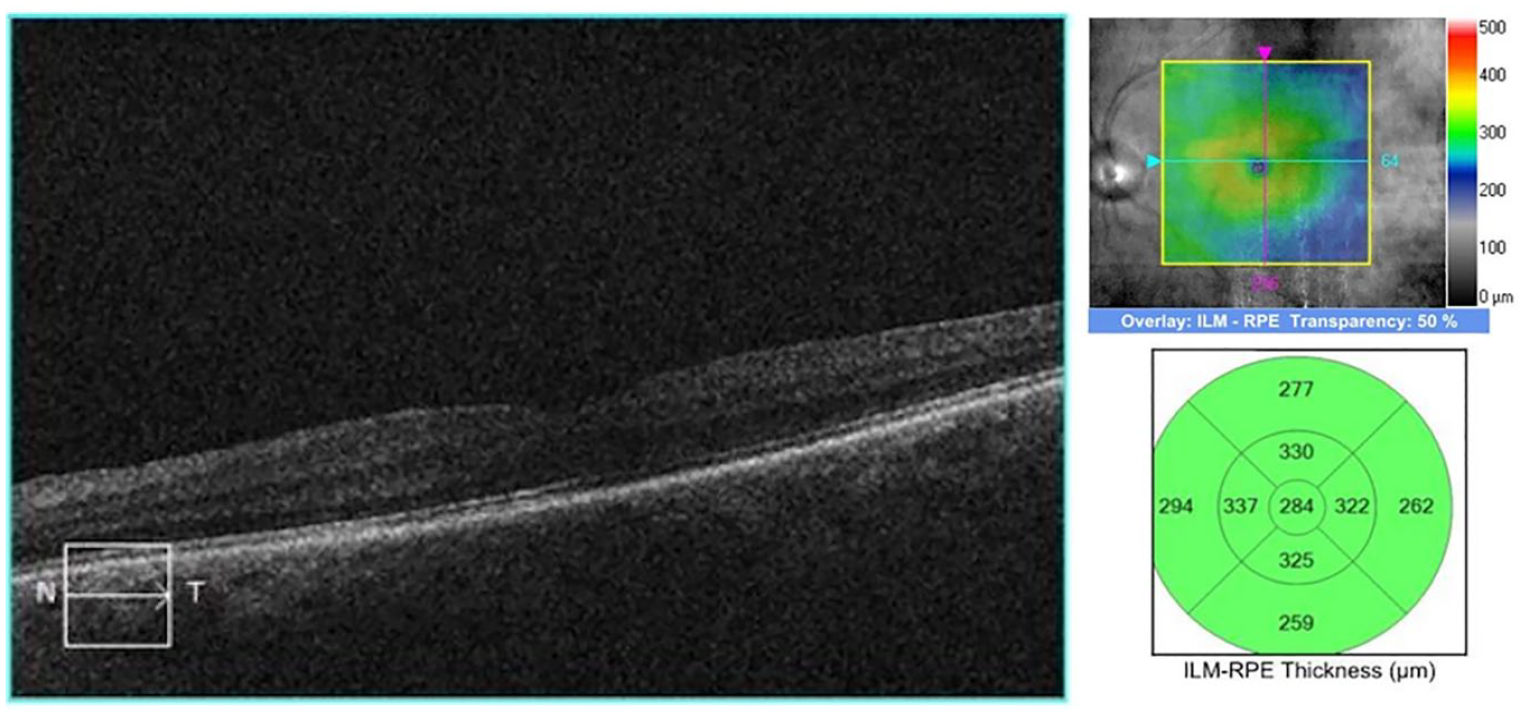
Optical coherence tomography of the left eye at most recent follow-up shows no recurrence of macular edema.
Conclusions
Ponesimod has been implicated in new-onset macular edema, henceforth termed ponesimod-associated macular-edema (PAME). It was first noted in a phase 2b core study of patients with RRMS, in which PAME was diagnosed in 4 patients. 9 Further instances of PAME were noted during the OPTIMUM phase III trial, in which PAME was diagnosed in 5 patients, constituting approximately 0.9% of the study’s treatment arm. 5
At present, no mechanism for PAME has been elucidated; however, its pathophysiology may be similar to that of FAME. Fingolimod, an S1P analogue, exerts a lymphocytic-trafficking blockade through an S1PR agonism. 8 Physiologically, S1P forms a concentration gradient used by immune cells in localizing to inflammation sites. 10 S1P analogues flood the existing gradient, resulting in lymphocytic cell stasis, and minimize inflammation, thereby aiding in symptomatic resolution. However, increased concentration of S1P analogue (ie, fingolimod and ponesimod) results in uncontrolled agonism of S1P receptors, which are expressed systemically.3,4 Agonism of S1P1R and S1P3R, in particular, has been implicated in angiogenesis, vascular tone, endothelial barrier regulation, and cell-junction preservation.4,6 This loss of appropriate cell adhesion likely disrupts the blood–retinal barrier, prompting vascular edema. 6 Although bilateral involvement would be expected from this systemic medication, our patient developed PAME in her left eye only.
Although CME can cause vision loss, PAME is reversible if diagnosed early and managed appropriately. Once PAME has been identified, ponesimod should be discontinued, if possible. Ponesimod’s relatively short half-life and rapid excretion make disease relapse a significant concern. Therefore, special consideration should be taken to consult the patient’s neurologist if modifying the patient’s MS treatment regimen is deemed necessary. 11
Although the discontinuation of ponesimod is likely sufficient to reverse early stages of PAME, patients may benefit from additional medical therapy. We believe PAME can be managed similarly to FAME, given the similarity between the compounds and the pathophysiology. Although there is no consensus on FAME management, supportive therapies—namely topical anti-inflammatory medications, corticosteroids, carbonic-anhydrase inhibitors, and antivascular endothelial growth factor (anti-VEGF) treatment—have been shown to aid resolution in small-scale studies.6,8
We show that PAME can be successfully managed by discontinuing ponesimod and providing supportive medical care with topical nonsteroidal anti-inflammatory drugs (NSAIDs) and corticosteroids. Our patient showed improvement after initiation of topical therapy, with subsequent worsening of the edema as the corticosteroid drops were tapered. This suggests that the topical therapy was contributing to the resolution of PAME. It is difficult to fully ascertain the role of topical therapy in our patient because the ponesimod was discontinued while the drops were simultaneously increased.
Given our patient was asymptomatic, we forwent more invasive management with subconjunctival corticosteroids, intravitreal corticosteroids, or anti-VEGF therapies. Previous studies of FAME, which is physiologically similar to PAME, have shown prompt resolution after sub-Tenon corticosteroid injection, a treatment that can be considered in the setting of refractory or difficult-to-treat macular edema.6,8 An alternate approach to treating our asymptomatic patient could have been to closely monitor her while continuing the ponesimod. An intervention could have then been made with topical medication, a sub-Tenon corticosteroid, and/or cessation of ponesimod if the PAME worsened or she developed visual symptoms.
Eye care providers should be aware of the potential complications of ponesimod use. Patients beginning ponesimod therapy should receive a pretreatment ophthalmic evaluation, including macular OCT scans as well as regular monitoring during the treatment course—a suggestion maintained by the US Food and Drug Administration for the less S1P1R-selective fingolimod and recommended by Janssen’s ponesimod prescribing information.8,11 Patients should further be informed that any painless vision loss during treatment requires urgent evaluation by their ophthalmologist. If macular edema is noted, transitioning patients from ponesimod treatment and starting supportive topical therapy with corticosteroids and NSAIDs is an effective PAME management algorithm.
Footnotes
Ethical Approval
This case report was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information were performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
Written informed consent was not required for this case report as no individually identifiable images or information are reported.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
