Abstract
Introduction
Although uveal melanoma is the most common intraocular tumor of the eye, it is rare in the overall population, with an age-adjusted incidence of 5.1 cases per million. 1 The effectiveness of local treatment control is typically good; however, uveal melanoma has a high mortality rate regardless of treatment type due to metastasis. After treatment, 50% of patients will develop metastasis, with a survival time after metastasis averaging 6 months to 1 year. 2
Many retrospective studies have identified prognostic risk factors of uveal melanoma in physical characteristics, such as tumor size, location, and histological characteristics. 3 More recently, genetic expression profiling (GEP) and preferentially expressed antigen in melanoma (PRAME) are commonly used as prognostic markers. These genetic tests can routinely be obtained during brachytherapy or laser ablation surgical treatment and requires only a small sample obtained by fine-needle aspiration.4 –6
GEP results are categorized into 2 classes. Class 1 confers a good prognosis, as it describes a tumor that rarely metastasizes. Class 1 can be further categorized as class 1A to show a low metastatic risk or class 1B to define an intermediate metastatic risk. Class 2 presents a worse prognosis because it describes a high risk for metastasis. 7
We report a unique case of an aggressive, recurrent uveal melanoma with scleral extension through a previous fine-needle biopsy site that had a change in GEP class. To our knowledge, a similar case has not been previously reported or studied.
Case Report
A 45-year-old Black man presented to the Ocular Oncology Clinic for a second opinion regarding regrowth of a previously treated choroidal melanoma. Two years prior to presentation, the patient was diagnosed with an inferior–temporal choroidal melanoma that had a basal diameter of 13.1 mm and maximum thickness of 11.5 mm. He was treated with iodine-125 plaque brachytherapy, and a fine-needle biopsy for GEP and PRAME (Castle Biosciences) was performed.
Biopsy results revealed a class 1A tumor and PRAME-negative genetics. Baseline abdominal magnetic resonance imaging and chest x-ray findings were negative for metastasis. The patient developed toxic tumor syndrome and was lost to follow-up for 9 months.
He presented again to the same ocular oncologist and was found to have neovascularization of the iris without glaucoma, white cataract, and a focal retinal detachment (RD). As the patient was comfortable, the patient elected for observation and treatment with topical steroids. Ultrasound biomicroscopy (UBM) showed no growth of the tumor, which was smaller than at presentation prior to treatment, with a basal diameter of 15.6 mm and thickness of 10.3 mm. The tumor was regressed and well-treated. For the next few months, examination findings were stable and serial UBMs did not change.
Approximately 6 months later, the patient started noticing darkening of the inferior sclera and presented to our clinic for a second opinion. The patient admitted that he did not follow up as scheduled with the previous ocular oncologist because he did not want surgery.
On examination, the patient had no light perception vision, a protruding pigmented lesion just inferior to the limbus (Figure 1), neovascular glaucoma, hyphema, and a white cataract obscuring the view posteriorly. UBM showed a solid collar-button lesion with a thickness of 10.2 mm and basal diameter of 14.0 mm as well as an extraocular extension, vitreous hemorrhage, and a total RD.
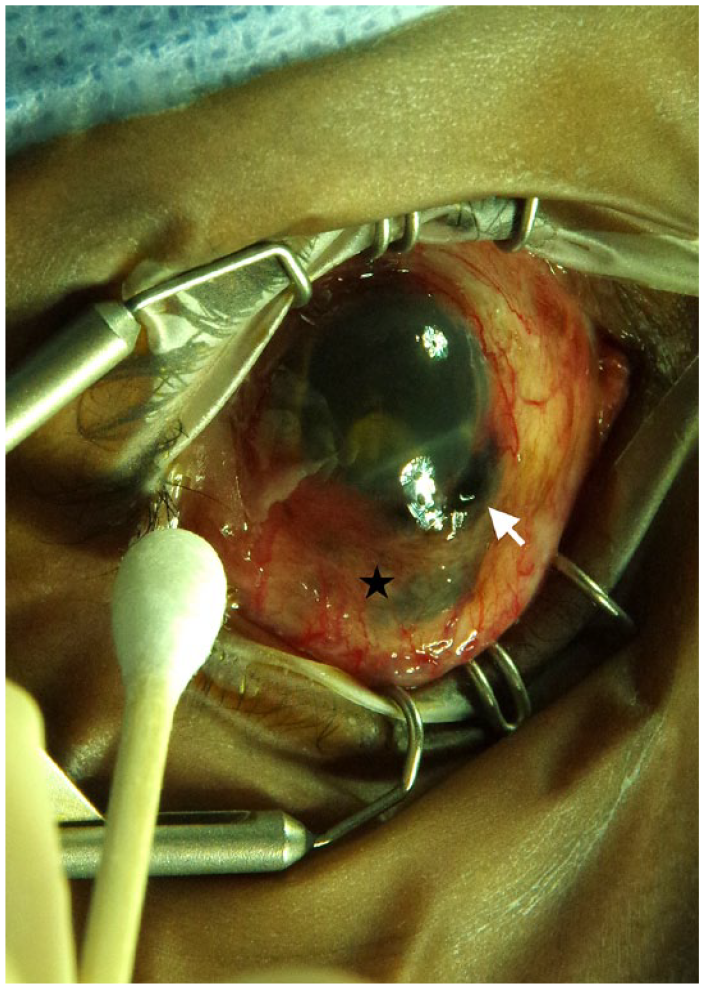
Extraocular extension of the uveal melanoma (white arrow) at the inferior limbus, likely at the site of previous fine-needle biopsy. Black star designates an area of scleral thinning.
After an extensive discussion, the patient elected for enucleation. Pathology of the specimen confirmed melanoma arising from a melanocytoma involving the iris, ciliary body, and choroid with gross extraocular extension posterior to the sclerocorneal limbus (Figure 2). At time of enucleation, a fine-needle biopsy was obtained for genetics. As there was new aggressive growth of the tumor, the case was discussed with Castle Biosciences and subsequent GEP analysis from the new biopsy of the tumor was requested. Repeat GEP analysis on the same tumor at different timepoints is not usually performed because GEP is thought to be a static measurement of tumor prognosis. Although the authors requested a second PRAME test, this was left to the discretion of Castle Biosciences and was not obtained. Castle Biosciences agreed to GEP analysis, and the results showed that the tumor that was previously classified as 1A was now classified as having 1B genetics.
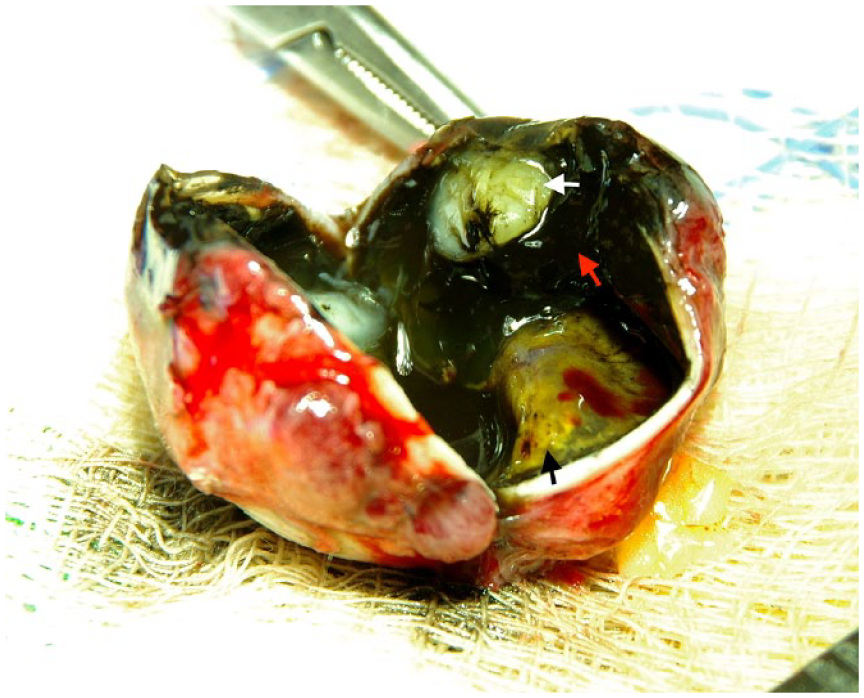
Gross pathology specimen with the globe cut partially to expose its contents. Uveal melanoma (black arrow), vitreous hemorrhage (red arrow), and a dense white cataract (white arrow) are shown.
Conclusions
This case presents interesting and unique findings that to our knowledge have not been previously reported. Three key points of the case are (1) aggressive recurrence of a previously treated and stable melanoma, (2) likely extraocular extension of the tumor at the previous biopsy site, and (3) a change in GEP class.
The patient had a melanoma previously treated with iodine-125 brachytherapy that was stable for little more than 1½ years, after which the tumor had aggressive, rapid regrowth. The Collaborative Ocular Melanoma Study (COMS) demonstrated that the risk for local treatment failure after iodine-125 brachytherapy after 5 years was relatively rare at 10.3% 8 ; however, the maximum tumor height in the COMS study was 10.0 mm and the patient’s initial tumor height was 11.5 mm. Subsequent retrospective studies reported local failure anywhere from 0% to 19% up to 3 years after brachytherapy of uveal melanomas with a tumor height up to 25.0 mm, with failure most common in large tumors and tumors close to the optic nerve.9 –11
Although failure is uncommon, it appears that most recurrence will occur within the first years after treatment. From 5 to 15 years, the annualized incidence risk for regrowth was 1.9% per year in one study. 12 The present case highlights the importance of continued follow-up with a thorough examination and serial ultrasonography at a close interval after brachytherapy. At present, there are no guidelines on follow-up after uveal melanoma treatment; however, we suggest close follow-up of 3 to 4 months early on with interval extensions no longer than 6 months.
The site of extrascleral extension had frank melanoma protruding from an anterior scleral area, which could represent protrusion through an area of postradiation scleral necrosis or protrusion through the previous fine-needle biopsy site. A 25-gauge needle is commonly used to obtain the biopsy transsclerally, and this area of weakness underlying the tumor may be an easy site for potential extension.
Smaller gauge needles have been considered, and Klofas et al 13 reported a series that showed a 27-gauge needle provided adequate sampling for prognostic testing but that needles larger than 27-gauge should be considered for cellular cytology. It may be beneficial to switch to a 27-gauge needle for genetic testing to minimize the wound size created during a biopsy. Furthermore, Burris et al 14 reported 2 cases of extrascleral extension of melanoma at sites of previous scleral manipulation: one at a previous fine-needle biopsy site and another at a previous trabeculectomy site. Although this is not common, it brings to light an important concept that extraocular extension via previous surgical incisions can occur and that this area should be examined at each subsequent visit.
Last, but possibly most important, is that subsequent GEP was performed and showed a change in the tumor class, which has not previously been reported or studied. In the initial study by Field et al, 4 GEP testing was found to be highly sensitive and is likely a more sensitive prognostic test in heterogenous tumors than chromosomal analysis. The authors showed that tumors with as little as 25% of mutations categorized as class 2 tumor cells were classified as class 2 based on their assay.
In a retrospective study by Augsburger et al, 15 biopsy samples from 2 sites of the same tumor showed that 11.3% of tumors had clearly discordant GEP analysis. As this was a large tumor, the change in GEP could be a result of discordance rather than evolution of genetics. More studies should be conducted to understand discordant class analysis and how it plays a role clinically. In addition, with how aggressively the tumor grew one could wonder whether this was a new separate tumor that grew, even though on pathology only a single solitary tumor was identified. Future studies should be performed in which subsequent GEP testing is done to see whether all tumors can have a GEP class change or whether a class change occurs due to the new growth of the tumor.
Although it has been shown that GEP analysis can be obtained after brachytherapy and or proton irradiation, there have not been studies showing the effect of radiation on GEP. 16 In the present case, the tumor changed from class 1A to class 1B, which portends a slightly worse prognosis. Retrospective studies have shown that there may not be a difference between metastasis rates of class 1A and class 1B tumors.17,18 In addition, PRAME was not repeated. Because it is independent of GEP class, a change in PRAME is likely a stronger suggestion of genetic class switching and likely has more clinical significance. 4 Future studies assessing the stability of PRAME should also be conducted. More important, this case may indicate that patients who failed primary therapy or who have a tumor with a change in its physical characteristics should undergo a new GEP and PRAME analysis of the tumor.
This case brings up many interesting questions and concerns that ultimately future studies will help elucidate. Important key points are that (1) tumor recurrence is rare but would occur within the first few years after treatment, (2) areas of previous scleral manipulation can be a site of extrascleral extension, (3) GEP class signatures can change within the same tumor, and (4) any tumor regrowth likely warrants subsequent GEP and PRAME testing.
Footnotes
Ethical Approval
This case report was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information were performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
Informed consent was obtained prior to performing the procedure, including permission for publication of all photographs and images included herein.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Murray is the Editor-in-Chief of the Journal of VitreoRetinal Diseases. All other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
