Abstract
Keywords
Introduction
Central retinal artery occlusion (CRAO) is a severe ocular condition with an estimated incidence of 1.3 per 100 000. 1 Characterized by acute painless monocular vision loss, this disease typically manifests with retinal whitening and a foveal cherry red spot. 2 Macular optical coherence tomography (OCT) shows a thickened, edematous inner retina with hyperreflectivity. 3 This is followed by atrophy of the neurosensory retina, reflecting permanent ischemic damage. 3 With no universally accepted means of treatment, CRAO is associated with irreversible vision loss.
Approximately 2.5% to 31.6% of CRAOs are further complicated by ocular neovascularization (ONV).4 –6 This is driven by proangiogenic factor expression in the setting of acute retinal hypoxia.6,7 Presentations of ONV include neovascular disc, neovascularization elsewhere, and neovascular iris (NVI). 7 Neovascularization of the iridocorneal angle is the preceding event for neovascular glaucoma (NVG), which represents an ophthalmic emergency.7,8
The timing of ONV onset has been reported 1 to 3 months after the initial CRAO.6,7,9 –11 Previous retrospective studies have also identified diabetes mellitus (DM) type 2, chronic kidney disease (CKD), and glaucoma as risk factors for post-CRAO ONV.9,11 However, to our knowledge no data describing the visual impact of this secondary complication have been published.
The aim of this retrospective cohort study was to model the visual prognosis of eyes with post-CRAO ONV using the time-based incidence of total blindness. Total blindness was defined as a best-corrected visual acuity (BCVA) of no light perception (NLP).
Methods
Ethical Approval
Institutional review board approval for this retrospective study was granted by Ochsner Medical Center (protocol #2022.85) and the Louisiana State University Health Sciences Center (protocol #4912). Informed consent was exempted for this study because it was a retrospective review with no identifiable patient information and/or images.
Patient Selection
The institutional electronic medical record was queried using ICD-9 and ICD-10 codes to identify patients diagnosed with CRAO between January 2012 and December 2022. This included patients seen in the emergency department, inpatient setting, and outpatient ophthalmology clinic. Inclusion criteria consisted of patients aged 18 years and older, diagnosed with CRAO via a dilated fundus examination and macular OCT, and with documented ONV after CRAO on manual chart review. Excluded were patients with alternative etiologies of ONV in the ipsilateral eye (including proliferative diabetic retinopathy, previous and/or concomitant central or branch retinal vein occlusion, exudative age-related macular degeneration, ocular ischemic syndrome, and/or sickle cell retinopathy) and those who had received panretinal photocoagulation (PRP) or antivascular endothelial growth factor (anti-VEGF) therapy prior to CRAO in the involved eye. 11 Also excluded were patients who were lost to follow-up after the initial detection of ONV and those with inadequate medical records resulting in an unknown date of ONV onset, unknown presenting and/or final BCVA, or unknown medical and/or surgical management.
Data Collection
Baseline clinical information included age, sex, race, phakic status, and laterality of the involved eye. Medical comorbidities recorded were hypertension, hyperlipidemia, DM, coronary artery disease, valvular heart disease, peripheral vascular disease, previous stroke, CKD, hemodialysis dependence, autoimmune and/or vasculitic disease, and smoking status.7,9 –11 Data pertaining to the initial CRAO consisted of the date of onset, presenting BCVA, etiology, and any diagnosis of temporal arteritis as defined by the 2022 American College of Rheumatology criteria. 12 Data related to secondary ONV included the time from the initial CRAO, clinical presentation of ONV, management (consisting of PRP, intravitreal anti-VEGF therapy and/or glaucoma surgical procedures), presenting BCVA, and total follow-up duration. The primary outcome measure was the final BCVA, with the time to first-documented NLP status where applicable. Secondary outcome measures included intractable eye pain, enucleation/evisceration, and/or the development of phthisis bulbi in the affected eye.
Statistical Analysis
Continuous data were reported using mean values with the SD. Categorical variables were reported as frequencies. Demographic and clinical factors between patients who reached NLP status and those who did not were compared using the Student t test (for continuous data) and the chi-square or Fisher exact test as applicable (for categorical data). The proportion of total blindness at the final follow-up was calculated. Kaplan-Meier reverse survival analysis was performed with NLP status as the event of interest using the Greenwood formula to derive 95% confidence intervals (CIs). 13 All tests were performed at a significance threshold of P < .05 using SPSS software (version 21, SPSS Inc, IBM Corp).
Results
Study Population
Of 345 patients with CRAO, 50 were identified with secondary ONV. Thirty-four eyes met the final inclusion criteria (Figure 1 and Table 1); the mean age was 72.0 ± 11.8 years, and the mean follow-up was 27.6 ± 29.1 months. Table 2 displays the demographic and comorbidity data. No cases in the ONV cohort were shown to be arteritic on the basis of a temporal artery biopsy. Eighteen cases (52.9%) were classified as thromboembolic based on the presence of retinal emboli, ipsilateral carotid artery disease, concomitant cerebrovascular incident, and/or echocardiographic thrombi. One CRAO case was attributed to severe iron-deficiency anemia, and the remainder (44.1%) were of unconfirmed nonarteritic (presumably also thromboembolic) etiology. 14
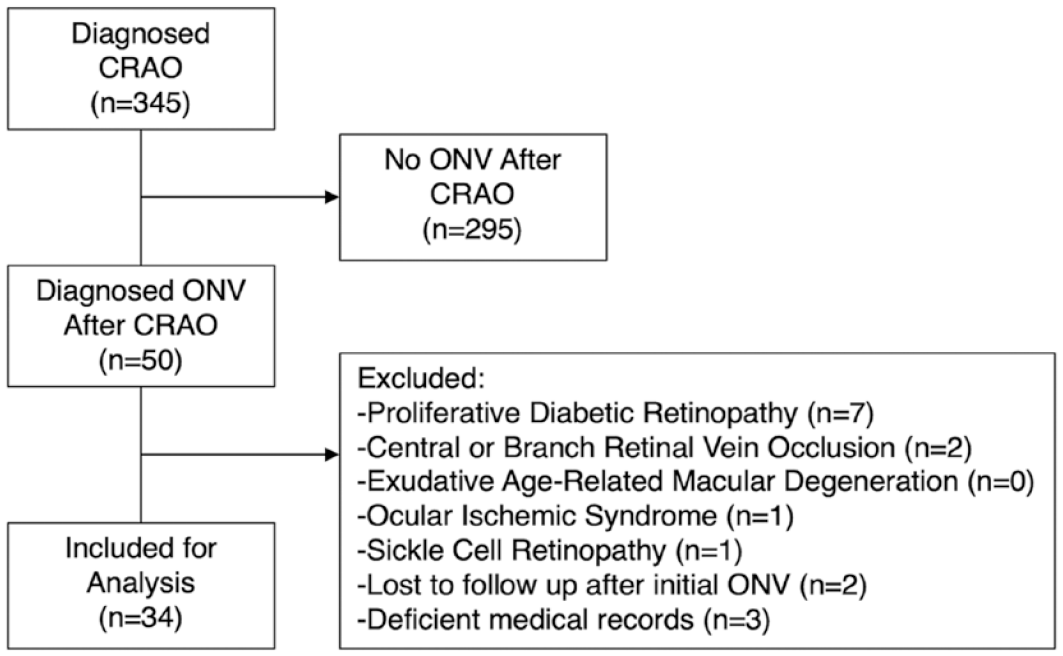
Patient selection flowchart.
Initial Clinical Presentation of Ocular Neovascularization.
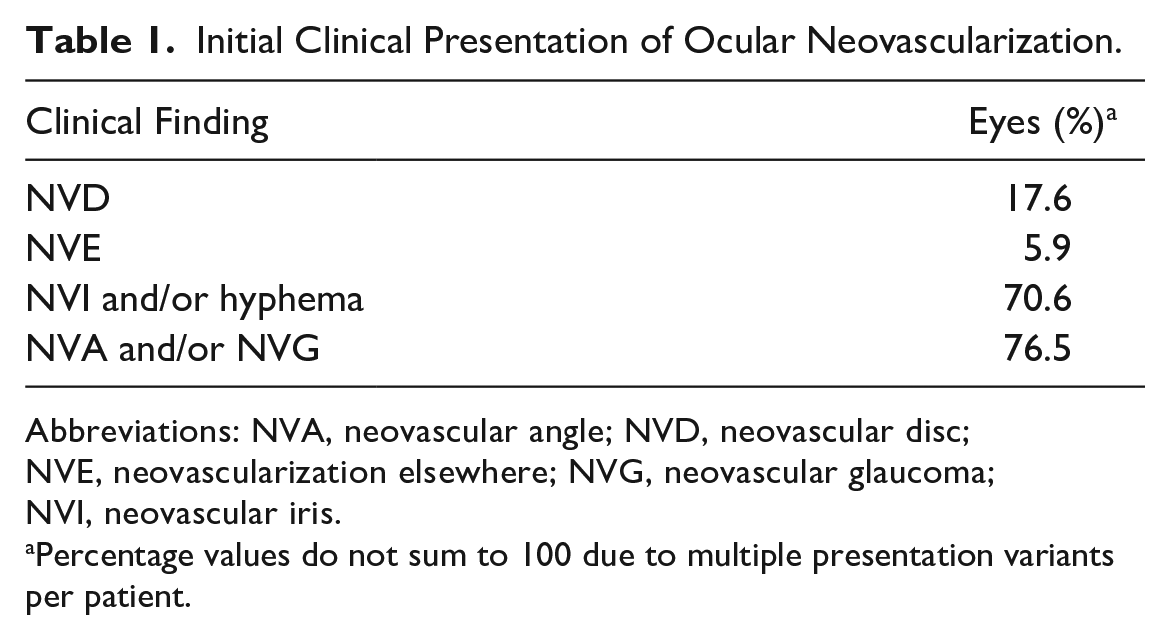
Abbreviations: NVA, neovascular angle; NVD, neovascular disc; NVE, neovascularization elsewhere; NVG, neovascular glaucoma; NVI, neovascular iris.
Percentage values do not sum to 100 due to multiple presentation variants per patient.
Baseline Demographic and Clinical Factors.

Abbreviation: CRAO, central retinal artery occlusion.
The mean time to ONV presentation was 3.3 ± 5.8 months from the initial CRAO. PRP was performed after ONV detection in 70.6% of patients. Intravitreal anti-VEGF therapy with bevacizumab 1.25 mg was used in 76.5% of cases, with a mean of 2.8 ± 5.6 injections per patient. Glaucoma drainage implant surgery or transscleral cyclophotocoagulation was performed in 32.4% of patients at a mean of 4.8 ± 6.9 months from the time of ONV presentation.
Total Blindness Incidence
Kaplan-Meier event analysis demonstrated a cumulative total blindness incidence of 25.1% (95% CI, 20.0%-30.2%) 4 months, 29.1% (95% CI, 22.4%-35.8%) 12 months, and 49.4% (95% CI, 27.2%-71.6%) 24 months from ONV onset among patients with CRAO (Figure 2). The mean time to total blindness was 12.5 ± 16.3 months from ONV onset. The incidence of total blindness among patients without ONV was 11.6% (95% CI, 10.8%-12.4%) 24 months and 18.5% (95% CI, 15.9%-21.1%) 60 months after the initial CRAO. Figure 3 shows the proportion of patients in the ONV cohort at each BCVA level. An overall shift toward lower BCVA values was noted from the time of ONV onset to the final follow-up.
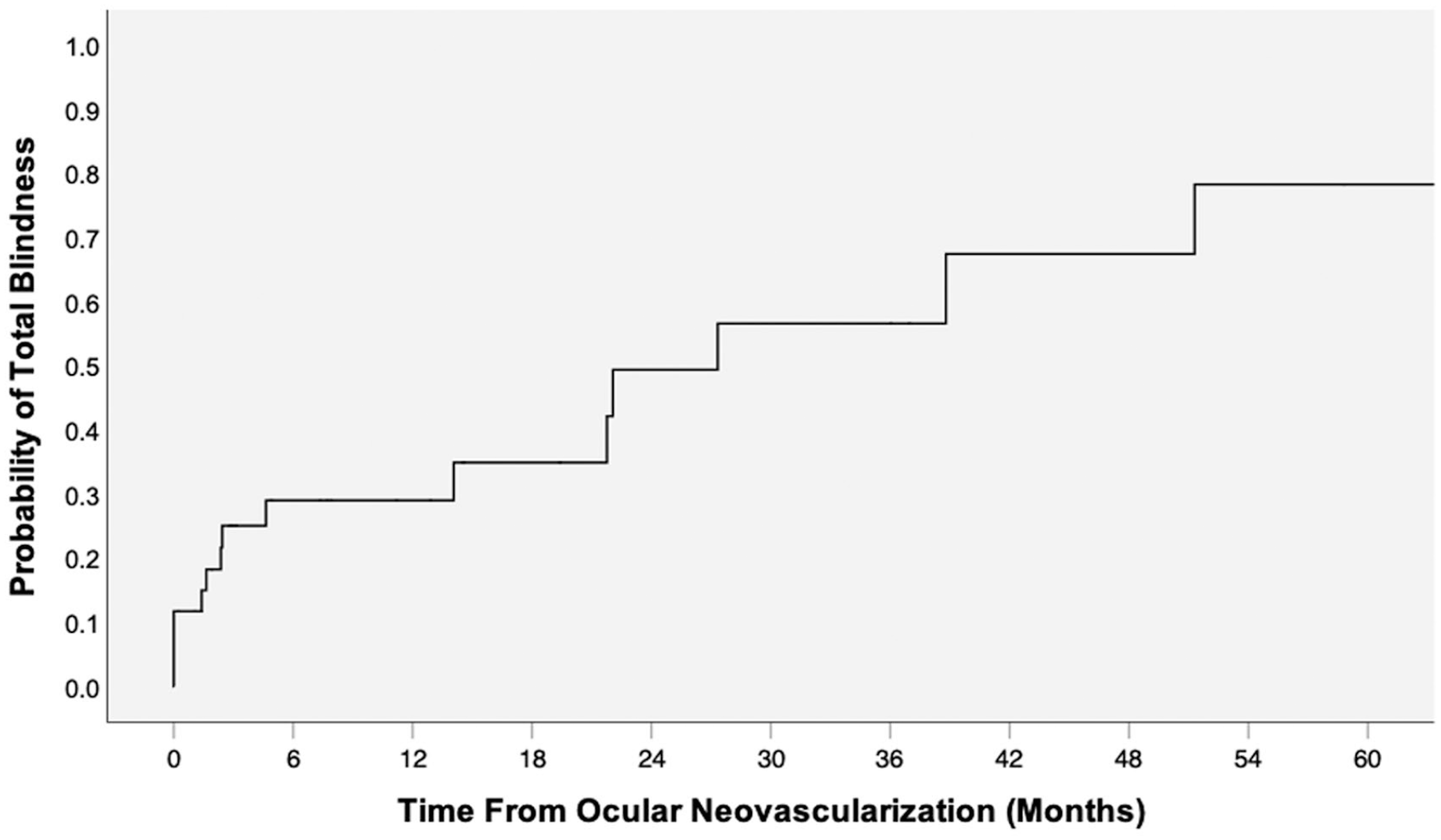
Time-based incidence of total blindness.
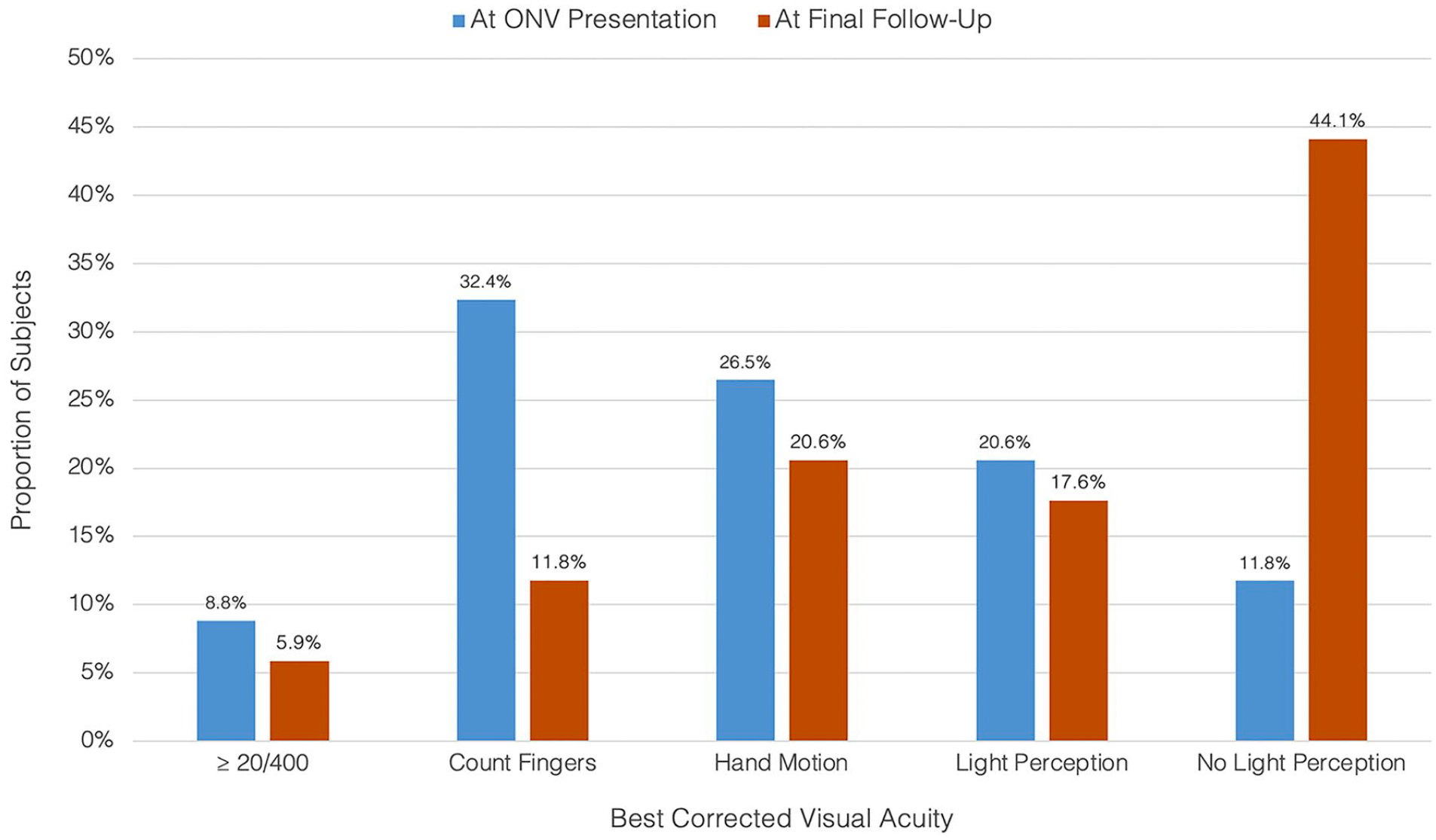
Visual acuity (VA) distribution at the onset of ocular neovascularization (ONV) vs at the final follow-up. The proportion of patients in the ONV cohort at each visual acuity (VA) level is shown at the time of ONV onset (left bars) and at the final follow-up (right bars). The distribution trend shows a shift toward lower VA values at the final follow-up.
Secondary Outcomes
At the final follow-up, the incidence of intractable eye pain was 23.5% among patients with post-CRAO ONV. Of these, 12.5% underwent eventual enucleation/evisceration (representing 2.9% of the total ONV cohort). No eye in the study cohort developed phthisis. Fifty percent of eyes with chronic pain had NLP vision. The incidence of chronic eye pain was lower among patients who had glaucoma surgery, although this was not statistically significant (9.1% vs 30.4%; P = .228).
Predictors of Total Blindness
There were no statistically significant differences (eg, age, sex, race, phakic status, and medical comorbidities) between patients with eyes reaching NLP status vs non-NLP status at the final follow-up, with the exception of laterality (73.3% right-sided vs 31.6%; P = .016). Glaucoma surgery was not associated with a significantly decreased incidence of NLP (36.4% vs 47.8%; P = .715). However, it was associated with a prolonged interval from ONV onset to NLP status (34.9 ± 6.5 months vs 4.4 ± 7.1 months; P < .001) (Table 3).
Visual Acuity and Procedural Details for Patients Receiving Glaucoma Surgery.
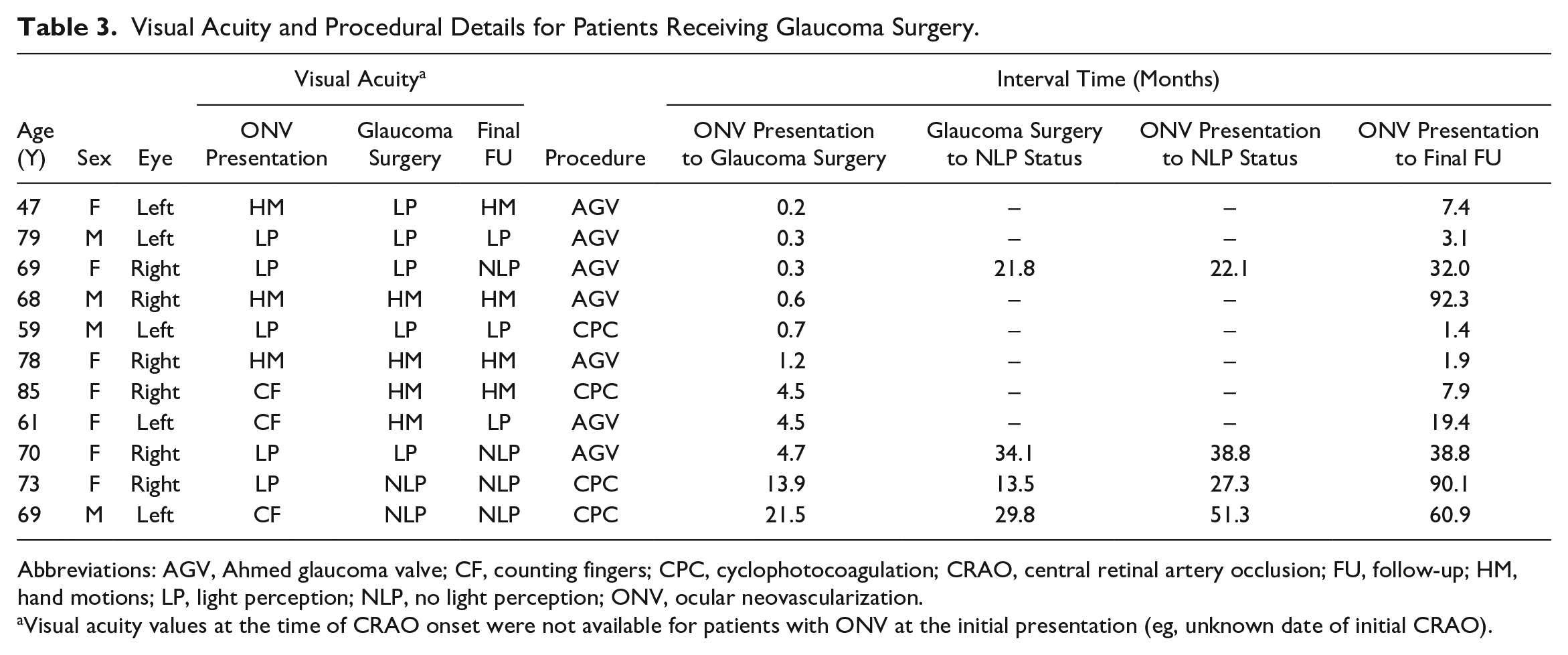
Abbreviations: AGV, Ahmed glaucoma valve; CF, counting fingers; CPC, cyclophotocoagulation; CRAO, central retinal artery occlusion; FU, follow-up; HM, hand motions; LP, light perception; NLP, no light perception; ONV, ocular neovascularization.
Visual acuity values at the time of CRAO onset were not available for patients with ONV at the initial presentation (eg, unknown date of initial CRAO).
Conclusions
To our knowledge, this represents the largest series of visual outcome data showing the clinical impact of post-CRAO ONV. Jung et al 10 found a 41.7% incidence of final NLP status among 12 patients with post-CRAO ONV, while Tanke et al 7 described a corresponding incidence of 21.4%. However, the timing of total blindness in both studies was not reported. In our cohort, 49.4% of included eyes eventually reached NLP status 24 months from ONV onset after CRAO. All cases of total blindness occurred within 5 years of ONV onset, with 53.3% taking place within the first 4 months. These findings underscore the importance of early follow-up after CRAO and provide empiric data to counsel patients about the potential visual consequences of undetected ONV.
NLP status represents a key clinical endpoint in CRAO, where up to 71.5% of patients have a final VA of 20/400 or worse after the initial ischemic episode. 15 Vision-related quality of life (QoL) has previously been shown to correlate with VA in the better-seeing eye. 16 However, Brown et al 17 recently found unilateral NLP status to be an independent predictor for decreased QoL regardless of VA in the better-seeing eye. Using the time-tradeoff method, the authors reported decreased utility scores among patients with NLP status compared to a matched control cohort (0.69 vs 0.80; P < .001). Given this clinical relevance, we elected to use total blindness as the event of interest in our survival analysis of eyes with post-CRAO ONV. Considering the already-severe vision loss following CRAO, preservation of any remaining visual potential can have significant implications for functional capacity and QoL. As an alternative clinical endpoint, eyes that developed ONV after CRAO were found to have a 23.5% incidence of intractable pain. This represents another important proxy for QoL, as shown by Qazi et al 18 with the validation of the Ocular Pain Assessment Survey.
NVG is known to cause ischemia of the optic nerve head and retinal tissue, leading to blindness in up to 90% of cases. 8 This is commensurate with our findings, in which 93.3% of patients who eventually reached NLP status developed NVG at some point. As discussed by Tanke et al, 7 even this may be an underestimate given that gonioscopy was not performed in a standardized fashion during follow-up examinations. This emphasizes the 2-stage pathophysiology of NVG, in which symptomatic closed-angle glaucoma results from the contraction of fibrovascular tissue in the iridocorneal angle.8,19 However, obstruction of the trabecular meshwork by rubeotic vessel infiltration may still cause insidious intraocular pressure (IOP) elevation before this occurs.8,19 In our cohort, glaucoma surgery was not associated with a significant decrease in the incidence of NLP (36.4% vs 47.8%; P = .715). However, it was associated with a considerably prolonged time from ONV onset to NLP status (34.9 ± 6.5 months vs 4.4 ± 7.1 months; P < .001) (Table 3).
The findings of this study have 2 main implications for the management of eyes with post-CRAO ONV. First, long-term IOP control represents a key measure to delay the onset of total blindness by addressing both the open-angle and closed-angle manifestations of NVG. Second, consideration should be given to extending the interval for undilated gonioscopy to 3 to 4 months after the initial CRAO, even in asymptomatic patients with no apparent NVI. Among our cohort, the mean time to ONV onset was 3.3 ± 5.8 months after CRAO. As 76.5% of these cases presented with NVG (Table 1), the paradigm of post-CRAO NVG as typically occurring within 30 days (compared to the “90-day glaucoma” phenomenon after ischemic CRVO) should be revisited.20,21 As proposed by Jung et al, 10 fluorescein angiography (FA) to confirm reperfusion may be warranted, with prophylactic PRP as an option if this fails to occur.
Right-sided laterality was associated with the development of total blindness among patients with post-CRAO ONV. This asymmetric predisposition has been reported by Li et al 22 in the context of primary CRAO based on a risk analysis of 44 622 unilateral cases from the IRIS registry. The authors proposed that this might stem from the anatomy of the aortic arch, whereby blood from the left ventricle passes the brachiocephalic trunk before supplying the left common carotid artery. As such, it is possible that emboli entering the systemic circulation have a greater probability of reaching the right common carotid artery (a branch of the brachiocephalic trunk). Given the high proportion of thromboembolic CRAO in our cohort (52.9% to 97.1%), we hypothesize that subsequent development of NVG with resultant vision loss may reflect a continuing process of systemic embolization through the right common carotid artery, exacerbating the process of VEGF secretion in an already-ischemic eye following the initial CRAO. Alternatively, continued embolization may result in secondary CRAO without NVG, extinguishing what little vision remains in affected eyes. To our knowledge, this represents the first preliminary data linking laterality not just to event incidence but also to subsequently worse visual outcomes.
This study is subject to several limitations. As a retrospective analysis, the possibility of selection bias from loss to follow-up cannot be excluded. Second, this was a multicenter study across 3 urban academic medical centers. As such, demographic differences may have existed between the study population and the general population. However, the incidence and timing of ONV in our study align with those of previously published cohorts, lending credence to the generalizability of the findings. Third, ONV management was not strictly standardized and reflected the individual practice patterns of retina specialists across the 3 included sites. Fourth, the degree of retinal ischemia demonstrated by FA would likely be a useful prognostic factor of the risk for NVG and subsequent vision loss. Although this was not incorporated in the present study because of variable imaging protocols between treating specialists and a lack of access to previous imaging studies (resulting from adoption of a new image archival system at our institution), it represents an important consideration for follow-up studies. Finally, no statistically significant demographic or clinical risk factors for total blindness were identified in our cohort. However, this was likely a function of the relatively small sample. Despite this limitation, ours represents the largest reported cohort of patients with post-CRAO ONV to date. Future studies with a larger patient population should focus on total blindness risk factor detection, which may further refine surveillance and treatment recommendations for CRAO.
Footnotes
Authors’ Note
Presented in part at the Association for Research in Vision and Ophthalmology annual meeting, April 2023. The authors confirm that they retain all rights to the above work.
Ethical Approval
Institutional review board approval for this retrospective study was granted by Ochsner Medical Center (protocol #2022.85) and the Louisiana State University Health Sciences Center (protocol #4912).
Statement of Informed Consent
Per institutional policy, informed consent was exempted for this study, being a retrospective review with no identifiable patient information and/or images.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Mazzulla is a consultant to Allergan PLC, Alimera Sciences Inc, and Apellis Pharmaceuticals unrelated to this study. None of the other authors reported financial disclosures related or unrelated to this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
