Abstract
Introduction
West Nile virus (WNV) is a single-stranded RNA virus that is the leading cause of mosquito-borne disease in the continental United States. 1 The virus was first identified in Uganda in 1937 2 and has been prevalent in Africa, Europe, the Middle East, and central Asia. In more recent years, WNV has emerged in temperate regions of North America. The first 2 cases of WNV encephalitis in North America were identified in New York City in late August 1999 by an infectious disease physician in the borough of Queens. 2
Especially prominent in moist, temperate areas, the WNV had not yet established a major foothold in dry, desert regions such as Arizona. 3 However, in Arizona in 2010 there were 166 confirmed cases of probable WNV. 3 The next decade followed suit with comparable numbers of WNV infections, with 2020 having a remarkably low number of cases (n = 11), in part as a result of low rainfall and COVID-19 lockdowns. 3
In 2021, after a heavy monsoon season, Maricopa County alone witnessed 1417 confirmed and probable cases of WNV, resulting in 113 fatalities across the state of Arizona. 3 Arizona as a whole witnessed 1644 confirmed or probable cases of WNV. 3 With the development of a WNV vaccine for humans making slow progress and no ongoing clinical trials for West Nile–specific therapies, this uptick of WNV in Arizona, especially Maricopa County, is concerning. 4
With changing climate patterns worldwide, the risk for contracting WNV will likely increase. Warmer temperatures associated with climate change can increase the breeding and biting rates of mosquitoes and lengthen incubation times of the virus within a mosquito. 5 Paired with changing precipitation patterns and a higher frequency of extreme weather events, the distribution and abundance of mosquitoes that transmit WNV will continue to rise. Changes in climate also affect bird migration and breeding patterns, which may also contribute to viral spread. 6 Proximity to infected birds will also enhance disease rates. Outbreaks of WNV as seen in Arizona in 2021 may become commonplace if this issue is not tackled.
According to US Centers for Disease Control and Prevention (CDC) criteria, laboratory diagnosis is generally accomplished by testing serum or cerebrospinal fluid (CSF) to detect WNV-specific immunoglobulin M (IgM) antibodies. 7 WNV-specific IgM antibodies are usually detectable 3 to 8 days after the onset of illness and persist for 30 to 90 days, although longer persistence is possible. Therefore, positive IgM antibodies occasionally may reflect a past infection. If serum is collected within 8 days of illness onset, the absence of detectable virus-specific IgM does not rule out the diagnosis of WNV infection, and the test may have to be repeated on a later sample. All positive results obtained with these assays should be confirmed by neutralizing antibody testing of acute-phase and convalescent-phase serum specimens at a state public health laboratory or the CDC. The presence of IgM antibodies alone is only evidence of previous infection. 7
Plaque-reduction neutralization tests can also confirm acute infection by yielding a fourfold or greater change in WNV-specific neutralizing antibody titer between acute-phase and convalescent-phase serum samples collected 2 to 3 weeks apart. Viral cultures and tests to detect viral RNA (eg, reverse transcriptase-polymerase chain reaction) can be performed on serum, CSF, and tissue specimens that are collected early in the course of the illness and if results are positive, can confirm an infection. However, the likelihood of detecting a WNV infection through molecular testing is fairly low. 7
WNV can present in various ways, ranging from febrile illness to neuroinvasive diseases such as encephalitis or meningitis. Symptomatic disease in humans occurs in approximately one fifth of infected individuals. 8 Neuroinvasive disease is reported to occur in 1% of infected persons and becomes more common with increasing age. 9 From 1999 to 2019, nearly one half of all reported cases of WNV were neuroinvasive, affecting the brain or nervous system. 7 Ocular manifestations can occur and usually coincide with severe neuroinvasive disease. 7 WNV can lead to acute anterior uveitis, occlusive retinal vasculitis, chorioretinitis, optic neuritis, and diplopia. 10 Ophthalmic sequelae may result in permanent vision loss, which further highlights the need to develop a specific treatment and/or vaccine. As the climate changes, outbreaks of WNV are anticipated to worsen in frequency and severity, making it paramount to tackle this disease with improved preventive and treatment strategies.
Here, we present the patient characteristics, clinical features, and visual outcomes of 7 cases of WNV-related chorioretinitis in Arizona over the past decade.
Methods
A chart search for individuals with “chorioretinitis” and “West Nile” in any of their clinical notes was performed from April 1, 2012, to February 1, 2023. Patients were included if they carried a diagnosis of chorioretinitis and a molecularly confirmed diagnosis of WNV with positive serology for IgM WNV and/or positive CSF for IgM WNV. Patients were excluded if they did not carry a definitive diagnosis of WNV chorioretinitis (5 patients) and/or did not have baseline testing with fundus photographs, spectral-domain optical coherence tomography (SD-OCT), and fluorescein angiography (FA) (2 patients). Based on these criteria, 7 of 14 patients with initial clinic visits between July 2019 and June 2022 were included.
All 7 patients were seen after hospitalization. They were diagnosed with chorioretinal lesions and were seen in our outpatient retina clinics. To our knowledge based on available hospital records and patient history, none of our patients received a dilated eye examination during the acute event of WNV encephalitis. Fundus imaging included color fundus photography, FA, SD-OCT, and possibly indocyanine green. The imaging was performed on different machines based on the satellite retina clinic.
This study was exempted by the Advarra Institutional Review Board using the US Department of Health and Human Services regulations found at 45 CFR 46.104(d)(4).
Results
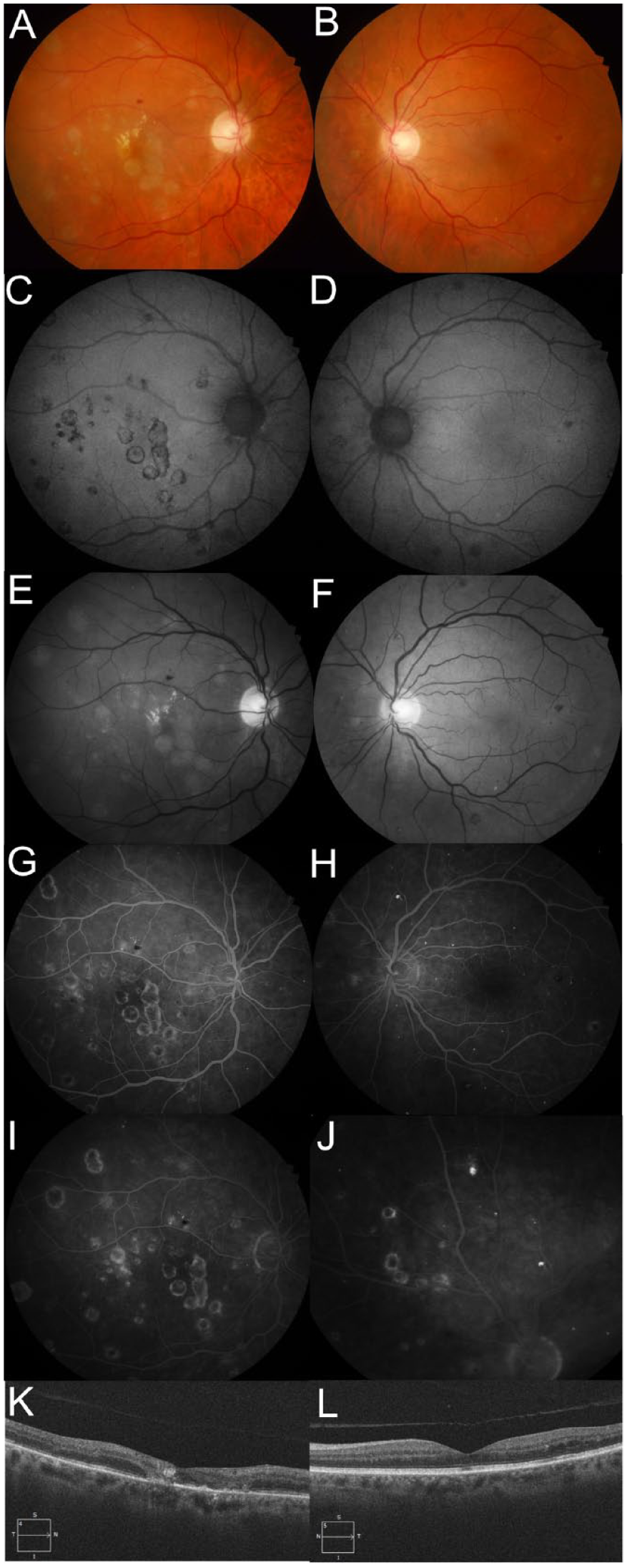
Table 1 summarizes the results. All 7 patients were referred from 2 weeks to 6 months after hospitalization for persistent visual changes. Patients were 59 years and older (range, 59-76; median, 71) and initially presented between July 2019 and June 2022. Five of 7 patients were men. Five of the 7 patients had a diagnosis of diabetes, and 1 patient had HIV with a nondetectable viral load.
Patient Characteristics, Cinical Features, and Visual Acuity Outcomes.
Abbreviations: DM, diabetes mellitus; FU, follow-up duration; HAART, highly active antiretroviral therapy.
The presenting visual acuity (VA) ranged from 20/20 to 20/40 (0 to 0. 3 logMAR; median, 0.3) in the better-seeing eye and 20/20 to 20/100 (0 to 0.7 logMAR; median, 0.5) in the worse-seeing eye. The final VA ranged from 20/20 to 20/40 (0 to 0.3 logMAR; median, 0.1) in the better-seeing eye and 20/20 to 20/70 (0 to 0.55 logMAR; median, 0.2) in the worse-seeing eye. The difference in VA improved an average of +0.1 logMAR in the better-seeing eye and +0.2 logMAR in the worse-seeing eye.
The time of follow-up from the initial visit to the final visit ranged from 1 to 24 months (median, 10 months). If there was an improvement in vision (5 patients), the improvement occurred within 1 to 2 months for the majority (4 of 5 patients). For 1 patient, the improvement could have been related to antivascular endothelial growth factor (anti-VEGF) injections to treat diabetic macular edema (DME).
The fovea was involved in 1 eye in 4 patients, and the macula was involved in at least 1 eye of all patients. Vitreous haze was present in 2 patients. None of the patients had anterior segment inflammation. Three patients used topical steroids. One patient (Case 2) was treated with anti-VEGF 2 months apart in 1 eye for DME.
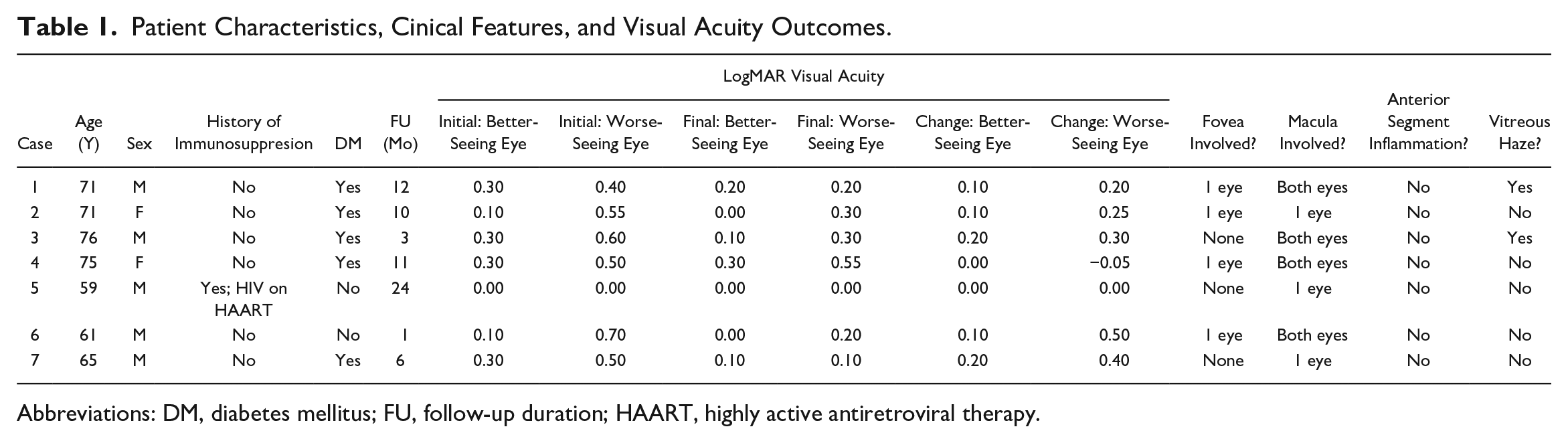
Classic nummular lesions in various stages were observed in many of the cases. Sometimes, lesions can be difficult to detect on examination. Case 3 serves as an example. A 76-year-old man presented with bilateral blurred vision for 2 weeks. His VA measured 20/80 OD and 20/40 OS. Pressures were normal in both eyes, and there was no anterior segment inflammation in either eye. Both eyes had 2+ vitreous haze (using Nussenblatt et al’s 11 standardized grading of vitreal inflammation). No discrete chorioretinal lesions were seen on examination or color photographs of both eyes (Figure 1, A and B). However, autofluorescence showed small chorioretinal lesions arranged in curvilinear streaks along the parafoveal vessels in both eyes (Figure 1, C and D). The small chorioretinal lesions showed hyperfluorescence with late staining on FA (Figure 1, E–H).
Case 3, initial visit. (A and B) Fundus photographs of the right eye and left eye, respectively, show a hazy view to the posterior pole along the inferior arcade resulting from vitritis and no obvious chorioretinal lesions. (C and D) Autofluorescence of the right eye and left eye, respectively, shows small chorioretinal lesions arranged in a curvilinear streak off superior and inferior parafoveal vessels. (E and F) Early fluorescein angiography (FA) of the right eye and left eye, respectively, at 1 minute shows more numerous small rings of hyperfluorescence along the superior and inferior parafoveal vessels than can be seen on other imaging, with more pronounced curvilinear streaks of small hyperfluorescent lesions in the left eye than in the right eye. (G and H) FA of late phase at 3 minutes for the right eye (G) and at 4 minutes for the left eye (H) shows late staining of the lesions with disc leakage.
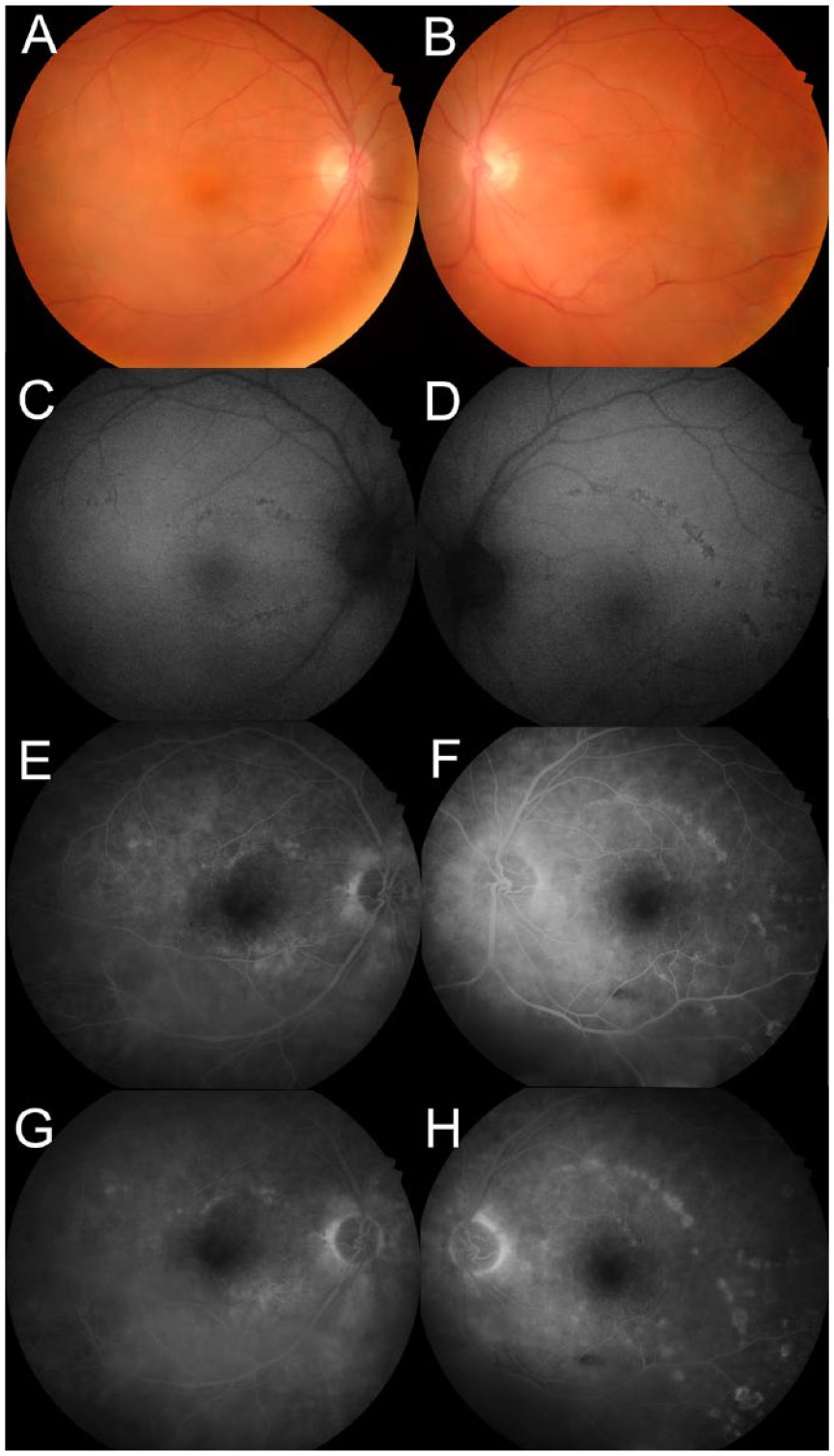
One month later, the patient’s VA improved to 20/40 OD and 20/25 OS with resolved vitreous haze bilaterally (Figure 2, A–D). The foveal centers in both eyes were spared (Figure 2, A–H).
Case 3, imaging 1 month after clinic presentation. (A–D) Nearly resolved vitreous haze in both eyes with persistent subtle chorioretinal scarring in both eyes. (E–H). Fluorescein angiography (FA) at 1 minute in the right eye (E) and left eye (F) shows persistent but lighter hyperfluorescence of the curvilinear chorioretinal scarring in the superior more than inferior parafoveal areas in both eyes. FA at 3 minutes 30 seconds in the right eye (G) and left eye (H) eyes shows late staining to a lesser degree than at the initial visit.
Conclusions
Vector-borne diseases such as WNV have been prevalent in tropical and subtropical environments and have expanded to other regions of the world in more recent years as the result of changes in climate and globalization. 12 WNV has spread to Arizona, and the state has recorded more than 1600 cases of confirmed or probable cases of WNV, the highest number seen in the state since 2005. 3
Although the majority of WNV illnesses manifest as subclinical infections or mild flu-like disease, some cases are neuroinvasive and can be lethal or leave an individual with significant morbidities, such as muscle paralysis and/or blindness. As a disease entity, a rise in WNV is important to the retina specialist given that nearly 80% of patients with neuroinvasive WNV will develop a multifocal chorioretinitis, the most common ocular manifestation of WNV. 13 Even though ocular involvement is self-limited, blindness can result from foveal involvement, choroidal neovascularization, vitreous hemorrhage, tractional retinal detachment, optic neuropathy, and/or retro-geniculate damage.
In our retina clinics, which serve patients from all parts of Arizona, a noticeable increase in the incidence of WNV chorioretinitis has been observed. Although referral patterns have not changed in our practice, the possibility of a referral bias cannot be excluded. On review of charts from April 2012 to February 2023, the majority of confirmed WNV cases with chorioretinitis were seen in the 4 years from July 2019 to June 2022. Only 1 of 8 patients who had a confirmed WNV chorioretinitis presented in 2012. (The 1 patient was excluded for inadequate baseline retina testing.) The uptrend in WNV chorioretinitis in our retina clinics reflects an overall rise in WNV outbreaks in Arizona in most recent years.
In this case series, all 7 patients had concomitant neuroinvasive encephalitis with hospitalization preceding clinical presentation. It is known that chorioretinitis can occur without coexisting meningoencephalitis; thus, the retina specialist should maintain high suspicion for WNV in the appropriate context of clinical retinal findings. Multimodal imaging with FA and fundus autofluorescence plays an important role in the diagnosis and in predicting the final visual prognosis, which depends on the level of macular involvement. Case 3, highlighted in Figures 1 and 2, emphasizes the critical role of multimodal imaging in some cases with subtle clinical features.
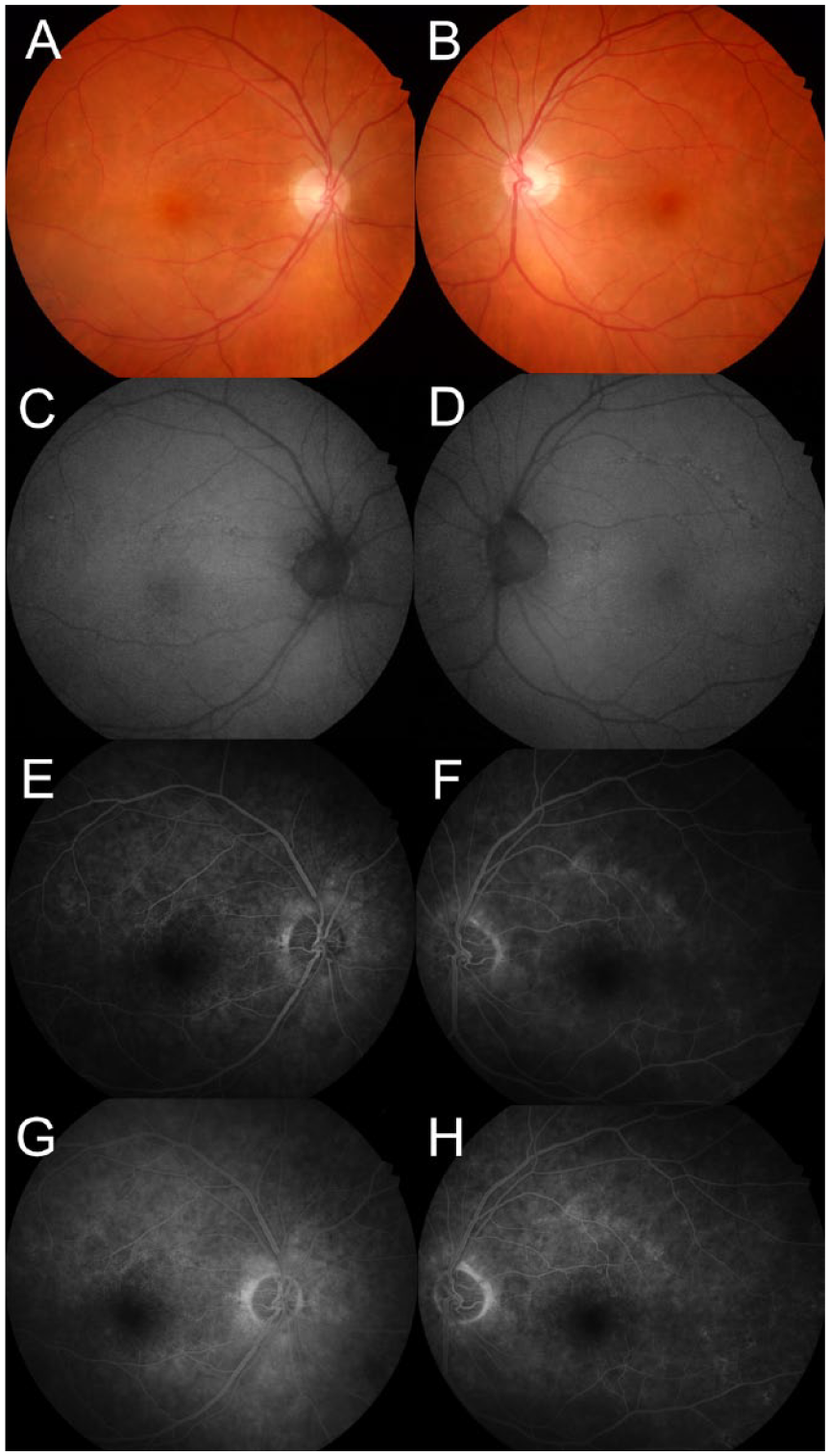
The pathogenesis of WNV chorioretinitis is still speculative. Some hypothesize neurotrophic spread along the nerve fiber layers and inner retina, which leads to the curvilinear pattern of lesions. 14 Other schools of thought hypothesize hematogenous spread resulting from deep retinal layer involvement on OCT, with sparing of the nerve fiber layer and inner retinal layers observed in cases of chorioretinitis without preceding or concomitant encephalitis. 15 Although the streak along the superior arcade in the left eye of Case 3 (Figure 1, D, F, and H) may be visualized as coursing along nerve fiber layers, other patterns of chorioretinal lesions such as the 2 confluent streaks tracking vertically across the horizontal raphe in the right eye of Case 2 (Figure 3) contradict the neurotrophic hypothesis and suggest a hematogenous process whereby unpredictable patchy involvement and perivascular patterns may emerge. The pathogenesis may involve a combination of the 2 processes; that is, neurotrophic and hematogenous.
Case 2. (A) Fundus photograph of the right eye shows multiple yellow–white chorioretinal lesions, some of which have coalesced into a vertical linear streak involving the foveal center macula. Others are found scattered in the macula and extending into the midperiphery in a perivascular pattern. Paracentral exudate and a couple of small intraretinal hemorrhages in the superior macula are seen. (B) Fundus photograph of the left eye shows a temporal macular intraretinal hemorrhage, a couple of yellow–white chorioretinal lesions nasal to the disc, and isolated lesions off the inferior arcade below the temporal macula. (C–F) Autofluorescence and red-free images emphasize the chorioretinal lesions, which show hyperautofluorescence surrounded by a ring of hypoautofluorscence in the right eye (shown in C). (G and H) Fluorescein angiography (FA) in the midphase at 30 seconds in the right eye and in the later phase at 1 minute in the left eye shows multiple nummular lesions with central hypofluorescence and a ring of hyperfluorescence. (I and J) FA of the right eye at 2 minutes (late phase) and an eccentric nasal photograph in the left eye at 6 minutes shows staining of the lesions. (K and L) An optical coherence tomography horizontal line scan through the foveal center of the right eye shows a thin retina, disorganized outer retinal layers, ellipsoid zone layer and retinal pigment epithelium disruption, and a focal area of inner retinal layer hyperreflectance correlating with the chorioretinal lesions on examination. The horizontal line scan through the central macula of the left eye shows no chorioretinal involvement but does show mild temporal intraretinal cystic fluid related to diabetes.
The literature has suggested diabetes mellitus (DM) is a risk factor for the development and severity of WNV chorioretinitis. 10 Interestingly, 5 of our 7 cases had a diagnosis of DM and 3 of these cases had lesions involving or abutting the foveal center in 1 eye. This finding further supports DM as a risk factor for the development of a more severe chorioretinitis. The patient with well-controlled HIV and undetectable viral loads had a milder degree of chorioretinitis with minimal macular involvement and maintained a VA of 20/20 OU. No articles were found in the literature on whether HIV modifies the risk for chorioretinitis development or severity; however, one may suspect immunodeficiency (ie, untreated HIV) can lead to more invasive disease. One epidemiologic study suggested that those immune to HIV through a C-C chemokine receptor type 5 mutation are more prone to be afflicted with symptomatic WNV infection. 16
Regarding treatment and final VA, although ocular manifestations are known to be self-limited, topical prednisolone or difluprednate was initiated in 3 cases that were thought to exhibit activity with or without vitreous haze. One cannot know whether VA improvement would have occurred to the same level without topical treatment. VA in the worse-seeing eye was still relatively good on the final visit (ranging from 20/20 to 20/70), even in eyes with foveal involvement. The duration of follow-up was dictated by the VA impact, provider decision-making, and patient adherence. The median duration of follow-up was 10 months from the initial visit to the final visit. Improvement in vision occurred within 1 to 2 months from the initial visit in 4 patients.
In conclusion, WNV chorioretinitis has become more prevalent in the past 4 years in our retina clinics across Arizona, which reflects the uptrend in suspected and confirmed systemic WNV cases across Arizona. If not lethal, neuroinvasive WNV could result in significant morbidity, including permanent vision loss from chorioretinal scarring or other neuroretinal complications.
Footnotes
Ethical Approval
This case report was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information was performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
Informed consent was not obtained for publication of the already de-identified retinal imaging acquired during clinic visits.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
