Abstract
Keywords
Introduction
Small-gauge vitrectomy with 27-, 25-, or 23-gauge instrumentation is the most common surgical approach to address macular diseases, including macular hole (MH), epiretinal membrane (ERM), and vitreomacular adhesion (VMA). Since the inception of small-gauge vitrectomy, the technology has progressed toward smaller instrumentation to limit surgical trauma, avoid the use of sutures, improve postoperative recovery and wound closure, and decrease postoperative complications.1 –12 Smaller instruments also present challenges of decreased fluid flow, increased flexibility, diminished lighting capability, and a decreased microforceps platform size, all of which potentially increase the complexity and length of surgical procedures.7 –9,13 –15
Results from the ASRS 2020 Preferences and Trends Membership Survey showed minimal use of 27-gauge vitrectomy since 2007 among surveyed surgical retina specialists. 16 Despite the potential safety benefits, the adoption of smaller 27-gauge vitrectomy over the more widely used larger 25-gauge vitrectomy has been slow, likely because of the perceived challenges associated with its use. Comparative investigation of 25-gauge and 27-gauge vitrectomy has been fairly limited, with mixed conclusions on whether smaller instruments increase case time.7 –9,12 –15,17,18
A major advantage of small-gauge vitrectomy is that the wound closures are often sutureless.6,19 Many techniques to facilitate wound closure have been described.12,17,20 –23 Conscientious wound-closure technique is important to avoid complications. We have extensive experience using both 25-gauge vitrectomy and 27-gauge vitrectomy and have used a systematic wound-closure approach to safely maximize sutureless closure. Smaller wounds close more readily without manipulation or sutures and represent a potential advantage over 27-gauge vitrectomy.
This study evaluated the effectiveness of small-gauge vitrectomy using 27-gauge instrumentation. This was performed by comparing the surgical duration, wound closure, and complication rate of 2 large consecutive cohorts having 25-gauge or 27-gauge vitrectomy for common indications.
Methods
Study Design
A single-center retrospective chart review was performed of consecutive patients who had 25-gauge or 27-gauge vitrectomy for MH, ERM, VMA, or a combination. Surgeries were performed between January 2013 and May 2015 in the 25-gauge group and between October 2014 and April 2017 in the 27-gauge group. Patients were excluded from analysis if they met any of the following criteria: combined surgery (cataract surgery, retinal detachment repair, glaucoma drainage implant, or intraocular lens exchange), surgery on platforms other than the Constellation (Alcon Laboratories, Inc), or previous pars plana or anterior vitrectomy. All surgeries were performed by the same surgeon (J.S.N.), who had more than 7 years of post-fellowship experience performing macular surgery before data collection. The study adhered to the tenets of the Declaration of Helsinki and received a waiver of authorization from the Adverra Institutional Review Board.
Patients
Data were collected at the nearest preoperative visit, intraoperatively, and at the 1-day postoperative visit. A complete medical, ophthalmic, and surgical history was recorded at every preoperative visit. Preoperative data were collected on age, sex, operative eye, lens status, indication for vitrectomy, best visual acuity (BVA), and intraocular pressure (IOP). Intraoperative data were obtained for the surgical duration (time between first trocar insertion and last trocar removal), wound-closure techniques, and procedural complications. Postoperative data were collected for the BVA, IOP, time to follow-up, and clinical course. The presence of postoperative hypotony (IOP ≤5 mm Hg), conjunctival chemosis, choroidal detachment, and wound leakage were also recorded.
All patients had a full ophthalmic examination at the preoperative and postoperative visits, including VA testing, tonometry, slitlamp microscopy, indirect ophthalmoscopy, and optical coherence tomography imaging. The BVA was evaluated using Snellen charts, checking both habitual corrections and pinhole vision, and converted to a logMAR notation for statistical analysis. The IOP was typically measured using a Tono-Pen. If the IOP was 6 mm Hg or lower or was unobtainable, Goldmann applanation tonometry was performed.
Surgical Technique
Each surgery was performed using a Constellation vitrectomy system. Trocars were inserted at a 30-degree angle through the conjunctiva and sclera, 3.5 to 4.0 mm from the limbus, depending on the lens status. The infusion was inserted in the inferotemporal trocar. With Biom visualization (Oculus Surgical Inc), a vitrectomy was performed. If the hyaloid was still attached, it was separated from the posterior pole using suction and gentle traction with the cutter. Triamcinolone was used to visualize the vitreous in some cases.
Vitrectomy was performed with an UltraVit cutter (Alcon Laboratories, Inc) using 5000 to 10 000 cpm, depending on the availability of the technology, with 0 to 650 mm Hg of suction. A complete vitrectomy was performed, removing as much of the peripheral vitreous as possible in all cases. All ERMs were peeled using an internal limiting membrane (ILM) forceps (Alcon Laboratories, Inc) under contact lens visualization (Bausch + Lomb). This was followed by peeling the central ILM with the aid of a highly diluted preparation of indocyanine green (ICG) (Diagnostic Green LLC) in dextrose water solvent. All macular holes underwent ICG-assisted ILM peeling in which the ILM was removed from the central macula surrounding the hole using the ILM forceps under contact lens visualization. The retinal periphery was inspected for breaks or other significant pathology, which were addressed with a laser, if indicated. Gas tamponade with isoexpansive sulfur hexafluoride or perfluoropropane was used if indicated after air–fluid exchange with a soft-tipped cannula.
Wound Closure
All wounds were closed using a systematic approach. The trocars were gently removed while gentle side pressure was placed on the wound to aid closure. On removal of the trocar, the wound was assessed for closure. If the wound was not closed, a series of sequential maneuvers was used until the wound was sealed. Closure was determined by visualizing apposition of the wound edges without reflux of gas or fluid during gentle palpation to ensure a normotensive IOP. The order of closure techniques was the same in both groups and included (1) 45 seconds of pressure perpendicular to the wound tract with a utility forceps, (2) trocar counterincision (perpendicular insertion of the trocar through the wound tract to appose wound edges), (3) placement of a 7-0 polyglactin transconjunctival suture, and (4) conjunctival cutdown (7-0 polyglactin suture closure of the scleral and conjunctival wounds). If visualization prevented a counterincision or transconjunctival suture, these maneuvers were deferred and the scleral wound was sutured after conjunctival cutdown.
Statistical Analysis
Statistical analyses were performed using SPSS software (version 21, SPSS, Inc). Values were listed as the mean ± SD or as a percentage. The chi-square test for independent variables was used to compare qualitative data. P values less than 0.05 were considered to represent statistically significant differences.
Results
The review identified 250 eyes of 250 patients meeting the criteria for analysis. The cohort consisted of 2 groups: 125 consecutive 25-gauge surgeries and 125 consecutive 27-gauge surgeries. Table 1 shows the general characteristics of both groups. The 2 groups did not significantly differ in any baseline characteristic.
Demographic Characteristics and Baseline Features of the 25-Gauge and-27 Gauge Vitrectomy Cohorts.
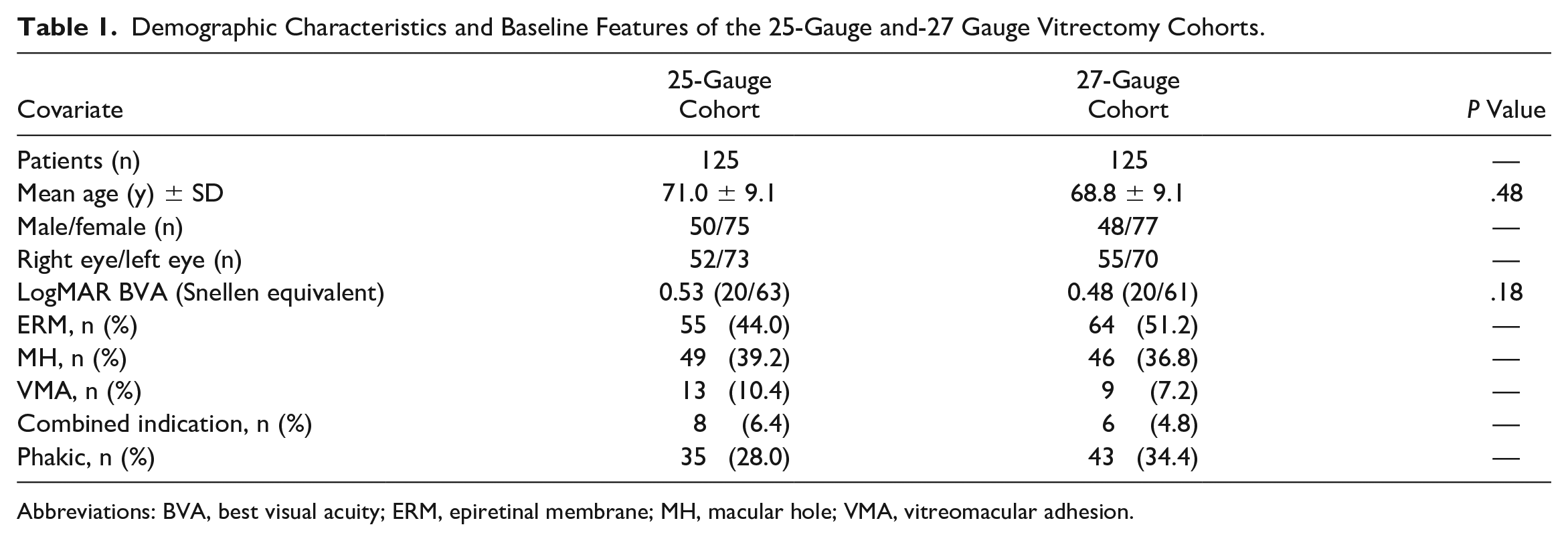
Abbreviations: BVA, best visual acuity; ERM, epiretinal membrane; MH, macular hole; VMA, vitreomacular adhesion.
Intraoperative Findings
Tables 2 and 3 show the intraoperative findings. The number of valved trocars used differed significantly between the groups (P < .01). There were 7 intraoperative retinal tears in the 25-gauge group and none in the 27-gauge group. No cases of choroidal detachment or cataract formation were observed intraoperatively in either group. The surgical duration was slightly longer in the 25-gauge group than in the 27-gauge group, although the difference was not statistically significant (P = .07). The surgical duration in valved 25-gauge cases was shorter than in nonvalved 25-gauge cases; however, the difference was not statistically significant (P = .09) (Table 3).
Intraoperative Findings for 25-Gauge and 27-Gauge Vitrectomy.
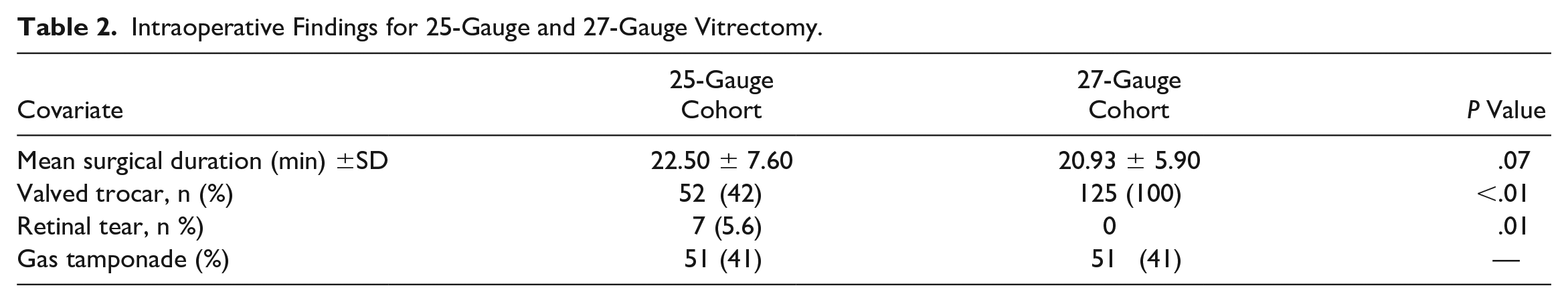
Comparison of Surgical Duration for Valved and Nonvalved 25-Gauge Vitrectomy.

Table 4 shows the intraoperative wound-closure findings. In the 25-gauge group, 364 supratemporal (ST), 121 supranasal (SN), and 125 inferotemporal (IT) of the 375 scleral wounds closed without additional manipulation. Three ST wounds required pressure for 45 seconds, 3 wounds required counterincision, and 1 wound required suture with cutdown. Two SN wounds required pressure for 45 seconds, and 2 wounds required a counterincision. No IT wounds required additional manipulation. In the 27-gauge group, 369 scleral wounds (122 ST, 122 SN, and 125 IT) closed without intervention. Two ST wounds and 2 SN wounds required pressure for 45 seconds, and 1 ST wound and 1 SN wound required a counterincision. No IT wound required additional manipulation. Chi-square analysis showed no statistical difference between the immediate closure rates between the 25-gauge group and 27-gauge group (P = .22).
Intraoperative Wound Closure for 25-Gauge and 27-Gauge Vitrectomy.
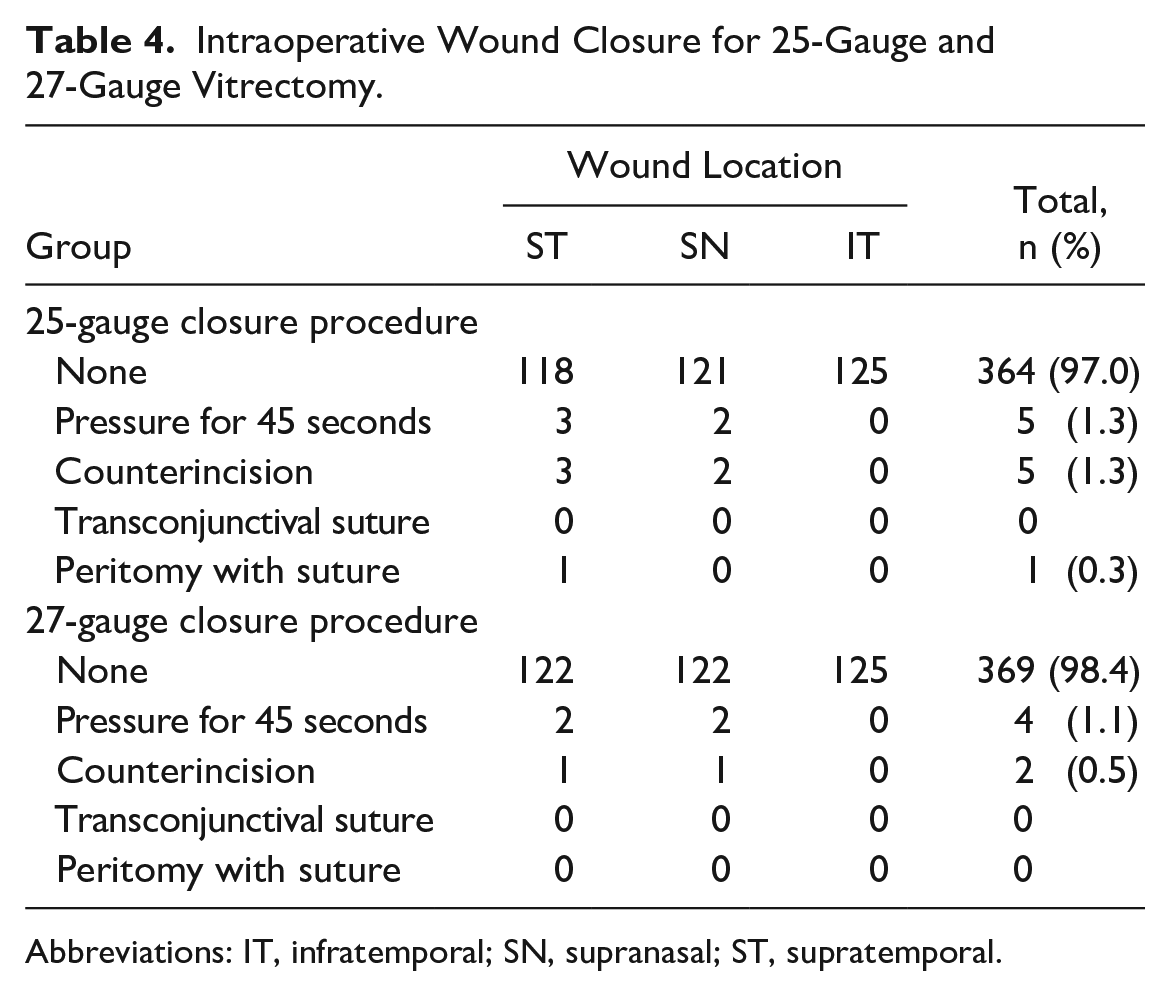
Abbreviations: IT, infratemporal; SN, supranasal; ST, supratemporal.
Postoperative Findings
Table 5 shows the findings 1 day postoperatively. The most common finding was hypotony followed by chemosis, with no significant difference between the 25-gauge group and 27-gauge group. No cases of subconjunctival gas formation, choroidal detachment, or wound leakage were found in either group. The mean IOP was 13.3 ± 5.4 mm Hg in the 25-gauge group and 13.8 ± 6.0 mm Hg in the 27-gauge group (P = .46). Three cases in the 25-gauge group did not have a recorded IOP at the 1-day postoperative follow-up visit.
Comparison of Postoperative Day 1 Findings for 25-Gauge and 27-Gauge Vitrectomy.
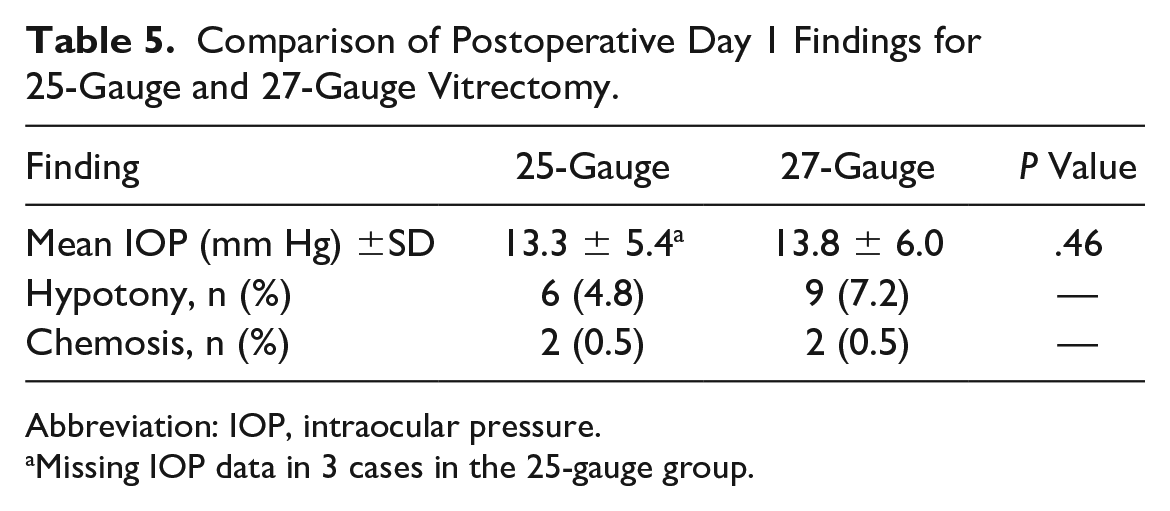
Abbreviation: IOP, intraocular pressure.
Missing IOP data in 3 cases in the 25-gauge group.
Conclusions
In 2010, Oshima et al 24 first investigated the clinical safety and practicality of 27-gauge instrumentation for small-gauge vitrectomy. The authors found no 1-day postoperative complications (hypotony, conjunctival swelling, subconjunctival gas formation, or scleral wound sealing) with 27-gauge vitrectomy, suggesting potential advantages over 25-gauge vitrectomy. Romano et al 12 and Rizzo et al 17 subsequently reported that the effectiveness and efficacy of 25-gauge vitrectomy and 27-gauge vitrectomy are equal. The findings of Romano et al 12 and Otsuka et al 14 also showed no significant increase in surgical duration with 27-gauge instruments. However, in multiple studies,7 –9,17,18 the surgical duration of 27-gauge vitrectomy was significantly longer than that of 25-gauge vitrectomy. It is imperative that larger studies be performed to validate previous findings.
Our study of small-gauge vitrectomy found that the 27-gauge and 25-gauge procedures were both safe and effective in addressing common macular pathology. Despite findings in previous reports, our experience showed that 27-gauge vitrectomy did not take longer than 25-gauge vitrectomy. Our analysis found that the duration of 27-gauge surgery was actually shorter than that of 25-gauge surgery, although the difference was not statistically significant. Nonvalved trocars were used in 73 of the 25-gauge cases. It was theorized that nonvalved trocars could have made surgery slightly more inefficient. Although the surgical duration for nonvalved cases was longer than that for valved cases, the difference was not statistically significant. Retinal tears were not associated with valved trocars.
The 7 retinal tears in the 25-gauge group required treatment and extended surgical duration. The mean case duration for the 7 cases was 30 minutes 17 seconds compared with 22 minutes 3 seconds for the 118 cases without retina breaks. The mean duration for the entire 125 cases in the 25-gauge cohort was 22 minutes 30 seconds. Dugel et al 25 reported greater involvement of surrounding tissues with larger instrumentation resulting from the longer membrane attraction distances and greater “sphere of influence.” It is possible that larger gauge instrumentation contributed to the retinal breaks. Six of the 7 breaks occurred with nonvalved trocars; however, this was not found to be statistically significant (P = .18). There is limited evidence to suggest intraoperative complications are more common with nonvalved trocars than with valved trocars. However, fluid reflux through unobstructed trocars could have increased vitreous traction on the peripheral retina, leading to retinal breaks.26 –28
Wound closure was more complicated in the 25-gauge cases, which could account for the slightly increased surgical duration for these cases. The smaller lumina and light fibers, increased instrument flexibility, and smaller forceps platforms in 27-gauge vitrectomy did not extend the surgical duration over that of 25-gauge vitrectomy.
The cases reported in this large series spanned 4 years 3 months, and improvements in technology and surgeon skill could have affected our results. Linear regression of case duration over time showed a slight decrease in time (25-gauge: y = −7E − 06x + 0.0184 [R2 = 0.0656]; 27-gauge: y = −3E − 06x + 0.0155 [R2 = 0.0177]) over the time period in both cohorts (Figure 1). There was a slightly greater decrease in case duration in the 25-gauge cohort than in the 27-gauge cohort. The development of higher cut-rate technology coinciding with the cases performed in this series could have contributed to improved cutter performance over time. Vitreous cut rates were not recorded for our cases. Surgeon skill likely increased over time; however, the impact cannot be independently parsed out from the data collected.
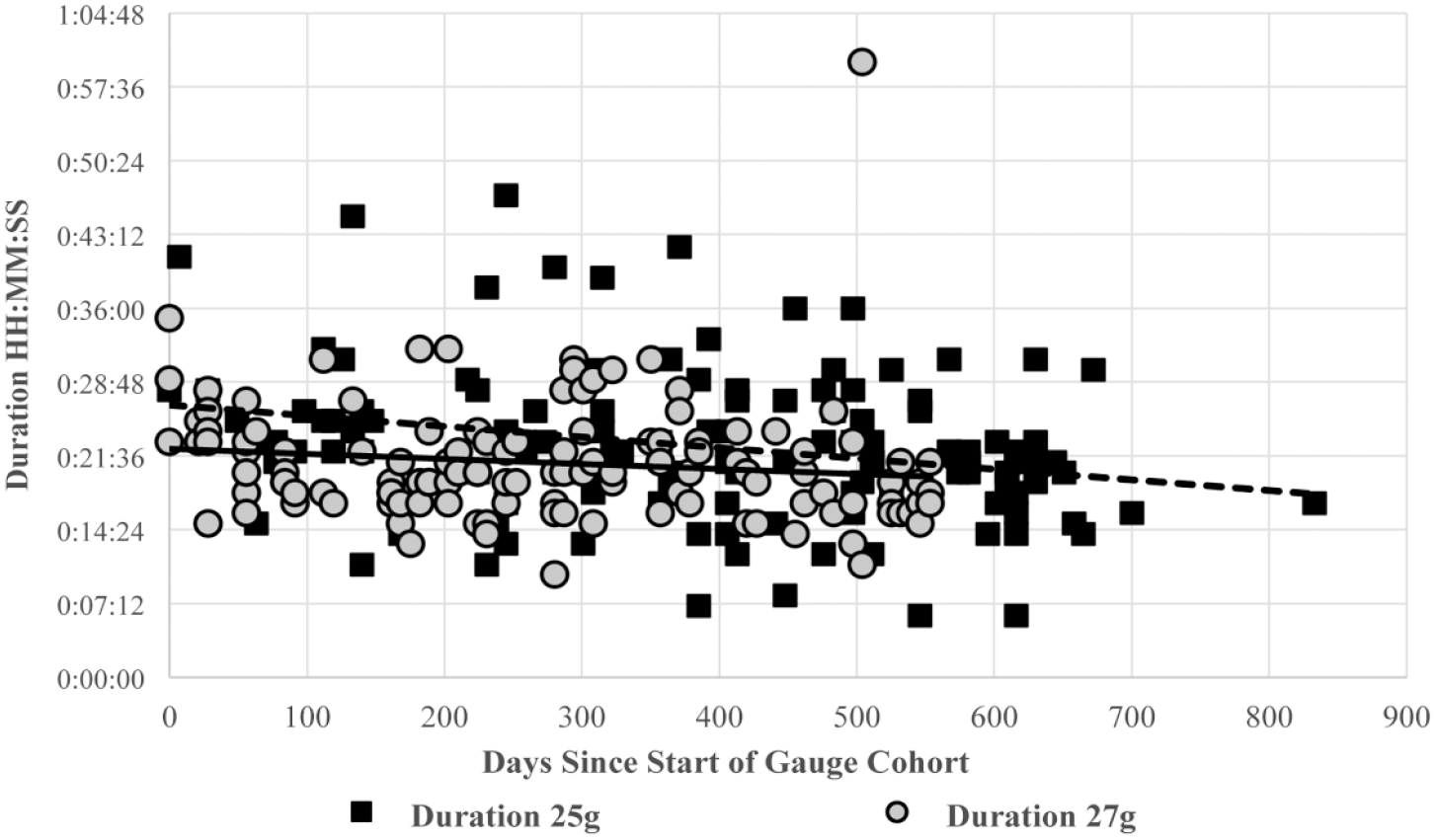
Scatterplot of case duration over time in the 25-gauge cohort and 27-gauge cohort shows a slight decrease in case duration over time in each cohort.
Although immediate closure was common for most wounds in both groups, it was significantly more common in the 27-gauge cases. Using our systematic approach, the larger 25-gauge wounds required more procedures in which additional closure was necessary. Hypotony on postoperative day 1 was slightly more common in the 27-gauge cohort. Postoperative hypotony is typically self-resolving and occurs in between 0% and 25% of sutureless vitrectomy cases.29,30 No wound leaks were noted with Seidel testing in hypotonus eyes. Hypotony was transient in all eyes and without deleterious sequelae. There was also an insignificant increase in IOP with 27-gauge vitrectomy than with 25-gauge vitrectomy at the 1-day postoperative visit. We hypothesize this was due to tighter wound seals with smaller gauge vitrectomy. These findings show that a higher rate of immediate wound closure is a significant benefit of using smaller surgical instruments.
Our experience has shown there are potential benefits to using smaller 27-gauge instruments over 25-gauge instruments in small-gauge vitrectomy for MH, ERM, and VMA. These benefits do not have to be realized at the cost of increased surgical time or serious complications.
Footnotes
Ethical Approval
This study was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information was performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
Informed consent was obtained before the procedure was performed, including permission for publication of all photographs and images included herein.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: research support from Alexion, Clearside, Gemeni Therapeutics, Genentech, Gyroscope Therapeutics, Iveric Bio, Kodiak Scientific, Novartis, NovoNordisk, Regeneron, and RegenxBio; consultant to Alcon, Apellis, Genentech, Iveric Bio, and Regeneron; speaker’s bureau participation for Apellis, Genentech, and Iveric Bio.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
