Abstract
Introduction
According to the US Centers for Disease Control and Prevention, the prevalence of diabetic retinopathy (DR) cases is projected to triple by 2050, afflicting 16 million US adults (from 5.5 million in 2005). 1 Prompt detection of DR is vital in preventing blindness. Although guidelines recommend annual DR screening, only 50% to 60% of patients with diabetes meet this recommendation. 2
Lack of DR screening is associated with low socioeconomic status, limited health literacy, barriers to medical care (eg, scarcity of providers, underinsured or uninsured status), and racial or ethnic minority status (in particular among African American, Hispanic, and Native American patients). 3 Primary care physicians also vary significantly in their adherence to DR screening guidelines. In addition, geography plays a role in DR screening adherence. Compared with metropolitan areas, rural locations have lower rates of DR screening as a result of the communities’ distance from providers, lower education and income, and lack of insurance. 4 Even in urban areas, especially inner cities and public housing communities, several studies have found limited access to appropriate eye care.5,6
Timely, consistent screening is essential because the risk for blindness increases as DR progresses to later stages. 7 Teleophthalmology screening for DR is becoming increasingly popular as an alternative screening method to the standard dilated eye examination by an eye care provider because it bypasses many existing barriers, such as limited access to eye doctors, the burden of additional in-person doctor visits, and pupil dilation requiring transportation assistance. 8 A population-based study observed that 82.2% of patients not receiving an annual dilated eye examination were seen by a primary care physician (PCP) in the past year, indicating the potential to significantly increase screening rates via teleophthalmology services in the primary care setting. 9 Primary care offices can provide diabetes screenings via teleophthalmology using nonmydriatic cameras to obtain fundus photographs, which are sent to an ophthalmologist for remote evaluation.
Teleophthalmology is an effective tool to increase DR screenings in areas of limited healthcare access and to efficiently triage patients who require further evaluation by an eye care provider.10 –12 Even in areas with high levels of health care access, teleophthalmology screening could potentially improve fundus examination compliance rates for patients with diabetes. 12 Ophthalmic screening at the point of primary care may offer timely evaluation and a reduction in doctor visits for an already treatment-burdened population as well as the opportunity for the primary care team to educate patients with diabetes on the importance of ophthalmic screenings in the preservation of this vital sense.
Although many studies have shown the success of teleophthalmology screening for DR, a few more closely examined the patients’ and providers’ experience with its implementation. Liu et al 13 conducted interviews with PCPs who described their rural teleophthalmology program as convenient and reliable. However, they also reported barriers with the program, such as insufficient reminders, difficulty tracking down patients who needed screening, and competing demands. Patients shared their concern of needing to pay out of pocket for the screening. In another study, 14 providers in an urban primary care setting were interviewed to understand their perceptions of the teleophthalmology screening program for DR. Investigators reported providers’ dissatisfaction with the program because they believed it did not address the root issues with screening this population, such as competing priorities and low health literacy. In addition, providers reported that the referral process imposed an increased burden. The experiences of providers and patients may offer valuable insights into the appropriateness of implementing a teleophthalmology program as well as the necessary logistical and operational processes to ensure its success.
Many studies have demonstrated the success of teleophthalmology for DR screening in various, mostly rural, locations worldwide.15 –18 However, there are still few studies of the role of teleophthalmology in urban settings. To our knowledge, no studies have focused on the implementation of DR teleophthalmology screening programs in New York City. Most notably, few studies have investigated obstacles (before and during the COVID-19 pandemic) faced by primary care services with implementation of teleophthalmology programs. The goals of this 2-part study were to (1) perform a pilot test examining the implementation of our teleophthalmology program for DR screening located at a large metropolitan hospital system in New York City and (2) to understand and highlight the challenges and barriers that primary care teams have encountered while participating in the program.
Methods
Part 1: Teleophthalmology Screening Program Cohort
This part of the study was a retrospective chart review of consecutive patients screened for DR by the teleophthalmology screening program at New York Presbyterian (NYP)/Weill Cornell Medicine (WCM) between August 2017 and June 2020. The study protocol was reviewed and approved by the Institutional Review Board, WCM College (WCMC).
Program Overview
The WCM Department of Ophthalmology deployed ultra-widefield (UWF) retinal fundus cameras (Optos) to the WCM divisions of General Internal Medicine and Endocrinology as well as to affiliated primary care practices. To date, 846 patients with diabetes had retinal imaging at the discretion of their internist or endocrinologist. The cohort included the first 300 patients with diabetes that had complete data at the time of the analysis. Trained ancillary personnel (eg, medical assistants) at individual practices obtained retinal images and transmitted them to the Department of Ophthalmology via Optos Advance and Epic Systems electronic health record software. A single retina specialist (S.K.) assessed the images for findings, including DR. The assessment and recommendations were sent to the referring endocrinologist or internist within 24 hours of image capture. Depending on the degree of retinopathy or other fundus findings, patients were scheduled for a follow-up examination by the Department of Ophthalmology or a repeat teleophthalmology screening with their provider between 1 month and 1 year from the time of image interpretation.
Data Collection
Given retinal imaging was at the discretion of the referring provider, data on how many screenings were offered or declined were not collected. Baseline variables data were collected, including age, sex, ethnicity, race, ZIP code, body mass index (BMI), hemoglobin A1c (HbA1c), diabetes type and duration, smoking status, and comorbidities (eg, hypertension, large-vessel disease). Teleophthalmology screening findings, including diagnosis and staging, and recommendations were recorded. For patients referred to the Department of Ophthalmology for in-person examination, the capture rate and the time between teleophthalmology screening and the office visit were determined. Of patients who presented for an in-person examination, the examination findings, including visual acuity (VA), intraocular pressure (IOP), fundus findings, and treatment, were recorded.
Statistical Analysis
Descriptive statistics (eg, mean, standard deviation) were calculated to summarize relevant demographic and teleophthalmology screening variables. Clinically relevant HbA1c levels (≥8 HbA1c, ≤7.9 HbA1c) were compared using a chi-square test of association in relation to a positive diagnosis for DR. The association between the duration of diabetes (≤9.9 years, ≥10 years) and a positive diagnosis of DR was also evaluated through the same statistical technique. These cutoff values were based on previous studies.19 –21 All P values were 2 sided with statistical significance evaluated at the 0.05 alpha level. All analyses were performed with SAS software (version 9.4, SAS Institute, Inc).
Part 2: Clinical Staff’s Perspective of Teleophthalmology Screening Program
An online survey was distributed to clinical staff (eg, physicians, nurses, medical assistants) in the Division of General Internal Medicine, Division of Endocrinology, and other NYP/WCM-affiliated primary care practices that previously or currently participated in the teleophthalmology screening program. The survey was distributed and completed in March 2022. The study protocol was reviewed and approved by the Institutional Review Board, WCMC.
Study Design
The web-based survey was distributed to the physicians and supporting clinical staff via email. The survey questions sought to identify the barriers encountered by the clinical staff in providing patients with diabetes with annual teleophthalmology screening examinations, their perceptions of the challenges patients face in completing ophthalmology referrals, and how the COVID-19 pandemic affected teleophthalmology screenings in their practices. Respondents were compensated with a $5.00 coffee shop gift card on survey completion.
Survey Design
The web-based survey was created via Qualtrics (Qualtrics). The survey questions (Supplemental Material) were adapted in part using previous surveys developed by Lindenmeyer et al 22 and Hipwell et al. 23 Before initiating the survey, providers and supporting staff were asked 3 screening questions regarding their consent, department affiliation, and familiarity with the teleophthalmology program. To be included in the study, respondents provided consent to participate in the study, were employed by 1 of the 3 listed departments, and indicated familiarity with the teleophthalmology screening program. Once enrolled, demographic information (eg, clinical role, years of experience, race, and ethnicity) was collected. The remainder of the survey included a mix of multiple-choice and open-ended questions about the teleophthalmology screening program at respondents’ practices, barriers experienced, COVID-19’s impact on the program, and perceptions of the program, DR screening, and patient experience.
A Likert scale was used for multiple-choice questions in which respondents were asked to assess the importance of DR screening, satisfaction with the teleophthalmology screening program, and perceived patient experience with the program. Because of the differences in responsibilities between the clinicians (eg, doctor, nurse practitioner) and supporting clinical staff (eg, medical assistant) for the program, display logic was used to display relevant questions for each participant based on his or her clinical role.
Statistical Analysis
Descriptive statistics (eg, frequency, mean, standard deviation, percentage) were calculated to summarize individual survey questions.
Results
Part I: Teleophthalmology Screening Program Cohort
Demographics
The cohort consisted of 124 women (41.3%) and 176 men (58.7%). The mean age was 51.9 ± 15.5 years. Table 1 shows the baseline demographics, including the mean BMI, mean HbA1c, diabetes status, insulin use, and comorbidities.
Patients’ Baseline Demographics (N = 300).

Abbreviations: BMI, body mass index; CVD, cerebrovascular disease; HbA1c; hemoglobin A1c.
The highest percentage of patients identified as White/Caucasian followed by Black/African American, other, Asian, American Indian/Alaskan, and Native Hawaiian/Pacific Islander; race was unspecified for 56 patients (18.6%). Regarding ethnicity, 39 patients (13.0%) identified as Hispanic/Latino. Most participants resided in New York City.
Diabetic Retinopathy Findings
Table 2 shows the teleophthalmology screening findings. Of the patients evaluated by UWF retinal imaging, 57 (19.0%) were diagnosed with DR, with the highest percentage having mild nonproliferative DR (NPDR) followed by PDR and moderate NPDR; none had severe NPDR. One patient was presumed to have macular edema based on the fundus images. An HbA1c of 8 or above (odds ratio [OR], 2.6; P < .01) and a duration of diabetes of 10 years or more (OR, 3.1; P < .01) were significant predictors of DR in this cohort. No associations were found between baseline demographics and DR, including age (P = .2), sex (P = .3), race (P = .5), or ZIP code (P = .6). Of the 57 patients diagnosed with DR, 1 had a retinal tear and 1 had a vitreous hemorrhage.
Teleophthalmology Screening Findings of Cohort (N = 3000).
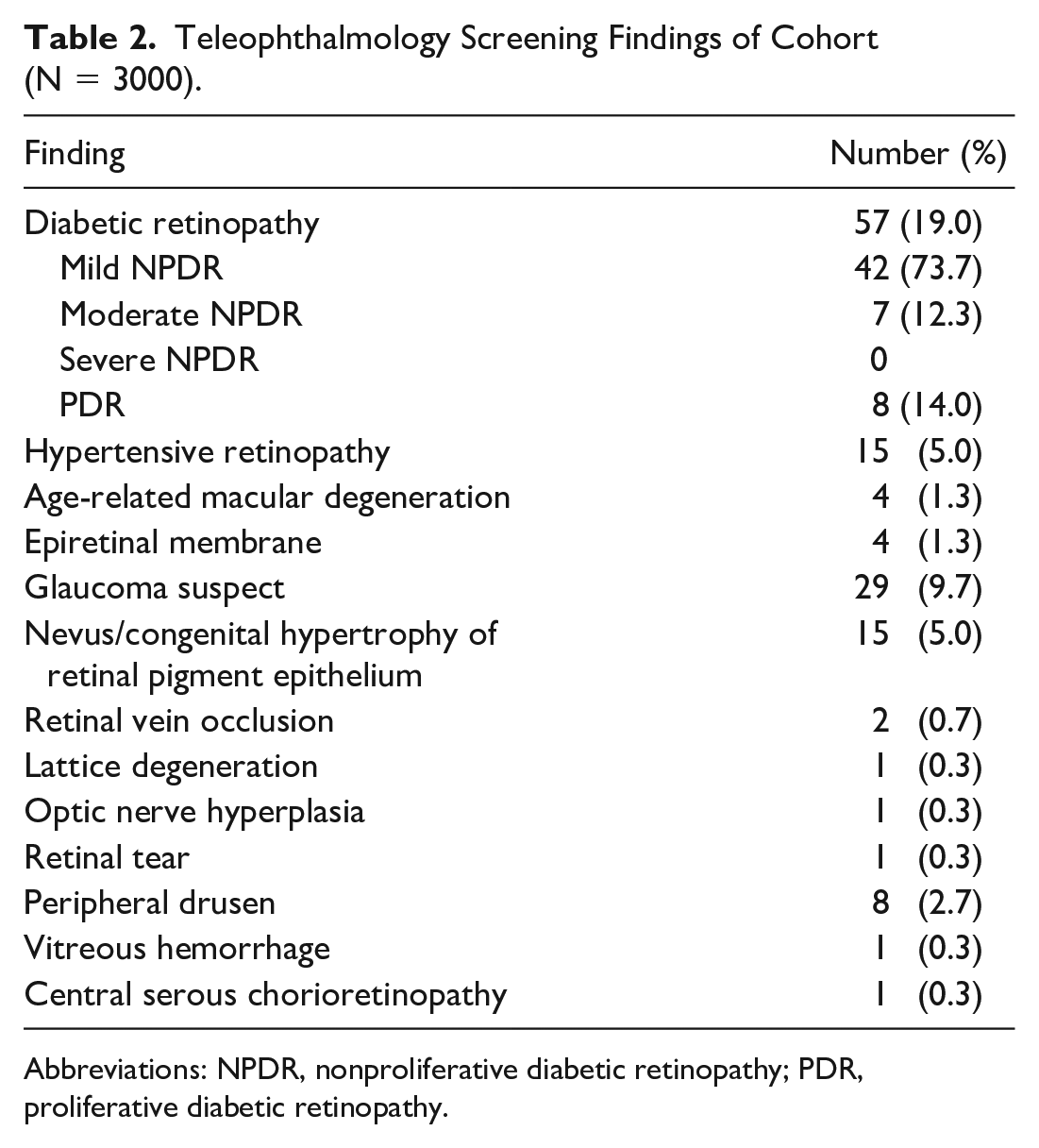
Abbreviations: NPDR, nonproliferative diabetic retinopathy; PDR, proliferative diabetic retinopathy.
Incidental Findings
Table 2 also shows the incidental findings, which included glaucoma suspect, choroidal nevus or congenital hypertrophy of the retinal pigment epithelium, age-related macular degeneration, retinal vein occlusion, lattice degeneration, and a retinal tear.
Recommendations for Patients With a Diabetic Retinopathy Diagnosis
The 57 patients diagnosed with DR via telescreening were recommended to continue monitoring with the teleophthalmology screening program or to be seen by ophthalmologists for an in-person examination and management (Figure 1). Twenty-six patients (45.6%) were recommended to continue monitoring via teleophthalmology. Of them, 24 (92.3%) had mild NPDR, 1 (3.9%) had moderate NPDR, and 1 (3.9%) had treated PDR. Thirteen (50.0%) of the 26 patients continued monitoring via the teleophthalmology screening program. Of the 13 who continued monitoring, 9 (69.2%) followed up in the recommended time of 6 or 12 months. Of the 13 patients who did not follow up via the teleophthalmology screening program, 4 were lost to follow-up, 2 had an order submitted by the provider not executed by the supporting staff, 4 continued to be seen by their provider but were not screened via the teleophthalmology screening program or ophthalmology department, 2 were seen at the Department of Ophthalmology, and 1 died.
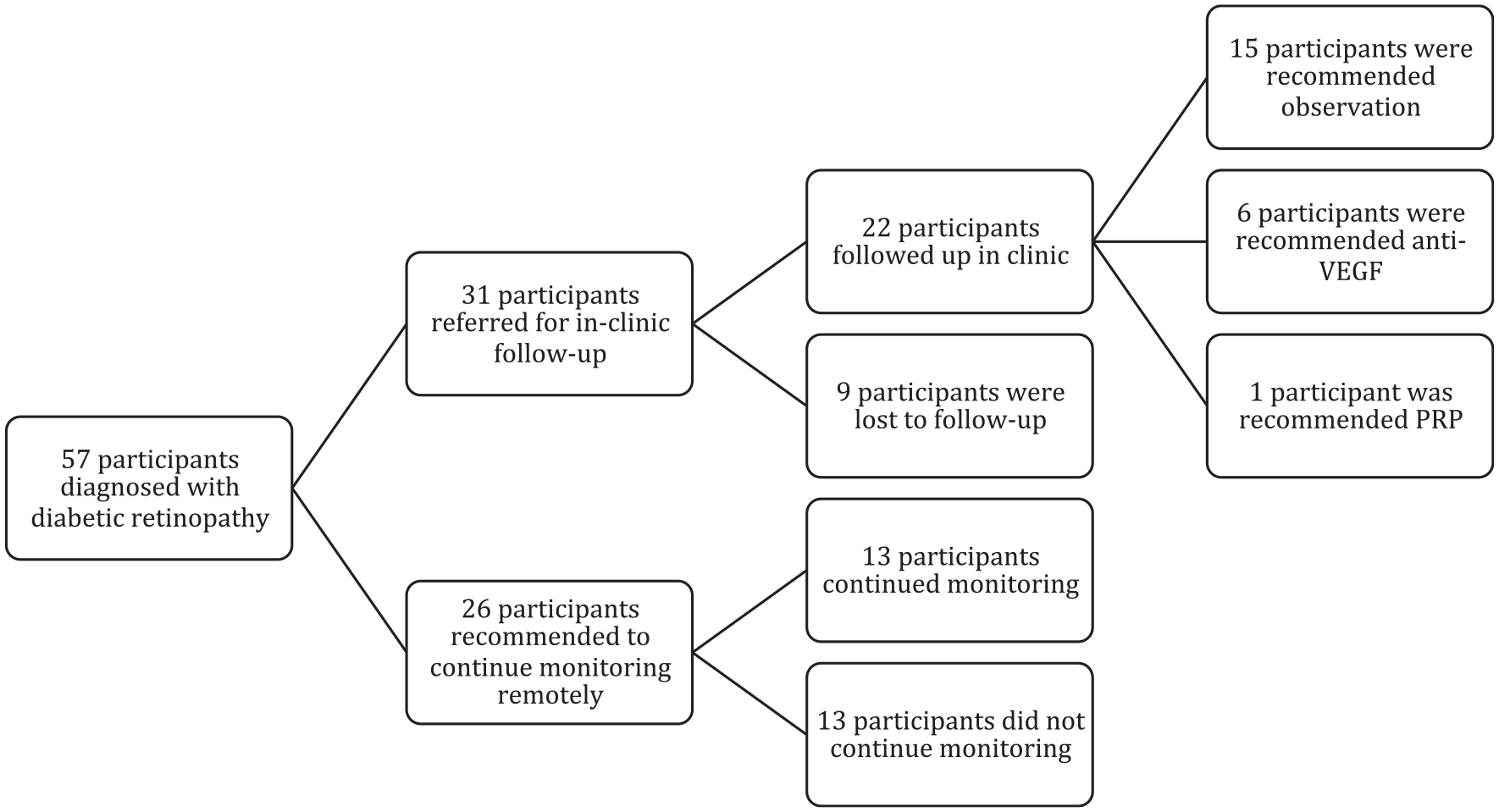
Teleophthalmology screening and treatment cascade.
Thirty-one patients (54.4%) with retinopathy diagnoses by screening were referred for an in-person follow-up at the clinic. Those diagnosed with mild or moderate NPDR were typically recommended to be seen within 12 months in the clinic, and those with severe NPDR or PDR were typically recommended to be seen within 3 months in the clinic. Of the 31 patients, 22 (71.0%) followed up at the clinic and 20 (64.5%) did so in the recommended time. The mean time between teleophthalmology screening and follow-up was 88.4 (range, 1-416 days). In the clinic, ophthalmologists reassessed staging and determined the presence of macular edema. Only 1 participant was diagnosed with mild NPDR on UWF retinal imaging and later with moderate NPDR in the clinic. Of the 22 evaluated in the clinic, 11 (50.0%) had mild NPDR, 4 (18.2%) had moderate NPDR, none had severe NPDR, and 7 (31.8%) had PDR, consistent with the initial teleophthalmology screening diagnoses. Six (27.3%) had macular edema, with 2 of them having center-involving macular edema. For the 22 evaluated, the average VA in the right eye was 20/30 (logMAR 0.19) and in the left eye was 20/32 (logMAR 0.2). The average IOP was 14.9 mm Hg (range, 9-23 mm Hg) and 14.8 mm Hg (range, 8-21 mm Hg), respectively.
Treatments
Of the 22 patients examined in the clinic, 15 (68.2%) were managed with initial observation. Six patients (27.3%) were recommended to initiate antivascular endothelial growth factor injection therapy, 4 of whom were treated with aflibercept and 1 with ranibizumab. Of the 5 treated patients, 3 had macular edema and PDR, 1 had macular edema and mild NPDR, and 1 had macular edema and moderate NPDR. One patient (4.6%) had panretinal photocoagulation treatment for PDR. No patient required surgery.
Part 2: Clinical Staff Perspectives of the Teleophthalmology Program
Demographics
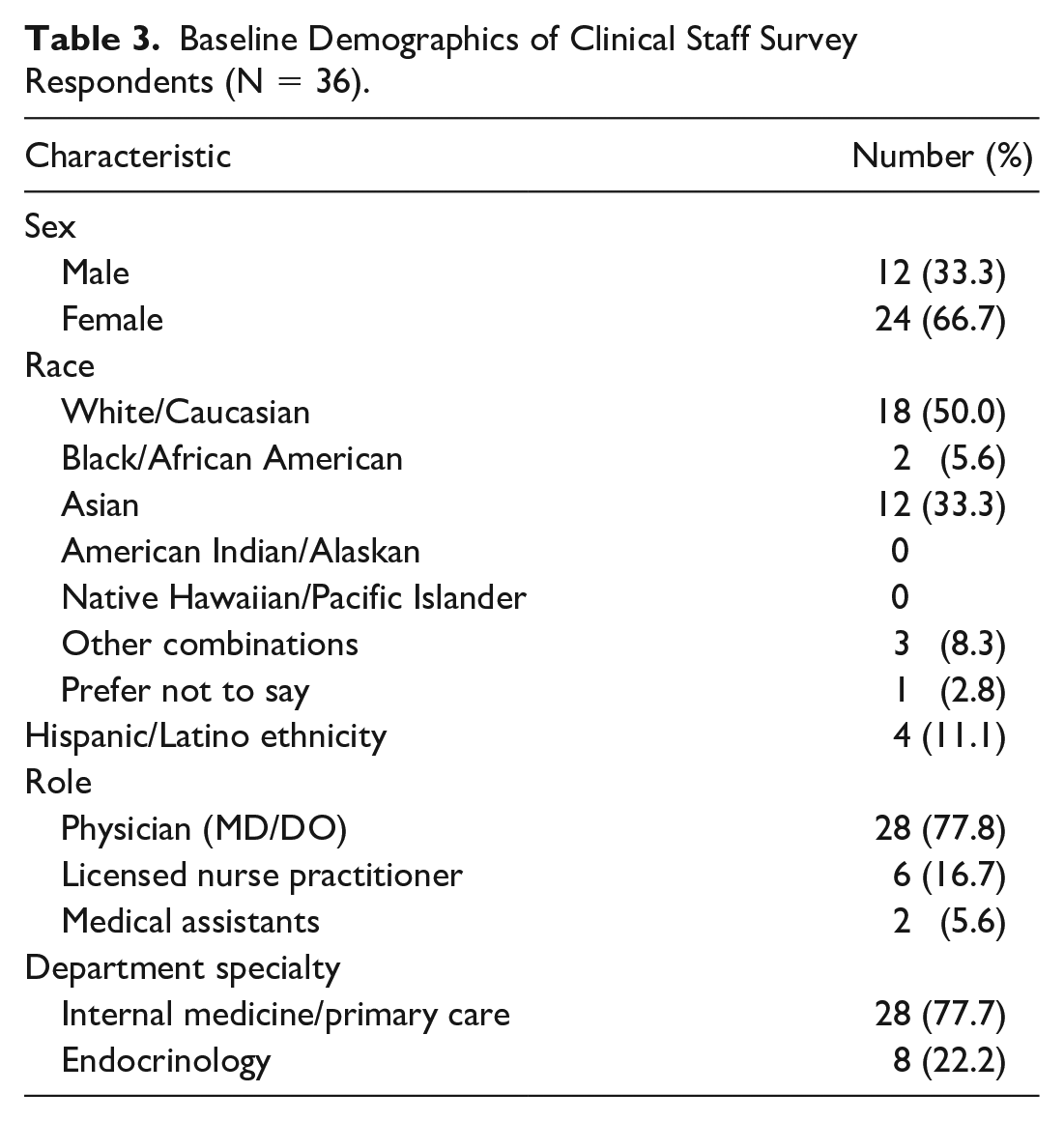
The survey was sent to approximately 300 providers and clinical staff at the NYP/WCM Divisions of General Internal Medicine and Endocrinology and affiliated primary care practices. Thirty-six respondents were determined to be eligible and completed the survey. Table 3 shows the baseline demographics. The survey respondents consisted of 28 (77.8%) physicians, 6 (16.7%) licensed nurse practitioners, and 2 (5.6%) medical assistants. Three providers screened more than 8 patients in a 1-month period. Two providers who screened 49% of the patients in the cohort did not participate in the survey.
Baseline Demographics of Clinical Staff Survey Respondents (N = 36).
Timing of Screening and Interpretation
Three respondents (9.7%) reported that the teleophthalmology encounter took less than 5 minutes, 8 (25.8%) reported it took 6 to 10 minutes, 7 (22.6%) reported 11 to 15 minutes, 6 (19.4%) reported 16 to 20 minutes, and 7 (22.6%) reported more than 20 minutes.
Fourteen providers (50.0%) reported a 1- to 2-day turnaround for the interpretation report, 12 (42.9%) reported 3 to 5 days, 1 (3.6%) reported 1 to 2 weeks, and 1 (3.6%) reported more than 2 weeks. Twenty-four providers (77.4%) “strongly agreed” or “somewhat agreed” they were satisfied with the interpretation report.
Diabetic Retinopathy Screening Preferences
Survey respondents were asked about the importance of DR screenings compared with other diabetes screening activities. Thirty respondents (90.9%) viewed annual eye examinations to be “very important” or “extremely important” relative to other diabetes screening activities. Meanwhile, 3 respondents (9.1%) believed annual eye examinations were “slightly important” or “moderately important.”
Providers were asked about their preferred mode of ophthalmology screening referrals. Twenty-two providers (71.0%) preferred in-person annual examinations; 12 (38.7%) encouraged patients to obtain an annual examination at their preferred ophthalmology practice, while 10 (32.3%) preferred submitting a referral to an ophthalmologist. Nine providers (29.0%) preferred a teleophthalmology screening encounter at their practice.
Perceptions of the Teleophthalmology Service
Survey respondents were asked about their satisfaction with the teleophthalmology screening program. Eighteen respondents (60.0%) “strongly agreed” with being satisfied with the program, 4 (13.3%) “somewhat agreed,” and 2 (6.7%) “somewhat” or “strongly disagreed”; the remaining 6 respondents (20.0%) selected “not applicable.” Survey respondents were asked whether patients were receptive to having an annual eye examination via the teleophthalmology screening program. Ten respondents (34.5%) “strongly agreed,” 9 (31.0%) “somewhat agreed,” 3 (10.3%) “neither agreed nor disagreed,” 1 “strongly disagreed,” and 6 (20.7%) selected “not applicable.”
Perceptions of Provider Barriers to Using the Teleophthalmology Service
Survey respondents were asked about the barriers they face with the teleophthalmology screening program at their respective practices, which included the following (respondent volume in parenthesis): changes due to COVID-19 (eg, reduced in-clinic patient volume) (14), workflow interruption (12), time-consuming for the staff (9), lack of staff (9), none (9), time-consuming for provider (4), time-consuming for patient (4), difficulty tracking patients who are due for annual examinations (4), and technical issues when obtaining or submitting retinal images (3) (Figures 2 and 3).
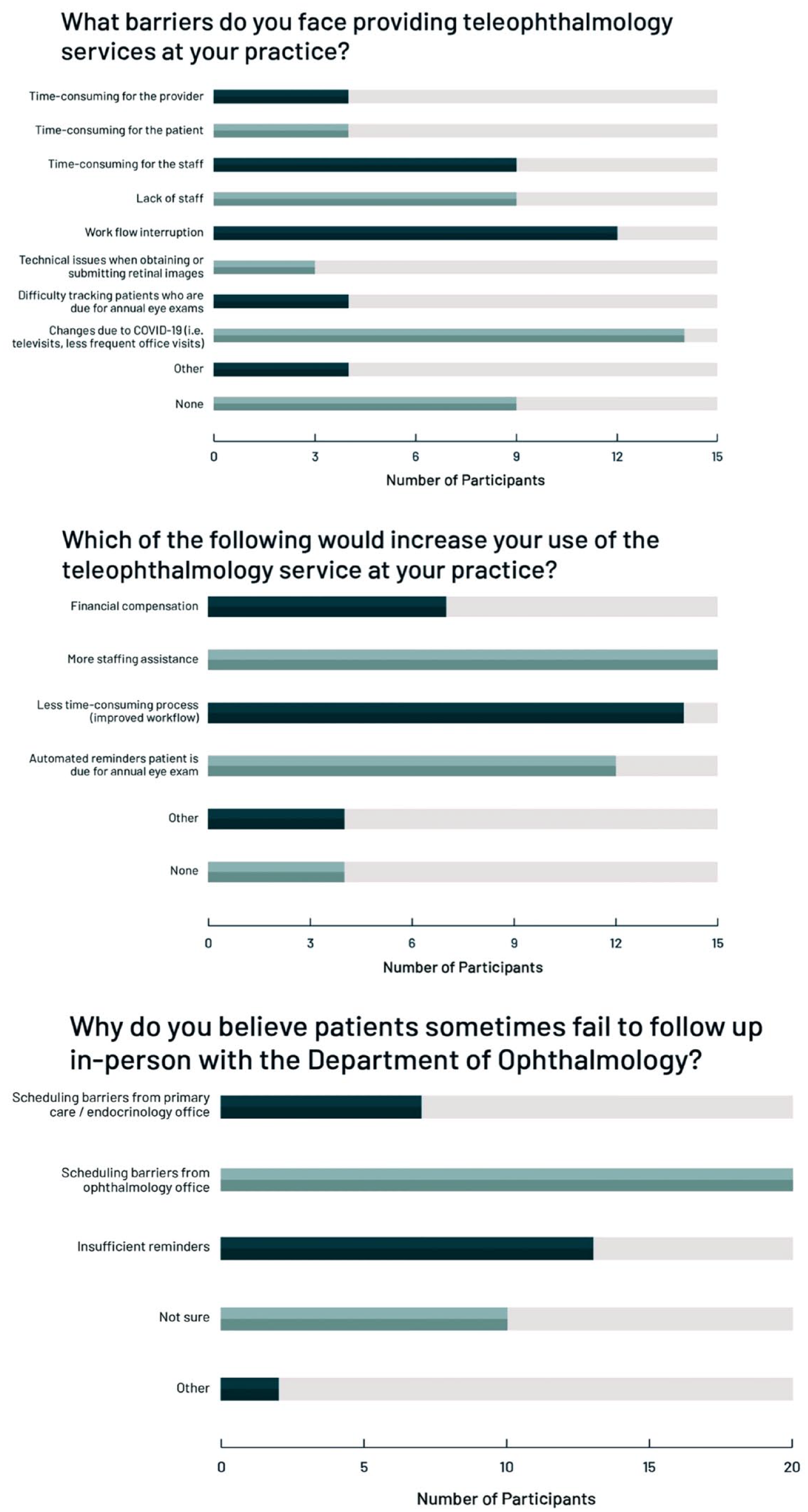
Clinical staff responses to survey questions.

Quotes from free-response questions.
Survey respondents were asked what changes would potentially increase their use of the teleophthalmology screening program. The following changes were selected: more staffing assistance (15), less time-consuming process (improved workflow) (14), automated reminders that the patient is due for annual eye screening (12), financial compensation (7), and none (4). Four participants who selected “other” suggested a functioning UWF retinal fundus camera, annual screening insurance coverage, and a redistribution of the associated administration work to the Department of Ophthalmology (Figures 2 and 3).
To assess how the COVID-19 pandemic affected volume, survey respondents were asked about the current volume of patients in their offices vs before the pandemic. Seven respondents (29.2%) reported it had not resumed at all to baseline, in other words, 0% resumption; 10 (41.7%) reported it resumed to or less than 50%, 1 (4.7%) reported it resumed to 75%, and 6 (25%) reported it resumed to 100%. In the free responses, respondents shared that COVID-19 resulted in fewer in-person visits and more video visits, thus decreasing in-office teleophthalmology screenings (Figure 3).
Perceptions of Screening Barriers for Patients
Survey respondents were asked why they believed patients might fail to follow up in person with the Department of Ophthalmology. Twenty-two respondents (66.7%) believed the patients understood the importance of an annual eye examination, 5 (15.2%) were unsure, and 6 somewhat disagreed (18.2%).
The following reasons were selected, with the number of participants who endorsed it in parentheses: scheduling barriers from the ophthalmology office (20), insufficient reminders (13), scheduling barriers from the primary care or endocrinology office (7), and not sure (10) (Figure 2). Other reasons included patient critiques of the ophthalmology office experience (eg, long wait time) and personal barriers (eg, transportation, work obligations).
Survey respondents were asked how the teleophthalmology program could be improved for patients. The following changes were recommended: faster interpretation of results, providers better emphasizing the importance of screening to patients, adequate UWF retinal fundus camera support, in-advance preparation to build screening encounter time into the visit, and annual insurance coverage for the encounter (Figure 3).
Conclusions
To our knowledge, our study is one of the first to examine the implementation of a teleophthalmology screening program for DR in New York City. Even in a metropolitan area with many ophthalmic offices, access to care can be difficult for patients given the treatment burdens they face.
The present pilot study suggests that a teleophthalmology screening program in a metropolitan area captures patients before retinopathy develops (243/300 [81.0%]) or at a very early stage in those with background DR (42/57 [73.7%]). It potentially offers an alternative to an in-person examination to increase DR screening rates and may allow for early diagnosis at a more modifiable stage when there is maximum opportunity for vision preservation with less-invasive measures and with better visual outcomes. Notably, screenings picked up patients with suspected glaucoma that required further ophthalmic evaluation to prevent unnecessary vision loss. There was overall favorable compliance with in-clinic follow-ups. Of the 31 patients who received teleophthalmology screening and were recommended to have a follow-up visit with an ophthalmologist, 22 (71.0%) completed the visit. This figure does, however, suggest room for improvement in removing friction associated with in-person follow-ups.
This study is also one of the first to examine barriers to teleophthalmology screening in the primary care setting. In our survey sample, the vast majority (90.9%) of primary care staff and providers said that annual screening for DR was “very important” and the majority (73.3%) of clinical staff respondents were “strongly” or “somewhat satisfied” with the teleophthalmology screening program. Despite this, 71.0% still preferred referring patients for in-person annual examinations over teleophthalmology screening.
Respondents identified multiple barriers to teleophthalmology screening. The majority of these related to workflow disruption, lack of staffing, or time spent obtaining the images. A majority of survey respondents (64.6%) stated that more than 10 minutes were spent per patient on this screening, suggesting room for improvement in staffing and staff training on image acquisition. In addition, survey respondents identified the COVID-19 pandemic as a major factor in performing screenings, mostly related to lower in-person clinic volumes.
Other studies have examined the implementation of teleophthalmology screening programs for DR in urban settings. In Austin, Texas, USA, a group analyzed the 2-year experience of a teleophthalmology screening program in an insured urban population. 24 The investigators reported screening 5764 patients with diabetes; 1152 of them (20.0%) had DR. Nearly 82% of patients who were recommended to see a specialist completed the follow-up visit. Comparatively, in our study, 19.0% of patients were diagnosed with DR and 71.0% completed the follow-up visit.
A large-scale teleophthalmology screening program was launched in 5 clinics in the Los Angeles County Department of Health Service, the largest publicly operated county safety net health care system in the US that mostly serves an underinsured and uninsured population, resulting in 21 222 screenings in 2 years. 12 Of the patients screened, DR was detected in 6888 (32.5%) and 4160 (19.6%) were referred to a specialist (moderate NPDR, severe NPDR, and PDR, as well as clinically significant macular edema). The investigators found that their program improved DR screenings from 40.6% to 56.9% and noted a reduction in wait times to be seen by an ophthalmologist as a result of eliminating DR-free patient visits. Most screenings in our cohort were negative for DR, similarly potentially decreasing the number of unnecessary in-person ophthalmology visits. In our pilot study and others, teleophthalmology DR screening programs in urban areas have shown promise as a potential solution to the need for increased compliance with DR screening in both insured and underserved communities.
In our experience, clinical staff faced challenges with the implementation of teleophthalmology screening, which affected the service’s use. These identified barriers highlight the need for systematic changes requisite for realizing the service’s full potential for cost-effective vision preservation at scale.
Similarly, other studies have evaluated the barriers the clinical team encountered with a teleophthalmology screening program. Ramchandran et al 25 performed a qualitative interview study to assess clinical staff experience with a teleophthalmology DR screening program in Rochester, New York, USA. Although the nursing staff and physicians viewed the program as convenient and valuable for the patients, they were often short staffed and were unable at times to incorporate the screening into their workflow. They also reported patients declining the service because of the extra 10 to 15 minutes it added to their visit without previous notification. Similar obstacles came to light in our study.
In a study by Bouskill et al, 26 medical assistants reported issues with scheduling follow-ups with the retina specialist. Likewise, of our survey respondents, 38.5% reported scheduling issues with the ophthalmology clinic to be a barrier for patients. Possible contributing factors include the scarcity of available appointments and issues with the clinic reaching the patient.
In another study, PCPs reported having insufficient reminders, time, and resources to discuss and refer patients to the program. 13 In our study, a third of survey respondents reported that automated reminders would improve their use of the service.
From the perspective of the clinical staff, it was challenging to incorporate the teleophthalmology screening service into their workflow. More than a quarter reported they would increase the use of such screening if there were more staffing assistance. A similar subset reported that a less time-consuming process would incentivize their use of the service. Ramchandran et al 25 found success in designating a clinician or nurse as an onsite champion in charge of tracking patients due for screening and training other employees on how to use the camera. They also trained the clinical staff on an implementation process to help them better incorporate the program into their workflow. Our institution’s teleophthalmology screening program could likewise benefit from increased formalized referral office-wide education and staff training as well as increased staffing, which could lead to decreased screening times, improved workflow, and increased use. Onsite champions at both the referral and ophthalmology offices who can maintain clear lines of communication, facilitate patient screening and scheduling, and troubleshoot technical issues will be key to our ongoing effort to optimize the utility of this service.
There are several limitations to this study. First, it is a retrospective review, which has its own biases and limitations, such as missed or inaccurate data, an inability to determine causation, and past data that might not reflect current data. Second, few providers frequently used the teleophthalmology screening process. Only 3 providers reported screening more than 8 patients in a 1-month period. In addition, most patients in the cohort were screened by 2 providers. Therefore, it is unclear whether the cohort is representative of the entire NYP/WCM health system.
Forty-six percent of physicians who participate in our teleophthalmology screening program completed the survey study, capturing referring physicians who only moderately use the service. Two of the PCPs who screened approximately one half of the patients in our cohort did not participate in the survey. Although our findings are useful for determining potential barriers to the use of teleophthalmology and solutions to increase program participation, they may over-represent negative experiences with the screening program as a result of response bias.
In conclusion, this study describes the implementation of a teleophthalmology DR screening program and barriers faced by providers who participate in the program in an urban setting at a large academic health system. Most patients had early or no retinopathy. This is a goal of a screening program; that is, to detect pathology early so it can be more easily managed, preventing more advanced disease and worse clinical outcomes. Most patients in our cohort continued monitoring through the teleophthalmology program or at our clinic. From our PCPs and endocrinologists, we learned that there is lower than ideal use of the screening program as a result of its workflow interruption and time consumption as well as suboptimal referral office staff resources, education, training, and incentivization.
Our findings highlight the weaknesses of teleophthalmology programs and draw attention to how their implementation can be improved to ensure their success. These insights will allow us to improve the program’s use and maximize our institution’s DR screening rate to allow us to maximally preserve vision in this at-risk population. In the future, we seek to perform a large-scale analysis of our teleophthalmology screening program and test various operational and logistical processes in an effort to improve our program.
Supplemental Material
sj-docx-1-vrd-10.1177_24741264231208253 – Supplemental material for Implementation of a Teleophthalmology Screening Program for Diabetic Retinopathy in New York City
Supplemental material, sj-docx-1-vrd-10.1177_24741264231208253 for Implementation of a Teleophthalmology Screening Program for Diabetic Retinopathy in New York City by Pamela Capellan, Alexander B. Dillon, Geoff Rodriguez, Jason Chua, M. Abdallah Mahrous, Kyle Kovacs, Sarah Van Tassel, Donald J. D’Amico, Szilard Kiss and Anton Orlin in Journal of VitreoRetinal Diseases
Footnotes
Ethical Approval
Both study protocols were reviewed and approved by the Institutional Review Board at Weill Cornell Medicine College.
Statement of Informed Consent
Informed consent was not required because this was a quality study under the institutions’ Diabetic Quality Institutional Review Board, which deemed the study exempt.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. D’Amico is a consultant to Alcon, Iveric Bio, and Aufbau Holdings; has equity in Iveric Bio and Aufbau Holdings; and has intellectual property with Aufbau Holdings. Dr. Kiss is consultant to Adverum, Alcon, Novartis, Optos, and Genentech/Roche; has received research funding from Allergan, Novartis, Optos, Genentech/Roche, and Regeneron; has equity in Adverum, Regenxbio, and Fortress Bio; has intellectual property with Gene Therapy for AMD, T-Cells for CMV Retinitis—assigned to Cornell University. None of the other declared potential conflicts.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Weill Cornell Medical College, the Area of Concentration program, and Dr. William Schpero at the Department of Population Health Sciences and in part by an unrestricted departmental grant from Research to Prevent Blindness, Inc. The statistician, Jason Chua, MPH, was partially supported by the following grant: Clinical and Translational Science Center at Weill Cornell Medical College (1-UL1-TR002384-01).
Dr. Schpero reported receiving grants from the Commonwealth Fund, the Laura and John Arnold Foundation, the Milbank Memorial Fund, the National Institute on Aging, the Patient-Centered Outcomes Research Institute, the Physicians Foundation, and the Robert Wood Johnson Foundation outside the submitted work.
Supplemental Material
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
