Abstract
Introduction
Subretinal hemorrhages, including large subretinal hemorrhages, are not uncommon in wet age-related macular degeneration (AMD). These hemorrhages typically remain confined to the subretinal space and resolve with therapy and time.
We report a case of wet AMD in which an acute subretinal hemorrhage from an active choroidal neovascular membrane (CNVM) resulted in foveal dehiscence and a full-thickness macular hole (MH). To our knowledge, this is the first report of a full-thickness MH developing in the setting of acute hemorrhage from classic wet AMD.
Case Report
An 84-year-old man presented with a history of bilateral wet AMD. At the time of initial conversion to wet AMD, each eye had exhibited classic leakage without polypoid changes on angiography. The left eye retained good vision (visual acuity [VA] 20/20) with intravitreal bevacizumab injections, and the treatment interval for this eye had been extended to 6 weeks with good effect. The right eye had initially been treated with bevacizumab. However, the fluid had persisted despite monthly bevacizumab injections, and the treatment regimen was switched to aflibercept. The eye had a good response to aflibercept on a treat-and-extend regimen, and the treatment frequency had been extended to every 8 weeks. The VA in the left eye was 20/80 (pinhole 20/50) (Figure 1A).
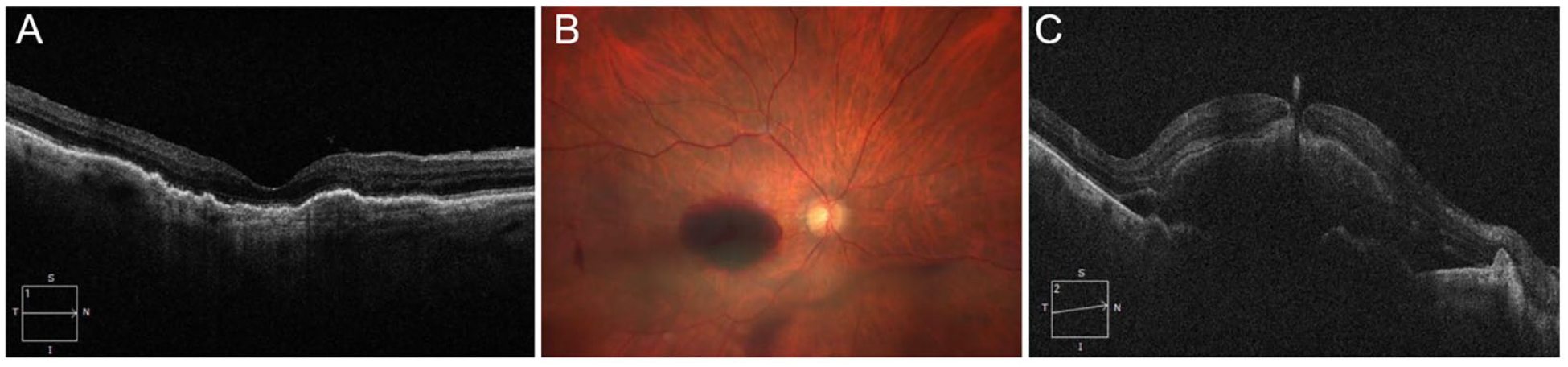
(A) Optical coherence tomography (OCT) of the right eye at baseline shows drusen and pigment epithelial detachments. There is no significant foveal epiretinal membrane or atrophic features. (B) Fundus photograph of the right eye 6 weeks after intravitreal aflibercept shows a vitreous hemorrhage and media opacity as well as a subretinal hemorrhage. (C) OCT shows a subretinal hemorrhage with a full-thickness macular hole and egress of the hemorrhage through the foveal defect.
Six weeks after the 12th aflibercept injection, the patient presented with sudden-onset central vision loss and new floaters in the right eye. The VA was 20/200. A dilated fundus examination showed a vitreous hemorrhage, MH, and subfoveal hemorrhage of approximately 4 disc areas (Figure 1B). Optical coherence tomography (OCT) showed a moderate-size subfoveal hemorrhage (subfoveal hemorrhage thickness 523 µm) with elevation of the neurosensory retina and foveal dehiscence with a full-thickness MH. There was a hemorrhage egressing from the foveal defect into the vitreous cavity (Figures 1C and 2A). The patient had an intravitreal injection of aflibercept.
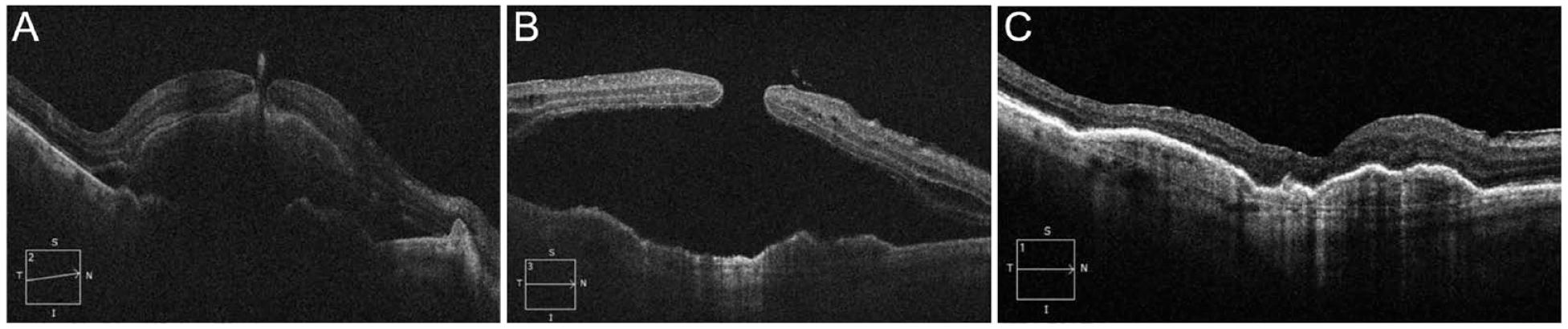
(A) Optical coherence tomography (OCT) scan of the right eye shows a subretinal hemorrhage from wet age-related macular degeneration and a full-thickness macular hole (MH). (B) The patient had an intravitreal aflibercept injection. Two weeks later, OCT showed resolution of the subretinal hemorrhage but a persistent MH with subretinal fluid. (C) The patient had a pars plana vitrectomy, internal limiting membrane peel, perfluoropropane gas tamponade, and prone positioning with subsequent closure of the MH.
Four weeks later, a follow-up examination showed a trace vitreous hemorrhage, a largely resolved subretinal hemorrhage, and a persistent MH (548 µm diameter) with a localized macular detachment (Figure 2B). The VA was 20/500. Several days later, the patient had a pars plana vitrectomy (PPV) with indocyanine green staining, membrane peeling, fluid–air exchange, and 10% perfluoropropane (C3F8) tamponade, followed by 5 days of prone positioning. The MH closed (Figure 2C), and the VA approximately 6 months postoperatively was 20/200 (pinhole 20/100).
Conclusions
An MH is a full-thickness defect in the neurosensory retina. The pathophysiology of MHs centers on a tractional etiology, with anteroposterior traction from an attached or separating posterior hyaloid and/or tangential traction from vitreous cortex, internal limiting membranes (ILMs), and/or epiretinal membranes (ERMs), resulting in foveal dehiscence.1,2
MH formation in the setting of AMD is not well studied or well described. These aging eyes with AMD are subject to the same tractional forces that underlie typical senile MHs (eg, vitreoretinal traction during an aberrant posterior vitreous detachment [PVD] process, ERM).3,4 In addition, the retina in eyes with AMD can have atrophic and degenerative changes that predispose to foveal dehiscence. 3 Most reports of MHs associated with wet AMD describe MH formation in conjunction with CNVM regression (ie, in the setting of fluid resolution after treatment with antivascular endothelial growth factor [anti-VEGF] therapy).5 –9 There are also many reports of MH formation during resolution of exudation in polypoidal choroidal vasculopathy treated with anti-VEGF 10 and during contraction of a treated myopic CNVM.11 –14 In such eyes, in addition to the tractional processes, tangential traction from a contracting CNVM may contribute to MH formation. In addition, intravitreal injections likely alter the fluid dynamics in the eye to promote the PVD process, which can contribute to MH formation. 15
We report a case in which wet AMD activity, rather than regression, produced an MH. In this case, an acute subretinal hemorrhage resulted in mechanical dehiscence of the fovea with simultaneous formation of a full-thickness MH. Our patient had no significant predisposing degenerative or atrophic changes, nor was there an ERM. In addition, the patient had developed a PVD years before the MH appeared. Therefore, in this case, the force of an acute and large subretinal hemorrhage likely created a posterior-to-anterior pushing force from the choroid toward the retina. This is akin to the posterior-to-anterior pulling force of the hyaloid or the anterior-to-posterior pulling force from a posterior staphyloma known to contribute to MH formation. It is not possible to determine in our case whether the hemorrhage erupted through the intact retina to directly cause the MH or whether the hemorrhage simply egressed through the hole once it formed (Figure 2A). Nevertheless, the presence of the MH enabled rapid release of the hemorrhage from the subretinal space, and OCT imaging 4 weeks later showed resolution of the subretinal hemorrhage (Figure 2B).
The incidence of subretinal hemorrhage in eyes being treated with anti-VEGF injections for wet AMD is unknown, with studies being small and limited. One recent registry study estimated the rate of visually significant subretinal hemorrhage in eyes having anti-VEGF therapy at approximately 1 per year per 200 patients. 16 In contrast, another study reported a rate of 8% for a new or increased subretinal hemorrhage in eyes with wet AMD being treated with ranibizumab. 17 MHs have been reported in conjunction with other conditions causing subretinal hemorrhages, such as retinal arterial microaneurysms (RAMs)18,19 and trauma.20 –22 In the setting of trauma, it is unknown whether the MHs were typical traumatic MHs or whether the subretinal hemorrhage contributed to MH formation.20 –22
Recently Pereira et al 23 described a patient with pathologic myopia, posterior staphyloma, and baseline myopic macular schisis who developed a myopic CNVM associated with the occurrence of a small, full-thickness MH. In that case, the anteroposterior traction from the staphyloma, along with weakened foveal integrity in the setting of myopic macular foveoschisis, may have contributed to foveal dehiscence and MH formation from the CNVM. In contrast, the patient we report here had wet AMD rather than myopic CNVM and exhibited no staphyloma or preexisting retinoschisis (Figure 1A). Moreover, compared with the relatively smaller hemorrhage in the Pereira et al case, our patient had a larger subretinal hemorrhage (Figure 1C).
The management of the concurrent subretinal hemorrhage and MH in previously reported cases such as RAM or trauma was based on the specific clinical and anatomic features and included observation, 22 pneumatic (C3F8) displacement of the submacular hemorrhage and prone positioning, 20 and PPV with ILM peeling (± inverted ILM flap) for the MH ± subretinal injection of recombinant tissue plasminogen injection for the submacular hemorrhage and gas tamponade.18,19,21 In each of these cases, the subretinal hemorrhage resolved 22 or was effectively displaced,18 –21 and the MH closed.18 –21
In the case reported here, given the absence of notable preexisting tractional forces, it is possible that treatment of the underlying wet AMD could have allowed sufficient hemorrhage and subretinal fluid resolution to reappose the retinal edges and achieve MH closure. However, in our case the MH persisted despite intravitreal aflibercept injections and an improvement in the hemorrhage. In fact, the MH began to recruit fluid, resulting in a localized macular detachment (Figure 2B). The patient therefore had PPV with wide ILM peeling, gas tamponade, and prone positioning, resulting in successful closure of the MH (Figure 2C) and improved VA. Few studies have evaluated the surgical outcomes after MH repair in eyes with concurrent wet AMD, although existing studies suggest lower rates of anatomic closure than with typical senile MHs, possibly because of the larger MH (as in our case) and/or degenerative changes that impair closure.24,25
In conclusion, we describe a case in which an acute subretinal hemorrhage from active wet AMD was associated with the development of a full-thickness MH. We propose that the hemorrhage resulted in a posterior-to-anterior mechanical force that led to foveal dehiscence and MH formation.
Footnotes
Ethical Approval
This case report was conducted in accordance of the Declaration of Helsinki. The collection and evaluation of protected patient health information was performed in a US Health Insurance Portability and Accountability Act–compliant manner
Statement of Informed Consent
Informed consent was obtained before all evaluations and imaging reported herein, as well as before treatments such as intravitreal injection and surgery.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
