Abstract
Introduction
Ritonavir is a potent cytochrome P450 34A (CYP34A) inhibitor often used in combination with other antiretroviral therapy medications to treat HIV. 1 Although it was originally developed as an inhibitor of HIV protease, its antiviral activity remains limited in managing HIV. It is widely used as a pharmacokinetic booster whereby inhibiting cytochrome P450 3A4 leads to increased bioavailability of other protease inhibitors. 2 Several clinical trials have demonstrated the efficacy of these therapeutic combinations in treating patients with HIV. 3
Ritonavir is now being used in conjunction with nirmatrelvir (ie, nirmatrelvir and ritonavir co-packaged as Paxlovid [Pfizer Laboratories]) to treat mild to moderate COVID-19 via emergency use authorization by the US Food and Drug Administration (FDA), and efficacy has been shown in clinical trials.4,5 A recent US FDA panel voted unanimously that the available efficacy and safety data support the use of nirmatrelvir/ritonavir for mild to moderate COVID-19 in individuals who are at high risk for progressing to severe disease, and the drug combination was approved for treatment of COVID-19 on May 25, 2023. 6 While cases of retinopathy in the setting of ritonavir exposure have been infrequently reported since 2011, 7 ritonavir’s efficacy in the treatment of mild to moderate COVID-19 warrants an understanding of potential toxicity associated with prior use. We describe 3 patients who experienced ritonavir-associated retinopathy related to prior HIV treatment.
Methods
A retrospective review of patient records was performed for data including ophthalmic examination findings and demographic and HIV clinical characteristics, such as history of ritonavir use and progression of maculopathy disease. Three patients with a history of HIV had been evaluated at the Emory Eye Center, Atlanta, GA, for bilateral vision loss in both eyes, and their paper medical records were reviewed for data collection.
Results
Case 1
A 62-year-old man was evaluated for retinopathy in both eyes of unclear origin. He was diagnosed with HIV at the age of 26 years, for which he was treated with antiretroviral therapy including lopinavir, ritonavir, emtricitabine, tenofovir disoproxil fumarate, raltegravir, and zidovudine. His medical history included syphilis and meningitis (probably viral) of unknown causes (Table 1).
Demographic and Clinical Characteristics.
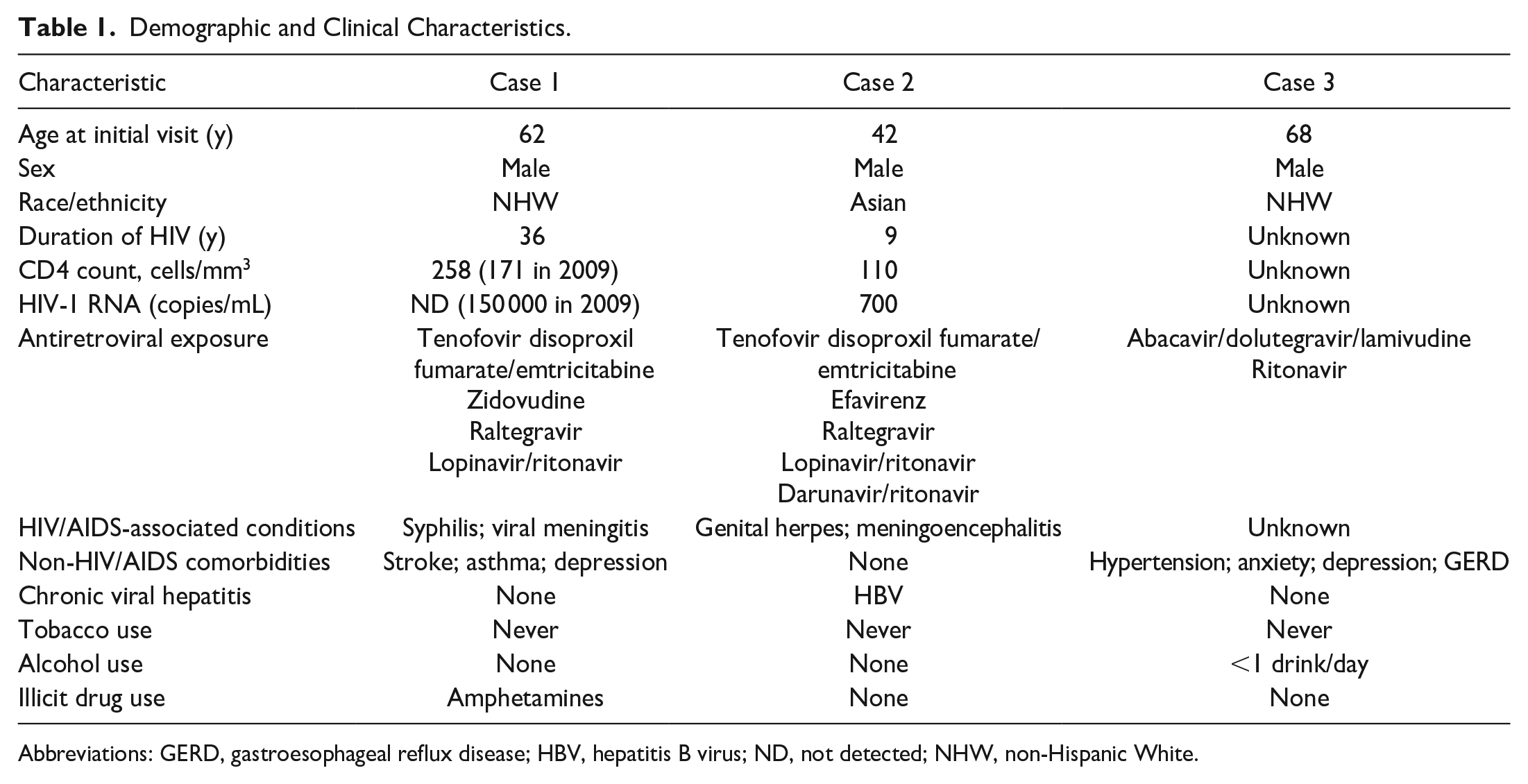
Abbreviations: GERD, gastroesophageal reflux disease; HBV, hepatitis B virus; ND, not detected; NHW, non-Hispanic White.
At the initial presentation, the best-corrected visual acuity (BCVA) was 20/200 OD and 20/80 OS. A fundus examination found bilateral macular atrophy in both eyes and areas of retinal pigment epithelium (RPE) hypertrophy in the right eye (Figure 1, A and B). Fundus autofluorescence revealed an area of hypoautofluorescence with an encircling stippled pattern in the right eye (Figure 1C) and with a diffuse stippled pattern in the left eye (Figure 1D). Optical coherence tomography (OCT) showed diffuse outer retinal atrophy with overlying epiretinal membrane in both eyes (Figure 1, E and F) and areas of subretinal hyperreflectivity in the right eye. A uveitis workup including a fluorescent treponemal antibody absorption test, a QuantiFERON-TB Gold test, and a check of angiotensin-converting enzyme levels was unremarkable for any infectious or inflammatory causes, and the findings from brain magnetic resonance imaging were normal.
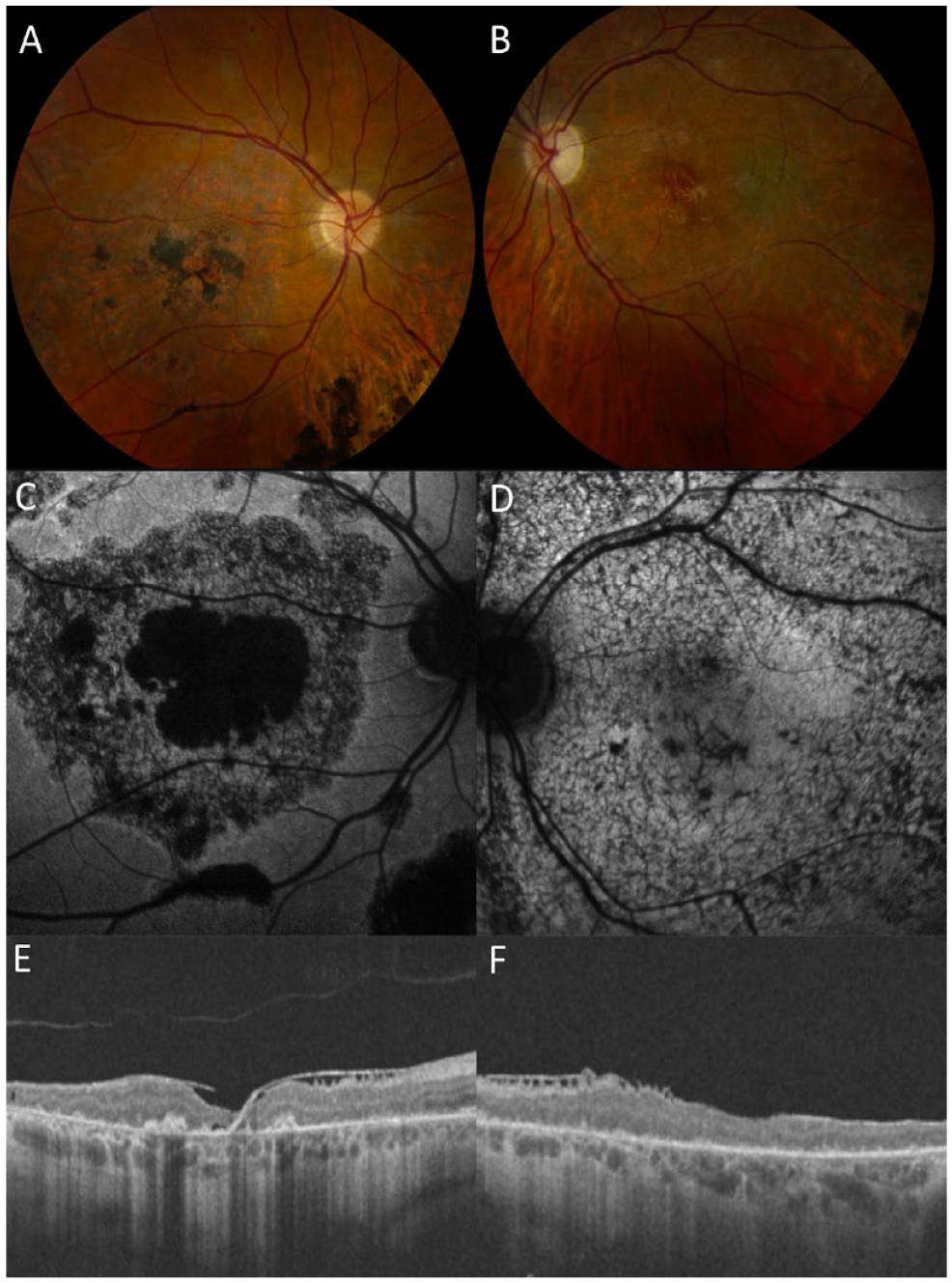
(A and B) Color fundus photographs demonstrated bilateral macular atrophy as well as areas of retinal pigment epithelium (RPE) hypertrophy in the right eye. Fundus autofluorescence revealed an area of hypoautofluorescence (C) with a surrounding stippled atrophic pattern in the right eye (D) and a diffuse stippled pattern in the left eye. (E and F) Optical coherence tomography showed diffuse outer retinal atrophy with overlying epiretinal membrane in both eyes and multiple RPE hypertrophy lesions in the right eye.
Given the characteristics of macular atrophy without evidence of intraocular inflammation and prior ritonavir use, the patient was diagnosed with bilateral ritonavir-associated retinopathy. Ritonavir was subsequently discontinued. At the follow-up 10 years after the initial visit, the BCVA was 20/150 OD and had deteriorated to counting fingers (CF) OS.
Case 2
A 42-year-old man was referred for waviness in his vision that had been ongoing for 1 week. He had been diagnosed with HIV 9 years prior to the referral and treated with multiple antiretroviral therapy regimens that included the agents emtricitabine, tenofovir disoproxil fumarate, efavirenz, raltegravir, lopinavir, darunavir, and ritonavir. His medical history was significant for chronic hepatitis B virus, genital herpes, and HIV meningoencephalitis (Table 1). The BCVA was 20/20 OD and 20/400 OS, and a slitlamp examination revealed a normal anterior segment.
A fundus examination found macular pigmentary changes in the right eye and a fibrotic scar with surrounding pigmentary changes in the left eye (Figure 2, A and B). Fundus autofluorescence showed central hyperautofluorescence in the right eye and central hypoautofluorescence with surrounding hyperautofluorescence in the left eye (Figure 2, C and D). OCT found outer retinal atrophy in both eyes that was more severe in the left eye than in the right eye (Figure 2, E and F). Further laboratory testing was negative for infectious or inflammatory causes. Given the findings on multimodal retinal imaging in the setting of prior ritonavir exposure, the patient was diagnosed with bull’s-eye maculopathy secondary to ritonavir toxicity.
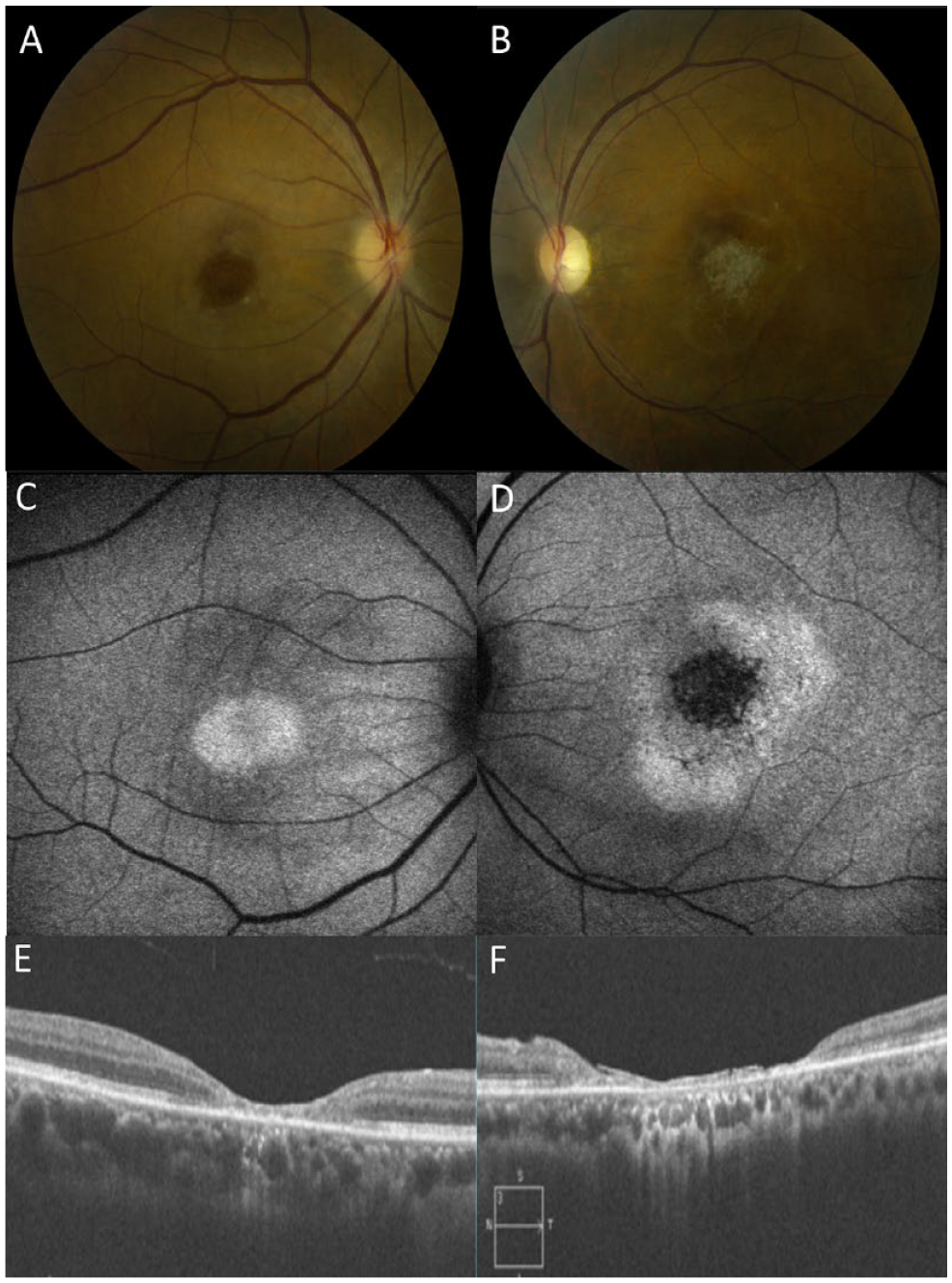
Color fundus photographs revealed (A) macular pigmentary changes in the right eye (B) and a fibrotic scar with surrounding pigmentary changes in the left eye. (C) Fundus autofluorescence showed a central hyperautofluorescence in the right eye (D) and a central hypoautofluorescence with surrounding hyperautofluorescence in the left eye. (E and F) Optical coherence tomography identified outer retinal atrophy in both eyes that was more severe in the left eye than in the right eye.
Ritonavir was discontinued after the patient transitioned to another therapeutic regimen for HIV. At the final follow-up 3 years after the initial presentation, the patient’s VA declined to 20/400 OD and CF at 6 inches OS with continued progression of outer retinal atrophy.
Case 3
A 68-year-old man presented with progressively worsening blurred vision for 2 years in both eyes prior to referral. His medical history was significant only for HIV, which had been treated with a combination of abacavir, dolutegravir, and lamivudine. He also had a history of ritonavir use as part of the HIV treatment (Table 1) and had reported occasional blurry vision while on ritonavir. At the initial visit, the BCVA was 20/30 OD and 20/30 OS. A slitlamp examination revealed a quiet anterior chamber. A fundus examination demonstrated central, well-demarcated retinal atrophy in both eyes (Figure 3, A and B). Fundus autofluorescence photographs identified a corresponding area of hypoautofluorescence with surrounding hyperfluorescence in both eyes (Figure 3, C and D). OCT showed outer retinal atrophy in both eyes that was more severe in the left eye than the right eye (Figure 3, E and F). Findings from a prior workup for infectious or inflammatory causes of uveitis were unremarkable.
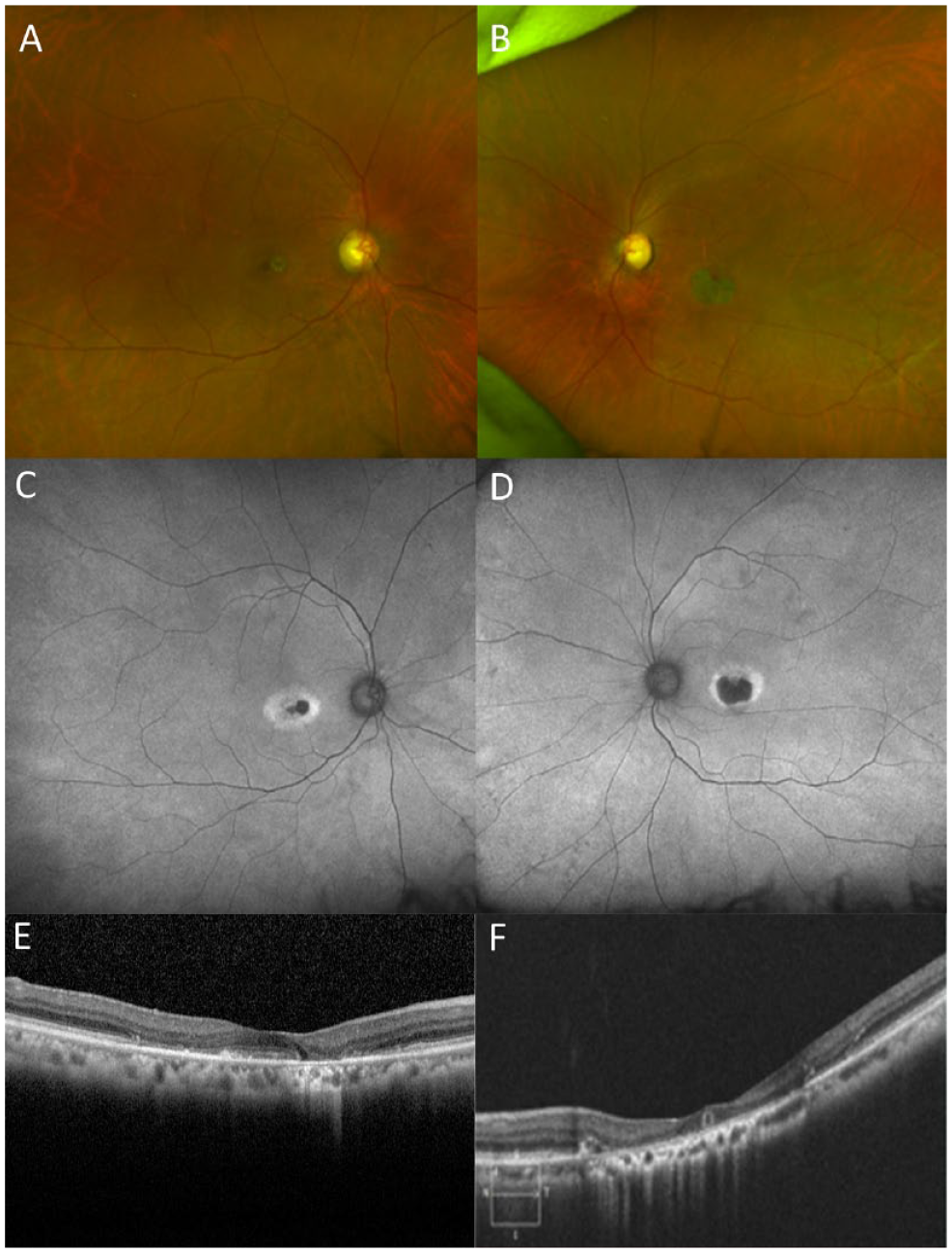
(A and B) Ultra-widefield fundus photographs demonstrated bilateral well-demarcated retinal atrophy in the central macula in both eyes. (C and D) Fundus autofluorescence photographs revealed a corresponding area of hypoautofluorescence with surrounding hyperfluorescence in both eyes. (E and F) Optical coherence tomography revealed outer retinal atrophy in both eyes with the left eye being more severe than the right eye.
Given the absence of intraocular inflammation and the pattern of macular atrophy, the patient was diagnosed with a drug-induced toxic maculopathy secondary to ritonavir. His VA showed moderate decline to 20/40 OD and 20/70 OS after 3 years of follow-up.
Conclusions
In this case series of 3 patients with HIV presenting with visual complaints, multimodal retinal imaging showed evidence of maculopathy and disease findings consistent with ritonavir-associated retinal toxicity. Although the differential diagnosis of RPE change and retinopathy is broad and may include HIV retinopathy, prior opportunistic infections, malignancy including lymphoma, and autoimmune retinopathy, drug toxicity remains in the differential diagnosis. Besides ritonavir, other medications used to treat HIV, including clofazimine, didanosine, efavirenz, and zidovudine, have previously been linked to retinal toxicity. 8 Although the use of ritonavir is limited in the treatment of persons living with HIV, evaluating for its retinal toxicity should be considered given the recent emergency authorization of ritonavir/nirmatrelvir in the treatment of COVID-19.
The pathophysiology of ritonavir-associated retinopathy remains poorly understood. Multiple mechanisms have been proposed to explain retinal changes in animal models that occur when they are exposed to ritonavir. In rodent studies, repeated administration of high doses of ritonavir led to lysosomal damage and excess accumulation of phospholipids in the retina. 9 Phospholipid-induced inflammatory reactions with resultant histopathological changes, including hypertrophy of the RPE and retinal degeneration in the setting of ritonavir exposure, have also been described.10,11 On electron microscopy, amorphous, granular inclusion bodies characteristic of phospholipid accumulation were observed in the retina. 10 Ritonavir can also cause retinal toxicity by inhibiting hypoxia-inducible factor 1α and vascular endothelial growth factor (VEGF). 12 Specifically, inhibition of VEGF led to choriocapillaris and photoreceptor degeneration in mice.12,13 Ritonavir has also been suspected to cause photoreceptor damage by depleting neurotrophic factors via VEGF-pathway inhibition. 13
Prior case series have documented ritonavir-associated retinopathy8,14 –21 and corroborate the disease phenotypes observed over the long-term follow-up of patients described in the present series. Given the recent US FDA approval of nirmatrelvir/ritonavir for mild to moderate COVID-19 in adult and pediatric patients,6,22 this series serves as a reappraisal of the literature to consider this toxicity in the COVID-19 era. Common fundus findings observed in prior series of persons with HIV8,14 –21 include macular atrophy with or without foveal involvement, midperipheral bone spicule-like pigmentary changes, RPE hypertrophy, and intraretinal crystal deposits. These findings are consistent with the macular atrophy pattern observed in our patients.
While the pathogenesis of ritonavir-associated retinopathy is unknown, the presence of intraretinal crystals has been observed with adaptive optics imaging, supporting the hypothesis that phospholipids may accumulate in this disease entity. 15 Besides the fundus findings, OCT has demonstrated the pattern of RPE and outer retinal atrophy as seen in our cases. Of note, 3 of 11 patients reported in the literature showed maculopathy with foveal atrophy, all of whom were taking ritonavir for a longer duration than other reported cases.8,15,17 This association suggests that ritonavir may also lead to toxicity related to duration in addition to a dose-related phenomenon as previously described. 16 Of note, 1 patient has been reported to show RPE thickening and inner-segment and outer-segment irregularity that resolved 2 weeks after ritonavir discontinuation. 21 This suggests that if detected early, immediate cessation of ritonavir may help prevent further damage to retinal structures. In addition, prior studies have shown patients with liver disease, such as hepatitis, may be predisposed to ritonavir-associated retinopathy, as observed in Case 2 in the present series.8,19,23 Therefore, careful consideration should be taken prior to using ritonavir in patients with COVID-19 who have liver comorbidities given that liver injury commonly occurs in patients with COVID-19. 24
While the current use of nirmatrelvir/ritonavir involves short-term, 5-day use, the near-term effects of ritonavir related to the retina have not been reported. Prior literature has shown that the shortest duration of therapy before retinopathy symptoms was 19 months. 20 With the ongoing COVID-19 pandemic, ritonavir is used much more widely in combination with nirmatrelvir among patients with and without HIV, and ocular symptoms should be monitored and assessed if a patient develops visual symptoms.
In summary, 3 patients with HIV developed retinopathy consistent with ritonavir-mediated damage. While their findings developed with extended use of ritonavir, the increasing use of combination nirmatrelvir/ritonavir for COVID-19 warrants a reminder of the potential toxicity associated with extended use. Ophthalmologists, primary care providers, and infectious disease providers should remain aware of this potential toxicity with long-term use of ritonavir, and near-term effects should be assessed in symptomatic individuals.
Footnotes
Authors’ Note
N.V. Nguyen and Dr. Kim contributed equally to this work as co–first authors.
Ethical Approval
This case series was conducted in accordance with the Declaration of Helsinki. The collection and evaluation of all protected patient health information was performed in a US Health Insurance Portability and Accountability Act–compliant manner.
Statement of Informed Consent
The written informed consent for patient information was not sought for the case series because all patient identifier information was not reported.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Eye Institute/National Institutes of Health under award number K23 EY030158 (Dr. Shantha) and R01 EY029594 (Dr. Yeh). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government. This research was supported by an Emory University Robert W. Woodruff Health Sciences Center Fund COVID-19 Synergy Award. Grant support was also provided by the Macula Society Retina Research Foundation, ARVO Mallinckrodt Young Investigator Grant, and the Stanley M. Truhlsen Family Foundation, Inc.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
